Editing of endogenous tubulins reveals varying effects of tubulin posttranslational modifications on axonal growth and regeneration
Figures
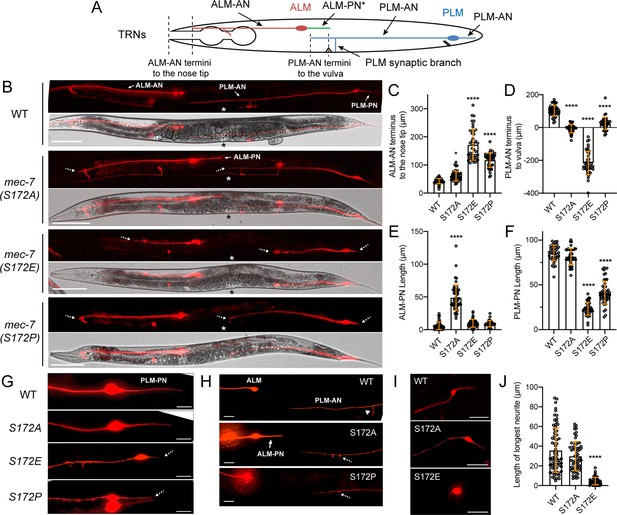
Substitution of MEC-7/β-tubulin S172 led to neurite growth defects in C. elegans TRNs.
(A) Neurite morphologies of ALM and PLM neurons. ALM-PN (green) is not prominent in the wild-type animals. ALM-AN and PLM-AN length are measured by the distance of their termini to the nose tip and the vulva (indicated by the distance between the dash lines). (B) TRN morphologies in wild-type animals, S172A, S172E, and S172P mutants. Specific neurites are indicated by arrows. Asterisks mark the position of the vulva. Dashed arrows point to the termini of neurites that are shortened in mutants. Scale bar = 100 μm. (C) The distance of ALM-AN terminus to the tip of the nose in various strains. The longer the distance, the shorter the ALM-AN. One and four asterisks indicate p<0.05 and 0.0001, respectively, in statistical significance when compared with the wild type in a post-ANOVA Dunnett’s test. (D) The distance from the PLM-AN terminus to the vulva in various strains. If the anteriorly directed PLM-AN grew past the vulva, the distance is positive. If PLM-AN cannot reach the vulva, the distance is negative. (E) Quantification of ALM-PN length. The wild-type animals have no or very short ALM-PN. (F) Quantification of PLM-PN length. (G) Representative images of PLM-PN in various strains. Dashed arrows indicate the neurite termini of shortened PLM-PN. (H) Representative images of ALM-PN in mec-7(S172A) mutants. Arrowhead indicates the synaptic branch of PLM-AN in the wild-type animals. Dashed arrows point to the branching defects where the synaptic branch failed to extend to the ventral cord. (I) TRNs extracted from the embryos of S172 mutants and cultured in vitro; they were identified by their expression of mec-17p::TagRFP among the embryonic cells. TRNs from S172E mutants had no or very short neurites. (J) The length of the longest neurite of the in vitro cultured TRNs from the S172 mutants. Scale bar = 20 μm.
-
Figure 1—source data 1
Numeric data for Figure 1B–E , and I.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig1-data1-v1.xlsx
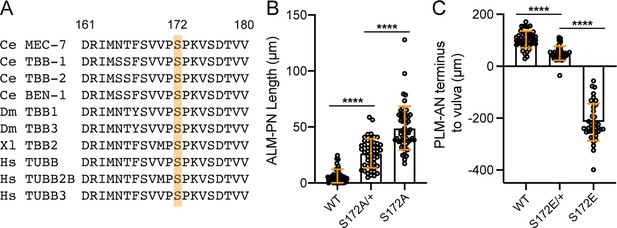
mec-7 S172A and S172E mutations are semidominant.
(A) Evolutionary conservation of S172 (highlighted) and flanking sequences among β-tubulin genes across species. Ce for C. elegans, Dm for Drosophila melanogaster, Xl for Xenopus laevis, and Hs for Homo sapiens. (B) The length of ALM-PN in day-one adults (three days after bleaching) of wild-type animals, mec-7(S172A)/+ heterozygotes, and mec-7(S172A) homozygotes. (C) The distance from the PLM-AN terminus to the vulva in the wild-type animals, mec-7(S172E)/+ heterozygotes, and mec-7(S172E) homozygotes. If PLM-AN grew past the vulva, the distance is positive. If PLM-AN cannot reach the vulva, the distance is negative. Four asterisks indicate p<0.0001 in a post-ANOVA Tukey’s HSD test.
-
Figure 1—figure supplement 1—source data 1
Numeric data for Figure 1—figure supplement 1B and C.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig1-figsupp1-data1-v1.xlsx
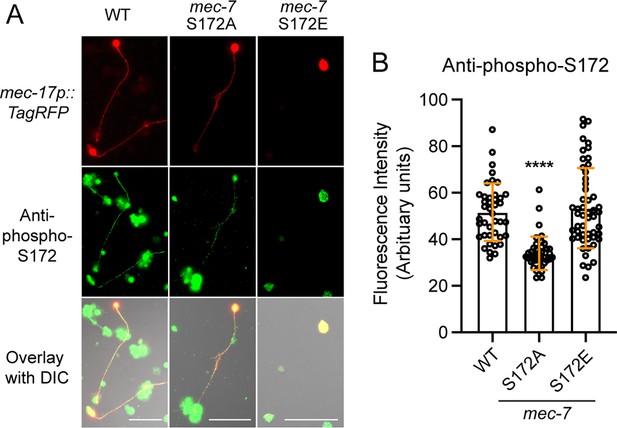
mec-7 S172 mutations altered β-tubulin S172 phosphorylation level.
(A) Anti-β-tubulin phospho-S172 antibody staining of in vitro cultured C. elegans embryonic cells from the indicated strains. Cells expressing the mec-17p::TagRFP were identified as the TRNs. Scale bar = 20 μm. (B) Fluorescent intensity in the axons were measured and quantified. Four asterisks indicate p<0.0001 in a Dunnett’s test comparing the mutant cells with the wild-type cells.
