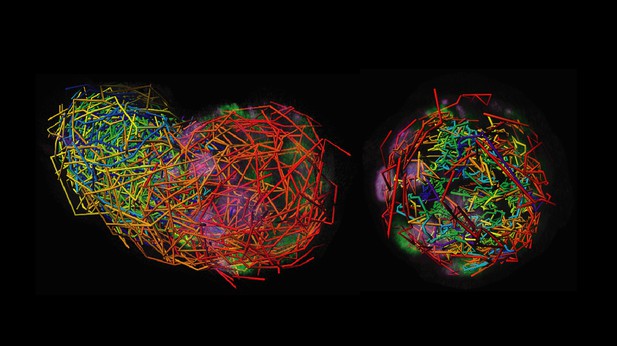
This image shows how individual cells have moved between the moment an embryo had four cells to the stage where it has grown to more than a hundred cells, including some that are specialized. Because the embryo moves and turns as it develops, the trajectories (left) need to be corrected (right) using a new piece of software called primed Track. The color gradient indicates the location of the individual cells at different time points, starting with blue (four-cell embryo) and ending with red. Image credit: Welling, Mohr et al. (CC BY 4.0)
A mouse embryo starts with one cell, which divides to create identical daughters that quickly start to multiply. Within three to four days, certain cells begin to specialize and take on specific roles. Scientists want to track these early events to understand how they give rise to an individual formed of huge numbers of cells organized in specialized tissues. To do so, researchers genetically manipulate embryos so that each cell produces fluorescent molecules that ‘glow’ under light. These embryos are grown inside a special microscope for several days. Images are taken regularly and then processed by specialized software that automatically tracks the fluorescent cells and their daughters over time. This helps reconstruct the history of each cell, and which structures they give rise to.
However, many embryos move and turn around between images, and so software packages often lose track of which cell was which. Taking images more frequently is not possible because each imaging event exposes the embryo to light, which can damage its fragile cells.
To address this problem, Welling, Mohr et al. made embryonic cells produce a special fluorescent marker, which is normally green but can be converted to red. Then, a technique known as primed conversion was used so that only one cell in a four-cell embryo would glow red. Welling, Mohr et al. designed a piece of software, baptized ‘primed Track’, that can use this red cell (and its daughters) to reorient the embryo during image analysis and reliably identify and match any mother cell to its daughters. The new approach means the experiments require fewer imaging events, but also fewer embryos because even the ones that move a lot can be studied. This should help scientists look into how early life processes give rise to specialized cells, and even explore the fate of cells in other tissues.




