Dual engagement of the nucleosomal acidic patches is essential for deposition of histone H2A.Z by SWR1C
Figures
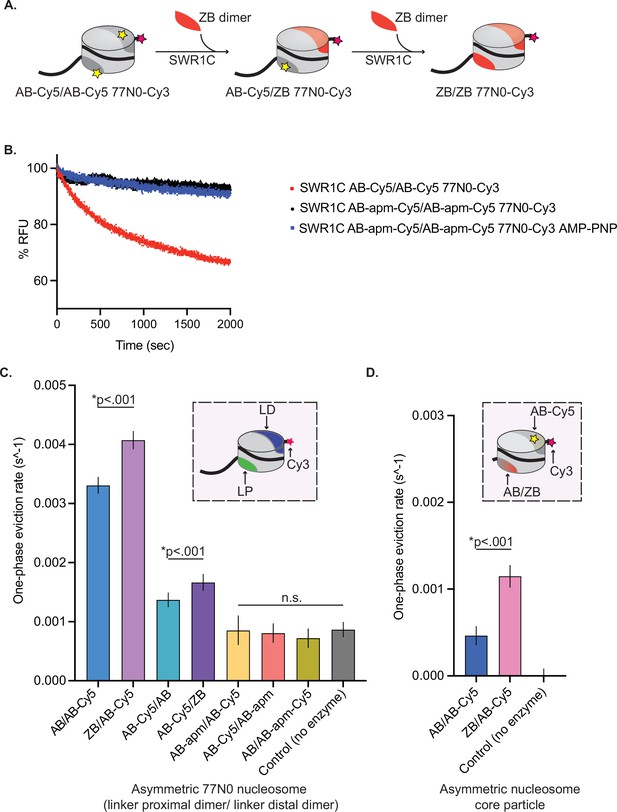
H2A.Z deposition requires each nucleosomal acidic patch.
(A) Schematic of SWR1C-mediated dimer exchange. Cylinder is representative of a nucleosome; light gray area of cylinder represents H3/H4 tetramer, dark gray area of cylinder represents AB heterodimer, yellow star represents Cy5, pink star represents Cy3, and solid black line represents DNA. The nucleosome undergoes two rounds of SWR1C-mediated dimer eviction where ZB heterodimers (orange) replace AB heterodimers. (B) 77N0-Cy3 nucleosomes were remodeled by SWR1C under single turnover conditions (30 nM SWR1C, 10 nM 77N0-Cy3 nucleosomes, 70 nM ZB dimers, 1 mM ATP or AMP-PNP). Dimer eviction is monitored by measuring Cy5 emission at 670 nm. SWR1C was able to perform dimer exchange on an AB-Cy5/AB-Cy5 nucleosome (red line) but was unable to exchange dimers on an AB-apm-Cy5/AB-apm-Cy5 nucleosome (black line). The AMP-PNP reaction contained the AB-apm-Cy5/AB-apm-Cy5 nucleosomal substrate. (C) Asymmetrically assembled 77N0-Cy3 and Cy3-0N77 nucleosomes were remodeled by SWR1C under single turnover conditions (50 nM SWR1C, 10 nM nucleosomes, 50 nM ZB dimers, 1 mM ATP) and one-phase eviction rates were calculated in Prism 9 and plotted. The dimer labels are listed in linker proximal (LP), linker distal (LD) order (see inset for template nucleosome represented as a cylinder with H3/H4 tetramer in light gray, LP dimer in green, LD dimer in dark blue, DNA as a solid black line with Cy3 (pink star) on the 0bp-linker side). Nucleosomes with a ZB dimer contralateral to the dimer being evicted had significantly faster rates for both linker distal (first two bars) and linker proximal (second two bars). Linker distal eviction was also significantly faster than linker proximal eviction. Rates for nucleosomes containing an AB-apm dimer contralateral to the dimer being evicted (fifth and sixth bars) were not significantly different from no enzyme controls (last bar) in either orientation. SWR1C was also unable to evict a dimer with an acidic patch mutant (seventh bar). (D) Asymmetrically assembled 0N0-Cy3 nucleosomes were remodeled by SWR1C under single turnover conditions (50 nM SWR1C, 10 nM nucleosomes, 50 nM ZB dimers, 1 mM ATP) and one-phase eviction rates were calculated in Prism 9 and plotted. Nucleosomes contained a single AB-Cy5 dimer and a contralateral AB or ZB dimer (see inset for template nucleosome represented as a cylinder with H3/H4 tetramer in light gray, AB-Cy5 dimer in dark gray, contralateral AB or ZB dimer represented by dark gray to orange gradient, 0N0-Cy3 DNA as a solid black line with Cy3 (pink star) label on one side). AB-Cy5/ZB 0N0-Cy3 nucleosomes had significantly faster eviction than AB-Cy5/AB 0N0-Cy3 nucleosomes. At least three independent nucleosome preparations were used for substrate, and error bars reflect 95% confidence intervals from at least three replicates.