-
Figure 1—figure supplement 2—source data 1
Numeric data for Figure 1—figure supplement 2B.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig1-figsupp2-data1-v1.xlsx
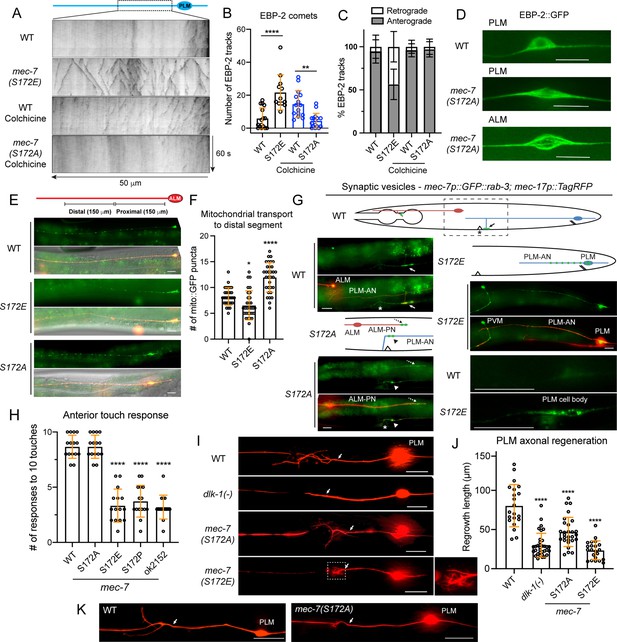
MEC-7/β-tubulin S172 phosphorylation regulates MT dynamics, cargo transport, mechanosensory function, and axonal regeneration.
(A) Representative kymographs of EBP-2::GFP dynamics in the PLM-AN of various strains. The wild-type animals and S172A mutants were subjected to a mild colchicine treatment to increase MT dynamics and imaged after a one-hour recovery. (B) Quantification of the number of EBP-2 tracks. One, two, and four asterisks indicate p<0.05, 0.01, and 0.0001, respectively, in statistical significance when compared with the wild type. (C) Percentages of retrograde and anterograde movement for the EBP-2 comets. (D) Comparison of the EBP-2::GFP signal in the cell body of wild-type and mec-7(S172A) animals. (E) Distribution of mitochondria in the ALM-AN indicated by the jsIs609 [mec-7p::mitoGFP] signal. (F) The quantification of the number of mitoGFP puncta in the distal segment of ALM-AN (150–300 μm away from the cell body). (G) Localization of the synaptic vesicles (GFP::RAB-3), which are indicated by green dots in the cartoon of S172 mutants. The fluorescent image for the wild type is a representative image of the region in the dashed box of the cartoon; arrows indicate the normal localization of RAB-3 signal to the synapses made by synaptic branch of PLM-AN in the ventral nerve cord posterior to the vulval position (indicated by the asterisk). In S172A mutants, RAB-3 is mistargeted to the ALM-PN (dashed arrow) and accumulates at the PLM-AN (arrowhead) when the synaptic branch fails to extend, and the axon hooks ventrally. In S172E mutants, RAB-3 signal was trapped in the cell body or the proximal segment of the neurite as PLM-AN is severely shortened. (H) Anterior touch responses of various mec-7 mutants; mec-7(ok2151) is a deletion allele. (I) Representative axonal regrowth of PLM-AN following laser axotomy in various strains; arrows indicate the cut site. (J) Quantification of PLM regrowth length for the regrowth cases. (K) Examples of reconnections after laser axotomy; arrows indicate the cut site. Scale bars = 20 μm for all panels.
-
Figure 2—source data 1
Numeric data for Figure 2B–C, F, H and J.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig2-data1-v1.xlsx
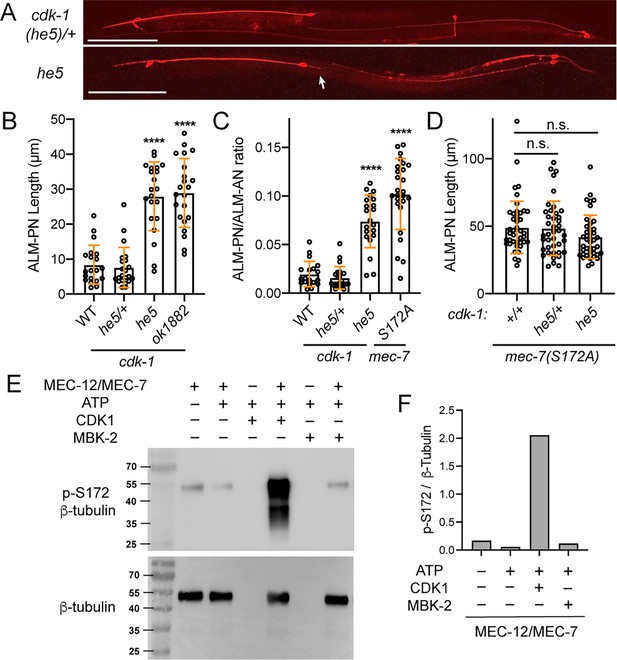
CDK-1 mediates MEC-7/β-tubulin S172 phosphorylation and regulates neurite growth.
(A) Images of TRN morphologies in cdk-1(he5)/+ heterozygotes and cdk-1(he5) homozygotes (escapers). Arrow points to the ectopic ALM-PN. Scale bar = 100 μm. (B) The length of ALM-PN in wild-type and cdk-1 mutants. ALM-PN length in the escapers of he-5 and ok1882 homozygous mutants were measured. (C) The ratio of ALM-PN length to ALM-AN length in various strains. (D) The ALM-PN length in animals carrying both mec-7(S172A) and cdk-1 mutations. (E) Western blot of the kinase reaction samples using anti-phospho-S172 (p–S172) antibodies. The kinase reaction was conducted in the MEM buffer with 475 nM CDK1/Cyclin B or 500 nM MBK-2m. The weak p-S172 signal in the first lane likely resulted from the nonspecific interaction of the anti-p-S172 antibodies with the unmodified MEC-12/MEC-7 heterodimer or the background phosphorylation level of the recombinant dimer. Anti-β-tubulin antibodies were used to ensure the equal loading of the tubulin dimers. (F) Quantification of the grey intensity ratio of p-S172 to β-tubulin signals in the western blot.
