Abstract
Many cells in high glucose repress mitochondrial respiration, as observed in the Crabtree and Warburg effects. Our understanding of biochemical constraints for mitochondrial activation is limited. Using a Saccharomyces cerevisiae screen, we identified the conserved deubiquitinase Ubp3 (Usp10), as necessary for mitochondrial repression. Ubp3 mutants have increased mitochondrial activity despite abundant glucose, along with decreased glycolytic enzymes, and a rewired glucose metabolic network with increased trehalose production. Utilizing Δubp3 cells, along with orthogonal approaches, we establish that the high glycolytic flux in glucose continuously consumes free Pi. This restricts mitochondrial access to inorganic phosphate (Pi), and prevents mitochondrial activation. Contrastingly, rewired glucose metabolism with enhanced trehalose production and reduced GAPDH (as in Δubp3 cells) restores Pi. This collectively results in increased mitochondrial Pi and derepression, while restricting mitochondrial Pi transport prevents activation. We therefore suggest that glycolytic-flux dependent intracellular Pi budgeting is a key constraint for mitochondrial repression.
Introduction
Rapidly proliferating cells have substantial metabolic and energy demands in order to increase biomass (1, 2). This includes a high ATP demand, obtained from cytosolic glycolysis or mitochondrial oxidative phosphorylation (OXPHOS), to fuel multiple reactions (3). Interestingly, many rapidly proliferating cells preferentially rely on ATP from glycolysis/fermentation over mitochondrial respiration even in oxygen-replete conditions, and is the well-known Warburg effect (4, 5). Many such cells repress mitochondrial processes in high glucose, termed glucose-mediated mitochondrial repression or the Crabtree effect (6, 7). This is observed in some tumors (5), neutrophils (8), activated macrophages (9), stem cells (10–12), and famously Saccharomyces cerevisiae (7). Numerous studies have identified signaling programs or regulators of glucose-mediated mitochondrial repression. However, biochemical programs and regulatory processes in biology evolve around key biochemical constraints (13). The biochemical constraints for mitochondrial repression remain unresolved (14, 15).
There are two hypotheses on the biochemical principles driving mitochondrial repression. The first proposes direct roles for glycolytic intermediates in driving mitochondrial respiration, by inhibiting specific mitochondrial outputs (16, 17). The second hypothesizes that a competition between glycolytic and mitochondrial processes for mutually required metabolites/co-factors such as pyruvate, ADP or inorganic phosphate (Pi) could determine the extent of mitochondrial repression (14, 15, 18). These are not all mutually exclusive, and a combination of these factors might dictate mitochondrial repression in high glucose. However, any hierarchies of importance are unclear (19), and experimental data for the necessary constraints for mitochondrial repression remains incomplete.
One approach to resolve this question has been to identify regulators of metabolic state under high glucose. Post-translational modifications (PTMs) regulated by signaling systems can regulate mitochondrial repression (20–22). Ubiquitination is a PTM that regulates global proteostasis (23, 24), but the roles of ubiquitination-dependent processes in regulating mitochondrial repression are poorly explored. Ubiquitination itself is determined by the balance between ubiquitination, and deubiquitinase (DUB) dependent deubiquitination (25). Little is known about the roles of DUBs in regulating metabolic states, making the DUBs interesting candidate regulators of mitochondrial repression.
In this study, using an S. cerevisiae DUB knock-out library-based screen, we identified the evolutionarily conserved deubiquitinase Ubp3 (mammalian Usp10) as required for mitochondrial repression in high glucose. Loss of Ubp3 resulted in mitochondrial activation, along with a reduction in the glycolytic enzymes-phosphofructokinase 1 (Pfk1) and GAPDH (Tdh2 and Tdh3). This consequently reroutes glucose flux and increases trehalose biosynthesis. This metabolic rewiring increases Pi release from trehalose synthesis, and decreases Pi consumption in glycolysis, to cumulatively increase Pi pools. Using ubp3Δ cells along with independent analysis of wild-type cells, and isolated mitochondrial fractions, we establish that glycolytic-flux dependent Pi allocations to mitochondria determines mitochondrial activity. Through these data, we propose how intracellular Pi balance as controlled by glycolytic flux, is a biochemical constraint for mitochondrial repression.
Results
A deubiquitinase deletion screen identifies Ubp3 as a regulator of glucose-mediated mitochondrial repression
In cells such as S. cerevisiae, high glucose represses mitochondrial activity as well as OXPHOS-dependent ATP synthesis (7, 26) (illustrated in Figure 1A). Our initial objective was to identify proteostasis regulators of glucose-mediated mitochondrial repression. We generated and used a deubiquitinase (DUB) deletion strain library of S. cerevisiae (Figure 1-figure supplement 1A), to unbiasedly identify regulators of mitochondrial repression by measuring the fluorescence intensity of a potentiometric dye Mitotracker CMXRos (illustrated in Figure 1B). Using this screen, we identified DUB mutants with altered mitochondrial membrane potential (Figure 1C, Figure 1-figure supplement 1 A). Note: WT cells in a respiratory medium (2% ethanol) were used as a control to estimate maximum mitotracker fluorescence intensity (Figure 1-figure supplement 1B).

A deubiquitinase deletion screen identifies Ubp3 as a regulator of glucose-mediated mitochondrial repression
A) Schematic depicting glucose-mediated mitochondrial repression (Crabtree effect).
B) Schematic describing the screen with a yeast DUB KO library to identify regulators of Crabtree effect.
C) Identifying DUB knockouts with altered mitochondrial potential. Heat map shows relative mitochondrial membrane potential of 19 deubiquitinase deletions in high glucose, from 2 biological replicates. Also see figure 1-figure supplements 1A, B.
D) The deubiquitinase activity of Ubp3 and repression of mitochondrial membrane potential. WT, ubp3Δ, and Ubp3C469A were grown inhigh glucose and relative mitochondrial membrane potential was measured. Data represent mean ± SD from three biological replicates (n=3). Also see figure 1-figure supplement 1 D.
E) Effect of loss of Ubp3 on ETC complex IV subunit Cox2. WT and ubp3Δ were grown in high glucose, and Cox2 was measured (western blot using an anti-Cox2 antibody). A representative blot (out of 3 biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD.
F) Basal oxygen consumption rate (OCR) in high glucose in ubp3Δ. WT and ubp3Δ were grown in high glucose, and OCR was measured. Basal OCR corresponding to ∼3 × 10^5 cells, from two independent experiments (n=2), normalized to the OD600 is shown. Bar graph representations are shown in the inset. Data represent mean ± SD.
G) Total ATP levels in ubp3Δ and WT. WT and ubp3Δ were grown in high glucose, and total ATP were measured. Data represent mean ± SD from three biological replicates (n=3).
H) Dependence of ubp3Δ on mitochondrial ATP. WT and ubp3Δ cells were grown in high glucose, and treated with 1 mM sodium azide for 45 minutes. Total ATP levels in sodium azide treated and untreated cells were measured. Data represent mean ± SD (n=3).
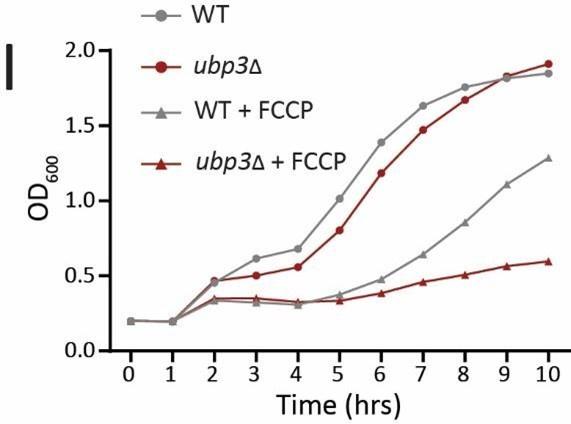
I) Requirement for mitochondrial respiration in high glucose in ubp3Δ. A growth curve of WT and ubp3Δ in high glucose in the presence of OXPHOS uncoupler FCCP (10 µM), and serial dilution growth assay in high glucose in the presence/absence of sodium azide (1 mM) are shown. Data represent mean ± SD (n=2). Also see figure 1-figure supplement 1 H, I.
Data information: **p<0.01, ***p<0.001.
A prominent ‘hit’ was the evolutionarily conserved deubiquitinase Ubp3 (Figure 1C), (homologous to mammalian Usp10) (Figure 1-figure supplement 1D). Due to its high degree of conservation across eukaryotes (Figure 1-figure supplement 1D) as well as putative roles in metabolism or mitochondrial function (27–29), we focused our further attention on this DUB. Cells lacking Ubp3 showed an ∼1.5-fold increase in mitotracker fluorescence (Figure 1C, Figure 1-figure supplement 1A, C). Cells with catalyticaly inactive Ubp3 (Ubp3C469A), showed increased mitochondrial potential comparable to ubp3Δ (Figure 1D). This data confirmed that Ubp3 catalytic activity of is required to fully repress mitochondrial activity under high glucose. The catalytic site mutation did not affect steady-state Ubp3 levels (Figure 1-figure supplement 1E).
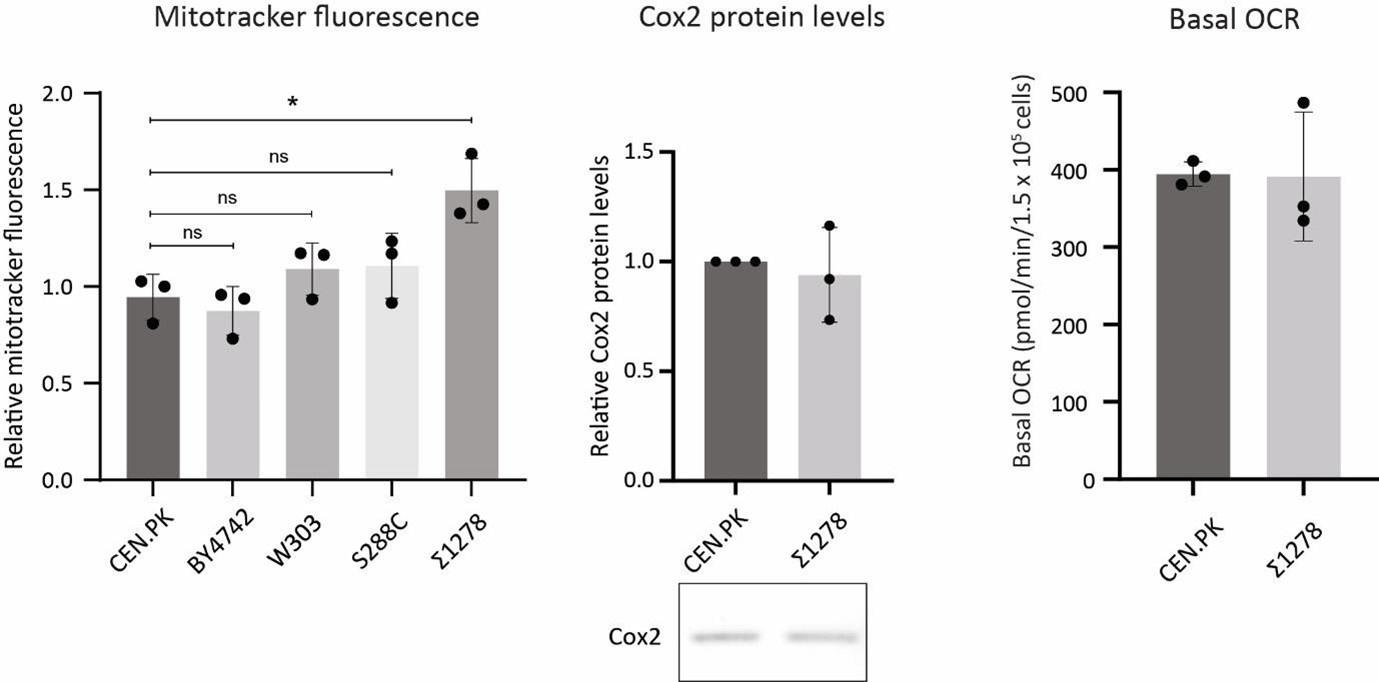
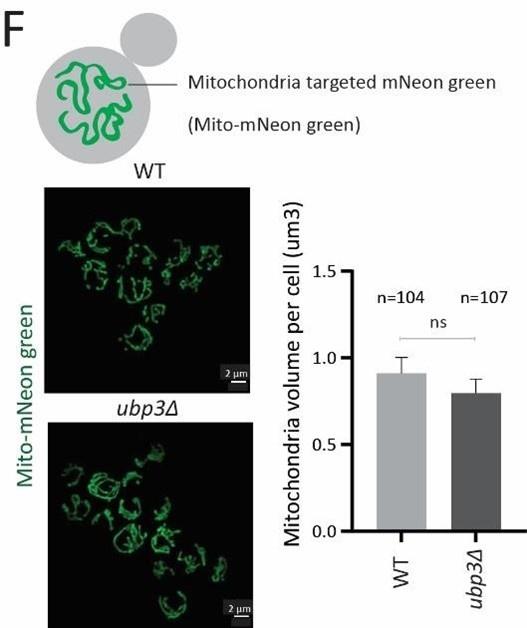
Next, to assess the requirement of Ubp3 for mitochondrial function, we quantified the electron transport chain (ETC) complex IV subunit Cox2 (30)(Figure 1E). ubp3Δ had higher Cox2 than WT (Figure 1E). As a control, we estimated total mitochondrial content in WT and ubp3Δ cells, using either estimates of the structural protein Tom70, or measuring the fluorescence-intensity in strains engineered with mitochondrial targeted mNeonGreen (31). There was no increase in the total mitochondrial volume (estimated by measuring the intensity of mitochondria targeted mNeon green) (Figure 1-figure supplement 1F) or mitochondrial outer membrane protein Tom70 (Figure 1-figure supplement 1G). This suggests that the increased Cox2 is not merely because of higher total mitochondrial content. We next measured the basal oxygen consumption rate (OCR) of ubp3Δ, and basal OCR was higher in ubp3Δ, indicating higher respiration (Figure 1F).