-
Figure 1—source data 1
Excel file containing raw data for experiments depicted in panels B–D of Figure 1.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig1-data1-v1.xlsx
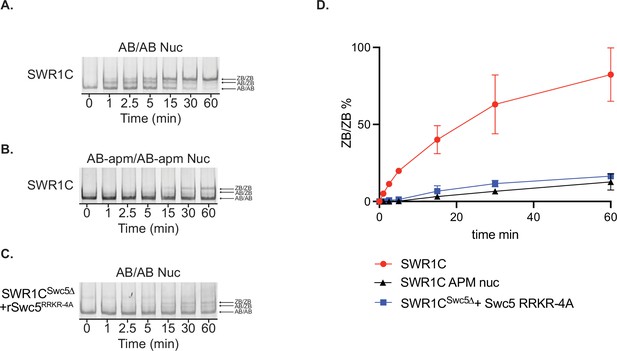
Gel-based assay for H2A.Z deposition.
SWR1C-mediated ZB deposition was measured by incorporation of H2AZ3x-FLAG/H2B dimers at various time points on 77N0 AB/AB (A) or 77N0 AB-apm/AB-apm (B) nucleosomes. SWR1CSwc5∆+rSwc5RRKR-4A-mediated ZB deposition on 77N0 AB/AB nucleosomes was also measured (C). Percent of remodeled nucleosome in each condition was quantified using ImageQuant and plotted in (D).
-
Figure 1—figure supplement 1—source data 1
Excel file containing raw data for experiments depicted in panel D of Figure 1—figure supplement 1.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig1-figsupp1-data1-v1.xlsx
-
Figure 1—figure supplement 1—source data 2
Zip file containing raw and annotated images for gels shown in panels A–C of Figure 1—figure supplement 1.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig1-figsupp1-data2-v1.zip
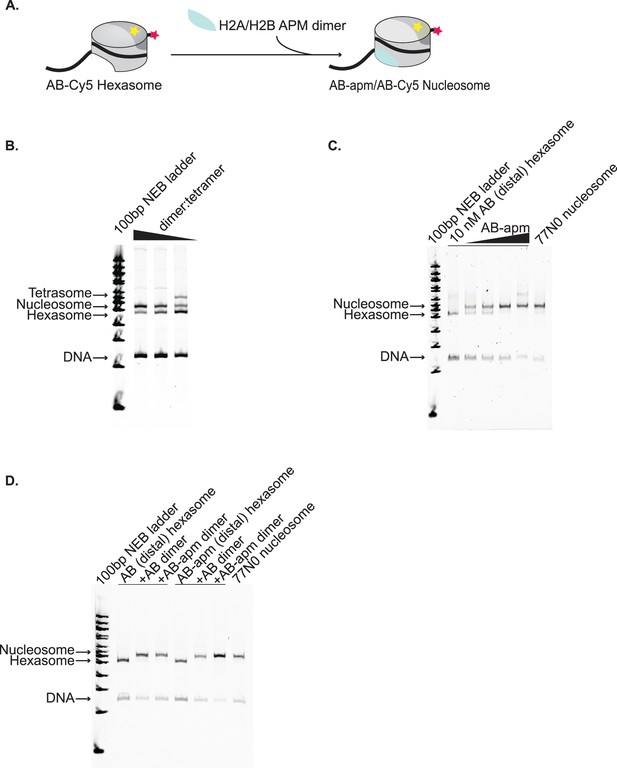
Strategy for assembly of asymmetric nucleosomes.
(A) Schematic of assembly of asymmetric nucleosomes from purified hexasomes. Hexasomes (represented by partial cylinder, H3/H4 tetramer in light gray, AB dimer in dark gray, Cy5 yellow star, 77N0 DNA by thick black line, and Cy3 by pink star) spontaneously assemble into nucleosomes upon addition of free dimer (AB-apm, shown added in light blue). (B–D) give representative examples of the checkpoints performed during assembly of asymmetric nucleosomes. (B) For each hexasome needed for biochemical assays, the ratio of dimer to tetramer was varied during reconstitution to optimally bias the reaction toward hexasomes and assessed by native-PAGE with a tetramer:DNA ratio of 1.4 and a dimer:DNA ratio of 1.4, 1.8, and 2.4. (C) After purification of hexasomes, contralateral dimer to be placed was titrated to determine optimal conversion to nucleosome and assessed by native-PAGE with a canonically reconstituted 77N0 or 0N0 nucleosome as appropriate added for comparison with a ratio of dimer to hexasome of 0.9, 1, 1.1, and 1.2. (D) As a final check before performing biochemical assays, each hexasome was run on a native-PAGE gel with nucleosomes formed by dimer addition and a canonically assembled nucleosome for comparison.
