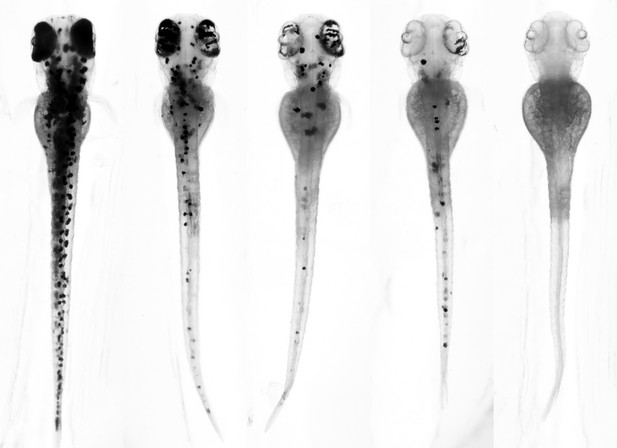
The typical phenotypes observed following the removal of the pigmentation gene oca2 using gene editing tools. Injecting zebrafish embryos with a Cas9 enzyme connected to a hei-tag most often led to the phenotype shown on the far right. Image credit: Thomas Thumberger (CC BY 4.0)
The genetic code stored within DNA provides cells with the instructions they need to carry out their role in the body. Any changes to these genes, or the DNA sequence around them, has the potential to completely alter how a cell behaves.
Scientists have developed various tools that allow them to experimentally modify the genome of cells or even entire living organisms. This includes the popular Cas9 enzyme which cuts DNA at specific sites, and base editors which can precisely change bits of genetic code without cutting DNA. While there are lots of Cas9 enzymes and base editors currently available, these often differ greatly in their activity depending on which cell type or organism they are applied to.
Finding a tool that can effectively modify the genome of an organism at the right time during development also poses a challenge. All the cells in an organism arise from a single fertilized cell. If this cell is genetically edited, all its subsequent daughter cells (which make up the entire organism) will contain the genetic modification. However, most genome editing tools only work efficiently later in development, resulting in an undesirable mosaic organism composed of both edited and non-edited cells.
Here, Thumberger et al. have developed a new ‘high efficiency-tag’ (also known as hei-tag for short) that can enhance the activity of gene editing tools and overcome this barrier. The tag improves the efficiency of gene editing by immediately shuttling a Cas9 enzyme to the nucleus, the cellular compartment that stores DNA. In all cases, gene editing tools with hei-tag worked better than those without in fish embryos and mouse cells grown in the laboratory. When Cas9 enzymes connected to a hei-tag were injected into the first fertilized cell of a fish embryo, this resulted in an even distribution of edited genes spread throughout the whole organism.
To understand how a gene affects an organism, researchers need to be able to edit it as early in development as possible. Attaching the ‘hei-tag’ to already available tools could help boost their activity and make them more efficient. It could also allow advances in medical research aimed at replacing faulty genes with fully functioning ones.




