Author response:
The following is the authors’ response to the original reviews
Thank you very much for the positive and constructive feedback on our manuscript. We have revised the manuscript accordingly and have added a substantial number of additional experiments and have extended the data.
Questions of the reviewers were focused mostly on mechanical insight into organoid formation, touching following aspects of lens organoid formation presented in the manuscript:
- Cellular arrangements/re-arrangements during the process of lens formation including potential contribution of differential adhesion-mediated cell sorting to the cellular arrangement in the organoid and characterization of individual contributions of lens- and retina- committed progenitors to this process.
- Activity of BMP and FGF signaling pathways during organoid formation, namely identification of tissue responding to the signaling withing forming organoids.
- Contribution of externally supplemented Matrigel to the differentiation process and cellular arrangements in ocular organoids.
To address those points in detail we included additional experiments that are now presented in revised version of the manuscript, namely in revised Figure 2-figure supplement 1 (addressing contribution of Matrigel); new Figure 4-supplement 1/Video S5 (addressing contribution of differential adhesion-mediated cell sorting); revised Figure 4/Video S6/Video S7 (addressing contribution of lens-committed progenitors); revised Figure 6 (addressing BMP and FGF signaling pathway activities).
Reviewer #1 (Evidence, reproducibility and clarity):
Summary
The authors focused on medaka retinal organoids to investigate the mechanism underlying the eye cup morphogenesis. The authors succeeded to induce lens formation in fish retinal organoids using 3D suspension culture with minimal growth factor-containing media containing the Hepes. At day 1, Rx3:H2B-GFP+ cells appear in the surface region of organoids. At day 1.5, Prox1+cells appear in the interface area between the organoid surface and the core of central cell mass, which develops a spherical-shaped lens later. So, Prox1+ cells covers the surface of the internal lens cell core. At day 2, foxe3:GFP+ cells appear in the Prox1+ area, where early lens fiber marker, LFC, starts to be expressed. In addition, foxe3:GFP+ cells show EdU+ incorporation, indicating that foxe3:GFP+ cells have lens epithelial cell-characters. At day 4, cry:EGFP+ cells differentiate inside the spherical lens core, whose the surface area consists of LFC+ and Prox1+ cells. Furthermore, at day 4, the lens core moves towards the surface of retinal organoids to form an eye-cup like structure, although this morphogenesis "inside out" mechanism is different from in vivo cellular "outside -in" mechanism of eye cup formation. From these data, the authors conclude that optic cup formation, especially the positioning of the lens, is established in retinal organoids though the different mechanism of in vivo morphogenesis.
Overall, manuscript presentation is nice. However, there are still obscure points to understand background mechanism. My comments are shown below.
Major comments
(1) At the initial stage of retinal organoid morphogenesis, a spherical lens is centrally positioned inside the retinal organoids, by covering a central lens core by the outer cell sheet of retinal precursor cells. I wonder if the formation of this structure may be understood by differential cell adhesive activity or mechanical tension between lens core cells and retinal cell sheet, just like the previous study done by Heisenberg lab on the spatial patterning of endoderm, mesoderm and ectoderm (Nat. Cell Biol. 10, 429 - 436 (2008)). Lens core cells may be integrated inside retinal cell mass by cell sorting through the direct interaction between retinal cells and lens cells, or between lens cells and the culture media. After day 1, it is also possible to understand that lens core moves towards the surface of retinal organoids, if adhesive/tensile force states of lens core cells may be change by secretion of extracellular matrix. I wonder if the authors measure physical property, adhesive activity and solidness, of retinal precursor cells and lens core cells. If retinal organoids at day 1 are dissociated and cultured again, do they show the same patterning of internal lens core covering by the outer retinal cell sheet?
The question, whether different adhesive activity is involved in cell sorting and lens formation is indeed very intriguing.