-
Figure 1—figure supplement 2—source data 1
Zip file containing raw and annotated images for gels shown in panels B–D of Figure 1—figure supplement 2.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig1-figsupp2-data1-v1.zip
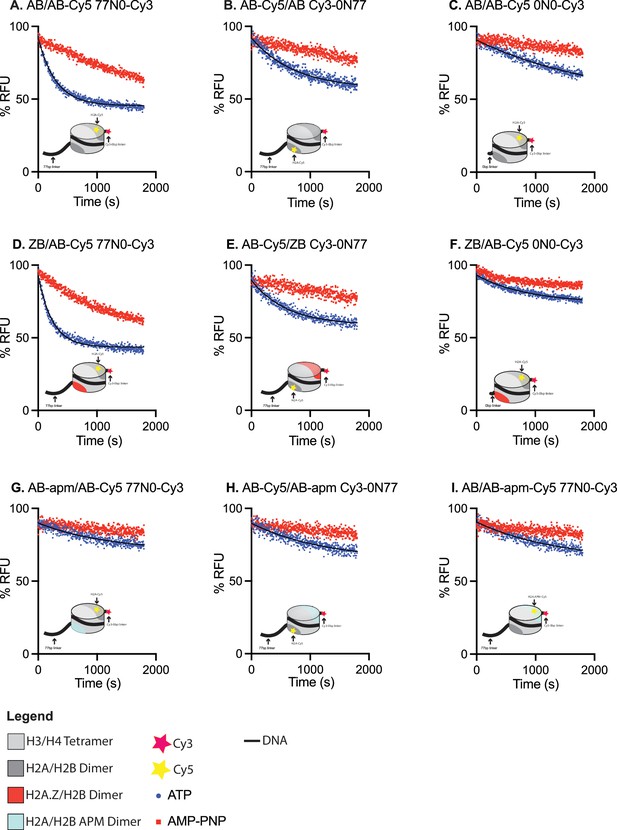
Förster-resonance energy transfer (FRET)-based dimer eviction.
(A–I) Graphs of dimer eviction data used to calculate rates in Figure 1C and D. Reactions were carried out under single turnover conditions (50 nM SWR1C, 10 nM nucleosomes, 60 nM ZB dimers) in the presence of ATP (blue) or AMP-PNP (red) for each of the asymmetric nucleosome (listed linker proximal/ linker distal/DNA template at top left of each graph) and loss of FRET was measured over time. (A–F) Rates for ATP reactions differed significantly from AMP-PNP while in (G–I) there was no significant difference. (A, D, F) have six replicates while the rest are done in triplicate. Note that the AMP-PNP reactions in panels A and D showed higher rates of FRET loss compared to all other substrates. This was reproducible in three different nucleosome preparations and in all replicates. This loss of FRET was not observed in the no enzyme control, indicating enzyme-dependent destabilization (see source data file). These higher ‘background’ rates are not observed when this same DNA template is reconstituted with Cy5-labeled octamers; thus, the apparent instability of these nucleosomes seems inherent to the hexosome assembly method.
-
Figure 1—figure supplement 3—source data 1
Excel file containing raw data for experiments depicted in panels A–I of Figure 1—figure supplement 3.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig1-figsupp3-data1-v1.xlsx
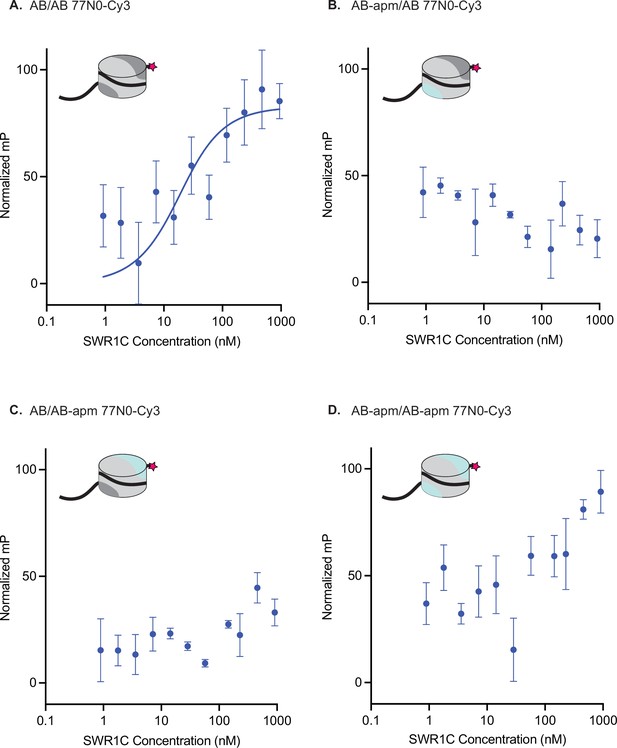
Nucleosomal acidic patches are required for SWR1C nucleosome binding.
Asymmetrically assembled 77N0-Cy3 nucleosomes were incubated with SWR1C serially diluted in concentration from approximately 1 µM to 1 nM. Fluorescence polarization (FP) values were collected and plotted by concentration for each nucleosome type. Each graph has a representative nucleosome at the top left (represented by a cylinder with H3/H4 tetramer in light gray, AB dimer in dark gray, AB-apm dimer in light blue, 77N0-Cy3 DNA as a solid black line with Cy3 (pink star) label on the 0bp-linker side). FP data were normalized between experiments by translation, which does not effect the binding curve fit so that plots can be compared at a glance. (A) AB/AB 77N0-Cy3 nucleosomes showed a Kd as determined by a fit to the Morrison equation (blue line). The Kd calculated for each individual replicate was 13.55, 13.32, 13.94, and 13.75. The average of those individual values is 13.64. The standard deviation is 0.27. A Kd was unable to be calculated for nucleosomes with a linker proximal acidic patch mutant (B), a linker distal acidic patch mutant (C), or both acidic patches mutated (D) at the range of concentrations of SWR1C tested suggesting at least a 10-fold reduction in binding affinity. Error bars reflect 95% confidence intervals from at least three replicates.