Next, we asked if mitochondrial ATP synthesis was higher in ubp3Δ. The total ATP levels in WT and ubp3Δ were comparable (Figure 1G). However, upon treatment with the ETC complex IV inhibitor sodium azide, ATP levels in WT were higher than ubp3Δ, contributing to ∼60% of the total ATP (Figure 1H). In contrast, the ATP levels in ubp3Δ after sodium azide treatment were ∼ 40% of the total ATP (Figure 1H). These data suggest a higher contribution of mitochondrial ATP synthesis towards the total ATP pool in ubp3Δ.
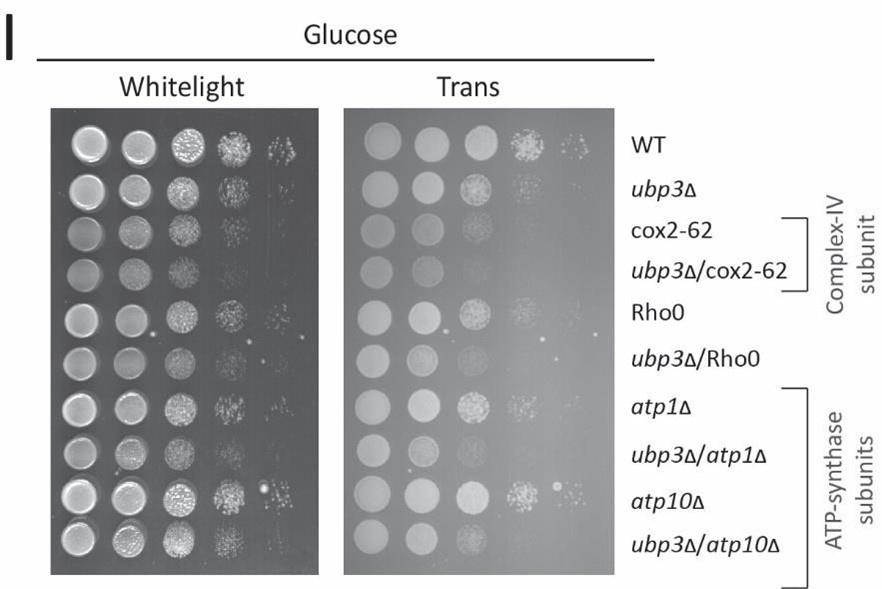
We next asked if ubp3Δ required higher mitochondrial activity for growth, using a series of mitochondrial activity inhibitors and comparing relative growth. In high glucose, WT cells show minimal growth inhibition in the presence of sodium azide, indicating lower reliance on mitochondrial function (Figure 1I). Contrastingly, ubp3Δ or Ubp3C469A show a severe growth defect in the presence of the mitochondrial ETC complex inhbitors sodium azide and oligomycin, and the mitochondrial OXPHOS uncoupler FCCP (Figure 1I, Figure 1-figure supplement 1H). Additionally, the loss of Ubp3 in respiration defective cox2-62 cells (32), or Rho0 cells (which lacks mitochondrial DNA) resulted in a severe growth defect (Figure 1-figure supplement 1I). Deletion of ATP synthase subunits Atp1 and Atp10 also results in a severe growth defect in ubp3Δ compared to WT (Figure 1-figure supplement 1I). Together, these results indicate that the loss of Ubp3 makes cells dependent on mitochondrial ATP synthesis in high glucose.
Collectively these data show that in 2% glucose, ubp3Δ have high mitochondrial activity, respiration, and rely on this mitochondrial function for ATP production and growth. We therefore decided to use ubp3Δ cells to start delineating requirements for glucose-mediated mitochondrial repression.
Key glycolytic enzymes decrease and glucose flux is rerouted in ubp3Δ cells
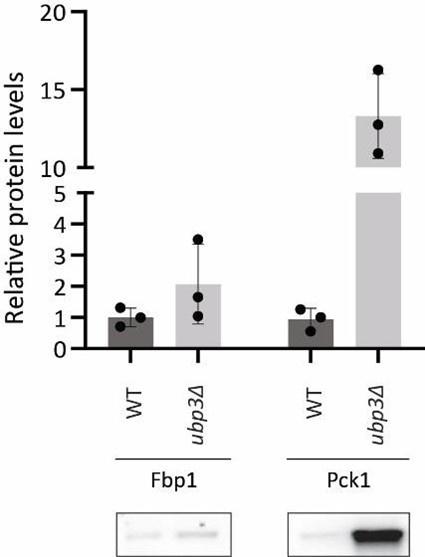
Glucose-6 phosphate (G6P) is the central node in glucose metabolism where carbon-allocations are made towards distinct metabolic arms, primarily: glycolysis, the pentose phosphate pathway (PPP), and trehalose biosynthesis (Figure 2A). We first compared amounts of two key (‘rate-controling’) glycolytic enzymes - Phosphofructokinase 1 (Pfk1), GAPDH isozymes (Tdh2, Tdh3) (3), along with the enolase isozymes (Eno1, Eno2) in WT and ubp3Δ cells. Pfk1, Tdh2, and Tdh3 substantially decreased in ubp3Δ (but not Eno1 and Eno2) (Figure 2B, Figure 2-figure supplement 1A). Since deubiquitinases can control protein amounts by regulating proteasomal degradation, we asked if the decrease in Pfk1, Tdh2 and Tdh3 in ubp3Δ is due to proteasomal degradation. To test this, we measured the levels of these enzymes in ubp3Δ after treatement with proteasomal inhibitor MG132. We did not observe any rescue in the levels of these enzymes in MG132 treated samples, suggesting that the decreased levels of these enzymes were not due to increased proteasomal degradation (Figure 2-figure supplement 1B). To further understand if these enzyme transcripts are altered in ubp3Δ, we measured the mRNA levels of PFK1, TDH2 and TDH3 in WT, ubp3Δ and Ubp3C469A cells. The transcripts of all the three genes in ubp3Δ and Ubp3C469A cells decreased (Figure 2-figure supplement 1C), suggesting that Ubp3 regulates the transcripts of these glycolytic enzyme genes. The reduction in the Pfk and GAPDH enzyme amounts was intriguing, because the Pfk and GAPDH steps are critical in determining glycolytic flux (3, 33). A reduction in these enzymes could therefore decrease glycolytic flux, and would reroute glucose (G6P) allocations via mass action towards other branches of glucose metabolism, primarily the pentose phosphate pathway as well as trehalose biosynthesis (Figure 2A). To assess this, we first measured the steady-state levels of key glycolytic and pentose phosphate pathway (PPP) intermediates, and trehalose in WT or ubp3Δ using targeted LC-MS/MS (Figure 2C, Figure 2-figure supplement 1D). Glucose-6/fructose-6 phosphate (G6P/F6P) increased in ubp3Δ (Figure 2C). Concurrently, trehalose, and the PPP intermediates ribose 5-phosphate (R5P) and sedoheptulose 7-phosphate (S7P), increased in ubp3Δ (Figure 2C).

Key glycolytic enzymes decrease and glucose flux is rerouted in ubp3Δ cells
A) A schematic illustrating directions of glucose-6-phosphate (G6) flux in cells. Glucose is converted to G6P, a precursor for trehalose, the pentose phosphate pathway (PPP), and glycolysis.
B) Effect of loss of Ubp3 on key glycolytic enzymes. WT and ubp3Δ were grown in high glucose and the Pfk1, Tdh2, and Tdh3 levels were measured by western blot using an anti-FLAG antibody. A representative blot (out of three biological replicates, n=3) and their quantification are shown. Data represent mean ± SD. Also see figure 2-figure supplement 1A.
C) Steady-state metabolite amounts in WT and ubp3Δ in high glucose. Relative steady-state levels of trehalose, major glycolytic, and PPP intermediates were estimated in WT and ubp3Δ. Data represent mean ± SD from three biological replicates (n=3). Also see Appendix Table S3.
D) Relative glycolytic and trehalose synthesis flux in WT and ubp3Δ. Relative 13C-label incorporation into trehalose and glycolytic intermediates, after a pulse of 1% 13C6 glucose is shown. Data represent mean ± SD from three biological replicates (n=3). Also see Appendix Table S3, figure 2-figure supplement 1 D, E.
E) Ethanol production in ubp3Δ. WT and ubp3Δ were grown in high glucose and ethanol in the media was measured. Data represent mean ± SD from three biological replicates (n=3).
F) Relative rate of ethanol production in WT vs ubp3Δ. WT and ubp3Δ were grown in high glucose (to OD600 ∼ 0.6), equal numbers of cells were shifted to fresh medium (high glucose) and ethanol concentration in the medium was measured temporally. Data represent mean ± SD from three biological replicates (n=3)
Data information: *p<0.05, **p<0.01, ***p<0.001.
Since steady-state metabolite amounts cannot separate production from utilization, in order to unambiguously assess if glycolytic flux is reduced in ubp3Δ cells, we utilized a pulse labeling of 13C6 glucose, following which the label incorporation into glycolytic and other intermediates was measured. Note that because glycolytic flux is very high in yeast, this experiment would require rapid pulsing and extraction of metabolites in order to stay in a linear range and avoid label saturation. We therefore established a very short time point of label-addition, quenching and metabolite extraction post 13C glucose pulse. Since flux saturates/reaches steady-state in seconds, we first ensured that the label incorporation into individual metabolites after the 13C glucose pulse was in the linear range, and for early glycolytic intermediates this was seconds after glucose addition. This new methodology is extensively described in materials and methods, with required controls shown in Figure 2-figure supplement 1F. WT and ubp3Δ cells were grown in high glucose, pulsed with 13C6 glucose, and the relative 13C label incorporation into glycolytic intermediates and trehalose were measured, as shown in the schematic (Figure 2-figure supplement 1E). In ubp3Δ, 13C label incorporation into G6P/F6P as well as trehalose substantially increased (Figure 2D). Contrastingly, 13C label incorporation into glycolytic intermediates F1,6BP, G3P, 3PG and PEP decreased, indicating decreased glycolytic flux (Figure 2D). We next measured ethanol concentrations and production rates, as an additional output of relative glycolytic rates. We observed decreased steady-state ethanol levels, as well as ethanol production rates in ubp3Δ (Figure 2F).
