Early evolution of the ecdysozoan body plan
Figures
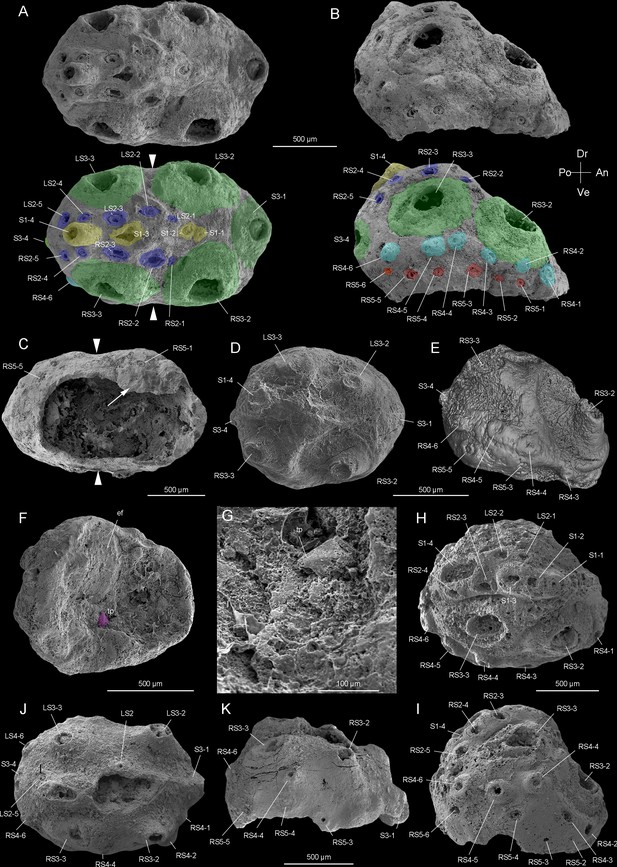
Beretella spinosa gen. et sp. nov. from Member 5 of the Yanjiahe Formation (Cambrian Stage 2), Yichang, Hubei Province, China.
(A‒C) Holotype, CUBar138-12. (A) Dorsal view showing the external ornament: (five sclerites at the midline in yellow (S1); flanked by two rows of sclerites in blue (S2); large broad-based conical sclerites in two dorsolateral pairs and one antero-posterior pairs in green (S3)); white arrows indicate lateral constriction. (B) Right lateral view showing two additional rows of six sclerites (S4 and S5, in light blue and pink, respectively). (C) Ventral view showing a large opening that may have accommodated the mouth (see the text) and an empty body cavity. (D‒G) CUBar75-45. (D) Dorsal view showing a broken S3. (E) Micro-CT image, right lateral view displaying S4. (F) Ventral view depicting a tiny projection in purple. (G) An enlargement of the projection of F. (H‒I) Paratype, CUBar171-5. (H) Right dorsal view showing S1‒S4. (I) Right-lateral view showing S4 and S5. (J‒K) Paratype CUBar121-8. (J) Dorsal view showing poorly preserved S1 and S2. (K) Right-lateral view showing S3‒S5. A, assumed anterior end (see text); ef, exotic fragment; D, assumed dorsal side; L, left; P, posterior end; R, right; tp, tiny spine; V, ventral side. The same abbreviations are used throughout the manuscript including supplementary files.
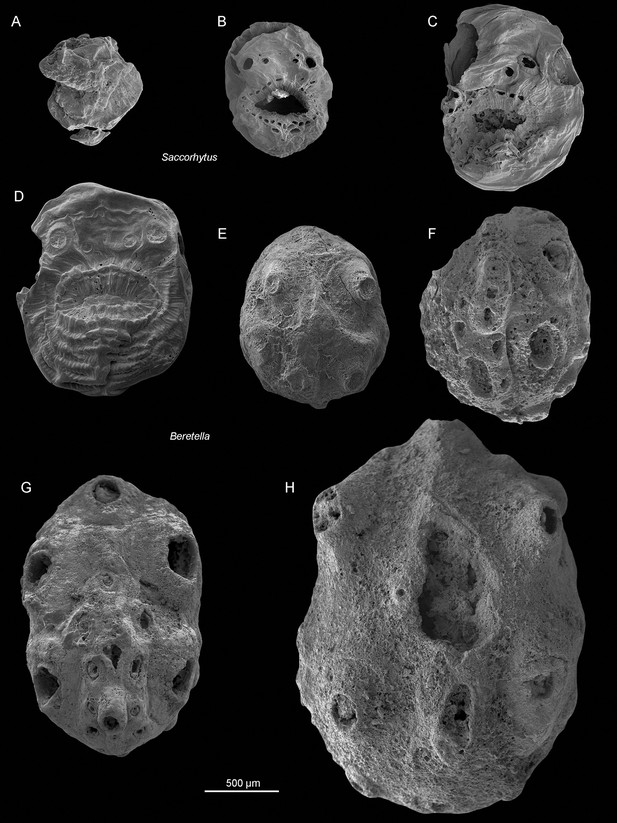
Size variation between Saccorhytus coronarius and Beretella spinosa.