To address this point, we included additional experiments in the revised manuscript. As proposed by the reviewer, we performed dissociation and re-aggregation experiments of day one organoids at the timepoint, when retinal cell fate is already established and first cells with early lens fate (Foxe3::GFP positive) start appearing (see new Figure 4-figure supplement 1).
After dissociation we followed Foxe3::GFP cells over time and observed that initially equally dispersed GFP+ lens-committed cells gradually sort and establish contact with other GFP+ cells, ultimately resulting in the formation of a central GFP+ sphere within a retinal neuroepithelium (AcTub+) localized on the surface of the organoid (see new Figure 4-figure supplement 1e and new Video S5). This data show that differential adhesive properties of lens/retinal precursor cells can enable the formation of a spherical lens in the center of the organoid. This is now clearly stated in the revised version of the manuscript.
(2) Optic cup is evaginated from the lateral wall of neuroepithelium of the diencephalon. In zebrafish, cell movement occurs from the pigment epithelium to the neural retina during eye morphogenesis in an FGF-dependent manner. How the medaka optic cup morphogenesis is coordinated? I also wonder if the authors conduct the tracking of cell migration during optic cup morphogenesis to reveal how cell migration and cell division are regulated in lens of the Medaka retinal organoids. It is also interesting to examine how retinal cell movement is coordinated during Medaka retinal organoids.
Looking into the detail of how optic cup-looking tissue arrangement of ocular organoids is achieved on cellular level is of course interesting. Our previous study showed that optic vesicles of medaka retinal organoids do not form optic cups (for details please see Zilova et al., 2021, eLife). We provide evidence that the formation of cup-looking structure of the ocular organoids presented here is mediated by the following processes: establishment of retina and lens domains at specific regions of the organoid – retina on the surface and lens in the center (see Figure 3-figure supplement 1d and Figure 3e, and Figure 4). Further, the dislocation of the centrally formed lens towards the organoid periphery results in the opening of the retina layer, moving the lens to the periphery while retinal cells stay static. We propose that the “cup-like” shape is acquired by an extrusion-like process of the lens from the center of the organoid.
To address the cellular mechanisms involved in this process, we included additional experiments and followed the movements of retinal and lens cells (see new Figure 4c and 4d, new Videos S6, S7 and S8). Retinal cells (tracked as nuclei of the Rx3::H2B-GFP transgenic line) established in the periphery display repeated short distance movements restricted to the retinal epithelium. These movements are characteristic for interkinetic nuclear migration as found in the developing retina. In contrast, Foxe3::GFP lens progenitor cells performed long distance movements from the center to the periphery of the organoid. This movement was accompanied by profound cell shape changes of lens progenitor cells, suggesting an active movement of lens cells to the organoid periphery. These movements are shown in new/extended figures and in new supplementary videos (new Figure 4c and 4d, new Videos S6, S7 and S8) in the revised version of the manuscript.
(3) The authors showed that blockade of FGF signaling affects lens fiber differentiation in day 1-2, whereas lens formation seems to be intact in the presence of FGF receptor inhibitor in day 0-1. I suggest the authors to examine which tissue is a target of FGF signaling in retinal organoids, using markers such as pea3, which is a downstream target of ERK branch of FGF signaling. Since FGF signaling promotes cell proliferation, is the lens core size normal in SU5402-treated organoids from day 0 to day 1?
Assessing the activity of FGF signaling (cross-reference to Reviewer #3) in the organoids is an important point that we have taken care of and included in the revised manuscript.
To address this point, we assessed which tissue/part of the organoid is responding to FGF signaling. To do so we analyzed the presence of phosphorylated ERK (pERK1/2) as FGF signaling target in ocular organoids from day 1 to day 2. At day 1, only low levels of FGF signaling activity were detectable in presumptive retinal or/and lens tissue (see revised Figure 6b). Only half a day later, a significant increase in FGF activity was observed specifically in the central region of the organoids (lens progenitor domain) (at day 1.5), prior to the onset of differentiation of lens fiber cells. This, together with inability of lens progenitor cells to differentiate to lens fiber cells in the presence of FGF inhibitor SU5402 provided during this critical period (day 1 to day 2) demonstrates that FGF signaling activity localized in the lens progenitor cells is required for lens fiber differentiation.