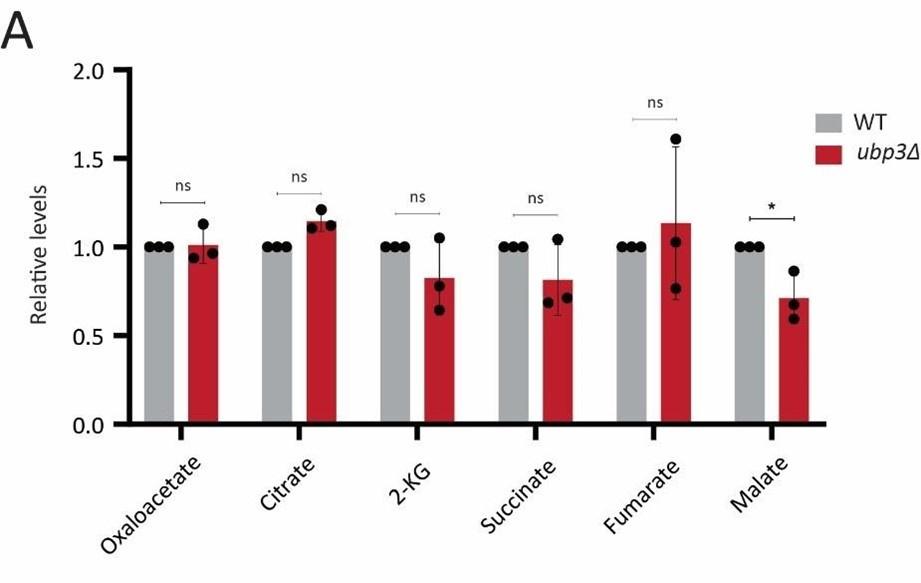
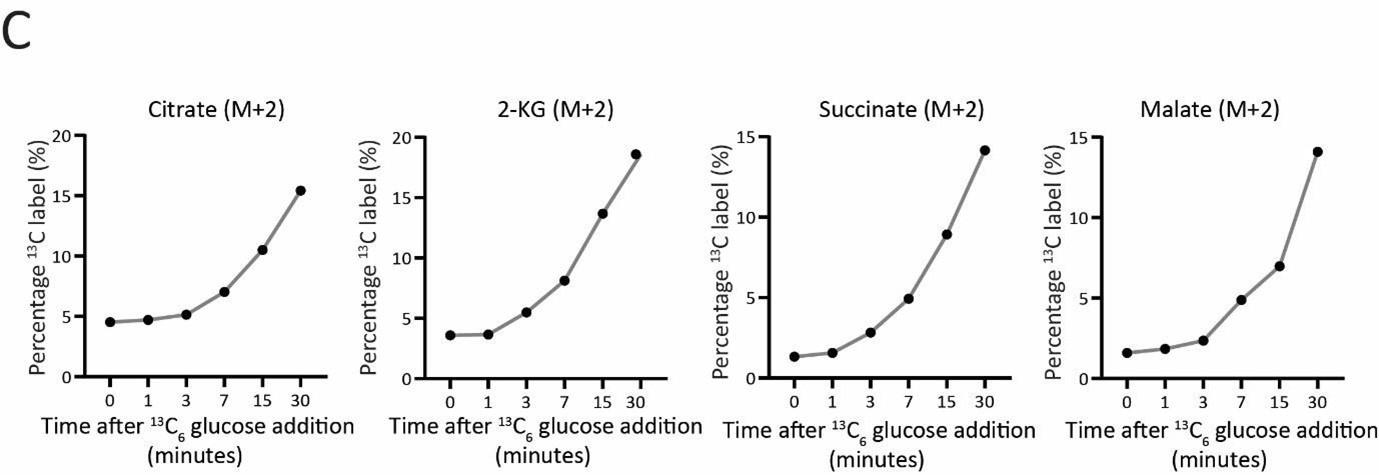
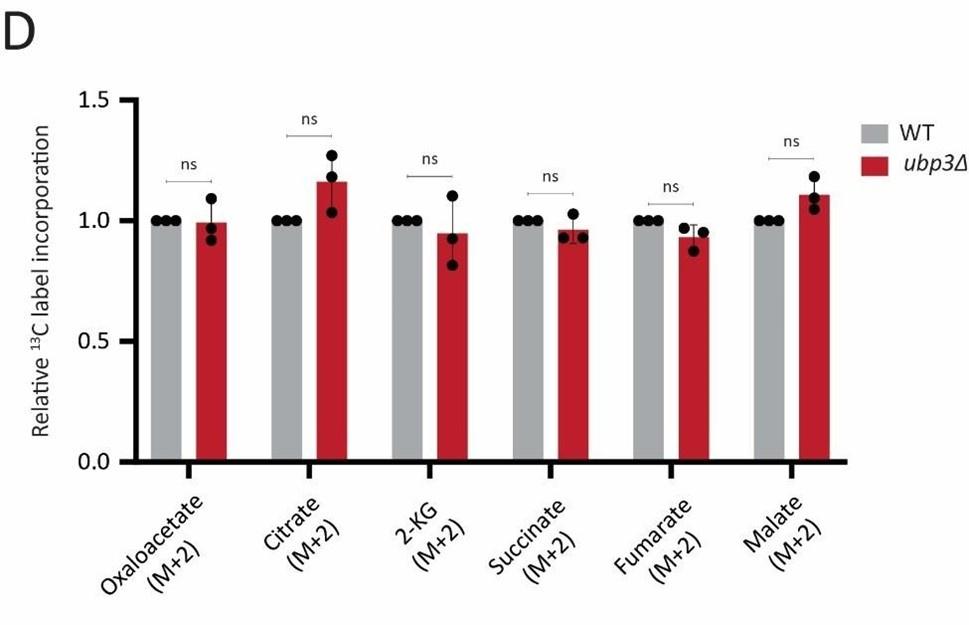
Glycolysis-derived pyruvate is transported to mitochondria and fuels the TCA cycle. Therefore, we asked if the decreased glycolytic rate result in a decrease in the TCA cycle flux as well. To test this, we first measured the steady state levels of TCA cycle intermediates in WT or ubp3Δ using targeted LC-MS/MS. We did not observe any significant change in the levels of TCA cycle intermediates in ubp3Δ, except malate which showed a significant decrease in ubp3Δ (Figure 2-figure supplement 2A). Next, in order to assess if TCA cycle flux reduces in ubp3Δ cells, WT and ubp3Δ cells were grown in high glucose, pulsed with 13C6 glucose, and the relative 13C label incorporation into TCA cycle intermediates was measured, as shown in the schematic (Figure 2-figure supplement 2B). The kinetics of 13C label incorporation in TCA cycle intermediates are shown in Figure 2-figure supplement 2C. We did not observe any significant change in the relative 13C label incorporation in TCA cycle intermediates in ubp3Δ. Therefore, these data suggest that the decreased glycolytic flux in ubp3Δ does not result in a decrease in TCA cycle flux. The increased respiration and mitochondrial activity in ubp3Δ cells is therefore driven via other factors.
Collectively, these results reveal that that reduced Pfk1 and GAPDH (in ubp3Δ) decrease glucose flux via glycolysis, which results in rewired glucose flux towards trehalose biosynthesis and the PPP.
Rerouted glucose flux results in phosphate (Pi) accumulation
We therefore asked if the proteomic state, as observed in ubp3Δ cells, could provide clues to explain the coupling between mitochondrial derepression and rerouted glucose flux. A recent study by Isasa et al., had systematically quantified the changes in protein levels in ubp3Δ cells (28). We therefore reanalyzed this extensive dataset, looking for changes in proteins that would correlate with these metabolic processes. Notably, we observed increased levels of proteins of the mitochondrial ETC and respiration, and decreased amounts of glucose metabolizing enzymes in ubp3Δ (Figure 3A). Additionally, multiple proteins involved in regulating phosphate (Pi) homeostasis were decreased in ubp3Δ (Figure 3A) (28). We had recently uncovered a reciprocal coupling of Pi homeostasis with the different arms of glucose metabolism, particularly trehalose biosynthesis (34, 35), and therefore wondered if a glycolytic-flux dependent change in Pi homeostatsis had any role in mitochondrial respiration. Our hypothesis was refined based on the reasoning given below.

Rerouted glucose flux results in phosphate (Pi) accumulation
A) Changes in protein levels in ubp3Δ (dataset from Isasa et al., 2015). ubp3Δ cells have an increase in proteins involved in mitochondrial respiration and decrease in proteins involved in glucose and phosphate metabolism.
B) Schematic showing maintenance of Pi balance during glycolysis. Trehalose synthesis from G6P releases Pi, and the conversion of G3P to 1,3BPG by GAPDH consumes Pi. In ubp3Δ, trehalose biosynthesis (which releases Pi) increases. ubp3Δ have decreased GAPDH, which will decrease Pi consumption. This increase in Pi release along with decreased Pi consumption could increase cytosolic Pi.
C) Intracellular Pi levels in WT and ubp3Δ. WT and ubp3Δ were grown in high glucose and the total free phosphate (Pi) levels were estimated. WT in high Pi (2% glucose, 10mM Pi) was a positive control. Data represent mean ± SD from three biological replicates (n=3). Also see figure 3-figure supplement 1A.
D) Pho regulon responses in WT and ubp3Δ. Protein levels of Pho84-FLAG and Pho12-FLAG were compared between WT grown in high glucose and in high Pi, ubp3Δ in high glucose with or without a shift to a no-Pi medium for one hour, by western blot. A representative blot (out of three biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD.
E) Contribution of trehalose synthesis as a Pi source. WT, tps2Δ, ubp3Δ, and ubp3Δtps2Δ were grown in high glucose and the total Pi levels were estimated. Data represent mean ± SD from three biological replicates (n=3). Also see figure 3-figure supplement 1B.
F) Loss of GAPDH isozymes Tdh2 and Tdh3 and effect on Pi. WT, ubp3Δ, and tdh2Δtdh3Δ were grown in high glucose and total Pi was estimated. Data represent mean ± SD from three biological replicates (n=3).
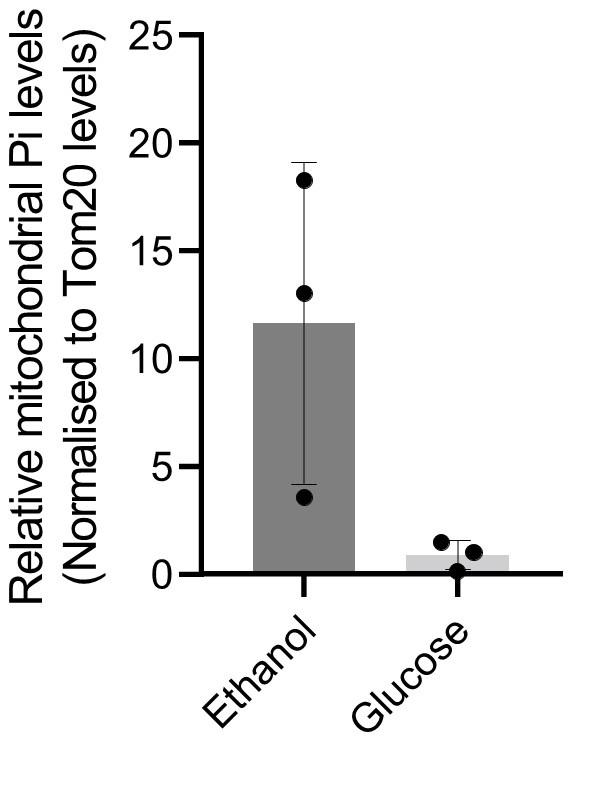
G) Pi levels in ubp3Δ and tdh2Δtdh3Δ cells in ethanol medium. WT, ubp3Δ, and tdh2Δtdh3Δ cells were grown in ethanol medium and the total Pi levels were estimated. Data represent mean ± SD from three biological replicates (n=3).
Data information: *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
One explanation for mitochondrial repression can be an internal competition for shared metabolites/co-factors between (cytosolic) glycolytic and mitochondrial processes, that mitochondria might not be sufficiently able to access (14, 18, 19). In this context, a plausible role for inorganic phosphate (Pi) in regulating mitochondrial repression can be hypothesized. Cytosolic glycolysis requires rapid, high consumption of net Pi (19, 36, 37), and this could possibly limit the Pi that is continuously available for mitochondrial use (18, 38), thereby repressing mitochondria. Contextually, the balance between reactions releasing vs consuming Pi could explain changes in global Pi levels (35). A continuous hub of Pi consumption is glycolysis. Here, GAPDH catalyzes G3P to 1,3BPG, converting ADP to ATP, while concurrently consuming a molecule of Pi (37, 39). This Pi that goes into ATP will subsequently be used for nucleotide biosynthesis, polyphosphate biosynthesis and protein phosphorylation (35, 40). Therefore, we can surmise that in high glycolytic flux, the production of ATP, nucleotides and polyphosphates is concurrent with Pi consumption (39, 41, 42). Could this reaction therefore limit cytosolic Pi for the mitochondria (as illustrated in Figure 3B)? Notably, ubp3Δ have reduced GAPDH levels and decreased glycolytic flux. Second, trehalose synthesis is a Pi releasing reaction, and a major source of free Pi that is critical for Pi homeostasis (34, 37). Flux through this reaction is also substantially higher in ubp3Δ. Therefore, these cells might have increased Pi release (via trehalose), coupled with decreased Pi consumption (via decreased GAPDH). We therefore asked if total Pi increases in ubp3Δ (Figure 3B)?
To test this, we directly assessed total Pi levels in ubp3Δ and WT. ubp3Δ cells had higher Pi than WT, and this was comparable to Pi in WT grown in excess Pi (Figure 3C). Similarly, Pi amounts also increased in Ubp3C469A (Figure 3-figure supplement 1A). Therefore, the loss of Ubp3 increases intracellular Pi levels. Next, we asked if ubp3Δ cells exhibit signatures of a ‘high Pi’ state. S. cerevisiae maintains internal Pi balance by controlling the expression of multiple genes collectively known as the Pho regulon (43). The Pho regulon is induced under Pi limitation, and repressed during Pi sufficiency (34, 43). We assessed two major Pho proteins (Pho84: a high-affinity membrane Pi transporter, and Pho12: an acid phosphatase) in WT and ubp3Δ in high glucose. ubp3Δ cells have lower amounts of Pho84 and Pho12 (Figure 3D). Further, upon shifting to low Pi for one hour, Pho84 and Pho12 increased in ubp3Δ. These data suggest that reduced Pho84 and Pho12 amounts in ubp3Δ are because of increased Pi, and not due to altered Pho regulon function itself (Figure 3D). These data clarify earlier observations from ubp3Δ which noted reduced Pho proteins (28). Therefore, ubp3Δ constitutively have higher Pi, and likely a consequent decrease in Pho proteins.
We next asked if the increased Pi in ubp3Δ is because of altered G6P allocations towards different end-points, particularly trehalose synthesis, which can be a major node of Pi restoration (34, 37). We assessed the contribution of increased trehalose synthesis towards the high Pi in ubp3Δ, by estimating Pi levels in the absence of trehalose 6-phosphate phosphatase (Tps2), which catalyzes the Pi-releasing step in trehalose synthesis. Notably, loss of Tps2 in ubp3Δ decreased Pi (Figure 3E). There was no additive difference in Pi between tps2Δ and ubp3Δtps2Δ (Figure 3E). As an added control, we assessed trehalose in WT and ubp3Δ in the absence of Tps2, and found no difference (Figure 3-figure supplement 1B). Therefore, increasing G6P flux towards trehalose biosynthesis is a major source of the increased Pi in ubp3Δ.
Since the major GAPDH isozymes, Tdh2 and Tdh3, are reduced in ubp3Δ, we directly asked if reducing GAPDH can decrease Pi consumption and increase Pi. To assess this, we generated tdh2Δtdh3Δ cells, which exhibit a growth defect, but are viable, permitting further analysis. tdh2Δtdh3Δ had higher Pi in high glucose (Figure 3F). Expectedly, we observed a decrease in ethanol in tdh2Δtdh3Δ (Figure 3-figure supplement 1C), along with an accumulation of F1,6 BP and G3P, and decreased 3PG and PEP (Figure 3-figure supplement 1D). However, G6P and trehalose levels between WT and tdh2Δtdh3Δ were comparable (Figure 3-figure supplement 1D, E). These data sugest that unlike in ubp3Δ, the increased Pi in tdh2Δtdh3Δ comes mainly from decreased Pi consumption (GAPDH step). To further assess the role of reduced glycolytic flux in increasing Pi (ubp3Δ and tdh2Δtdh3Δ), we measured the Pi in these cells growing in a gluconeogenic medium – 2%, ethanol. In this scenario, the GAPDH catalyzed reaction will be reversed, converting 1,3BPG to G3P, which should release and not consume Pi. Compared to WT, the Pi levels decreased in ubp3Δ and tdh2Δtdh3Δ (Figure 3G), suggesting that the changes in Pi in these mutants is driven by the relative change in Pi release vs consumption.