(A-D) Saccorhytus coronarius; ELIXX25-62, ELIXX48-64, ELIXX61-27, and ELIXX65-296, respectively. (E-G) Beretella spinosa; CUBar75-45, CUBar171-5, CUBar138-12, and CUBar121-8, respectively. All specimens at the same scale (500 µm). All SEM images. ‘A-C’ are new photographs of published specimens (Han et al., 2017).
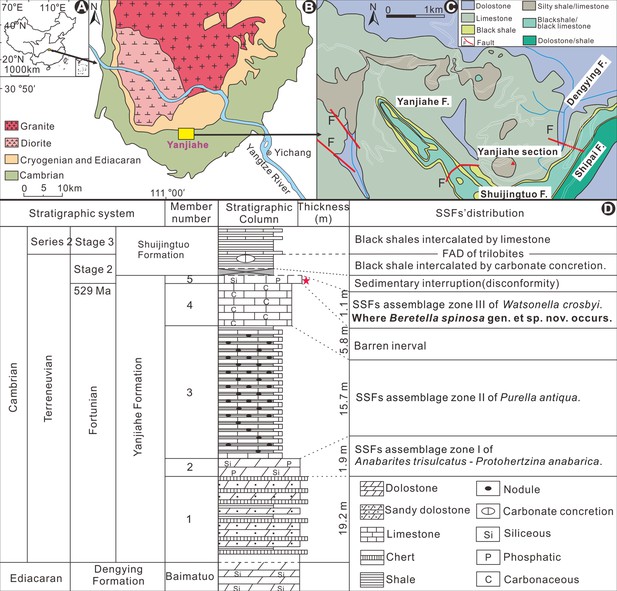
Origin of fossil material.
(A-D) Geological background and locality map and stratigraphy (Guo et al., 2019). The red star indicates Member 5 of the Yanjiahe Formation where Beretella spinosa was found; this fossiliferous horizon is correlated with the bottom of Cambrian Stage 2 (Terreneuvian; ca. 529 Ma).

Typical Small Shelly Fossils (SSFs) found associated with Beretella spinosa in Member 5 of the Yanjiahe Formation.
(A) CUBar21-4, Watsonella crosbyi. (B) CUBar206-6, Aldanella attleborensis.
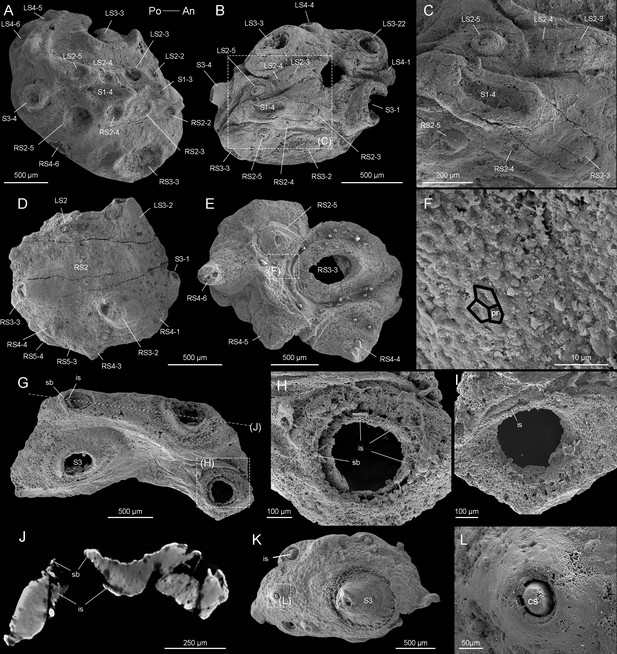
Beretella spinosa gen. et sp. nov.