By day 2, FGF activity was detected in both lens and retinal tissue of the organoid. Similar patterns of FGF activity were observed in embryos at 2 days post fertilization (see revised Figure 6b).
The treatment with the FGF signaling inhibitor SU5402 from day 0 to day 1 did have no impact on the core size of organoid the dimension of which were fully comparable to the control (please see Figure 6d).
(4) Fig. 3f and 3g indicate that there is some cell population located between foxe3:GFP+ cells and rx2:H2B-RFP+ cells. What kind of cell-type is occupied in the interface area between foxe3:GFP+ cells and rx2:H2B-RFP+ cells?
That is for sure an interesting question. We are aware of this population of cells. We currently do not have data that clarify the fate of those cells with the required certainty. Rather than speculating, we are currently following up on that question by scRNA sequencing, however we see that beyond the scope of the current manuscript.
(5) Fig. 5e indicates the depth of Rx3 expression at day 1. Is the depth the thickness of Rx3 expressing cell sheet, which covers the central lens core in the organoids? If so, I wonder if total cell number of Rx3 expressing cell sheet may be different in each seeded-cell number, because thickness is the same across each seeded-cell number, but the surface area size may be different depending on underneath the lens core size. Please clarify this point.
The referee is right, figure 5e indicates the thickness of the cell sheet expressing Rx3 positioned at the surface of the organoid. Indeed, the number of Rx3-expressing cells (and lens cells) scales with the size of the organoid as stated in the submitted manuscript. We have taken care to remove ambiguities related to that point in the revised version of the manuscript.
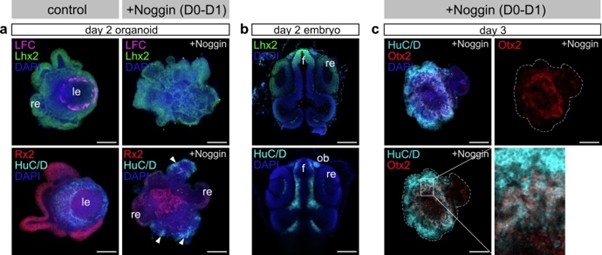
(6) Noggin application inhibits lens formation at day 0-1. BMP signaling regulates formation of lens placode and olfactory placode at the early stage of development. It is interesting to examine whether Noggin-treated organoid expands olfactory placode area. Please check forebrain territory markers.
What tissue differentiates at the expense of the lens in BMP inhibitor-treated organoids is of course an intriguing question.
To address this point, we labeled Noggin treated organoids at day 2 and day 3 with forebrain and olfactory placode markers. We could identify an increase in the domains expressing Lhx2, HuC/D and Otx2 in Noggin-treated organoids, showing a shift of the preferential differentiation of the neurons of anterior forebrain identity (see attached figure for reviewer). However, the available markers Lhx2, HuC/D and Otx2 found in the olfactory placode are in addition also co-expressed in further neuronal cell types of the anterior forebrain. While the speculation is tempting, the shift in expression does not allow to conclusively state the expansion of the olfactory placode.
Author response image 1.
Expression of forebrain and olfactory placode markers.
I have no minor comments
Referees cross-commenting
I agree that all reviewers have similar suggestions, which are reasonable and provided the same estimated time for revision.
Reviewer #1 (Significance):
Strength:
This study is unique. The authors examined eye cup morphogenesis using fish retinal organoids. Eye cup normally consists of the lens, the neural retina, pigment epithelium and optic stalk. However, retinal organoids seem to be simple and consists of two cell types, lens and retina. Interestingly, a similar optic cup-like structure is achieved in both cases; however, underlying mechanism is different. It is interesting to investigate how eye morphogenesis is regulated in retinal organoids,under the unconstrained embryo-free environment.