These results collectively indicate that the combined effect of increased Pi release coming from trehalose synthesis, and decreased Pi consumption from reduced GAPDH increase Pi levels in ubp3Δ.
Mitochondrial Pi availability correlates with mitochondrial activity in ubp3Δ
We therefore wondered if this observed Pi increase from the combined rewiring of glucose metabolism resulted in more Pi becoming accessible to the mitochondria. This would effectively result in Pi budgeting between cytosolic glycolysis and mitochondria based on the relative flux in different arms of glucose metabolism, by increasing Pi availability for the mitochondria (Figure 4A). To test if this were so, we first asked if the mitochondria in ubp3Δ have increased Pi. Mitochondria were isolated by immunoprecipitation from WT and ubp3Δ, the isolation efficiency was analyzed (Figure 4-figure supplement 1A), and relative Pi levels compared. Pi levels were normalised to Idh1 (isocitrate dehydrogenase) in isolated mitochondria, since Idh1 protein levels did not decrease in ubp3Δ (Figure 4-figure supplement 1B), and consistent with (28). Mitochondrial Pi was higher in ubp3Δ (Figure 4B). We next asked if the increased mitochondrial activity in ubp3Δ is a consequence of high Pi. If this were so, tdh2Δtdh3Δ should partially phenocopy ubp3Δ with respect to mitochondrial activity. Consistently, tdh2Δtdh3Δ have higher mitotracker intensity as well as Cox2 levels compared to WT (Figure 4-figure supplement 1C, Figure 4C). Also consistent with this, a high basal OCR in tdh2Δtdh3Δ was observed (Figure 4D), together indicating high mitochondrial activity in tdh2Δtdh3Δ, which is comparable to ubp3Δ.

Mitochondrial Pi availability correlates with mitochondrial activity in ubp3Δ A) A hypothetical mechanism of cytosolic free Pi controling mitochondrial activity by regulating mitochondrial Pi availability.
B) Mitochondrial Pi amounts in WT vs ubp3Δ. Mitochondria were isolated by immunoprecipitation from WT and ubp3Δ and mitochondrial Pi estimated. Mitochondrial Pi levels (normalised to Idh1) are shown. Data represent mean ± SD from three biological replicates (n=3). Also see figure 4-figure supplement 1A, B.
C) Cox2 protein in tdh2Δtdh3Δ. WT, ubp3Δ, and tdh2Δtdh3Δ were grown in high glucose and Cox2 protein was estimated. A representative blot (out of three biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD.
D) Basal OCR levels in tdh2Δtdh3Δ. WT, ubp3Δ, and tdh2Δtdh3Δ were grown in high glucose and basal OCR was measured from two independent experiments (n=2). Data represent mean ± SD. Also see figure 4-figure supplement 1C.
E) Comparative Pi amounts and Cox2 levels in ubp3Δ, tdh2Δtdh3Δ, WT cells. WT cells were grown in high glucose, ubp3Δ and tdh2Δtdh3Δ were grown in high glucose and low Pi, and Cox2 protein was estimated. A representative blot (out of three biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD. Also see figure figure 4-figure supplement 1D, F.
F) Pi amounts and basal OCR in ubp3Δ and tdh2Δtdh3Δ vs WT cells. WT cells were grown in high glucose, ubp3Δ and tdh2Δtdh3Δ were grown in high glucose and low Pi, and basal OCR was measured from three independent experiments (n=3). Data represent mean ± SD.
G) Effect of loss of mitochondrial Pi transporter Mir1 on Cox2protein. WT, ubp3Δ, mir1Δ, and mir1Δubp3Δ were grown in high glucose and Cox2 amounts compared. A representative blot (out of three biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD.
H) Relationship of mitochondrial Pi transport and basal OCR in WT vs ubp3Δ. WT, ubp3Δ, mir1Δ, and mir1Δubp3Δ cells were grown in high glucose and basal OCR was measured from two independent experiments (n=2). Data represent mean ± SD.
Data information: *p<0.05, **p<0.01, ****p<0.0001.
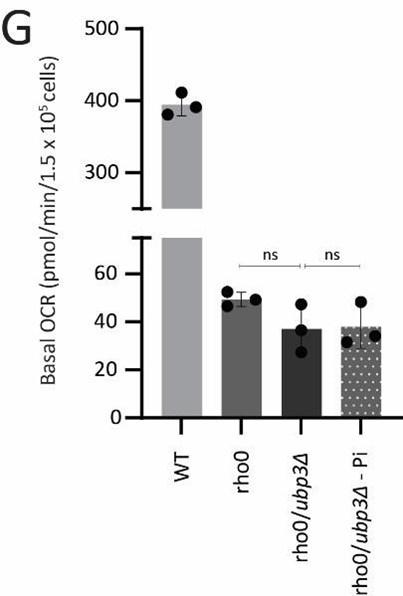
We next asked if higher Pi is necessary to increase mitochondrial activity in ubp3Δ and tdh2Δtdh3Δ. Since glycolysis is defective in ubp3Δ and tdh2Δtdh3Δ, it is necessary to distinguish the effect of high Pi vs. only the effect of low glycolysis in activating mitochondria. Logically, if decreased glycolysis (independent of Pi) is sufficient to activate mitochondria, bringing down the Pi levels in ubp3Δ to that of WT should not affect mitochondrial activity. Notably, ubp3Δ grown in low (1 mM) Pi have Pi levels similar to WT in standard (normal Pi) medium (Figure 4-figure supplement 1D). ubp3Δ grown in low Pi also had decreased ethanol, suggesting reduced glycolysis (Figure 4-figure supplement 1E). Therefore, we used this condition to further understand the role of Pi in inducing mitochondrial activity. Mitotracker fluorescence decreased in both ubp3Δ and tdh2Δtdh3Δ in low Pi (Figure 4-figure supplement 1F). Note: basal mitotracker fluorescence in WT also decreases in low Pi, which is consistent with a required role of Pi for mitochondrial activity (Figure 4-figure supplement 1F). Similarly, Cox2 levels were reduced in both ubp3Δ and tdh2Δtdh3Δ (Figure 4E). Consistent with both reduced mitotracker intensity and Cox2 levels, basal OCR also decreased in both ubp3Δ and tdh2Δtdh3Δ in low Pi (Figure 4F). As an additional control, we used Rho0 strains (which have no mitochondrial DNA and therefore lack functional ETC) to compare basal OCR. The expectation in these strains is that basal OCR will not change if Pi changes. Consistently, we did not observe any significant difference in basal OCR in WT, ubp3Δ and ubp3Δ in low Pi in a Rho0 strain background (which lacks mitochondrial DNA) (Figure 4-figure supplement 1G). These data collectively suggest that high intracellular Pi is necessary to increase mitochondrial activity in ubp3Δ and tdh2Δtdh3Δ.
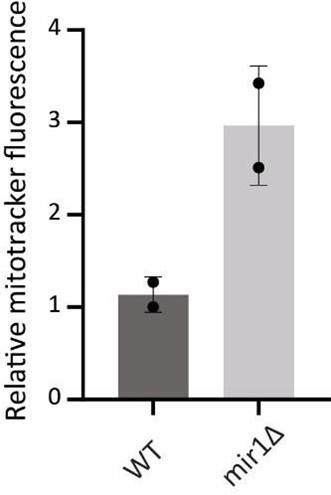
Next, we asked how mitochondrial Pi transport regulates mitochondrial activity. Mir1 and Pic2 are mitochondrial Pi transporters, with Mir1 being the major Pi transporter (44, 45). We first limited mitochondrial Pi availability in ubp3Δ by knocking-out MIR1. In mir1Δ, the increased Cox2 observed in ubp3Δ was no longer observed (Figure 4G). Consistent with this, we observed no further increase in the basal OCR in mir1Δubp3Δ compared to mir1Δ (Figure 4H). Furthermore, mir1Δ showed decreased Cox2 as well as basal OCR even in WT cells (Figure 4G, Figure 4H). These data together suggest that mitochondrial Pi transport is critical for increasing mitochondrial activity in ubp3Δ, and in maintaining basal mitochondrial activity even in high glucose.
As a control, no significant increase in Mir1 and Pic2 was observed in ubp3Δ (Figure 4-figure supplement 1H), suggesting that ubp3Δ do not increase mitochondrial Pi by merely increasing the Pi transporters, but rather by increasing available Pi pools.
Taken together, these data suggest the possibility that increased cytosolic Pi increases the mitochondrial Pi pool, which increases mitochondrial activity. Decreasing mitochondrial Pi by either reducing total Pi, or by reducing mitochondrial Pi transport decreases mitochondrial activity.
Mitochondrial Pi availability constrains mitochondrial activity under high glucose
So far, these data suggest that the cytosolic Pi available for the mitochondria can determine the extent of mitochondrial activity. Therefore, we further investigated if mitochondrial Pi allocation was a necessary constraint for glucose-mediated mitochondrial repression.
To test this, we first asked how important mitochondrial Pi transport was to switch to increased respiration. In WT yeast, low glucose or glycolytic inhibition will result in increased respiration (22). What happens therefore if we restrict mitochondrial Pi in this context? For this, we measured the basal OCR in WT and mir1Δ after switching from high (2%) to low (0.1%) glucose. We observed a significant increase in the basal OCR in WT but not in mir1Δ (Figure 5A). The alternate scenario is after glycolytic inhibition. We assessed the role of mitochondrial Pi in this context, by inhibiting glycolysis using 2-deoxyglucose (2DG). WT, but not mir1Δ, increased their OCR (respiration) upon a 1-hour treatment with 2DG (Figure 5B). Consistent with this, mitotracker fluorescence increased with an increase in 2DG in WT, but not in mir1Δ (Figure 5-figure supplement 1A). We further asked if the mitochondrial Pi transporter itself glucose repressed, and therefore assessed Mir1 amounts in high and low glucose. Mir1 levels are higher upon a shift to low glucose, and in cells grown in 2% ethanol, suggesting that Mir1 is glucose repressed (Figure 5C, Figure 5-figure supplement 1B). These data suggest that mitochondrial Pi transport is necessary for increasing mitochondrial activity after glucose derepression.

Mitochondrial Pi availability constrains mitochondrial activity under high glucose
A) Relationship of mitochondrial Pi transport and respiration after glucose removal. WT and mir1Δ cells were cultured in high (2%) glucose and shifted to low (0.1%) glucose for 1 hour. The normalized basal OCR, from two independent experiments (n=2) are shown. Data represent mean ± SD.
B) Requirement of mitochondrial Pi transport for switch to respiration upon glycolytic inhibition by 2DG. WT and mir1Δ cells were cultured in high glucose and treated with or without 0.25% 2DG for 1 hour. Basal OCR was measured from two independent experiments (n=2). Data represent mean ± SD. Also see figure 5-figure supplement 1A.
C) Glucose-dependent regulation of Mir1. Cells (with Mir1-HA) were grown in high glucose and shifted to low glucose (0.1% glucose) for 1 hour, and Mir1 levels compared. A representative blot (out of three biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD. Also see figure 5-figure supplement 1B.
D) Increasing Pi concentrations and mitochondrial activity in isolated mitochondria. Mitochondria were isolated from WT cells grown in high glucose, incubated with 1 mM pyruvate, 1 mM malate, 0.5 mM ADP and 0-50 mM KH2PO4. The mitochondrial activity was estimated by mitotracker fluorescence intensity, and intensities relative to the sample with 0 mM KH2PO4 is shown. Data represent mean ± SD from three biological replicates (n=3).
E) Effect of overexpressing Mir1 on Cox2 protein. WT (containing empty vector) and Mir1 overexpressing (Mir1OE) cells were grown in high glucose and Cox2 levels were estimated. A representative blot (out of three biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD. Also see figure S5F.
F) Effect of overexpressing Mir1 on basal OCR. The basal OCR in WT (containing empty vector) and Mir1OE in high glucose was measured from three independent experiments (n=3). Data represent mean ± SD.
Data information: *p<0.05, **p<0.01, ***p<0.001.