-
Figure 3—source data 1
Numeric data for Figure 3B–D , and F.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig3-data1-v1.xlsx
-
Figure 3—source data 2
Labeled uncropped western blot image for the blots shown in Figure 3E and Figure 3—figure supplement 3.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig3-data2-v1.pdf
-
Figure 3—source data 3
Unlabeled uncropped western blot image for the blots shown in Figure 3E and Figure 3—figure supplement 3.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig3-data3-v1.zip
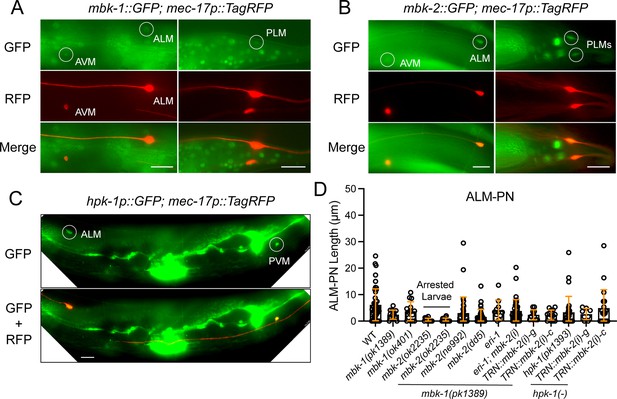
Expression and phenotypes of the minibrain homologs, mbk-1, mbk-2, and hpk-1 in the TRNs.
(A) The expression of cmIs6 [mbk-1::GFP] in the TRNs labeled by mec-17p::TagRFP. Circles indicate the expression of GFP in TRNs, including ALM, PLM, and AVM neurons. (B) The expression of cmEx6 [mbk-2p::GFP] in the TRNs. (C) The expression of unkEx194 [hpk-1Bp::GFP] in TRNs. Scale bar = 20 μm for all panels. (D) Quantifications of ALM-PN length in various strains. ALM-PN was measured in the arrested larvae of the mbk-2(ok2235) single and mbk-1(pk1389); mbk-2(ok2235) double mutants; the rest were measured in adults. Both ne992 and dd5 are temperature-sensitive alleles and were first grown at 15 °C to L1 stage and then shifted to 25 °C and scored at adult stage. mbk-2(i) indicates feeding RNAi done in the CGZ963 mbk-1(pk1389) X; eri-1(mg366ts) IV; uIs115 IV strain for enhanced RNAi. TRN::mbk-2(i)-g and -c indicate double strand RNA expressed from the TRN-specific mec-17 promoter against the genomic DNA and cDNA of mbk-2, respectively.
-
Figure 3—figure supplement 1—source data 1
Numeric data for Figure 3—figure supplement 1D.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig3-figsupp1-data1-v1.xlsx
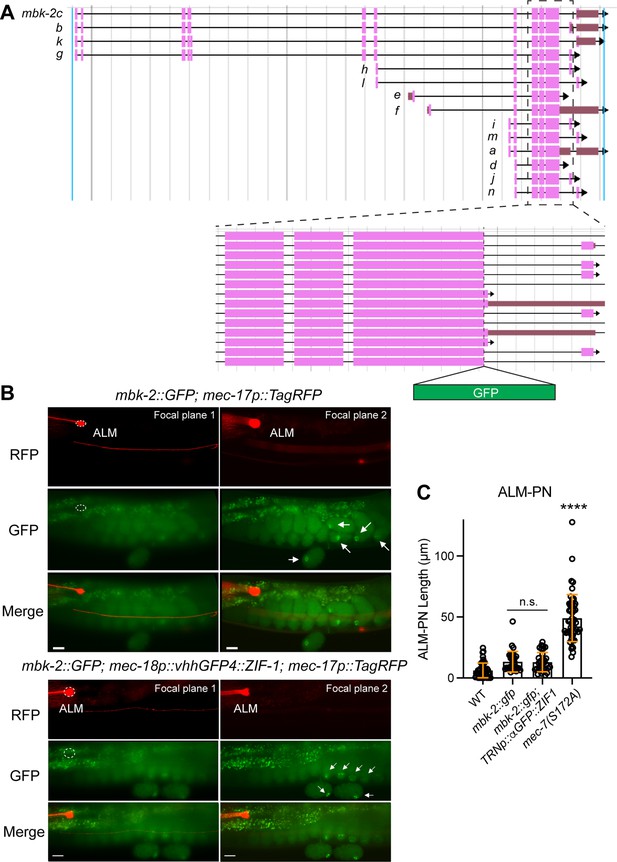
Potential TRN-specific degradation of MBK-2 does not result in the growth of long ALM-PN.
(A) Gene structure of mbk-2 downloaded from https://wormbase.org/ (WS292); the exon structures of 14 isoforms are shown. Dashed box is enlarged to show the GFP insertion site. (B) Expression of MBK-2::GFP in adult animals with or without the TRN-specific expression of ZIF-1 fused with anti-GFP nanobodies (vhhGFP4::ZIF-1). No GFP signals (dashed circles) were detected in TRNs labeled by mec-17p::TagRFP, but GFP signals (arrows) were found in the embryos (with potential concentration in the P lineage). (C) Quantification of ALM-PN length in various strains. n.s. indicates no statistical significance in a post-ANOVA Tukey’s HSD test. Four asterisks indicate p<0.0001 in comparison with the wild-type animals.