-
Figure 2—source data 1
Excel file containing raw data for experiments depicted in panels A–D of Figure 2.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig2-data1-v1.xlsx
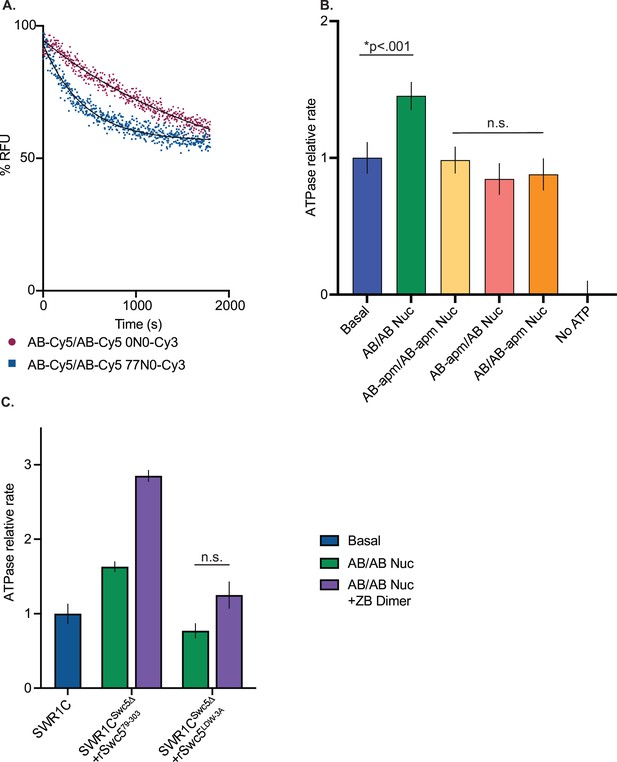
Dimer eviction and ATPase assays.
(A) Graph of dimer eviction data carried out at high enzyme concentrations (200 nM SWR1C, 10 nM nucleosomes, 250 nM ZB dimers, 1 mM ATP) on AB/AB-Cy5 0N0-Cy3 nucleosomes (maroon) or AB/AB-Cy5 77N0-Cy3 nucleosomes (blue). SWR1C evicted AB-Cy5 dimers significantly faster in the presence of linker DNA. (B) ATPase activity of SWR1C was measured by phosphate-binding protein assay in the presence of various asymmetrically assembled 77N0 nucleosomes and normalized to basal activity. AB/AB nucleosomes (green bar) stimulated the ATPase rate as compared to basal SWR1C activity (blue bar). SWR1C activity in the presence of nucleosomes containing a linker proximal (yellow), distal (salmon) acidic patch mutant, or both (orange) acidic patches mutated did not differ significantly from basal SWR1C rates. (C) Nucleosomal stimulation of SWR1C ATPase activity with (purple bars) or without (green bars) ZB dimers added was measured for SWR1C, SWR1CSwc5∆+rSwc579–303, and SWR1CSwc5∆+rSwc5LDW-3A using a phosphate sensor assay. Calculated rates were normalized to basal SWR1C activity (blue bar). Stimulation of ATPase activity was restored to the Swc5Δ complex by the Swc579–303 derivative, but not by the Swc5LDW-3A derivative.
-
Figure 2—figure supplement 1—source data 1
Excel file containing raw data for experiments depicted in panels A–C of Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig2-figsupp1-data1-v1.xlsx
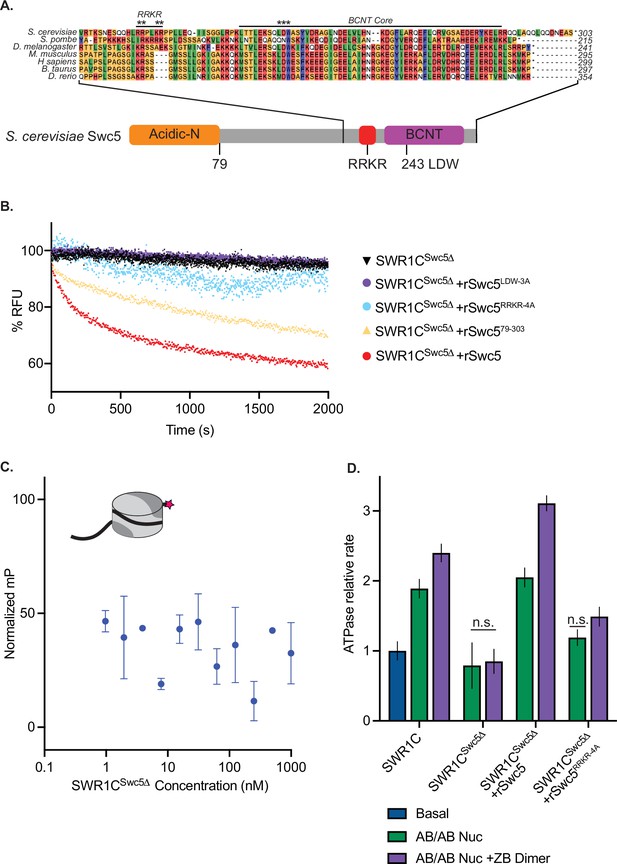
Swc5 contains a key arginine-rich motif.
