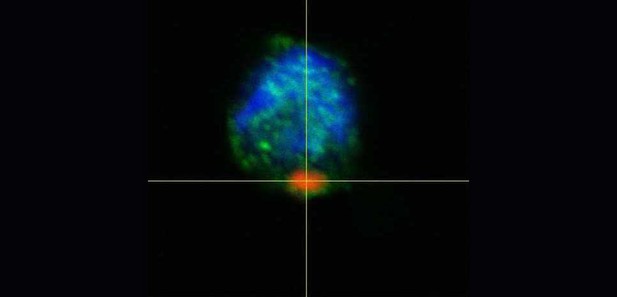
Laser scanning confocal microscopy shows that AAT polymers (red) are present in close proximity with CX3CR1 proteins (green), suggesting that the two entities interact. Image credit: Tumpara et al. (CC BY 4.0)
Proteins can lose their structure and form polymers because of mutations or changes in their immediate environment which can lead to cell damage and disease. Interestingly, polymers formed by a variety of proteins can reduce the levels of CX3C chemokine receptor 1 (CX3CR1 for short) that controls the behaviour of immune cells and is implicated in a range of illnesses.
Inherited ZZ alpha-1 antitrypsin deficiency is a rare genetic condition that highly increases the risk of liver and lung diseases. This disorder is characterised by mutant alpha-1 antitrypsin proteins (AAT for short) reacting together to form polymers; yet it remains unclear how the polymers affect different cells or organs, and lead to diseases. To investigate this question, Tumpara et al. examined whether polymers of mutant AAT influence the level of the CX3CR1 protein in specific classes of immune cells.
Experiments revealed that in people with AAT deficiency, certain blood immune cells express lower levels of CX3CR1. Regardless of age, clinical diagnosis, or treatment regimen, all individuals with ZZ alpha-1 antitrypsin deficiency had AAT polymers circulating in their blood: the higher the levels of polymers measured, the lower the expression of CX3CR1 recorded in the specific immune cells. When Tumpara et al. added polymers of mutant AAT to the immune cells of healthy donors, the expression of CX3CR1 dropped in a manner dependent on the polymer concentration. According to microscopy data, AAT polymers occurred inside cells alongside the CX3CR1 protein, suggesting that the two molecular actors interact. In the future, new drugs that remove these polymers, either from inside cells or as they circulate in the body, could help patients suffering from conditions associated with this abnormal protein aggregation.