-
Figure 3—figure supplement 2—source data 1
Numeric data for Figure 3—figure supplement 2C.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig3-figsupp2-data1-v1.xlsx
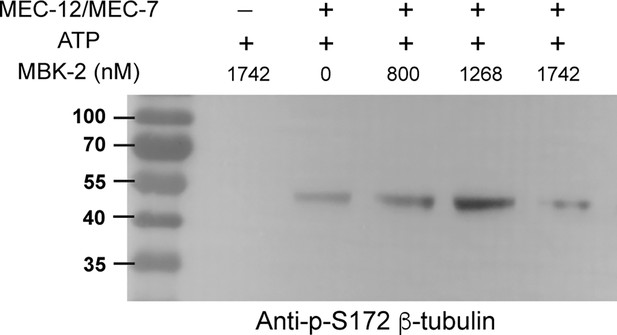
Kinase assay for MBK-2 in BRB80 buffer.
500 nM recombinant MEC-12/MEC-7 heterodimer and 1 mM ATP were incubated with various concentration of MBK-2. Western blot of the kinase reaction samples was done using the anti-phospho-S172 antibodies.
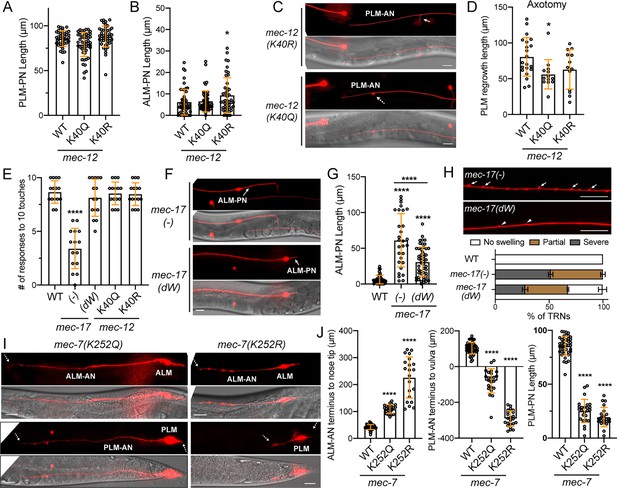
The effects of tubulin acetylation on neurite development.
(A) The PLM-PN length in mec-12 K40 mutants. (B) The length of ALM-PN in mec-12 K40 mutants. (C) 20% of PLM-AN failed to extend the synaptic branch in mec-12(K40Q) mutants. Arrow points to the normal synaptic branch in mec-12(K40R) mutants and the dashed arrow indicates the branching defects. See Figure 1H for the wild-type control. (D) Quantification of the PLM regrowth length after laser axotomy in mec-12 K40 mutants. (E) Anterior touch responses of mec-17 and mec-12 mutants. The deletion allele ok2109 was used for mec-17(-) and the unk126 (G121W & G123W) allele was used for mec-17(dW). (F) Comparison of ALM-PN in mec-17(-) and mec-17(dW) mutants. (G) Quantification of ALM-PN lengths in mec-17 mutants. Four asterisks indicate p<0.0001 in a post-ANOVA Tukey’s honestly significant difference (HSD) test. (H) Comparison and quantification of the neurite swelling and looping phenotypes in mec-17(-) and mec-17(dW) mutants (ALM-AN is shown). For the quantification, severe swelling (arrows) is defined by having two or more bumps taller than 1 μm within 100 μm length of axon; partial swelling (arrowheads) is defined by having smaller or fewer bumps than the severe ones; no swelling is defined by having no >0.5 μm bumps in the entire axon. (I) The ALM and PLM morphologies in mec-7 K252 mutants. Dashed arrows point to the termini of the shortened ALM-AN or PLM-AN or PLM-PN. See Figure 1B for the wild-type control. (J) Quantification of the shortening of the three neurites. For ALM-AN, the distance from the neurite ending to the nose tip was measured; the bigger the gap, the shorter the ALM-AN. For PLM-AN, the distance from the neurite terminus to the vulva was measured. Positive value means the neurite grew past the vulva and negative value means the neurite did not reach the vulva. Scale bars = 20 μm for all panels.
-
Figure 4—source data 1
Numeric data for Figure 4A–B, D–E, G–H , and J.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig4-data1-v1.xlsx

Evolutionary conservation of α-tubulin K40 and β-tubulin K252 residues.
(A) Comparison of K40 (highlighted) and the flanking sequences (a.a. 31–40) in MEC-12 with other α-tubulin genes across species. Ce for C. elegans, Dm for Drosophila melanogaster, Xl for Xenopus laevis, and Hs for Homo sapiens. (B) The conservation of K252 (highlighted) and the adjacent sequences (a.a. 241–260) among β-tubulin genes across species.
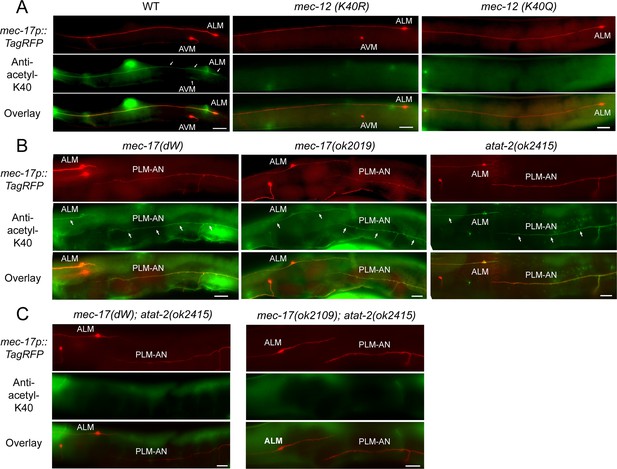
mec-12 K40R and K40Q mutations and the combined loss of mec-17 and atat-2 affect α-tubulin K40 acetylation.
(A) Anti-acetyl-K40 staining of the wild-type, mec-12(K40R), and mec-12(K40Q) animals. Only the wild-type animals showed the staining in TRNs. Neither mec-12(K40R) nor mec-12(K40Q) mutants showed any staining. Arrows point to the staining signal in cell bodies or axons. Scale bar = 20 μm. (B–C) Anti-acetyl-K40 staining of acetyltransferase mutants. mec-17(dW), mec-17(-), and atat-2(-) single mutants showed clear staining signal, whereas the mec-17; atat-2 double mutants showed no signal at all. Arrows point to the staining signal in the neurites. Scale bar = 20 μm.