(A) Alignment of SWR1C subunit Swc5 homologs reveals a conserved arginine in a region we named the RRKR motif in S. cerevisiae adjacent to the essential BCNT region and conserved LDW residues (asterisks are used to denote residues mutated in RRKR-4A and LDW-3A mutants). A linear schematic of this gene with the acidic N-terminal domain in orange, gene body in gray, RRKR motif in red, and BCNT region in purple shows relative locations of these domains. (B) Swc5 and Swc5 derivatives were reconstituted into SWR1CSwc5∆ complexes to measure their activity on dimer exchange using Förster-resonance energy transfer (FRET) at single turnover conditions (30 nM SWR1C, 10 nM 77N0-Cy3 AB-Cy5/AB-Cy5 nucleosomes, 70 nM ZB dimers, 1 mM ATP). The relative fluorescence unit (RFU) for each reaction was normalized and plotted as a function of time. SWR1CSwc5∆ was unable to carry out dimer exchange (black triangles), and recombinant Swc5 (rSwc5) rescued activity (red circles). SWR1CSwc5∆ reconstituted with Swc5 containing mutations in the LDW (rSwc5LDW-3A) or RRKR motifs (rSwc5RRKR-4A) showed no dimer eviction activity (purple and blue circles, respectively), while an N-terminal truncation of Swc5 (rSwc579–303) had a reduction in activity compared to full-length Swc5 (yellow triangles). (C) Asymmetrically assembled 77N0-Cy3 nucleosomes were incubated with SWR1CSwc5∆ serially diluted in concentration from approximately 1 µM to 1 nM. Fluorescence polarization values were collected and plotted by concentration. A representative nucleosome is shown at the top left (represented by a cylinder with H3/H4 tetramer in light gray, AB dimer in dark gray, 77N0-Cy3 DNA as a solid black line with Cy3 (pink star) label on the 0bp-linker side). (D) Nucleosomal stimulation of SWR1C ATPase activity with (purple bars) or without (green bars) ZB dimers added was measured for complexes containing SWR1C, SWR1CSwc5∆, SWR1CSwc5∆+rSwc5, and SWR1CSwc5∆+rSwc5RRKR-4A using a phosphate sensor assay. Calculated rates were normalized to basal SWR1C activity (blue bar). Stimulation of ATPase activity was lost in the SWR1CSwc5∆ complex and reduced in the SWR1CSwc5∆+rSwc5RRKR-4A complex. Error bars reflect 95% confidence intervals from at least three replicates.
-
Figure 3—source data 1
Excel file containing raw data for experiments depicted in panels B, C, and D of Figure 3.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig3-data1-v1.xlsx
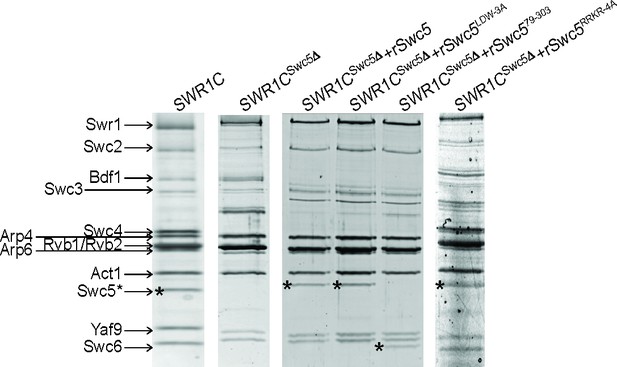
Reconstitution of SWR1C with Swc5 derivatives.
(A) Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) gel showing wildtype SWR1C, SWR1CSwc5∆, and SWR1CSwc5∆ with various recombinant Swc5 reincorporated (Swc5 denoted by asterisk).
-
Figure 3—figure supplement 1—source data 1
Zip file containing raw and annotated images for gels (numbered 1 through 4 from left to right) shown in Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig3-figsupp1-data1-v1.zip
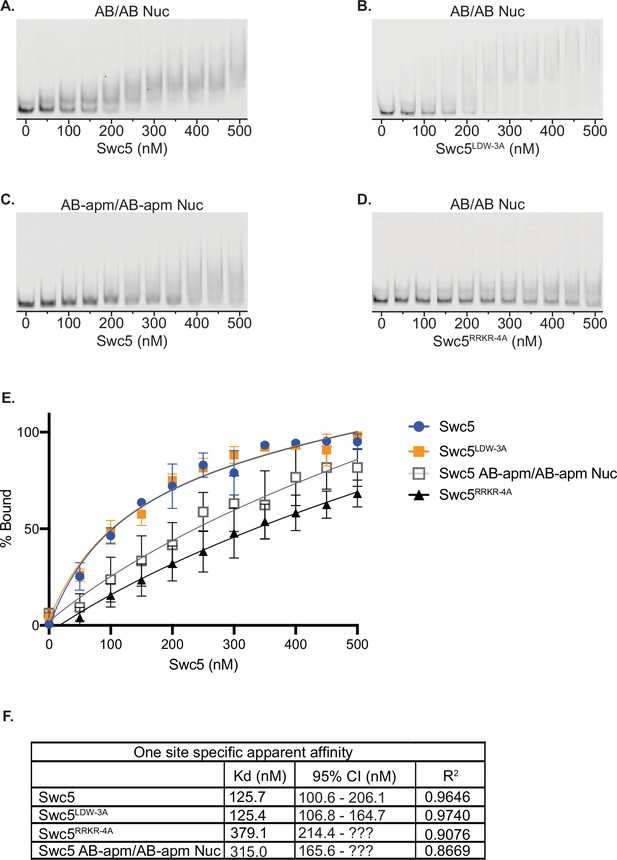
Swc5 is a nucleosome-binding subunit.