(A) CUBar99-19, dorsal view showing an ornament S1‒S4. (B, C) CUBar136-9, general dorsal view and details. (D) CUBar136-11, dorsal view showing S1‒S5. (E, F) CUBar73-15 general view and details of the cuticular polygonal reticulation in black. (G‒J) CUBar128-27. (G) General view. (H, I) details of outer and inner surface of the bi-layered structure of the cuticular wall as seen in broken conical sclerites. (J) Micro-CT section showing possibly sclerite infilling. (K, L) CUBar99-18, cuticular fragment, general view and details of large sclerite (central feature represents possible phosphatic infilling). is, infilling sclerite; pr, polygonal reticulation; sb, sclerite base.
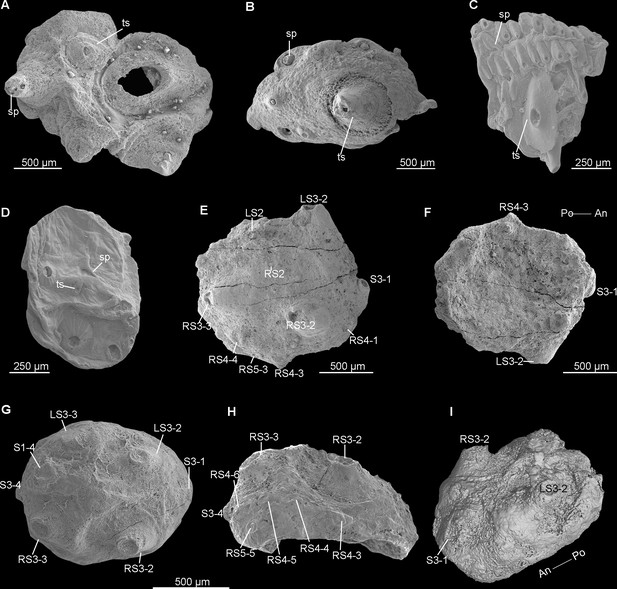
Truncated sclerites in early Cambrian saccorhytids and scalidophoran worms.
(A, B) Beretella spinosa (CUBar73-15 and CUBar99-18, respectively). (C) Undetermined scalidophoran worm (ELIXX57-320). (D) Saccorhytus coronarius (ELIXX34-298). (E-I) Beretella spinosa. (E, F) CUBar136-11, dorsal view showing arrangement of S1‒S5; ventral view. (G-I) CUBar75-45. (G) Dorsal view showing two pairs of S3 pointing outwards (tip broken). (H) Lateral-right view showing RS4 and RS5. (I) Micro-CT image, intermediate dorsal view showing S3-1. All SEM images except ‘I’. ‘D’ is a new photograph of a published specimen (Han et al., 2017). Sp, spiny sclerite; ts, truncated sclerite. (Rich media 3- Micro-CT render of CUBar128-27).

Saccorhytus coronarius, multi-layered secondarily phosphatized cuticle.
(A-C) ELIXX45-20, general view and close-ups showing chevron patterns on inner and outer layers. D-F, ELIXX58-336, general view and close-ups showing bi-layered cuticle (not on a sclerite) with chevron pattern on both layers. (G-L) ELIXX65-116, showing bi-layered cuticular structure; general view (SEM), Micro-CT section parallel to the specimen’s external surface, and close-up in I. (J-N) ELIXX99-420. (J-L) General view and details showing two- or three-layered cuticular structures. (M, N) Micro-CT transverse and vertical sections through the cuticle (see location in j) showing tri-layered cuticular structures. All SEM images except ‘H, L, M, N’. ‘A’ is a new photograph of a published specimen (Han et al., 2017). il, inner layer; ilc, inner layer chevron; Mo, mouth; ol, outer layer; olc, outer layer chevron. (Rich media 4. Micro-CT render of ELIXX65-116, Rich media 5. Micro-CT render of ELIXX99-420).