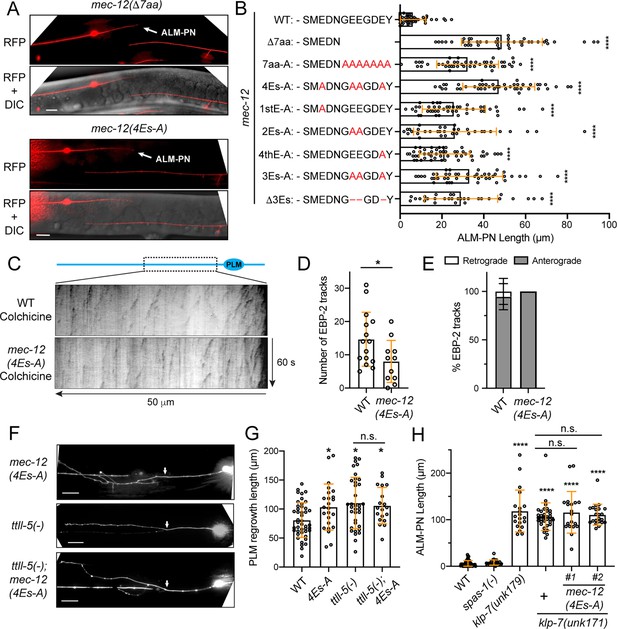
Tubulin polyglutamylation regulates neurite growth and regeneration.
(A) The growth of ectopic ALM-PN (arrow) in mec-12 mutants lacking the polyglutamylation sites. (B) The quantification of ALM-PN length in various mec-12 mutant strains. The changes made to the last twelve amino acids of MEC-12 in the various alleles are shown. (C) Kymographs showing EBP-2::GFP dynamics in wild-type and mec-12(4Es-A) animals after a mild colchicine treatment and a one-hour recovery. (D) Quantification of the number of EBP-2 comets in mec-12(4Es-A) mutants. (E) The percentage of retrograde and anterograde EBP-2 movements. (F) PLM regrowth following laser axotomy in mec-12(4Es-A) and ttll-5(tm3360) mutants and the double mutants; arrows indicate the cut site. (G) The quantification of PLM regrowth length in various strains. (H) ALM-PN lengths in spas-1(tm683), klp-7(unk179; Δ11bp), klp-7(unk171; Δ14bp), and klp-7; mec-12(4Es-A) double mutants. unk171 was generated by gene editing in both the wild-type and the mec-12(4Es-A) background, and #1 and #2 were two independent lines. See Figure 5—figure supplement 2 for the details of the klp-7 alleles. Scale bars = 20 μm for all panels; n.s. indicates no statistical significance in a post-ANOVA Tukey’s HSD test.
-
Figure 5—source data 1
Numeric data for Figure 5B, D–E , and G–H.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig5-data1-v1.xlsx

Sequence divergence of α-tubulin C-terminal tails in C. elegans.
Comparison of the amino acid sequences of the C-terminal region of the α-tubulin isotypes. The last twelve amino acids of MEC-12 are highly divergent among the isotypes (boxed region).
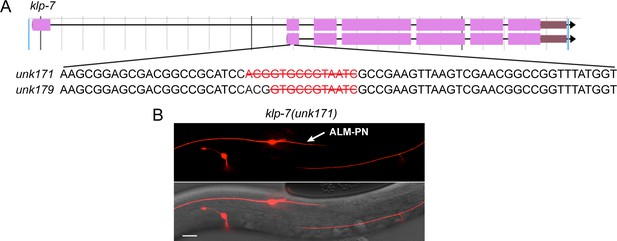
Mutations in klp-7 result in the growth of an ectopic ALM-PN.
(A) The gene structure of klp-7 (from https://wormbase.org/) and the nucleotides deleted in unk171 and unk179 alleles, which were generated by CRISPR/Cas9-mediated gene editing. (B) The ectopic ALM-PN in klp-7(unk171) mutants.
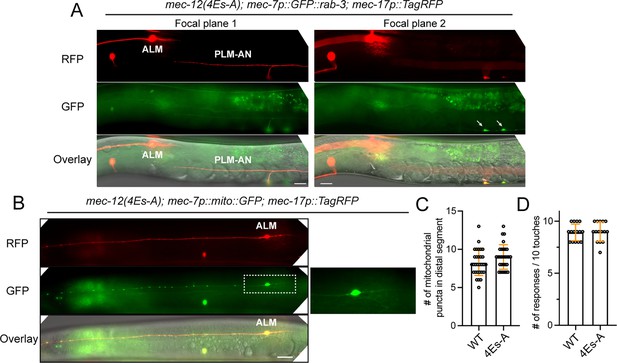
The lack of cargo transport and mechanosensory defects in mec-12(4Es-A) mutants.
(A) The normal localization of synaptic vesicles (GFP::RAB-3) to the synaptic branch (arrows) of PLM-AN in mec-12(4Es-A) mutants. Focal plane 1 focuses on the neurites, while focal plane 2 focuses on the ventral cord. (B) Distribution of mitochondria in the ALM-AN of mec-12(4Es-A) mutants. (C) Quantification of the number of mito::GFP puncta in the distal segment (150~300 μm from the cell body) of ALM-AN. (D) Anterior touch response in the mec-12(4Es-A) mutants.