Gel mobility shift assays were performed with 5 nM 0N0 nucleosomes and recombinant Swc5 ranging in concentration from 0 to 500 nM, in 50 nM increments. Swc5 was able to bind AB/AB nucleosomes (A) but showed reduced binding of AB-apm/AB-apm nucleosomes (B). Swc5LDW-3A bound AB/AB nucleosomes (C) whereas Swc5RRKR-4A showed reduced binding (D). Percent of nucleosomes bound for each condition was calculated using ImageQuant and plotted by Swc5 concentration (E) and specific binding Kd values were predicted along with 95% confidence intervals (CIs) predicted in Prism 9 (F).
-
Figure 4—source data 1
Excel file containing raw data for experiments depicted in panel E of Figure 4.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig4-data1-v1.xlsx
-
Figure 4—source data 2
Zip file containing raw and annotated images for gels shown in panels A–D of Figure 4.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig4-data2-v1.zip
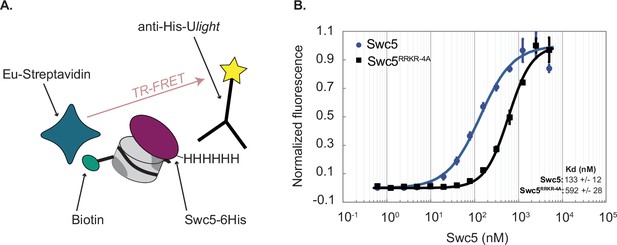
Time-resolved Förster-resonance energy transfer (TR-FRET) assay confirms Swc5 nucleosome-binding activity.
(A) Schematic of the TR-FRET assay. Recombinant 6His-Swc5 is labeled with a ULight alpha 6His acceptor antibody, and biotinylated nucleosomal DNA is labeled with an Eu-streptavidin acceptor fluorophore. The Förster-resonance energy transfer (FRET) signal increases as 6His-Swc5 binds to the nucleosome. (B) TR-FRET assay was performed with 2 nM 31N1 nucleosomes and recombinant Swc5 or Swc5RRKR-4A ranging in concentration from 0.6 to 6000 nM. Recombinant wildtype Swc5 bound with an apparent Kd of ~133 nM, while the Swc5RRKR-4A derivative bound with an apparent Kd of ~592 nM. Note that the Swc5RRKR-4A assays did not reach full saturation, and thus the measured Kd is likely an under-estimate of the reported value. Kd values were determined from triplicate titrations of each Swc5 variant and are reported as means ± standard error of the mean.
-
Figure 5—source data 1
Excel file containing raw data for experiments depicted in panel B of Figure 5.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig5-data1-v1.xlsx
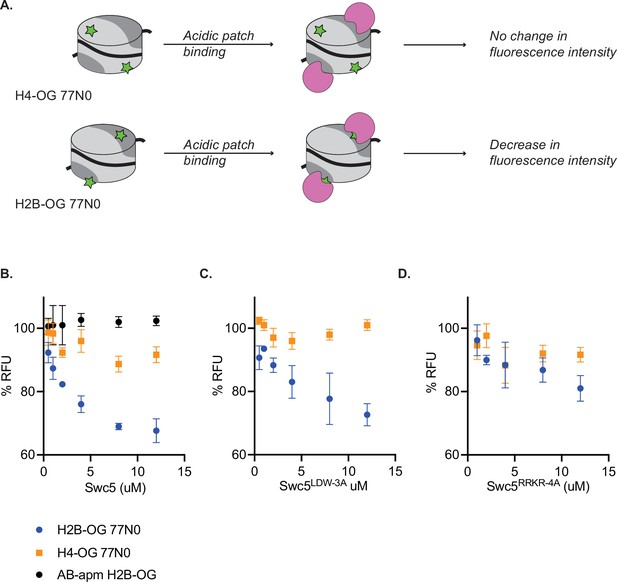
Swc5 interacts with the nucleosome acidic patch.