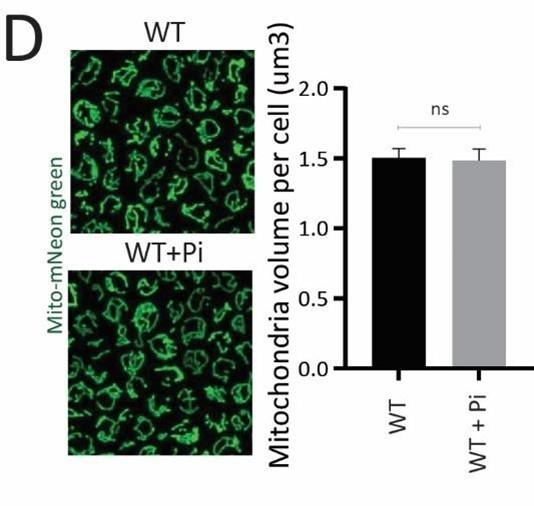
We next asked whether just adding external Pi was sufficient to increase mitochondrial activity, when cells are in high glucose. In medium supplemented with excess Pi, the internal Pi increases as seen earlier (Figure 3C). Therefore, a simplistic assumption would be that the addition of external Pi to cells in high glucose would also increase mitochondrial Pi. However, an alternate possibility presents itself whererin since glycolytic flux is already high in glucose, supplementing Pi will continue to fuel glycolysis. Indeed, this was originally obseved by Harden and Young in 1908, where adding Pi increased fermentation (46). In such a scenario, mitochondrial Pi will not increase. We estimated mitochondrial Pi in this condition where excess Pi was externally supplemented. Notably, cells grown in high Pi had decreased mitochondrial Pi (Figure 5-figure supplement 1C), without any changes in total mitochondria volume or amounts (Figure 5-figure supplement 1D). Furthermore, directly adding Pi to cells growing in high glucose also decreased basal OCR (Figure 5-figure supplement 1E), consistent with decreased mitochondrial Pi. These data indicate that in high glucose, simply supplementing Pi will not increase Pi access to the mitochondria. We therefore now asked, if we inhibit glycolytic flux and then supplement Pi, what would happen to mitochondrial activity. For this, we treated cells with 2DG, and subsequently added Pi and measured the OCR (Figure 5-figure supplement 1F). In this case, supplementing Pi increased the basal OCR (Figure 5-figure supplement 1F). Collectively, these data suggest that a combination of decreasing glycolysis and increasing Pi can together increase respiration. Next, in order to directly test mitochondrial activation based on external Pi availability, we isolated mitochondria, and estimated activity in vitro upon adding increasing Pi. Mitochondrial activity increased with increased Pi, with maximum activity observed with 25 mM Pi supplemented (Figure 5D). In a complementary experiment, we overexpressed the Mir1 transporter in wild-type cells, to increase Pi within mitochondria (Figure 5-figure supplement 1G). Mir1-OE cells have higher Cox2 levels and basal OCR (Figure 5E, 5F). Therefore, increasing Pi transport to mitochondria is sufficient to increase mitochondrial activity in high glucose.
Mitochondrial pyruvate transport is also required for mitochondrial respiration (47). We asked where Pi availability stands in a hierarchy of constraints for mitochondrial derepression, as compared to mitochondrial pyruvate transport. We measured the amounts of the Mpc3 subunit of the mitochondrial pyruvate carrier (MPC) complex (47, 48). Mpc3 protein increases in ubp3Δ (Figure 5-figure supplement 1H). This also correlated with the unimpaired TCA cycle flux (Figure 2-figure supplement 2D) and the increased mitochondrial activity. Interestingly, in ubp3Δ grown in low Pi, Mpc3 further increased (Figure 5-figure supplement 1H), but as shown earlier this condition cannot increase OCR or mitochondrial activity (Figure 4F, Figure 4-figure supplement 1F). Basal Mpc3 levels decrease in mir1Δ, but upon shifting to 0.1% glucose, Mpc3 increases in both WT and mir1Δ, with higher levels in mir1Δ (Figure 5-figure supplement 1I). Therefore, even where Mpc3 is high (ubp3Δ in low Pi, and mir1Δ in low glucose), mitochondrial activity remains low if Pi is restricted (Figure 4F, 4H). There was also no decrease in basal OCR in mpc3Δ in high glucose, and the basal OCR increased to the same level as of WT after shifting to 0.1% glucose (Figure 5-figure supplement 1J). Since Mpc3 changes with mitochondrial Pi availability (Figure S5G, S5H), we also measured Mpc3 in the Mir1OE. No further changes in Mpc3 were observed in Mir1OE (Figure 5-figure supplement 1K), indicating that increasing mitochondrial Pi alone need not increase Mpc3. Overall, although Mpc3 levels corelate with decreased glycolysis (ubp3Δ - Figure 5-figure supplement 1H, ubp3Δ in low Pi- Figure 5-figure supplement 1H, low glucose - Figure 5-figure supplement 1I), increased Mpc3 alone cannot increase mitochondrial activity and respiration in the absence of adequate mitochondrial Pi.
Collectively, mitochondrial Pi availability constrains glucose-mediated mitochondrial repression. Increasing available pools of Pi to enter the mitochondria is sufficient to induce mitochondrial activity.
Repression of mitochondrial respiration via Pi budgeting is conserved in Ubp3 mutants across diverse yeast genetic backgrounds
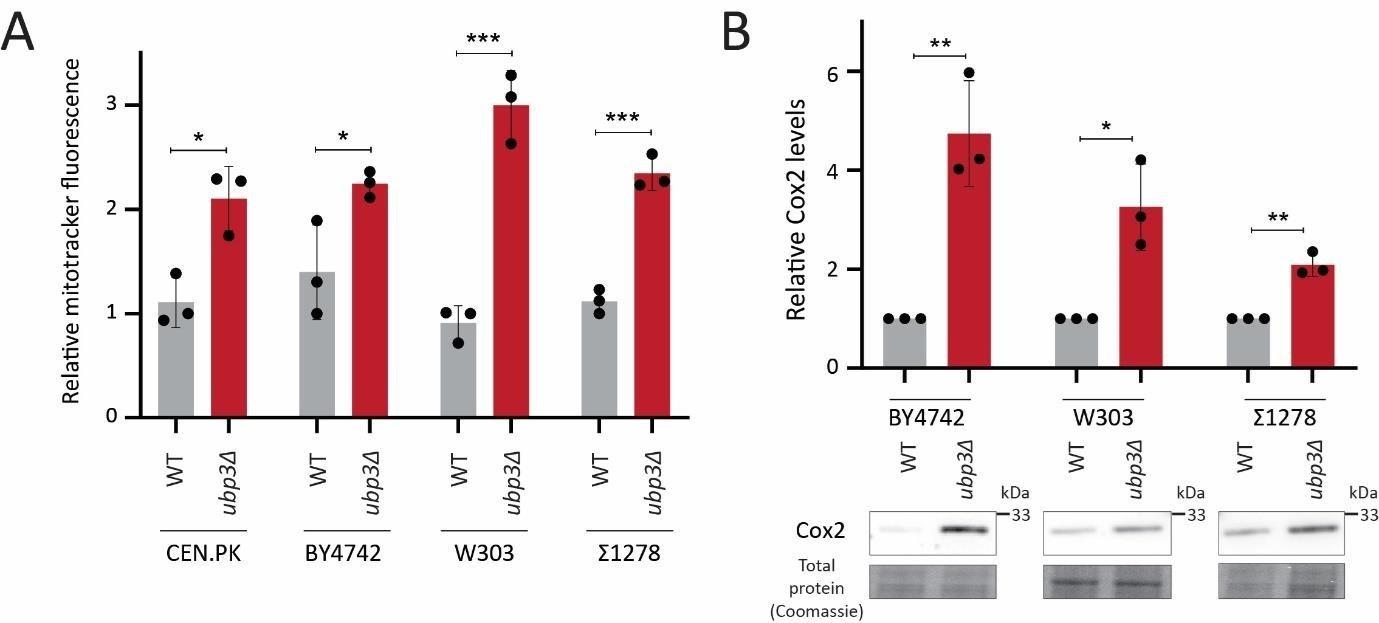
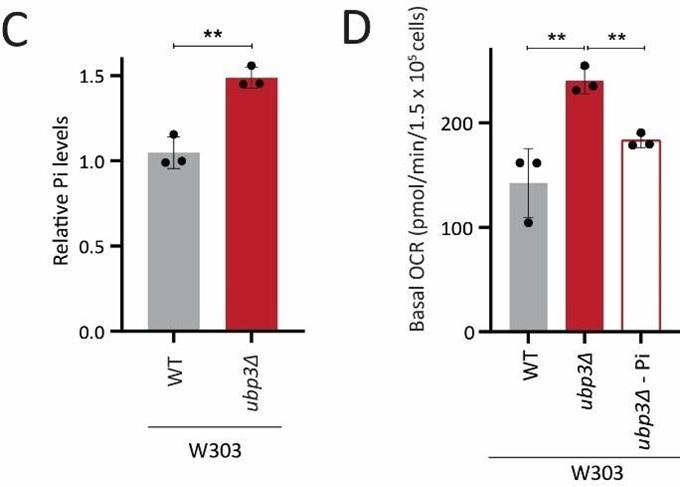
So far, we have identified a role for intracellular Pi budgeting as a constraint for mitochondrial activity under high glucose. These were all carried out using a robust, prototrophic yeast strain from a CEN.PK background. S. cerevisiae however, while Crabtree positive, have tremendous genetic diversity (49). We therefore asked if this mitochondrial repression through Pi budgeting (mediated by Ubp3 function) is conserved across other strains of S. cerevsiae as well. To test this, we generated Ubp3 deletion mutants in different genetic backgrounds of S. cerevsiae, including BY4742, W303 and Σ1278. In all these strain backgrounds, we observed a significant increase in mitotracker fluorescne intensity and Cox2 protein levels in ubp3Δ (Figure 6A, B). This suggests the role of Ubp3 as a regulator of mitochondrial repression, independent of the genetic background of the yeast (S. cerevsiae) strain. To further assess if loss of Ubp3 shows a concurrent increase in Pi levels, we measured the total Pi levels in WT and ubp3Δ in a W303 strain background. We observed a significant increase in total Pi levels in ubp3Δ cells in this strain (Figure 6C), similar to what we observed in the CEN.PK strain (Figure 3C). Finally, to test if the altered Pi budgeting regulates the mitochondrial activity in these cells, we measured the basal OCR in WT, ubp3Δ and ubp3Δ in low (1 mM) Pi medium, in the W303 strain background. Consistent with the increase in mitotracker fluorescence intensity and Cox2 protein levels, we observed a significant increase in the basal OCR in ubp3Δ cells (Figure 6D). This increase was not observed in ubp3Δ in a low Pi medium (Figure 6D). This suggests that the role of altered Pi budgeting in regulating mitochondrial respiration is conserved in other genetic backgrounds of S. cerevisiae.

Repression of mitochondrial respiration via Pi budgeting is conserved in Ubp3 mutants across diverse yeast genetic backgrounds
A) Effect of loss of Ubp3 on mitochondrial membrane potential in different yeast strains. WT and ubp3Δ cells (in CEN.PK, BY4742, W303 and Σ1278 strains of S. cerevisiae) were grown in high glucose and relative mitochondrial membrane potential was measured. Data represent mean ± SD from three biological replicates (n=3).
B) Effect of loss of Ubp3 on ETC complex IV subunit Cox2. WT and ubp3Δ (in BY4742, W303 and Σ1278 strains of S. cerevisiae) were grown in high glucose, and Cox2 was measured. A representative blot (out of 3 biological replicates, n=3) and their quantifications are shown. Data represent mean ± SD.
C) Intracellular Pi levels in WT and ubp3Δ in W303 strain background. WT and ubp3Δ (in W303 strain background) were grown in high glucose and the total free phosphate (Pi) levels were estimated. Data represent mean ± SD from three biological replicates (n=3).
D) Effect of low Pi on the basal OCR in WT and ubp3Δ cells in W303 strain background. WT cells were grown in high glucose and ubp3Δ were grown in high glucose and low Pi, and basal OCR was measured. Data represent mean ± SD (n=3).
E) Requirement of mitochondrial Pi transport for growth after 2DG treatment. Shown are serial dilution growth assays in high glucose in the presence and absence of 0.1% 2DG, using WT and mir1Δ cells. The results after 40hrs incubation/30°C are shown.
F) A model illustrating how mitochondrial Pi availability controls mitochondrial activity. In high glucose, the decreased Pi due to high Pi consumption in glycolysis, along with the glucose-mediated repression of mitochondrial Pi transporters, decreases mitochondrial Pi availability. This reduces mitochondrial activity. In low glucose, increased mitochondrial Pi transporters and lower glycolytic flux increases mitochondrial Pi, leading to enhanced mitochondrial activity. In ubp3Δ cells in high glucose, high trehalose synthesis and lower glycolytic flux results in an increase in Pi. This increases mitochondrial Pi availability and thereby the mitochondrial activity.
Data information: *p<0.05, **p<0.01, ***p<0.001.
Finally, we asked how important mitochondrial Pi transport was for growth, under glycolytic inhibition. Consistent with the requirement for mitochondrial Pi transport to increase mitochondrial respiration upon glycolytic inhibition (Figure 5B), WT cells only exhibit a slightly decreased growth in the presence of 2DG (Figure 6E). In contrast, mir1Δ show a severe growth defect upon 2DG treatment (Figure 6E), revealing a synergetic effect of combining 2DG with inhibiting mitochondrial Pi transport. Therefore, the combined inhibition of glycolysis and mitochondrial Pi transport restricts the growth of glycolytic cells.
Collectively, our data suggests a conserved role for intracellular Pi bugeting in regulating mitochondrial repression in high glucose and the role of mitochondrial Pi transport in regulating adaptation for growth under glycolytic inhibition.