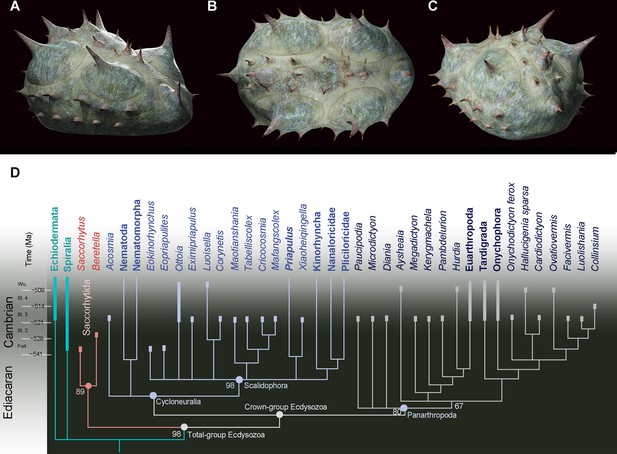
Position of Beretella spinosa in the animal tree based on cladistic analysis.
(A-C), artistic three-dimensional reconstructions of Beretella spinosa in the anterolateral (A), dorsal (B), and posterolateral views (C). (D) Phylogenetic tree obtained from cladistic analyses using maximum likelihood. Saccorhytus and Beretella join in a clade (new phylum Saccorhytida) resolved as the sister-group of all other ecdysozoans; numbers at key nodes denote probability. Fossil and extant taxa are in italics and bold, respectively. Known fossil record indicated by thicker vertical bars (after Shu and Han, 2020).
-
Figure 3—source code 1
The dataset (matrix) for cladistic analysis.
- https://cdn.elifesciences.org/articles/94709/elife-94709-fig3-code1-v1.zip
-
Figure 3—source data 1
Characters description for cladistic analysis.
- https://cdn.elifesciences.org/articles/94709/elife-94709-fig3-data1-v1.docx
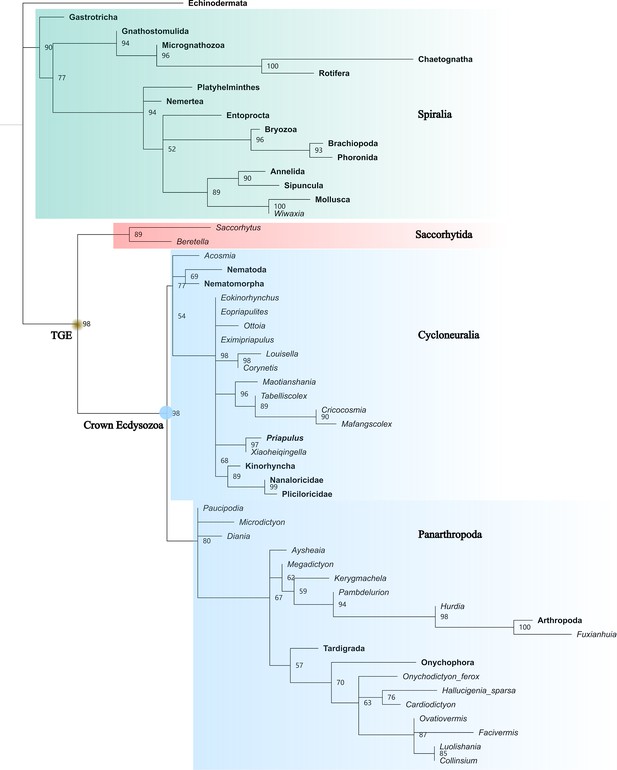
Full maximum likelihood tree generated by IQTREE.
50% majority rule consensus. Total-group Ecdysozoa (TGE, yellow dot) contains Saccorhytida, Cycloneuralia, and Panarthropoda. Saccorhytida is resolved as the sister group of the crown-group Ecdysozoa (blue dot, Cycloneuralia and Panarthropoda). Cycloneuralia is well resolved as a monophyletic group including Scalidophora, Nematoida, and Acosmia. The relationships of cycloneuralian ingroups are unclear. Numbers close to nodes are probabilities. Taxa in bold represent extant species.