Limitation:
Description is OK, but analysis is not much profound. It is necessary to apply a bit more molecular and cellular level analysis, such as tracking of cell movement and visualization of FGF signaling in organoid tissues.
Advancement:
The current study is descriptive. Need some conceptual advance, which impact cell biology field or medical science.
Audience:
The target audience of current study are still within ophthalmology and neuroscience community people, maybe translational/clinical rather than basic biology. To beyond specific fields, need to formulate a general principle for cell and developmental biology.
Reviewer #2 (Evidence, reproducibility and clarity):
In this study from Stahl et al., the authors demonstrate that medaka pluripotent embryonic cells can self-organise into eye organoids containing both retina and lens tissues. While these organoids can self-organize into an eye structure that resembles the vertebrate eye, they are built from a fundamentally different morphogenetic process - an "inside-out" mechanism where the lens forms centrally and moves outward, rather than the normal "outside-in" embryonic process. This is a very interesting discovery, both for our understanding of developmental biology and the potential for tissue engineering applications. The study would benefit from some additional experiments and a few clarifications.
The authors suggest that the lens cells are the ones that move from the central to a more superficial position. Is this an active movement of lens cells or just the passive consequence of the retina cells acquiring a cup shape? Are the retina cells migrating behind the lens or the lens cells pushing outwards? High-resolution imaging of organoid cup formation, tracking retina cells in combination with membrane labeling of all cells would help elucidate the morphogenetic processes occurring in the organoids. Membrane labeling would also be useful as Prox1 positive lens cells appear elongated in embryos while in the organoids, cell shapes seem less organised, less compact and not elongated (for example as shown in Fig 3f,g).
Looking into the detail of how the optic cup-like arrangement of ocular organoids is achieved on the cellular level is indeed highly interesting. In the revised manuscript we now provide evidence that the formation of cup-like structure of the ocular organoids presented here is mediated by the following processes: establishment of retina and lens domains at distinct regions of the organoid – retina on the surface and lens in the center (see Figure 3-figure supplement 1d and Figure 3e, and Figure 4). Further, the dislocation of the centrally formed lens towards the organoid periphery results in the opening of the retina layer, moving the lens to the periphery while retinal cells stay static. We propose that the cup-like shape is acquired by an extrusion process of the lens from the center of the organoid.
To address cellular mechanisms involved in this process, we included additional experiments and followed the movements of retinal and lens cells (see new Figure 4c and 4e, new Videos S6, S7 and S8).
Retinal cells (tracked as nuclei of the Rx3::H2B-GFP transgenic line) display repeated short distance movements within the retinal epithelium. These movements are characteristic for interkinetic nuclear migration as found in the developing retina.
In contrast, Foxe3::GFP lens progenitor cells performed long distance movements from the center to the periphery of the organoid. This movement was accompanied by profound cell shape changes of lens progenitor cells, suggesting an active movement of lens cells to the organoid periphery.
These movements are shown in new/extended figures and in new supplementary videos (new Figure 4c and 4e, new Videos S6, S7 and S8) in the revised version of the manuscript.
The organoids could be a useful tool to address how cell fate is linked to cell shape acquisition. In the forming organoids, retinal tissue initially forms on the outside, while non-retinal tissue is located in the centre; this central tissue later expresses lens markers. Do the authors have any insights into why fate acquisition occurs in this pattern? Is there a difference in proliferation rates between the centrally located cells and the external ones? Could it be that highly proliferative cells give rise to neural retina (NR), while lower proliferating cells become lens?