(A) Schematic of Oregon green based fluorescence quenching assay. Top shows a nucleosome (represented as a cylinder with H3/H4 tetramer in light gray, AB dimers in dark gray, and 0N0 DNA as a solid black line) with an Oregon green label (green star) on H4. Oregon green is exquisitely sensitive to changes in its local environment. Binding of an acidic patch interacting protein (pink) does not interact with Oregon green label on H4, resulting in no change in fluorescence intensity. A label on H2B near the acidic patch (bottom) is affected by an acidic patch interacting protein, resulting in decreased fluorescence intensity. (B) Swc5 was added to 10 nM H4-OG AB/AB 0N0 nucleosomes (orange squares), H2B-OG AB/AB 0N0 nucleosomes (blue circles), and H2B-OG AB-apm/AB-apm 0N0 nucleosomes (black circles) resulting in quenching of the AB/AB H2B-OG 0N0 nucleosomes but not the H4-OG AB/AB 0N0 nucleosomes or the H2B-OG AB-apm/AB-apm 0N0 nucleosomes. (C) Swc5LDW-3A similarly quenched H2B-OG AB/AB 0N0 nucleosomes with no change in H4-OG AB/AB 0N0 nucleosomes while Swc5RRKR-4A (D) did not show significant quenching in either condition. Error bars reflect standard deviations from at least three replicates.
-
Figure 6—source data 1
Excel file containing raw data for experiments depicted in panels B–D of Figure 6.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig6-data1-v1.xlsx
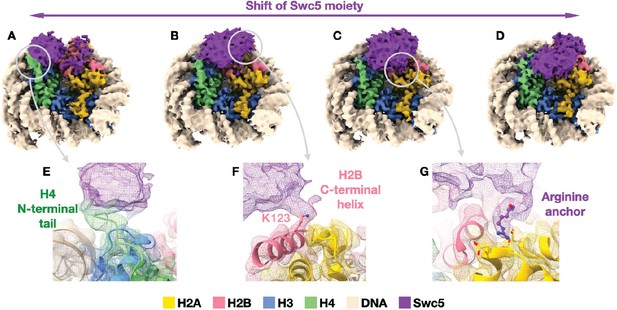
Cryo-EM structure of Swc5/nucleosome complex.
(A–D) Reconstructions of selected particle subsets obtained from 3D classification that was focused on the Swc5 moiety (purple) illustrate the dynamic nature of Swc5 binding to the nucleosome. Three interactions persist among the 3D classes: (E) Swc5 interaction with the N-terminal tail of histone H4 (green), (F) Swc5 interaction with the C-terminal helix of histone H2B including K123, and (G) an Arginine anchor bound into the acidic patch of the histone core. The gray circles and arrows denote the reconstructions and locations from where the cut-out panels originate.
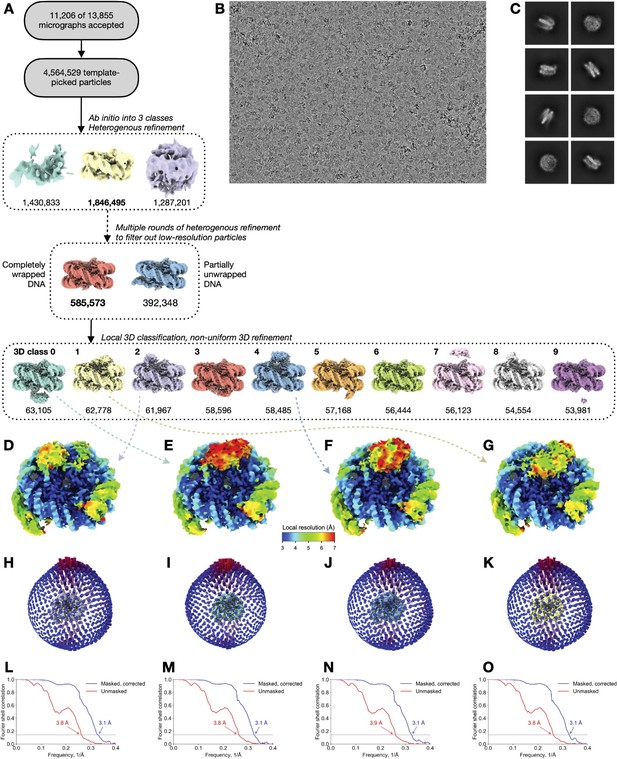
Cryo-EM analysis of Swc5/nucleosome complex.
(A) Flowchart of data processing and particle classification. The 3D classes marked with letters E–G correspond to the four selected reconstructions in the panels below and in Figure 6. (B) Representative cryo-EM micrograph. (C) Selected class averages from 2D classification of the 1,846,495 nucleosomal particles obtained after the first round of heterogenous 3D refinement. (D–G) Selected 3D reconstructions (the same as in Figure 6) colored by local resolution. (H–K) Euler angle distributions of the particles used in the reconstructions in panels D–G. The view angle is the same as in panels D–G. (L–O) Fourier shell correlation plots of independently refined half maps corresponding to reconstructions in panels D–G. Resolution is determined at FSC = 0.143.
Conformational heterogeneity of Swc5.
The 3D class depicted in Figure 7C and Figure 7—figure supplement 1F, J, N was subjected to cryoSPARC’s 3D Variability Analysis. Variability was solved in two modes, filtered to 7 Å, and a white noise model was used. The animation displays the primary component of heterogeneity in the subset – Swc5 moiety swinging between H4 N-terminal tail on the left and H2A/B dimer on the right.

Swc5 arginine-rich region is key for SWR1C activity in vivo.