Discussion
In this study, we highlight a role for Pi budgeting between cytosolic glycolysis and mitochondrial processes (which compete for Pi) in constraining mitochondrial repression (Figure 6F). Ubp3 controls this process (Figure 1), by maintaining the amounts of the glycolytic enzymes Pfk1 and GAPDH (Tdh2 and Tdh3) and thereby allows high glycolytic flux. At high fermentation rates, glycolytic enzymes levels at maximal activity maintain high glycolytic rates (effectively following zero-order kinetics), and therefore, changes in the enzyme levels will have a direct, proportionate effect on flux (50). The loss of Ubp3 decreases glycolytic flux, resulting in a systems-level, mass-action based rewiring of glucose metabolism where more G6P is routed towards trehalose synthesis and PPP (Figure 2, Figure 6F). Indeed, inhibiting just phosphofructokinase can reroute glucose flux from glycolysis to PPP (51–53), and our study now permits contextualized interpretations of these results. Such reallocations of glucose flux will collectively increase overall Pi, coming from the combined effect of increased Pi release from trehalose synthesis, and decreased Pi consumption via reduced GAPDH. This altered intracellular Pi economy increases Pi pools available to mitochondria, and increasing mitochondrial Pi is necessary and sufficient to increase respiration in high glucose (Figure 4, Figure 5, Figure 6). Mitochondrial Pi transport maintains mitochondrial activity in high glucose, and increases mitochondrial activity in low glucose (Figure 4, Figure 5). Finally, the mitochondrial Pi transporter Mir1 itself decreases in high glucose (Figure 5). Therefore, this glucose-dependent repression of Mir1 also restricts mitochondrial Pi availability (and thereby activity) in high glucose.
Traditionally, loss-of-function mutants of metabolic enzymes are used to understand metabolic state regulation. This approach negates nuanced investigations, since metabolic enzymes are often essential for viability. Furthermore, metabolic pathways have multiple, contextually regulated nodes, through which cells maintain their metabolic state. Therefore, alternate approaches to identify global regulators of metabolic states (as opposed to single enzymes), might uncover ways via which multiple nodes are simultaneously tuned, and can reveal unanticipated systems-level principles of metabolic state rewiring. In this study of glucose-mediated mitochondrial repression, the loss of the DUB Ubp3 decreases glycolytic flux by reducing the enzymes at two critical nodes in the pathway - Pfk1 and GAPDH. Unlike loss of function mutants, a reduction in amounts will only rewire metabolic flux. By ‘hitting’ multiple steps in glycolysis simultaneously, ubp3Δ have decreased Pi consumption, as well as increased Pi release. Such a cumulative phenomenon reveals more than inhibiting only GAPDH, where increased Pi comes only from reduced Pi consumption, and not from increased trehalose biosynthesis. Our serendipitous identification of a regulator which regulates multiple steps in glucose metabolism to change the metabolic environment, now suggests a general basis of mitochondrial regulation that would have otherwise remained hidden. Separately, finding the substrates of Ubp3 and whether Ubp3 directly regulates glycolytic enzymes are exciting future research questions requiring concurrent innovations in accessible chemical-biological approaches to study DUBs.
Because phosphates are ubiquitous, it is challenging to identify hierarchies of Pi-dependent processes in metabolic state regulation (35). Phosphate transfer reactions are the foundation of metabolism, driving multiple, thermodynamically unfavorable reactions (54, 55). Contextually, the laws of mass action predict that the relative rates of these reactions will regulate overall Pi balance, and contrarily the Pi allocation to Pi-dependent reactions will determine reaction rates (35, 37). Additionally, cells might control Pi allocations for different reactions via compartmentalizing Pi in organelles, to spatially restrict Pi availability (56–58). Our data collectively suggest a paradigm where the combination of factors regulates mitochondrial Pi and thereby activity. In glycolytic yeast cells growing in high glucose, mitochondrial Pi availability becomes restricted due to higher utilization of Pi in glycolysis compared to mitochondria. Consistent with this, a rapid decrease in Pi upon glucose addition has been observed (18, 19, 39). Interesting, in vitro studies with isolated mitochondria from tumor cells also find that decreasing Pi levels decreases respiration (19), which would be consistent with this scenario. Further, supplementing Pi correlates with decreased mitochondrial repression in tumors (18, 59). By increasing Pi through a systems-level rewiring of glucose metabolism (such as in ubp3Δ cells), cells can collectively increase mitochondrial access to Pi. This Pi budgeting determines mitochondrial activity. Supplementing Pi under conditions of low glycolysis (where mitochondrial Pi transport is enhanced), as well as directly supplementing Pi to isolated mitochondria, increases respiration (Figure 5, Figure 5-figure supplement 1). Therefore, in order to derepress mitochondria, a combination of increased Pi along with decreased glycolysis is required. An additional systems-level phenomenon that might regulate Pi transport to the mitochondria is the decrease in cytosolic pH upon decreased glycolysis (60, 61). The cytosolic pH in highly glycolytic cells is ∼7, and decreasing glycolysis results in cytosolic acidification (60, 61). Therefore, under conditions of decreased glycolysis (2DG treatment, deletion of Ubp3, and decreased GAPDH activity), cytosolic pH becomes acidic. Since mitochondrial Pi transport itself is dependent on the proton gradient, a low cytosolic pH would favour mitochondrial Pi transport (62). Therefore, under conditions of decreased glycolysis (2DG treatment, or loss of Ubp3, or decreased GAPDH activity), where cytosolic pH would be acidic, increasing cytosolic Pi might indirectly increase mitochondria Pi transport, thereby leading to increased respiration. Alternately, increasing mitochondrial Pi transporter amounts can achieve the same result, as seen by overexpressing Mir1 (Figure 5). A similar observation has been reported in Arabidopsis, reiterating an evolutionarily conserved role for mitochondrial Pi in controlling respiration (63). Relatedly, glycolytic inhibition can suppress cell proliferation in Warburg-positive tumors (64, 65), or inflammatory responses (66). However, these cells survive by switching to mitochondrial respiration (67, 68), requiring alternate approaches to prevent their proliferation (69). Inhibiting mitochondrial Pi transport in combination with glycolytic inhibition could restrict the proliferation of Warburg/Crabtree positive cells.
Since its discovery in the 1920s, the phenomenon of accelerated glycolysis with concurrent mitochondrial repression has been intensely researched. Yet, the biochemical constraints for glucose-mediated mitochondrial repression remains unresolved. One hypothesis suggests that the availability of glycolytic intermediates might determine the extent of mitochondrial repression. F1,6BP inhibits complex III and IV of the electron transport chain (ETC) in Crabtree positive yeast (14–17). Similarly, the ratio between G6P and F1,6BP regulates the extent of mitochondrial repression (16, 17). Although G6P/F6P accumulates in ubp3Δ (Figure 2C), this is not the case in tdh2Δtdh3Δ (GAPDH mutant) (Figure 3-figure supplement 1D), suggesting that G6P/F6P accumulation in itself is not the criterion to increase mitochondrial activity. Separately, the competition for common metabolites/co-factors between glycolysis and respiration (such as ADP, Pi or pyruvate) could drive this phenomenon (14, 18). Here we observe that Mpc3 mediated mitochondrial pyruvate transport alone cannot increase respiration. An additional consideration is the possible contribution of changes in ADP in regulating mitochondrial activity, where the use of ADP in glycolysis might limit mitochondrial ADP. Therefore, when Pi changes as a consequence of glycolysis, it coud be imagined that a change in ADP balance can coincidentally occur. However, prior studies show that even though cytosolic ADP decreases in the presence of glucose, this does not limit mitochondrial ADP uptake, or decrease respiration, due to the very high affinity of the mitochondrial ADP transporter (14, 19). These collectively reiterate the importance of Pi access and transport to mitochondria in constraining mitochondrial respiration. Indeed, this interpretation can also contextually explain observations from other model systems where mitochondrial Pi transport seems to regulate respiration (70, 71).
We parsimoniously suggest that Pi access to the mitochondria as a key constraint for mitochondrial repression under high glucose. In a hypothetical scenario, a single-step event in evolution, reducing mitochondrial Pi transporter amounts, and/or increasing glycolytic flux (to deplete cytosolic Pi), will result in whole-scale metabolic rewiring to repress mitochondria. More elaborate regulatory events can easily be imagined as subsequent adaptations to enforce mitochondrial repression. Given the central role played by Pi, something as fundamental as access to Pi will constrain mitochondrial repression. Concurrently, the rapid incorporation of Pi into faster glycolysis can give cells a competitive advantage, while also sequestering Pi in the form of usable ATP. Over the course of evolution, this could conceivably drive other regulatory mechanisms to enforce mitochondrial repression, leading to the currently observed complex regulatory networks and signaling programs observed in the Crabtree effect, and other examples of glucose-dependent mitochondrial repression.
Materials and methods
Statistics and graphing
Unless otherwise indicated, statistical significance for all indicated experiments were calculated using unpaired Student’s t-tests (GraphPad prism 9.0.1). Graphs were plotted using GraphPad prism 9.0.1
Yeast strains, media, and growth conditions
A prototrophic CEN.PK strain of Saccharomyces cerevisiae (WT) (72) was used unless mentioned otherwise. Strains are listed in appendix table S1. Gene deletions, chromosomal C-terminal tagged strains were generated by PCR mediated gene deletion/tagging (73). Mitochondria targeted Mneon strain (Mito-mNeon green) is described in (31). The cox2-62 strain is described in (32). Media compositions, growth conditions and CRISPR-Cas9 based mutagenesis are described in extended methods (SI appendix).
Mitotracker fluorescence
Mitotracker fluorescence was measured using Thermo VarioscanTM LUX multimode plate reader (579/599 excitation/emission). Detailed protocol is described in extended methods. Mitotracker fluorescence were normalized using OD600 of each sample and relative fluorescence intensity calculated.
Protein extraction and Western blotting
Total protein was precipitated, extracted using trichloroacetic acid (TCA) as described earlier (74). Blots were quantified using ImageJ software. Detailed protocol is described in extended methods.
Basal OCR measurement
The basal OCR was measured using Agilent Seahorse XFe24 analyzer. Basal OCR readings were normalized for cell number (using OD600 of samples) in each well. The detailed methods are described in extended methods.
Mitochondrial volume estimation
High-resolution 3D fluorescence experiments were performed on an inverted confocal laser scanning microscope (Carl Zeiss LSM 780 or Olympus FV3000). For each imaging field of view, sequential z-stacks were acquired for each excitation channel. 488 nm laser excitation for mNeonGreen and 561 nm laser excitation for Mitotracker CMXros dye were used respectively. Images taken were deconvolved and analyzed further in ImageJ software with custom-written routines. Mitochondria segmentation and quantification was done using the Mitochondria Analyzer plugin (75) in Imagej. For visualization, maximum intensity projection of 3D images was used.
RNA extraction and RT-qPCR
The RNA extraction was done using the hot phenol extraction method as described in (74). The isolated RNA was DNase treated, and used for cDNA synthesis. Superscript III reverse transcriptase enzyme (Invitrogen) was used for cDNA synthesis and RT-qPCR was performed using KAPA SYBR FAST qRT PCR kit (KK4602, KAPA Biosystems). Taf10 was used as a control for normalisation and the fold change in mRNA levels were calculated by 2^-ddct method.
ATP, ethanol and Pi measurements
ATP levels were measured by ATP estimation kit (Thermo Fisher A22066). Ethanol concentration in the medium was estimated using potassium dichromate based assay described in (76) with modifications. Pi was estimated using a malachite green phosphate assay kit (Cayman chemicals, 10009325). Detailed sample collection and assay protocols are described in extended methods.
Metabolite extraction and analysis by LC-MS/MS
The steady state levels and relative 13C label incorporation into metabolites were estimated by quantitative LC-MS/MS methods as described in (77). Detailed methodology is extensively described in extended methods. Peak area measurements are listed in supplement file 1.
Mitochondrial isolation
Mitochondria was isolated by immunoprecipitation as described in (78, 79) with modifications. The detailed protocol is described in extended methods (SI appendix). The eluted mitochondria were used in malachite green assay for Pi estimation, boiled with SDS-glycerol buffer for western blots or incubated with mitotracker CMXROS with mitochondria-activation buffer for mitotracker assays.
Supporting information
Data availability
The LC-MS/MS peak area for all the metabolites can be found in supplement file 1. This study includes no data deposited in external repositories.
Acknowledgements
We thank all SL lab members, Benjamin Tu, Anand Bachhawat and Vijay Jayaraman for comments and suggestions. We acknowledge the NCBS/inStem/CCAMP mass spectrometry facility for LC-MS/MS support. We thank Aayushee Khanna for help with experiments and Gaurav Singh for help with microscopy. V.V acknowledges funding support from DST-INSPIRE fellowship (IF170236) from the Department of Science and Technology (DST), Govt. of India, S.N and SA acknowledges intramural funding from the DBT-inStem PhD program. S.L acknowledges support from the DST SERB CRG grant CRG/2019/004772, a DBT-Wellcome India Alliance Senior Fellowship (IA/S/21/2/505922), and institutional support from inStem.
Supplement figure legends

DUB screen details and further characterization of Ubp3 functions
A) A list of the known/identified S. cerevisiae deubiquitinase enzymes is shown. The DUBs which were used in this study, and the mitotracker intensity of two biological replicates (n=2) relative to WT are shown.
B) WT cells grown in a respiratory, ethanol medium and mitotracker fluorescence intensity compared to glucose medium. WT cells were grown in high glucose medium (2% glucose) and ethanol medium (2% ethanol), and the mitochondrial membrane potential was measured. Data from two independent experiments (n=2) is shown. Data represent mean ± SD.
C) Representative images of WT and ubp3Δ cells treated with mitotracker red CMXROS.
D) Schematic representation of the conserved domains of Ubp3 and its mammalian ortholog Usp10. The catalytic cysteine of Usp10 and Ubp3 is highlighted.
E) The catalytically inactive mutant of Ubp3, Ubp3C469A does not have altered Ubp3 protein levels. WT and Ubp3C469A cells containing endogenous Ubp3 tagged with 3xFLAG (C terminus) were grown in high glucose and the protein levels of Ubp3 were measured. A representative blot (out of three biological replicates, n=3) and their quantification are shown. Data represent mean ± SD.