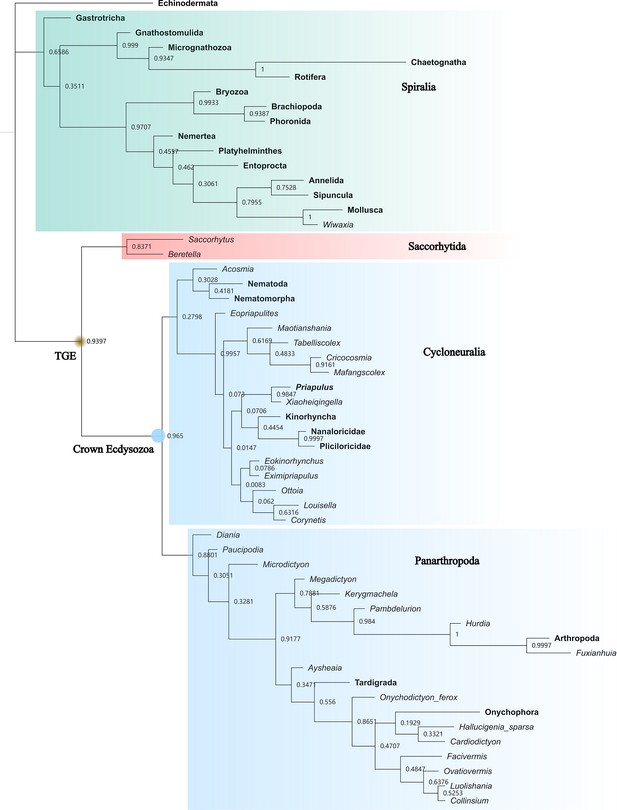
Bayesian inference tree generated by MrBayes.
50% majority rule consensus. Total-group Ecdysozoa (TGE, yellow dot) contains Saccorhytida, Cycloneuralia, and Panarthropoda. Saccorhytida is resolved as the sister group of the crown-group Ecdysozoa (blue dot, Cycloneuralia and Panarthropoda). Cycloneuralia is resolved as a monophyletic group including Scalidophora and Nematoida but the probabilities of Cycloneuralia and Nematoida +Acosmia nodes are very low. Acosmia is resolved as a stem member of Nematoida. Numbers close to nodes are posterior probabilities. Taxa in bold represent extant species.
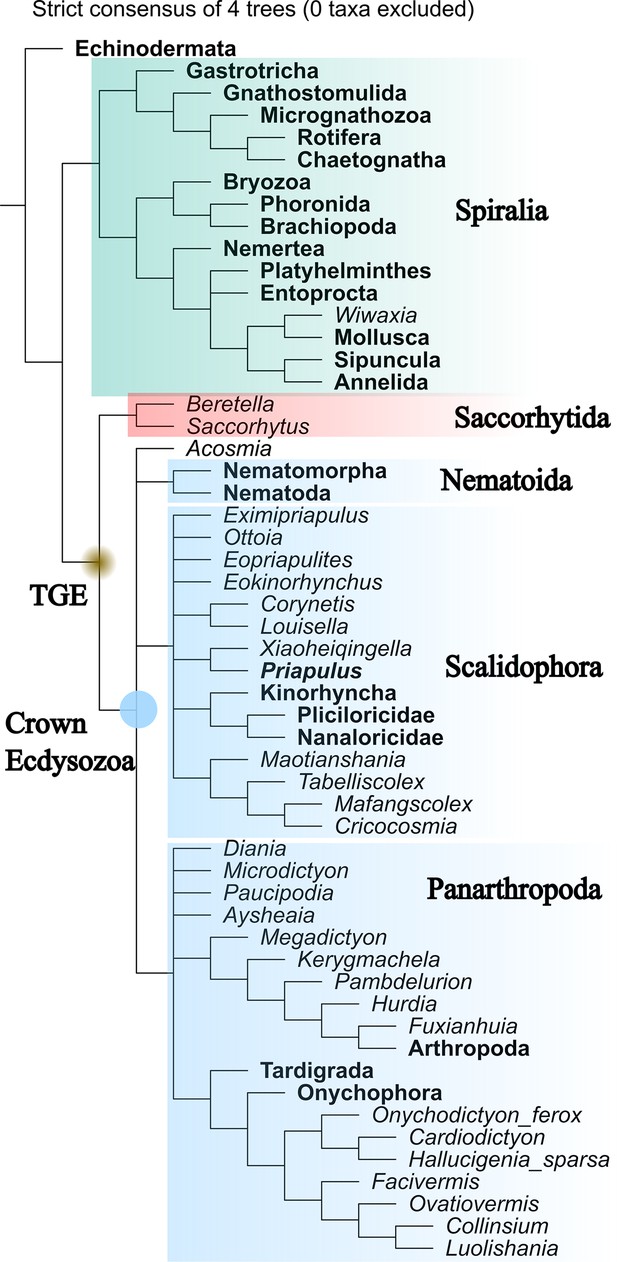
Maximum parsimony tree generated by TNT (equal weight).