Genetic complementation using a swc5∆ strain (MATa his3Δ1 leu2Δ0 met15Δ0 ura3Δ0 swc5Δ::kanMX4) transformed with an empty pRS416 CEN/ARS URA3 vector (top row in both panels) or the same vector with various alleles of SWC5. Cultures were grown to an OD of 1 and spotted with serial dilution onto plates with synthetic complete media lacking uracil without (left) or with (right) 2% formamide.
-
Figure 8—source data 1
Zip file containing raw and annotated images for gels (referred to as left and right panel) shown in Figure 8.
- https://cdn.elifesciences.org/articles/94869/elife-94869-fig8-data1-v1.zip
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (S. cerevisiae) | W1588-4C swr1::SWR1– 3xFLAG-P-kanMx-P htz1Δ::natMX | Ed Luk (SUNY Stony Brook) | yEL190 | |
| Strain, strain background (S. cerevisiae) | W1588-4C swr1::SWR1– 3xFLAG-P-kanMx-P htz1Δ::natMX swc5Δ::hphMX | This paper | CY2535 | Available from Peterson lab |
| Strain, strain background (S. cerevisiae) | MATa his3Δ1 leu2Δ0 met15Δ0 ura3Δ0 YBR231c::kanMX4 | Euroscarf | Y03371 | |
| Strain, strain background (E. coli) | Rosetta 2 (DE3) | Novagen | 70954 | |
| Strain, strain background (E. coli) | DH5alpha | Thermo Fisher Scientific | EC0112 | |
| Antibody | ULight alpha-6xHIS acceptor antibody | Perkin Elmer | TRF0134-D | Mouse monoclonal anti poly Histidine tag used at 1:20 molar ratio to His-tagged Swc5 variants |
| Recombinant DNA reagent | pRS416 (plasmid) | ATCC | 87521 | CEN/ARS/URA3 vector |
| Recombinant DNA reagent | CP1566 (plasmid) | This paper | SWC5 version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | CP1567 (plasmid) | This paper | swc5 R219A version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | CP1568 (plasmid) | This paper | swc5 R214A, R215A version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | CP1569 (plasmid) | This paper | swc5 K218A, R219A version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | CP1579 (plasmid) | This paper | swc5 RRKR-4A version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | CP1580 (plasmid) | This paper | swc5 LDW-3A version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | CP1581 (plasmid) | This paper | swc5 79–303 version of pRS416 Available from Peterson lab | |
| Recombinant DNA reagent | pQE80L (plasmid) | QIAGEN | 6x-HIS tag expression vector | |
| Recombinant DNA reagent | p601 | Widom lab | Lowary, P. T. & Widom, J. New DNA sequence rules for high affinity binding to histone octamer and sequence-directed nucleosome positioning. J. Mol. Biol. 276, 19–42 (1998). | |
| Sequence-based reagent | 77 w-N | This paper | PCR primers | GTACCCGGGGATCCTCTAGAGTG Ordered from Integrated DNA technologies (IDT) |
| Sequence-based reagent | 0 w-N | This paper | PCR primers | ACAGGATGTATATATCTGACACGTGCC Ordered from Integrated DNA technologies (IDT) |
| Sequence-based reagent | Cy3-0w-N | This paper | PCR primers | Cy3-ACAGGATGTATATATCTGACACGTGCC Ordered from Integrated DNA technologies (IDT) |
| Sequence-based reagent | N-77s | This paper | PCR primers | GATCCTAATGACCAAGGAAAGCATGATTC Ordered from Integrated DNA technologies (IDT) |
| Sequence-based reagent | N-0s | This paper | PCR primers | CTGGAGAATCCCGGTGCCGA Ordered from Integrated DNA technologies (IDT) |
| Sequence-based reagent | N-0s-Cy3 | This paper | PCR primers | Cy3-CTGGAGAATCCCGGTGCCGA Ordered from Integrated DNA technologies (IDT) |
| Peptide, recombinant protein | 3x-FLAG | Sigma-Aldrich | F4799 | |
| Peptide, recombinant protein | Phusion high-fidelity DNA polymerase | New England Biolabs | M0530 | |
| Peptide, recombinant protein | Streptavidin-Eu | Perkin Elmer | 1244-360 | |
| Chemical compound | Cy3 maleimide | Lumiprobe | 41080 | |
| Chemical compound | Cy5 maleimide | Lumiprobe | 43080 | |
| Chemical compound | Oregon green 488 maleimide | Thermo Fisher Scientific | O6034 | |
| Commercial assay, kit | Phosphate sensor | Thermo Fisher Scientific | PV4406 | |
| Software | Prism 9 | GraphPad Software, LLC | Version 9.2.0 | |
| Software | cryoSPARC | Structura Biotechnology Inc | Version 4 | Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. CryoSPARC: Algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14 (2017). |
| Software | ChimeraX | UCSF | Pettersen, E. F. et al. UCSF ChimeraX: Structure visualization for researchers, educators, and developers. Protein Sci. 30 (2021). | |
| Other | Planetary ball mill | Retsch | PM-100 | See Method section for SWR1C preparation |
| Other | Mini-prep cell apparatus | Bio-Rad | 1702908 | See Method section for nucleosome reconstitution |