F) The total mitochondrial volume does not change in ubp3Δ cells. WT and ubp3Δ cells with mitochondria targeted mNeon green were imaged, and the total mitochondrial volume per cell (n=104 for WT, n=107 for ubp3Δ) was calculated. A representative image and quantification are shown. Data represent mean ± SD.
G) Tom70 protein amounts do not change in ubp3Δ cells. WT and ubp3Δ cells containing endogenous Tom70 tagged at the C terminus with a 3xFLAG epitope were grown in high glucose and the protein levels of Tom70 were measured by western blot. A representative blot (out of three biological replicates, n=3) and their quantification are shown. Data represent mean ± SD.
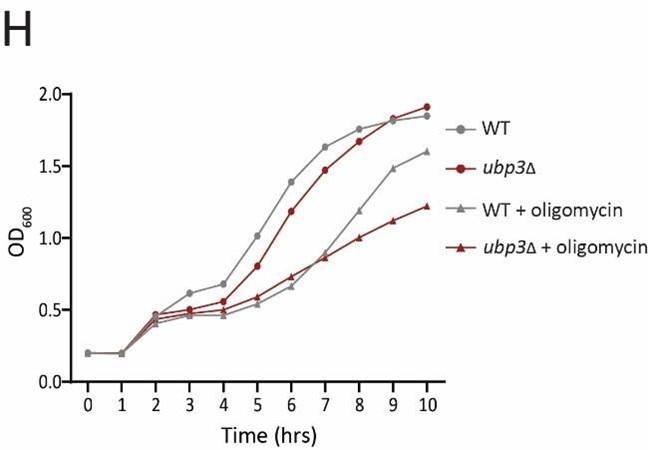
H) Dependence of ubp3Δ on mitochondrial respiration. WT and ubp3Δ cells were grown in high glucose with or without mitochondrial inhibitor oligomycin (25 µM), and the OD600 was measured at different time points. Data represent mean ± SD (n=2).
I) Dependence of ubp3Δ on functional mitochondrial respiration. Ubp3 deletions were done in respiration defective strains -Rho0, cox2-62, atp1Δ and atp10Δ, and serial dilution growth assay was done in high glucose. The results after 24 hours of incubation at 30°C are shown.

Further estimates of glycolytic enzymes and flux
A) Loss of Ubp3 does not change the protein levels of enolase isozymes Eno1 and Eno2. WT and ubp3Δ cells containing Eno1 and Eno2 C terminus endogenously tagged with a 3x FLAG epitope, were grown in high glucose and protein amounts were estimated. A representative blot (out of three biological replicates, n=3) is shown in the lower panel, quantifications shown in the upper panel. Data represent mean ± SD.
B) The decreased levels of glycolytic enzymes in ubp3Δ cells is not because of proteasomal degradation. WT and ubp3Δ were grown in high glucose and ubp3Δ cells were treated with or without MG132 (100 μM) for 30 minutes. Pfk1, Tdh2, and Tdh3 levels were measured by western blot using an anti-FLAG antibody. A representative blot (out of three biological replicates, n=3) and their quantification are shown. Data represent mean ± SD.
C) Loss of Ubp3 results in decreased transcription of PFK1, TDH2 and TDH3 genes. WT, ubp3Δ and Ubp3C469A cells were grown in high glucose and the mRNA levels of PFK1, TDH2 and TDH3 were analysed by RT-qPCR. The fold changes in mRNA levels are shown. Data represent mean ± SD.
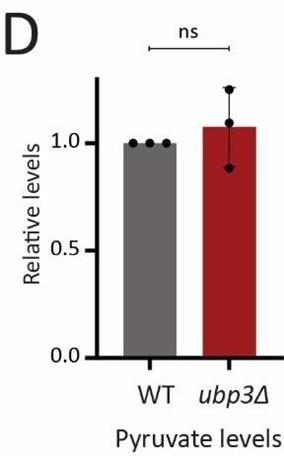
D) Steady-state pyruvate levels in WT and ubp3Δ in high glucose. Data represent mean ± SD from three biological replicates (n=3). Also see Appendix Table S3.
E) Schematic showing the experimental design for measuring 13C label incorporation into glycolytic intermediates and trehalose using a 13C6 glucose-pulse.
F) Changes in 13C label incorporation into glycolytic intermediates and trehalose with time, and linearity of label incorporation after a pulse of 1% 13C6 glucose is shown.

Estimation of steady state levels and flux of TCA cycle intermediates
A) Steady-state TCA metabolite amounts in WT and ubp3Δ in high glucose. Relative steady-state levels of TCA cycle intermediates were estimated in WT and ubp3Δ. Data represent mean ± SD from three biological replicates (n=3). Also see Appendix Table S3.
B) Schematic showing the experimental design for measuring 13C label incorporation into TCA cycle intermediates using a 13C6 glucose-pulse.
C) Changes in 13C label incorporation into TCA cycle intermediates with time, and linearity of label incorporation after a pulse of 1% 13C6 glucose is shown.
D) Relative TCA cycle flux in WT and ubp3Δ. Relative 13C-label incorporation into TCA cycle intermediates, after a pulse of 1% 13C6 glucose is shown. Data represent mean ± SD from three biological replicates (n=3). Also see Appendix Table S3.
Data information: *p<0.05

Comparisons of phosphate, ethanol and other metabolites in WT, ubp3Δ and GAPDH mutants
A) Loss of Ubp3 deubiquitinase activity and Pi levels. WT, ubp3Δ, and Ubp3C469A cells were grown in high glucose and the total free phosphate (Pi) levels were estimated. Data from two independent experiments (n=2) is shown. Data represent mean ± SD. B) Trehalose levels in WT and ubp3Δ cells in the absence of Tps2. tps2Δ and ubp3Δtps2Δ cells were grown in high glucose, and the trehalose levels were estimated using targeted LC-MS/MS. Data represent mean ± SD from three biological replicates (n=3).
C) Loss of GAPDH isozymes Tdh2 and Tdh3 and ethanol production. WT and tdh2Δtdh3Δ cells were grown in high glucose and the ethanol concentration in the medium was measured. Data are represented as mean ± SD from three biological replicates (n=3). Note: The WT shown here is same as the WT in Figure 2E, the ethanol assays in Figure 2E and Figure S3C were done together and a common WT control was used.
D) Steady state levels of glycolytic intermediates: tdh2Δtdh3Δ cells have unaltered steady state levels of G6P/F6P, increased levels of F1,6 BP and G3P and decreased levels of 3PG and PEP. WT and tdh2Δtdh3Δ cells were grown in high glucose and the steady state amounts of glycolytic intermediates were estimated using targeted LC-MS/MS. Data represent mean ± SD from three biological replicates (n=3). Also see Appendix Table S3.
E) Trehalose levels in tdh2Δtdh3Δ cells. WT and tdh2Δtdh3Δ cells were grown in high glucose and trehalose amounts were estimated using targeted LC-MS/MS. Data represent mean ± SD from three biological replicates (n=3).
Data information: *p<0.05, **p<0.01.

Mitochondrial Pi estimation characterizations and correlations of mitochondrial activity with Pi availability
A) Lack of vacuolar contamination in mitochondria isolated by immunoprecipitation. Mitochondria were isolated from WT cells grown in high glucose and the protein levels of Tom20, Cox2, Idh1 and Vph1 and were measured in cell lysate and immunoprecipitated mitochondria by western blot.
B) Idh1 levels in ubp3Δ cells and WT cells grown in high Pi. WT cells were grown in high glucose, or high glucose and high Pi medium (10mM Pi) and ubp3Δ cells were grown in high glucose, and the Idh1 protein levels were estimated by western blot. A representative blot (out of three biological replicates, n=3) is shown in the lower panel. Quantification is shown in the upper panel. Data represent mean ± SD.
C) Mitochondrial membrane potential in tdh2Δtdh3Δ cells. WT, ubp3Δ, and tdh2Δtdh3Δ cells were grown in high glucose and the mitochondrial membrane potential was measured. Data represent mean ± SD from three biological replicates (n=3).
D) Pi levels in ubp3Δ cells grown in low Pi (1mM Pi) vs WT cells. WT cells were grown in high glucose and ubp3Δ cells were grown in high glucose or high glucose-low Pi, and the total Pi levels were estimated. Data represent mean ± SD from three biological replicates (n=3).
E) Ethanol production in ubp3Δ cells grown in a low Pi medium. WT cells were grown in high glucose and ubp3Δ cells were grown in high glucose or high glucose-low Pi, and the ethanol concentration in the medium was measured. Data represent mean ± SD from three biological replicates (n=3).
F) Pi amounts and mitochondrial membrane potential in WT, ubp3Δ, and tdh2Δtdh3Δ cells. The cells were grown in high glucose or high glucose-low Pi, and the mitochondrial membrane potential was measured. Data from two independent experiments (n=2) is shown. Data represent mean ± SD.
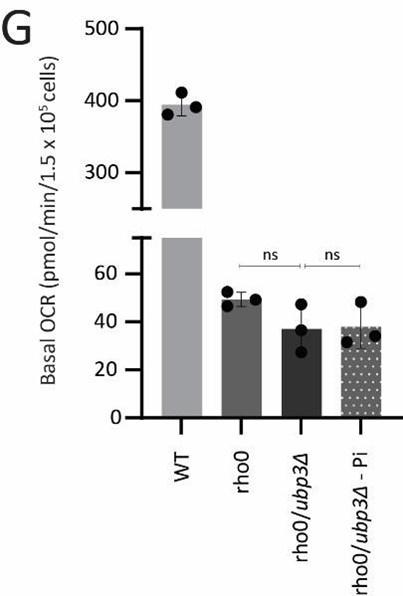
G) The Pi mediated change in basal OCR in ubp3Δ is dependent on mitochondrial respiration. WT and Rho0 cells were grown in high glucose, Rho0/ubp3Δ were grown in high glucose and low Pi, and basal OCR was measured from three independent experiments (n=3). Data represent mean ± SD.
H) The protein levels of mitochondrial Pi transporters Mir1 and Pic2 in ubp3Δ cells. WT and ubp3Δ cells containing endogenously tagged Mir1 and Pic2 at their C terminus with a 6x HA epitope tag were grown in high glucose and Mir1 and Pic2 proteins were measured by western blot. A representative blot (out of three biological replicates, n=3) is shown in the lower panel. Quantification is shown in the upper panel. Data represent mean ± SD.
Data information: *p<0.05, **p<0.01, ***p<0.001.

Mitochondrial phosphate and pyruvate transport relationships with mitochondrial activity
A) Requirement of Mir1 for switching to increased mitochondrial activity upon 2-deoxyglucose (2-DG) treatment. WT and mir1Δ cells were grown in high glucose and treated with 0.1% and 0.25% 2-DG for one hour. The mitochondrial membrane potential was measured from two independent experiments (n=2). Data represent mean ± SD.
B) Mir1 protein levels and glucose repression. Cells containing Mir1 endogenously tagged at the C terminus with a 6x HA epitope tag were grown in high glucose or a respiratory medium (2% ethanol). Mir1 protein was estimated by western blot. A representative blot (out of three biological replicates, n=3) is shown in the lower panel. Quantification is shown in the upper panel. Data represent mean ± SD.
C) Effect of supplementing Pi in the medium on mitochondrial Pi. WT cells were grown in high glucose, and high glucose, high Pi medium (10 mM Pi), mitochondria was isolated and the mitochondrial Pi levels were estimated. The mitochondrial Pi levels normalised to Idh1 protein levels is shown. Data represent mean ± SD from three biological replicates (n=3). Also see supplementary figure S4B.
D) Effect of supplementing Pi on the total mitochondrial volume. WT cells with mitochondria targeted mNeon green were grown in high glucose, and high glucose, high Pi medium (10 mM Pi), cells were imaged and the total mitochondrial volume per cell was calculated.
E) Increasing Pi concentration in a high glucose medium results in a decrease in basal OCR. WT cells were grown in high glucose medium (YPD-Pi medium), supplemented with 5 mM Pi and at OD600∼0.6, the medium was supplemented with Pi (10 mM and 25 mM final concentrations). The basal OCR was measured one hour after Pi supplementation. Data represent mean ± SD from three biological replicates (n=3).
E) Effect of increasing the Pi concentration in a high glucose medium, in the presence of 2DG, on basal OCR. WT cells were grown in high glucose medium (YPD-Pi medium), supplemented with 5 mM Pi and at OD600∼0.6, the medium was supplemented with Pi (25 mM final concentration) and 2DG (0.25%) for one hour and the basal OCR was measured. Data represent mean ± SD from three biological replicates (n=3).
F) Overexpression of Mir1. C terminal 6x HA epitope-tagged Mir1 was expressed under G6PD promoter in cells with Mir1 endogenously tagged at the C terminus with a 6x-HA epitope tag. WT cells (carrying an empty vector, and expressing Mir1-HA under the endogenous Mir1 promoter) and Mir1-OE cells were grown in high glucose. The protein levels of Mir1 were estimated by western blot, and clones with an ∼2-fold increase in Mir1 were selected. A representative blot (out of three biological replicates, n=3) is shown in the lower panel. Quantification is shown in the upper panel. Data represent mean ± SD.
G) Mpc3 protein levels in ubp3Δ cells. WT and ubp3Δ cells containing endogenously tagged Mpc3 at the C terminus with a 3x FLAG epitope tag, were cultured in high glucose (2% glucose) and high glucose-low Pi (2% glucose, 1 mM Pi). The Mpc3 protein levels were measured by western blot. A representative blot (out of three biological replicates, n=3) is shown in the lower panel, quantifications shown in the upper panel. Data represent mean ± SD.