We agree with the reviewer that this is a highly interesting question and in the revised manuscript we followed the advice and dedicated a part of the discussion to this topic. We believe that the arrangement is due to the induction of central lens fates by signal emanating from the retinal epithelium and discuss the role of the diffusion limit and the potential contribution of BMB and FGF signaling to this arrangement. Additional experiments addressing the target tissues of FGF and BMP signaling in the organoid have been provided in response to Reviewer #1. Interfering with FGF signaling that is essential for lens fiber cell differentiation interestingly did not impact on the lens size arguing against an immediate proliferative effect. Although the analysis of the respective proliferation rates at the surface or in the central region of the organoid might show some differences, we do not have any indications, that the proliferation rate itself would be instructive or superior to the cell fate decisions.
What happens in organoids that do not form lenses? Do these organoids still generate foxe3 positive cells that fail to develop into a proper lens structure? And in the absence of lens formation, does the retina still acquire a cup shape?
Lens formation is primarily dependent on the acquisition/specification of Foxe3-expressing lens placode progenitors. In the absence of Foxe3-expression, a lens does not develop. Once Foxe3-expressing progenitors are established, a lens is formed in unperturbed conditions (measured by the presence of expression of crystallin proteins). Organoids that do not have a lens, do not contain Foxe3-expressing cells.
In the absence of a lens, the organoid is composed of retinal neuroepithelium, that does not form an optic cup like shape (for details of such phenotypes please see Zilova et al., 2021, eLIFE). We took care to state that clearly in the revised manuscript.
The author suggest that lens formation occurs even in the absence of Matrigel. Is the process slower in these conditions? Are the resulting organoids smaller? While there are indeed some LFC expressing cells by day2, these cells are not very well organised and the pattern of expression seems dotty. Moreover, LFC staining seems to localise posterior to the LFC negative, lens-like structure (e.g. Fig.S1 3o'clock). How do these organoids develop beyond day 4? Do they maintain their structural integrity at later stages?
The role of HEPES in promoting organoid formation is intriguing. Do the authors have any insights into why it is important in this context? Have the authors tried other culture conditions and does culture condition influence the morphogenetic pathways occurring within the organoids?
We thank the reviewer for pointing this out. In the revised manuscript we made sure to be sufficiently clear in the wording and description of our observation. Indeed, Matrigel is not required for the acquisition of lens fate, which can be demonstrated by the expression of lensspecific markers. However, the presence of Matrigel has a profound impact on structural aspects of organoid formation. Matrigel is essential for organization of retinal-committed cells to form a retinal epithelium (Zilova et al., 2021, eLife). The absence of the structure of the retinal epithelium indeed negatively impacts on the cellular organization and the overall lens structure.
To clarify the contribution of the Matrigel to the organoid organization, we performed additional experiments (see revised Figure 2-figure supplement 1c-f). As mentioned above, the absence of Matrigel impacts on the organization and thickness of retinal neuroepithelium (Rx2+, Figure 2-figure supplement 1c). However, measurement of the lens in organoids at day 2 and day 5 showed that size of the lens is not impacted upon in the absence of Matrigel (Figure 3-figure supplement 1d-e). Additionally, taking advantage of the Foxe3::GFP lens reporter line, we measured the onset of lens-specific gene expression in organoids with and without Matrigel. In both conditions, with and without Matrigel supplementation, Foxe3::GFP expression was initiated at 25 hours post aggregation (see revised Figure 4b).
The role of the HEPES in lens formation is indeed very intriguing and currently under investigation. HEPES is mainly used to regulate the pH of the culture media which on its own might have an impact on multiple cellular processes. It will require a significant time investment to address the potential HEPES triggered molecular mechanisms impacting on lens formation (cross reference with Reviewer #3), which goes beyond the scope of the current manuscript.
Referees cross-commenting
Pleased to see that all the other reviewers are positive about the study and raise similar concerns and comments
Reviewer #2 (Significance):
This is a very interesting paper, and it will be important to determine whether this alternative morphogenetic process is specific to medaka or if similar developmental routes can be recapitulated in organoid cultures from other vertebrate species.