-
Figure 5—figure supplement 3—source data 1
Numeric data for Figure 5—figure supplement 3C–D.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig5-figsupp3-data1-v1.xlsx
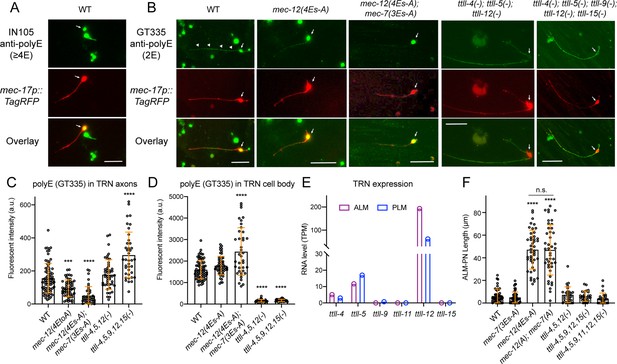
Substitution of glutamates in MEC-12 and MEC-7 C-terminal tails eliminate tubulin polyglutamylation in the axons.
(A) Antibody staining of in vitro cultured TRNs (labeled by RFP) with IN105 anti-polyglutamylation antibodies that recognize a chain of four or more glutamates. (B) Antibody staining of in vitro cultured TRNs from various strains with GT335 anti-polyglutamylation antibodies that recognize the branchpoint and a side chain of two glutamyl units. CGZ1554 ttll-4(tm3310); ttll-5(tm3360); ttll-12(unk185); uIs115 and CGZ1475 ttll-4(tm3310); ttll-5(tm3360) ttll-9(tm3889) ttll-15 (tm3871); ttll-12(unk185); uIs115 were used in the experiment. Arrows point to the TRN cell bodies and arrowheads point to the staining signal in the axon. (C–D) Quantification of the fluorescent intensity (in arbitrary units or a.u.) for the polyE staining of TRN axons and cell bodies using GT335. (E) RNA level of the ttll genes in ALM and PLM neurons according to the L4 stage single-cell transcriptomic data obtained from https://wormbase.org/. (F) ALM-PN length in mec-7, mec-12, and ttll mutant strains. CGZ1554, CGZ1475, and CGZ1474 ttll-4(tm3310); ttll-11(tm4059); ttll-5(tm3360) ttll-9(tm3889) ttll-15(tm3871); ttll-12(unk185); zdIs5 were used. Scale bars = 20 μm for all panels. Three and four asterisks indicate p<0.001 and 0.0001, respectively, in a post-ANOVA Dunnett’s test comparing all mutants with the wild-type animals.
-
Figure 6—source data 1
Numeric data for Figure 6C–F.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig6-data1-v1.xlsx
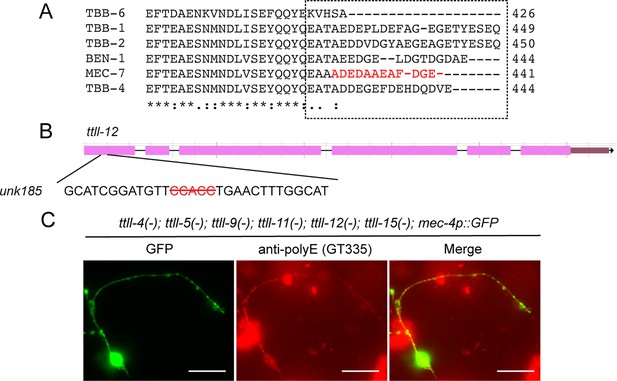
Sequence divergence of the C-terminal tail among β-tubulin isotypes and the molecular details of ttll-12 CRISPR allele.
(A) Comparison of the amino acid sequences of the C-terminal region of the β-tubulin isotypes in C. elegans. The last 12 amino acids of MEC-7 are divergent among the isotypes (boxed region). (B) Five nucleotides (red with strikethrough) were deleted in ttll-12(unk185) allele. (C) Anti-polyglutamylation staining of CGZ1474 ttll-4(tm3310) III; ttll-11(tm4059) IV; ttll-5(tm3360) ttll-9(tm3889) ttll-15(tm3871) V; ttll-12(unk185) II; zdIs5[mec-4p::GFP] I with GT335 antibodies and rhodamine-conjugated rabbit anti-mouse secondary antibodies. The staining pattern is similar to the quintuple mutants shown in Figure 6B.
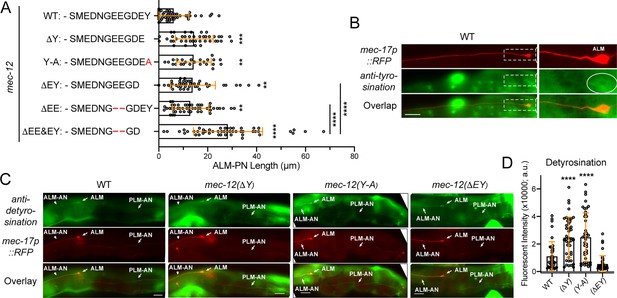
The effects of tubulin detyrosination and Δ2 modification on neurite growth.
(A) The length of ALM-PN in strains deleting the terminal tyrosine or the last two residues. Two, three, and four asterisks correspond to p<0.05, 0.001, and 0.0001, respectively, in a post-ANOVA Tukey’s HSD test. (B) The weak staining of wild-type TRNs with the anti-tyrosinated α-tubulin antibodies. (C) The staining of the wild-type animals and the various mec-12 mutants with anti-detyrosinated α-tubulin antibodies. Arrows point to the ALM cell bodies, ALM-AN, and PLM-AN (zoom in to see the signal). (D) Quantification of the fluorescent intensity of the anti-detyrosination staining signal in the axons. Arbitrary units (a.u.) were used.
-
Figure 7—source data 1
Numeric data for Figure 7A and D.
- https://cdn.elifesciences.org/articles/94583/elife-94583-fig7-data1-v1.xlsx
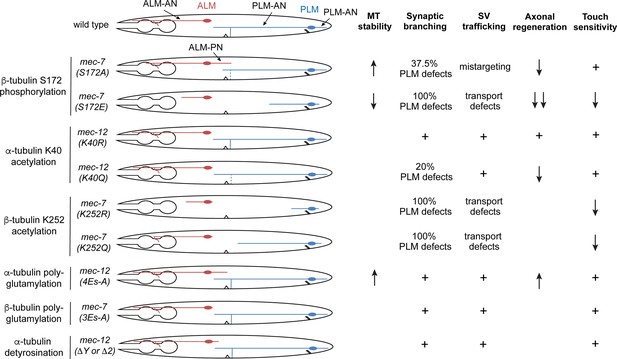
Summary of the phenotypes of the tubulin PTM site mutants.