Strict consensus of four most parsimonious trees. Total-group Ecdysozoa (TGE, yellow dot) contains Saccorhytida, Acosmia, Nematoida, Scalidophora, and Panarthropoda. Saccorhytida is resolved as the sister group of the crown-group Ecdysozoa (blue dot, Acosmia, Nematoida, Scalidophora, and Panarthropoda). Note that Cycloneuralia does not appear as a monophyletic clade. The relationships of crown-group ecdysozoan ingroups are unclear. Taxa in bold represent extant species.
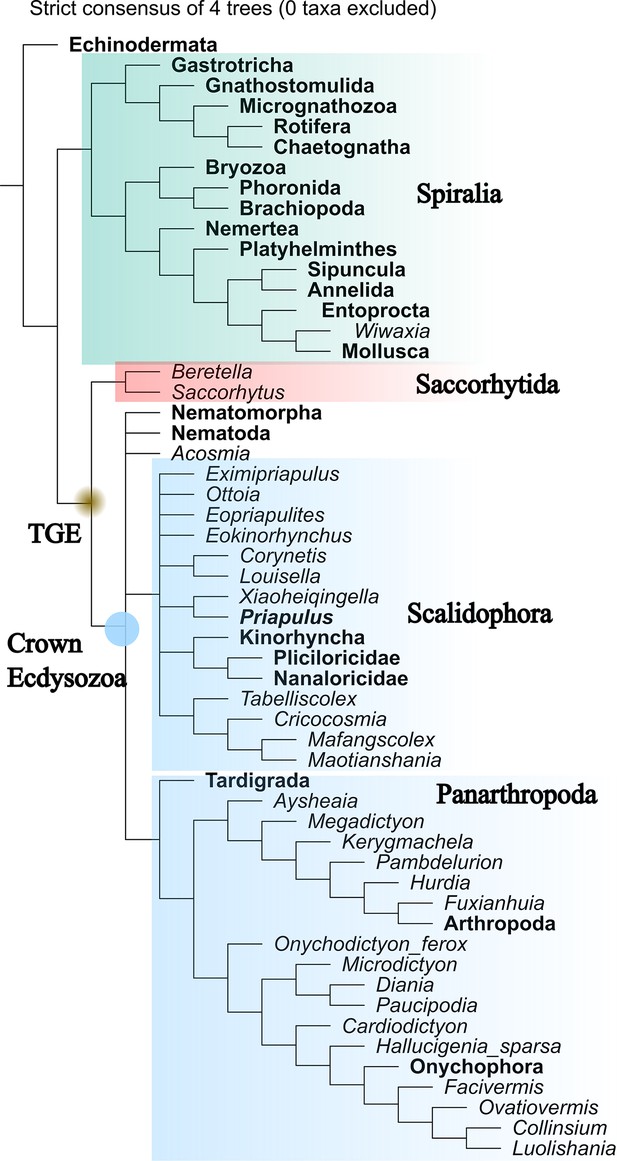
Maximum parsimony tree generated by TNT (implied weight, k=3).
Strict consensus of four most parsimonious trees. Total-group Ecdysozoa (TGE, yellow dot) contains Saccorhytida, Acosmia, Nematoda, Nematomorpha, Scalidophora, and Panarthropoda. Saccorhytida is resolved as the sister group of the crown-group Ecdysozoa (blue dot, Acosmia, Nematoda, Nematomorpha, Scalidophora, and Panarthropoda). Note that Cycloneuralia does not appear as a monophyletic clade. The relationships of crown-group ecdysozoan ingroups are unclear. Taxa in bold represent extant species.