Reviewer #3 (Evidence, reproducibility and clarity):
Summary:
The manuscript by Stahl and colleagues reports an approach to generate ocular organoids composed of retinal and lens structures, derived from Medaka blastula cells. The authors present a comprehensive characterisation of the timeline followed by lens and retinal progenitors, showing these have distinct origins, and that they recapitulate the expression of differentiation markers found in vivo. Despite this molecular recapitulation, morphogenesis is strikingly different, with lens progenitors arising at the centre of the organoid, and subsequently translocating to the outside.
Comments:
The manuscript presents a beautiful set of high quality images showing expression of lens differentiation markers over time in the organoids. The set of experiments is very robust, with high numbers of organoids analysed and reproducible data. The mechanism by which lens specification is promoted in these organoids is, however, poorly analysed, and the reader does not get a clear understanding of what is different in these experiments, as compared to previous attempts, to support lens differentiation. There is a mention to HEPES supplementation, but no further analysis is provided, and the fact that the process is independent of ECM contradicts, as the authors point out, previous reports. The manuscript would benefit from a more detailed analysis of the mechanisms that lead to lens differentiation in this setting.
We followed the reviewer’s advice and have included a systematic analysis of the contribution of ECM (Matrigel) to the process of lens formation. In the revised manuscript we made sure to be sufficiently clear in the wording and description of our observation. Indeed, Matrigel is not required for the acquisition of lens fate, which can be demonstrated by the expression of lensspecific markers. However, the presence of Matrigel has a profound impact on structural aspects of organoid formation. Matrigel is essential for organization of retinal-committed cells to form a retinal epithelium (Zilova et al., 2021, eLIFE). The absence of the structure of the retinal epithelium in turn indeed negatively impacts on the cellular organization and the overall lens structure.
To clarify the contribution of the Matrigel to the organoid organization, we performed additional experiments (see revised Figure 2-figure supplement 1c-f). As mentioned above, the absence of Matrigel impacts on the organization and thickness of retinal neuroepithelium (Rx2+, Figure 2-figure supplement 1c). However, measurement of the lens in organoids at day 2 and day 5 showed that size of the lens is not impacted upon by the absence of Matrigel (Figure 3-figure supplement 1d-e).
Additionally, taking advantage of the Foxe3::GFP lens reporter line, we measured the onset of lens-specific gene expression in organoids with and without Matrigel. In both conditions (with and without Matrigel supplementation), Foxe3::GFP expression was initiated at 25 hours post aggregation (see revised Figure 4b).
The role of the HEPES in lens formation is indeed intriguing and currently under investigation. HEPES is mainly used to adjust the pH of the culture media, which, on its own might have an impact on multiple cellular processes. It will require a significant time investment to address the potential HEPES triggered molecular mechanisms impacting on lens formation (cross reference with Reviewer #3), which clearly goes beyond the scope of the current manuscript.
The markers analysed to show onset of lens differentiation in the organoids seem to start being expressed, in vivo, when the lens placode starts invaginating. An analysis of earlier stages is not presented. This would be very informative, allowing to determine whether progenitors differentiate as placode and neuroepithelium first, to subsequently continue differentiating into lens and retina, respectively. Could early placodal and anterior neural plate markers be analysed in the organoids? This would provide a more complete sequence of lens vs retina differentiation in this model.
We have taken care to show according stages in embryo and organoid side by side. We provide additional data to highlight the expression of Rx3::H2B-GFP (retina) and Foxe3::GFP (lens and lens placode) markers in earlier developmental stages. For the presumptive eye field within the region of the anterior neural plate (S16, late gastrula) Rx3 represents one of the earliest markers (see revised Figure 3-figure supplement 1). Already before an apparent lens placode is formed (see revised Figure 3d) Foxe3::GFP expression is detected within the presumptive lens ectoderm, demonstrating that Foxe3 is ideally suited as an early marker for placodal progenitors in medaka. The onset of Rx3 and Foxe3-driven reporters is clearly early enough to support the claim about the separate origin of the lens (placodal) and retinal (anterior neuroectoderm) tissues within the ocular organoids now represented in the revised figures.
