Researchers have shown how pre-existing immune cells in the blood may help individuals develop rapid and strong protection against hepatitis B infection following vaccination.
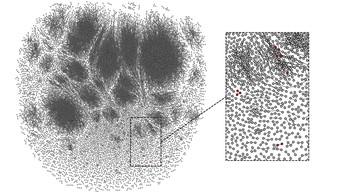
This image provides an overview of the T-cell response in one of the study participants. Shown in red are the vaccine-specific T cells present following vaccination. Image credit: Elias, Meysman, Bartholomeus et al.
(CC BY 4.0)
Their methods, described today in eLife, may lead to new ways for scientists and clinicians to predict which individuals will be protected by vaccination against hepatitis B infection, and who may have a delayed or inadequate immune response. It may also help them better understand how individuals develop natural immunity to pathogens or immunity following vaccination.
Immune cells called CD4 T cells help the body recognise pathogens. They also recognise vaccine antigens that help the body identify and develop immunity to the pathogens. Once a CD4 T cell recognises a pathogen or a vaccine antigen, it develops into a memory cell ready to defend the body quickly against future infections.
“Some people also have pre-existing memory CD4 T cells that recognise pathogens or vaccine antigens that they haven’t even been exposed to before. We wanted to find out if having pre-existing memory CD4 T cells that recognise the hepatitis B vaccine surface antigen helps individuals develop immunity after vaccination,” explains George Elias, who worked on the study while he was a PhD student at the Vaccine and Infectious Disease Institute, University of Antwerp, Belgium. Elias is a co-first author of the study alongside Pieter Meysman and Esther Bartholomeus (University of Antwerp).
To answer this question, the team recruited 34 people who had never been exposed to hepatitis B or vaccinated against it. They collected blood samples from these individuals before and after they were given two doses of the hepatitis B vaccine. They then used next-generation sequencing and machine learning methods to analyse the memory CD4 T cells in these individuals before vaccination and at three time points following vaccination.
Their analysis revealed that participants with a higher number of pre-existing memory CD4 T cells that recognised the vaccine antigen developed immunity more quickly and produced more antibodies following vaccination. They also showed that using immunosequencing to analyse the participants’ pre-existing memory CD4 T cells could predict whether they would rapidly produce strong immunity after hepatitis B vaccination.
The authors say that the ability of CD4 T cells to recognise multiple antigens may explain why even people who have never been exposed to hepatitis B or its vaccine have pre-existing memory cells that recognise the virus. These memory cells may have been generated in response to environmental exposures or to microbes that live in the human body, but more research is needed to better understand their origins.
“Our analysis has uncovered a role for pre-existing memory CD4 T cells in mounting a quick and powerful immune response to the hepatitis B vaccine,” concludes Benson Ogunjimi, Co-Founder and Biomedical Coordinator of the Antwerp Unit for Data Analysis and Computation in Immunology and Sequencing (AUDACIS), and a co-senior author of the study alongside Viggo Van Tendeloo and Kris Laukens (AUDACIS). “We’ve shown that pre-existing memory CD4 T cells could affect the outcome of vaccination, which may have significant implications for immunity and vaccine development efforts.”
Ogunjimi and his co-authors have launched a company based on the next-generation sequencing methods described in their work. ImmuneWatch works to provide in-depth sequencing and analysis of the human T-cell response, and aims to transform this data into clinically actionable insights for the development of improved vaccines and immune therapies.
More information about ImmuneWatch is available at https://www.immunewatch.com.
Media contacts
Emily Packer
eLife
e.packer@elifesciences.org
+441223855373
About
About eLife
eLife is a non-profit organisation created by funders and led by researchers. Our mission is to accelerate discovery by operating a platform for research communication that encourages and recognises the most responsible behaviours. We review selected preprints in all areas of biology and medicine, including Immunology and Inflammation, and Microbiology and Infectious Disease, while exploring new ways to improve how research is assessed and published. eLife receives financial support and strategic guidance from the Howard Hughes Medical Institute, the Knut and Alice Wallenberg Foundation, the Max Planck Society and Wellcome. Learn more at https://elifesciences.org/about.
To read the latest Immunology and Inflammation research published in eLife, visit https://elifesciences.org/subjects/immunology-inflammation.
And for the latest in Microbiology and Infectious Disease, see https://elifesciences.org/subjects/microbiology-infectious-disease.
About the University of Antwerp
The University of Antwerp is a research university where pioneering, innovative research is conducted at an international level. Research and education are closely linked. Educational innovation is a constant focus, and special care is also taken to welcome and guide each of the 20,000 students spread across our nine faculties.
The University of Antwerp is not an island: we build bridges to secondary education, to industry and, by extension, to society as a whole. With over 6,000 members of staff, the University of Antwerp is one of the most important employers in Antwerp, Flanders’ largest city. For more information, visit www.uantwerp.be.