Cartoons represent the morphologies of the ALM and PLM neurons in various mec-7 and mec-12 mutants. Severe shortening of neurites often indicates phenotypes of antimorphic alleles, while the growth of a prominent ALM-PN is characteristic of neomorphic alleles. Dashed lines indicate partial defects of forming the PLM synaptic branch. For synaptic vesicle (SV) trafficking, mistargeting means that SV is trafficked to ALM-PN; transport defects mean that SV is mostly trapped in cell bodies or proximal segment of the neurite. MT stability is measured by EBP-2 comets; increased EBP-2 dynamics indicates reduced MT stability, and reduced EBP-2 dynamics indicates elevated MT stability. Blank space means phenotype not determined; “+” means phenotype similar to the wild-type animals.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (C. elegans) | mec-12 | NA | WormBase ID: WBGene00003175 | |
| Gene (C. elegans) | mec-7 | NA | WormBase ID: WBGene00003171 | |
| Gene (C. elegans) | klp-7 | NA | WormBase ID: WBGene00002219 | |
| Gene (C. elegans) | cdk-1 | NA | WormBase ID: WBGene00000405 | |
| Gene (C. elegans) | mbk-2 | NA | WormBase ID: WBGene00003150 | |
| Strain, strain background (C. elegans N2) | uIs115 [Pmec-17::RFP] IV | PMID: 26460008 | TU4065 | |
| Strain, strain background (C. elegans N2) | mec-7(u1137; S172A) X; uIs115 [mec-17p::TagRFP] IV | This study | TU6235 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(u1136; S172E) X; uIs115 [mec-17p::TagRFP] IV | This study | TU6234 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(u1056; S172P) X; uIs115 [mec-17p::TagRFP] IV | This study | TU6549 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | uIs115; juIs338 [mec-4p::ebp-2::GFP +ttx-3p::RFP]. | PMID: 33378215 | CGZ562 | |
| Strain, strain background (C. elegans N2) | mec-7(u1136; S172E) X; uIs115 IV; juIs338 [mec-4p::ebp-2::GFP +ttx-3p::RFP]. | This study | CGZ563 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(u1137; S172A) X; uIs115 IV; juIs338 [mec-4p::ebp-2::GFP +ttx-3p::RFP]. | This study | CGZ564 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | uIs134 [mec-17p::TagRFP] V; jsIs821[mec-7p::GFP::rab-3] X | This study | TU5595 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(u1136; S172E) X; jsIs821 X; uIs134 V | This study | CGZ941 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(u1137; S172A) X; jsIs821 X; uIs134 V | This study | CGZ942 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | jsIs973 [mec-7p::mRFP +unc-119(+)] III. jsIs609 [mec7p::mtGFP +lin-15(+)] X. | PMID: 23051668 | NM4244 | |
| Strain, strain background (C. elegans N2) | mec-7(u1137; S172A) X; jsIs973 [mec-7p::mRFP +unc-119(+)] III. jsIs609 [mec7p::mtGFP +lin-15(+)] X | This study | CGZ1019 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(u1136; S172E) X; jsIs973 [mec-7p::mRFP +unc-119(+)] III. jsIs609 [mec7p::mtGFP +lin-15(+)] X | This study | CGZ1020 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | dlk-1(ju476) I; uIs115 [mec-17p::TagRFP] IV | This study | CGZ263 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | uIs134; cmIs6 [(pBR104) mbk-1::GFP +pNC4.21]. | This study | CGZ70 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | uIs134; cmEx6 [mbk-2p::GFP; rol-6(D)] | This study | CGZ64 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | unc-119(ed3) III; unkEx194 [hpk-1Bp::GFP; unc-119(+)] | This study | CGZ915 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-1(pk1389); uIs115 | This study | CGZ62 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-2(dd5); uIs134 | This study | CGZ76 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-2(ne992ts); uIs134 | This study | CGZ93 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-2(ok2235) IV/nT1 [qIs51] (IV;V); zdIs5 [mec-4p::GFP] I | This study | CGZ127 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-1(pk1389) X; mbk-2(dd5ts) IV; uIs134 V | This study | CGZ366 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-1(pk1389) X; mbk-2(ne992ts) IV; uIs134 V | This study | CGZ367 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-1(pk1389) X; mbk-2(ok2235) IV/nT1 [qIs51] (IV;V); zdIs5 I | This study | CGZ828 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-1(pk1389) X; mbk-2(ok2235) IV/nT1 [qIs51] (IV;V); uIs31 III | This study | CGZ829 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mbk-1(pk1389) X; uIs115 IV; unkEx101[mec-17p::mbk-2-dsRNA; ceh-22p::GFP] | This study | CGZ404 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | cdk-1(he5)/hT2; uIs115 IV | This study | CGZ1178 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | cdk-1(ok1882) III/hT2 [bli-4(e937) let-?(q782) qIs48] (I;III); uIs134 V | This study | CGZ97 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | cdk-1(he5)/hT2; mec-7(u1137; S172A) X; uIs115 IV | This study | CGZ1394 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | hpk-1(pk1393); uIs134 | This study | CGZ66 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk119; K40Q) III; uIs115 IV | This study | CGZ814 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk120; K40R) III; uIs115 IV | This study | CGZ815 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-17(unk126; dW) IV; uIs115 IV | This study | CGZ1017 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-17(ok2109) IV;uIs134 V | This study | CGZ895 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-17(ok2109) IV; atat-2(ok2415) X;uIs134 V | This study | CGZ1098 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-17(unk126; dW) IV; atat-2(ok2415) X; uIs115 IV | This study | CGZ1099 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | atat-2(ok2415) X; uIs115 IV | This study | CGZ1100 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(unk143; K252Q) X; uIs115 IV | This study | CGZ1138 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(unk144; K252R) X; uIs115 IV | This study | CGZ1139 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-7(unk135; 3Es-A) X; uIs115 IV | This study | CGZ1097 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk124; Δ7aa) III; uIs115 IV | This study | CGZ1015 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk133; 7aa-A) III; uIs115 IV | This study | CGZ1095 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk136; 4Es-A) III; uIs115 IV | This study | CGZ1000 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk148; 1stE-A) III; uIs115 IV | This study | CGZ1175 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk155; 2Es-A) III; uIs115 IV | This study | CGZ1184 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk153; 4thE-A) III; uIs115 IV | This study | CGZ1182 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk138; 3Es-A) III; uIs115 IV | This study | CGZ1133 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk150; Δ3E) III; uIs115 IV | This study | CGZ1176 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk122; ΔY) III; uIs115 IV | This study | CGZ847 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk123; Y-A) III; uIs115 IV | This study | CGZ1014 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk121; ΔEY) III; uIs115 IV | This study | CGZ846 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk131; ΔEE) III; uIs115 IV | This study | CGZ1093 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk151; ΔEE&EY) III; uIs115 IV | This study | CGZ1177 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk136; 4Es-A) III; mec-7(unk135; 3Es-A) V; uIs115 IV | This study | CGZ896 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | spas-1(tm683) V; uIs115 IV | This study | CGZ1179 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | spas-1(ok1608) V; uIs115 IV | This study | CGZ1180 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | klp-7(unk179; Δ11bp) III; uIs115 IV | This study | CGZ1183 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | klp-7(unk171; Δ14bp) III; uIs115 IV | This study | CGZ1259 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | klp-7(unk171; Δ14bp) III; mec-12(unk136; 4Es-A) III; uIs115 IV | This study | CGZ1260 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk136; 4Es-A) III; uIs115 IV; juIs338 | This study | CGZ1256 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk136; 4Es-A) III; uIs115 IV; jsIs609 X | This study | CGZ1257 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | mec-12(unk136; 4Es-A) III; uIs115 IV; jsIs821 X | This study | CGZ1258 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | ttll-5(tm3360); uIs115 | This study | CGZ159 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | ttll-5(tm3360) V; mec-12(unk136; 4Es-A) III; uIs115 IV | This study | CGZ1393 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | ttll-4(tm3310) III; ttll-5(tm3360) V; ttll-12(unk185; Δ5bp) II; uIs115 IV | This study | CGZ1554 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | ttll-4(tm3310) III; ttll-5(tm3360) ttll-9(tm3889) ttll-15 (tm3871) V; ttll-12(unk185; D5nt) II; uIs115 IV | This study | CGZ1475 | Chaogu Zheng lab |
| Strain, strain background (C. elegans N2) | ttll-4(tm3310) III; ttll-11(tm4059) IV; ttll-5(tm3360) ttll-9(tm3889) ttll-15(tm3871) V; ttll-12(unk185 d5nt) II; zdIs5 I | This study | CGZ1474 | Chaogu Zheng lab |
| Antibody | Rabbit polyclonal anti-phospho-S172 | abcam | ab76286 | 1:100 for staining worms; 1:1000 for staining cells; 1:3000 for WB |
| Antibody | Mouse monoclonal anti-acetyl-K40 | abcam | ab24610 | Same as above |
| Antibody | Mouse monoclonal anti-polyE GT335 | AdipoGen Life Science | AG-20B-0020-C100 | Same as above |
| Antibody | Rabbit polyclonal anti-polyE chain IN105 | AdipoGen Life Science | AG-25B-0030-C050 | Same as above |
| Antibody | Rabbit polyclonal anti-glycylated tubulin Gly-pep1 | AdipoGen Life Science | AG-25B-0034-C100 | Same as above |
| Antibody | Rat monoclonal anti-tyrosinated α-tubulin | Sigma | MAB1864-I | Same as above |
| Antibody | Mouse monoclonal anti-detyrosinated α-tubulin | Sigma | MAB5566 | Same as above |
| Antibody | Goat anti-Mouse IgG, Alexa Fluor Plus 488 | Thermo Fisher | A32723 | 1:1000 for staining worms and cells |
| Antibody | Goat anti-Rat IgG, Alexa Fluor 488 | Thermo Fisher | A11006 | 1:1000 |
| Antibody | TRITC-Goat Anti-Mouse IgG | Jackson ImmunoResearch | 115-025-164 | 1:1000 |
| Antibody | Alexa Fluor 488 Goat Anti-Rabbit IgG | Jackson ImmunoResearch | 111-545-003 | 1:1000 |
| Antibody | TRITC-Goat Anti-Rabbit IgG | Jackson ImmunoResearch | 111-025-003 | 1:1000 |
| Antibody | Alexa Fluor 488 Goat Anti-Mouse IgG | Jackson ImmunoResearch | 115-545-003 | 1:1000 |
| Recombinant DNA reagent | mec-17p::mbk-2-sense | This study | CGZ#194 | Chaogu Zheng lab |
| Recombinant DNA reagent | mec-17p::mbk-2-antisense | This study | CGZ#195 | Chaogu Zheng lab |
| Recombinant DNA reagent | hpk-1bp::GFP | This study | CGZ#261 | Chaogu Zheng lab |
| Recombinant DNA reagent | hpk-1cp::GFP | This study | CGZ#262 | Chaogu Zheng lab |
| Recombinant DNA reagent | L4440-mbk-2(gDNA) RNAi | This study | CGZ#338 | Chaogu Zheng lab |
| Recombinant DNA reagent | L4440-mbk-2(cDNA) RNAi | This study | CGZ#339 | Chaogu Zheng lab |
| Recombinant DNA reagent | L4440 empty control | This study | CGZ#378 | Chaogu Zheng lab |
| Software, algorithm | LAS X Life Science Microscope Software Platform | Leica Microsystems | N/A |
Additional files
-
Supplementary file 1
CRISPR targets and repair templates used for gene editing.
- https://cdn.elifesciences.org/articles/94583/elife-94583-supp1-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/94583/elife-94583-mdarchecklist1-v1.docx