The analysis of BMP and Fgf requirement for lens formation and differentiation is suggestive, but the source of these signals is not resolved or mentioned in the manuscript. Are BMP4 and Fgf8 expressed by the organoids? Where are they coming from?
Assessing the activity of BMP and FGF signaling (cross-reference to Reviewer #1) in the organoids is an important point that we have taken care of and included in the revised manuscript.
To address this point, we assessed which tissue/part of the organoid is responding to BMP and FGF signaling. To do so we analyzed the presence of phosphorylated SMAD1/5/8 (pSMAD1/5/8) and phosphorylated ERK (pERK1/2) as BMP and FGF signaling target in ocular organoids from day 1 to day 2. BMP signaling activity was detected in the center (region of establishment of lens-committed progenitors (Figure 3e)) of the organoid at day 1 (see revised Figure 6a). At day 1, only low levels of FGF signaling activity were detectable in presumptive retinal or/and lens tissue (see revised Figure S6b). Only half a day later, a significant increase in FGF activity was observed specifically in the central region of the organoids (lens progenitor domain, at day 1.5), prior to the onset of differentiation of lens fiber cells. This, together with inability of lens progenitor cells to differentiate to lens fiber cells in the presence of FGF inhibitor SU5402 provided during this critical period (day 1 to day 2) demonstrates that FGF signaling activity localized in the lens progenitor cells is required for lens fiber differentiation.
By day 2, FGF activity was detected in both lens and retinal tissue of the organoid. Similar patterns of FGF activity were observed in embryos at 2 days post fertilization (see revised Figure S6b).
The treatment with the FGF signaling inhibitor SU5402 from day 0 to day 1 did have no impact on the core size of organoid the dimension of which were fully comparable to the control (please see Figure 6b).
Related to the presence of the corresponding ligands we can state that they are indeed expressed in the organoids at the matching stages based on RNA seq and RT-PCR analyses, however we could not find them specifically localized. This may be due to a widespread, ubiquitous expression or may simply relate to technical problems.
While we can state with confidence that the ligands are present at the relevant time points and trigger the downstream pathways in a localized manner, the question whether the response is due to a localized signal or localized competence remains to be addressed.
The fact that the lens becomes specified in the centre of the organoid is striking, but it is for me difficult to visualise how it ends up being extruded from the organoid. Did the authors try to follow this process in movies? I understand that this may be technically challenging, but it would certainly help to understand the process that leads to the final organisation of retinal and lens tissues in the organoid. There is no discussion of why the morphogenetic mechanism is so different from the in vivo situation. The manuscript would benefit from explicitly discussing this.
Following the shift of the lens in vivo is indeed very relevant suggestion and we have taken care to address this in the revised manuscript.
To clarify this process, we included additional experiments and followed the movements of lens cells (see new Figures 4c, 4d and 4e, new Videos S6 and S7). Foxe3::GFP lens progenitor cells were found to actively move over long distances from center to the organoid periphery. This movement was accompanied by profound cell shape changes of lens progenitor cells with the active extension of lamellipodia and filopodia strongly arguing for an active movement of lens cells to the organoid periphery (cross-reference with Reviewer #1 and Reviewer #2).
Referees cross-commenting
We all seem to have similar comments and concerns. I think overall the suggestions are feasible and realistic for the timeframe provided.
Reviewer #3 (Significance):
This study describes a reproducible approach to differentiate ocular organoids composed of lens and retinal tissues. The characterisation of lens differentiation in this model is very detailed, and despite the morphogenetic differences, the molecular mechanisms show many similarities to the in vivo situation. The manuscript however does not highlight, in my opinion, why this model may be relevant. Clearly articulating this relevance, particularly in the discussion, will enhance the study and provide more clarity to the readers regarding the significance of the study for the field of organoid research, ocular research and regenerative studies.