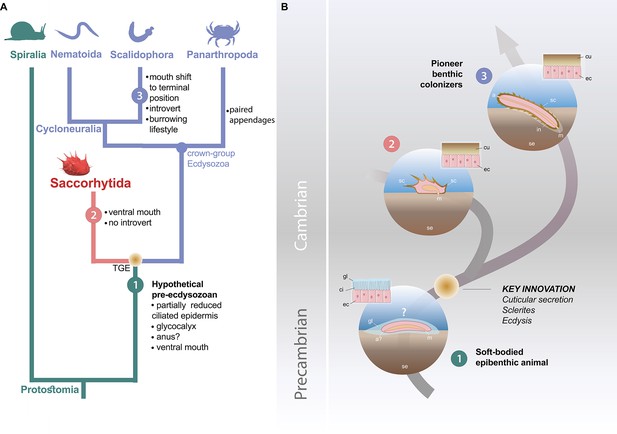
Possible evolutionary scenario to explain the origin and early evolution of ecdysozoans.
(A) Summary tree (see Figure 3—figure supplements 1–4) showing saccorhytids as a sister-group of Cycloneuralia (Nematoida plus Scalidophora)+Panarthropoda; main morphological features of each group listed along each branch. (B) Potential evolutionary pathway to evolve Saccorhytida and crown-group Ecdysozoa. Numbers in green, red and blue circles designate pre-ecdysozoan (Spiralia), Saccorhytida and Cycloneuralia, respectively. Light brown gradient (circle) to emphasize ecdysis and sclerite secretion seen as key evolutionary steps. 1, Hypothetical pre-ecdysozoan animal with a ciliated epidermis and glycocalyx. 2, Saccorhytid exemplified by Beretella with a cuticle bearing sclerites. 3, Crown-group ecdysozoan exemplified by a scalidophoran worm with an elongated shape, a differentiated head (introvert) and trunk, sclerites, a through gut, a terminal mouth and abilities to burrow into bottom sediment. Animals not to scale. Abbreviations: a, anus; a?, uncertain status of anus; ci, cilia; cu, cuticle; ec, epidermal cell; gl, glycocalyx (mucous layer); m, mouth; in, introvert; sc, sclerite; se, sediment; TGE, total-group Ecdysozoa. Silhouettes from phylopic.org. (CC BY 3.0 or public domain): Spiralia (by Martin R. Smith), Nematoida (by Birgit Lang), Scalidophora (by Fernando Carezzano), and Panarthropoda (by Harold N Eyster). Saccorhytida generated from reconstruction of Figure 3.
Videos
Animation of holotype of Beretella spinosa.
Animation of holotype of Beretella spinosa without color.
Additional files
-
Supplementary file 1
Meaurements of Saccorytida and ancestral character state reconstruction of Cycloneuralia.
(a) Measurements of Beretella. L, length; W, width; H, height; ae, anterior end; B, body; pe, posterior end; PP, polygonal net-like pattern; tp, tiny spine; VO, ventral opening;?, no accurate measurement possible. (b) Length/width ratio of Beretella and Saccorhytus. (c) Similarities and differences between Beretella and Saccorhytus. AP, antero-posterior; DV, dorso-ventral side; LR, left-right. (d) Ancestral character state reconstructions for the topology where Cycloneuralia is monophyly. Values of ancestral character state reconstructions. 0=absence of character, 1=presence of character, P=posterior probability. TGE, total-group Ecdysozoa; CGE, crown-group Ecdysozoa, SA, Saccorhytida. (e) Ancestral character state reconstructions for the topology where Cycloneuralia is paraphyletic. Values of ancestral character state reconstructions. 0=absence of character, 1=presence of character, P=posterior probability. TGE, total-group Ecdysozoa; CGE, crown-group Ecdysozoa, SA, Saccorhytida.
- https://cdn.elifesciences.org/articles/94709/elife-94709-supp1-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/94709/elife-94709-mdarchecklist1-v1.docx