H) Mpc3 protein amounts upon shifting WT and mir1Δ cells to a low-glucose medium. WT and mir1Δ cells containing Mpc3-FLAG, were cultured in high glucose (2% glucose) and shifted to low (0.1%) glucose for 1 hour. Mpc3 protein was measured by western blot. A representative blot (out of three biological replicates, n=3) is shown in the lower panel, quantifications shown in the upper panel. Data represent mean ± SD.
I) Requirement of Mpc3 for the increase in basal OCR upon shifting to low glucose. WT and mpc3Δ cells were cultured in high glucose (2% glucose) and shifted to low (0.1%) glucose for 1 hour. The basal OCR from two independent experiments (n=2), normalized to the OD600 is shown. Data represent mean ± SD.
J) Mpc3 levels and Mir1 overexpression. WT cells and Mir1-OE cells containing endogenously tagged Mpc3 ate the C terminus with a 3x FLAG tag were grown in high glucose. The protein levels of Mpc3 were estimated by western blot. A representative blot (out of three biological replicates, n=3) is shown in the panel. Quantification is shown in the upper panel. Data represent mean ± SD. Data information: *p<0.05, **p<0.01, ***p<0.001.
References
- 1.Metabolic regulation of cell growth and proliferationNat. Rev. Mol. Cell Biol 20:436–450Google Scholar
- 2.Driving the Cell Cycle Through MetabolismAnnu. Rev. Cell Dev. Biol 28:59–87Google Scholar
- 3.Lehninger principles of biochemistryGoogle Scholar
- 4.The metabolism of carcinoma cells 1J. Cancer Res 9:148–163Google Scholar
- 5.Understanding the Warburg Effect: The Metabolic Requirements of Cell ProliferationScience (80-.) 324:1029–1033Google Scholar
- 6.Observations on the carbohydrate metabolism of tumoursBiochem. J 23:536–545Google Scholar
- 7.The Crabtree effect: a regulatory system in yeastJ. Gen. Microbiol 44:149–156Google Scholar
- 8.The cancer metabolic reprogramming and immune responseMol. Cancer 20:1–21Google Scholar
- 9.The immunologic Warburg effect: Evidence and therapeutic opportunities in autoimmunityWiley Interdiscip. Rev. Syst. Biol. Med 12:1–17Google Scholar
- 10.Cancer stem cell theory and the warburg effect, two sides of the same Coin?Int. J. Mol. Sci 15:8893–8930Google Scholar
- 11.The emerging facets of non-cancerous Warburg effectFront. Endocrinol. (Lausanne) 8:1–7Google Scholar
- 12.Energy Metabolism Regulates Stem Cell PluripotencyFront. Cell Dev. Biol 8:1–16Google Scholar
- 13.Biochemical evolution : the pursuit of perfectionGarl. Sci Google Scholar
- 14.The Warburg and Crabtree effects: On the origin of cancer cell energy metabolism and of yeast glucose repressionBiochim. Biophys. Acta - Bioenerg 1807:568–576Google Scholar
- 15.The Crabtree and Warburg effects: Do metabolite-induced regulations participate in their induction?Biochim. Biophys. Acta - Bioenerg 1857:1139–1146Google Scholar
- 16.Mitochondrial Oxidative Phosphorylation Is Regulated by Fructose 1,6-Bisphosphate: A POSSIBLE ROLE IN CRABTREE EFFECT INDUCTION?*. J. Biol. Chem 283:26948–26955Google Scholar
- 17.The role of glycolysis-derived hexose phosphates in the induction of the Crabtree effectJ. Biol. Chem 293:12843–12854Google Scholar
- 18.Phosphate mediation of the Crabtree and Pasteur effectsScience (80-.) 178:127–133Google Scholar
- 19.Multisite control of the Crabtree effect in ascites hepatoma cellsEur. J. Biochem 268:2512–2519Google Scholar
- 20.Post-translational modifications on yeast carbon metabolism: Regulatory mechanisms beyond transcriptional controlBiochim. Biophys. Acta - Gen. Subj 1850:620–627Google Scholar
- 21.Post-translational modifications and the Warburg effectOncogene 33:4279–4285Google Scholar
- 22.Nutritional control of growth and development in yeastGenetics 192:73–105Google Scholar
- 23.THE UBIQUITIN SYSTEMAnnu. Rev. Biochem 67:425–79Google Scholar
- 24.The Ubiquitin CodeAnnu. Rev. Biochem 81:203–229Google Scholar
- 25.Ubiquitin: Structures, functions, mechanismsBiochim. Biophys. Acta - Mol. Cell Res 1695:55–72Google Scholar
- 26.Enzymic analysis of the crabtree effect in glucose-limited chemostat cultures of Saccharomyces cerevisiaeAppl. Environ. Microbiol 55:468–77Google Scholar
- 27.Cdc48 and Ufd3, new partners of the ubiquitin protease Ubp3, are required for ribophagyEMBO Rep 11:548–554Google Scholar
- 28.Multiplexed, Proteome-Wide Protein Expression Profiling: Yeast Deubiquitylating Enzyme Knockout StrainsJ. Proteome Res 14:5306–5317Google Scholar
- 29.The Catalytic Activity of the Ubp3 Deubiquitinating Protease Is Required for Efficient Stress Granule Assembly in Saccharomyces cerevisiaeMol. Cell. Biol 36Google Scholar
- 30.Assembly of mitochondrial cytochrome c-oxidase, a complicated and highly regulated cellular processAm. J. Physiol. - Cell Physiol 291:1129–1147Google Scholar
- 31.DarT-mediated mtDNA damage induces dynamic reorganization and selective segregation of mitochondriaJ. Cell Biol 221Google Scholar
- 32.Mitochondrial Translation of Saccharomyces cerevisiae COX2 mRNA Is Controlled by the Nucleotide Sequence Specifying the Pre-Cox2p Leader PeptideMol. Cell. Biol 21:2359–2372Google Scholar
- 33.Quantitative determinants of aerobic glycolysis identify flux through the enzyme GAPDH as a limiting stepElife 3:1–18Google Scholar
- 34.A tRNA modification balances carbon and nitrogen metabolism by regulating phosphate homeostasisElife 8:1–33Google Scholar
- 35.Cycles, sources, and sinks: Conceptualizing how phosphate balance modulates carbon flux using yeast metabolic networksElife 10:1–14Google Scholar
- 36.The importance of inorganic phosphate in regulation of energy metabolism of Streptococcus lactisJ. Biol. Chem 256:1861–1866Google Scholar
- 37.Lost in transition: Start-up of glycolysis yields subpopulations of nongrowing cellsScience (80-.) 343Google Scholar
- 38.Metabolic Requirement for Inorganic Phosphate by the Rabbit Proximal Tubule: EVIDENCE FOR A CRABTREE EFFECT Metabolic Requirement for Inorganic Phosphate by the Rabbit Proximal Tubule EVIDENCE FOR A CRABTREE EFFECTJ Clin Invest 70:53Google Scholar
- 39.Evidence for trehalose-6-phosphate-dependent and-independent mechanisms in the control of sugar influx into yeast glycolysisMol. Microbiol 20:981–991Google Scholar
- 40.Why nature chose phosphate to modify proteinsPhilos. Trans. R. Soc. B Biol. Sci 367:2513–2516Google Scholar
- 41.Regulation of amino acid, nucleotide, and phosphate metabolism in Saccharomyces cerevisiaeGenetics 190:885–929Google Scholar
- 42.Phosphate Homeostasis − A Vital Metabolic Equilibrium Maintained Through the INPHORS Signaling PathwayFront. Microbiol 11:1367Google Scholar
- 43.New aspects on phosphate sensing and signalling in Saccharomyces cerevisiaeFEMS Yeast Res 6:171–176Google Scholar
- 44.Isolation and characterization of the gene for a yeast mitochondrial import receptorNat. 1990 347:488–491Google Scholar
- 45.Yeast mitochondria lacking the phosphate carrier/p32 are blocked in phosphate transport but can import preproteins after regeneration of a membrane potentialMol. Cell. Biol 16:6524–6531Google Scholar
- 46.The alcoholic Ferment of Yeast-juice.Part III.-The Function of Phosphates in the Fermentation o f Glucose hy Yeast-juiceProc. R. Soc. London. Ser. B 80:299–311Google Scholar
- 47.Differential Regulation of Mitochondrial Pyruvate Carrier Genes Modulates Respiratory Capacity and Stress Tolerance in YeastPLoS One 8:e79405Google Scholar
- 48.Regulation of mitochondrial pyruvate uptake by alternative pyruvate carrier complexesEMBO J 34:911–924Google Scholar
- 49.Genome evolution across 1,011 Saccharomyces cerevisiae isolatesNature 556:339–344Google Scholar
- 50.An excess of glycolytic enzymes under glucose-limited conditions may enable Saccharomyces cerevisiae to adapt to nutrient availabilityFEBS Lett https://doi.org/10.1002/1873-3468.14484Google Scholar
- 51.PFK1 Glycosylation Is a Key Regulator of Cancer Cell Growth and Central Metabolic PathwaysScience 337Google Scholar
- 52.Rewiring of embryonic glucose metabolism via suppression of PFK-1 and aldolase during mouse chorioallantoic branchingDev 144:63–73Google Scholar
- 53.Examining Escherichia coli glycolytic pathways, catabolite repression, and metabolite channeling using Δpfk mutantsBiotechnol. Biofuels 9:1–13Google Scholar
- 54.Why Nature Chose PhosphatesScience (80-.) 235:1173–1178Google Scholar
- 55.Why nature really chose phosphateQ. Rev. Biophys 46:1–132Google Scholar
- 56.Phosphate Transport in Yeast Vacuoles *J. Biol. Chem 272:20408–20413Google Scholar
- 57.Lessons from protozoans: Phosphate sensing and polyphosphate storage in fungiPLOS Pathog 18:e1010298Google Scholar
- 58.Depletion of mitochondrial inorganic polyphosphate (polyP) in mammalian cells causes metabolic shift from oxidative phosphorylation to glycolysisBiochem. J 478:1631–1646Google Scholar
- 59.The Inhibition of Respiration by Glucose, Fructose, and Mannose in the Ehrlich Mouse Ascites TumorCancer Res 16:364–368Google Scholar
- 60.Intracellular pH is a tightly controlled signal in yeastBiochim. Biophys. Acta - Gen. Subj 1810:933–944Google Scholar
- 61.Cytosolic pH is a second messenger for glucose and regulates the PKA pathway through V-ATPaseEMBO J 29:2515–2526Google Scholar
- 62.Redundancy in the function of mitochondrial phosphate transport in Saccharomyces cerevisiae and Arabidopsis thalianaMol. Microbiol 51:307–317Google Scholar
- 63.Overexpression of Mitochondrial Phosphate Transporter 3 Severely Hampers Plant Development through Regulating Mitochondrial Function in ArabidopsisPLoS One 10:e0129717Google Scholar
- 64.HuangGlycolysis inhibition for anticancer treatment :4633–4646Google Scholar
- 65.2-Deoxy-D-Glucose inhibits aggressive triple-negative breast cancer cells by targeting glycolysis and the cancer stem cell phenotypeSci. Rep 9:1–11Google Scholar
- 66.Glycolysis – a key player in the inflammatory responseFEBS J 287:3350–3369Google Scholar
- 67.Glycolytic suppression dramatically changes the intracellular metabolic profile of multiple cancer cell lines in a mitochondrial metabolism-dependent mannerSci. Rep 9:1–15Google Scholar
- 68.Tumor Cells Switch to Mitochondrial Oxidative Phosphorylation under Radiation via mTOR-Mediated Hexokinase II Inhibition - A Warburg-Reversing EffectPLoS One 10:e0121046Google Scholar
- 69.Mitochondria-targeted drugs synergize with 2-deoxyglucose to trigger breast cancer cell deathCancer Res 72:2634–2644Google Scholar
- 70.Regulation of mitochondrial respiration by inorganic phosphate; comparing permeabilized muscle fibers and isolated mitochondria prepared from type-1 and type-2 rat skeletal muscleEur. J. Appl. Physiol 105:279–287Google Scholar
- 71.The mitochondrial phosphate carrier: Role in oxidative metabolism, calcium handling and mitochondrial diseaseBiochem. Biophys. Res. Commun 464:369–375Google Scholar
- 72.An interlaboratory comparison of physiological and genetic properties of four Saccharomyces cerevisiae strainsEnzyme Microb. Technol 26:706–714Google Scholar
- 73.Additional modules for versatile and economical PCR-based gene deletion and modification in Saccharomyces cerevisiaeYeast 14:953–961Google Scholar
- 74.The E3 ubiquitin ligase Pib1 regulates effective gluconeogenic shutdown upon glucose availabilityJ. Biol. Chem 294:17209–17223Google Scholar
- 75.INNOVATIVE METHODOLOGY A pipeline for multidimensional confocal analysis of mitochondrial morphology, function, and dynamics in pancreatic  -cellsAm. J. Physiol. - Endocrinol. Metab 318:87–101Google Scholar
- 76.A Rapid Spectrophotometric Method for Quantitative Determination of Ethanol in Fermentation ProductsOrient. J. Chem 35:744–750Google Scholar
- 77.A versatile LC-MS / MS approach for comprehensive, quantitative analysis of central metabolic pathways [version 1 ; peer review : 2 approved]:1–15Google Scholar
- 78.Isolation of mitochondria from Saccharomyces cerevisiae using magnetic bead affinity purificationPLoS One 13:1–15Google Scholar
- 79.Rapid immunopurification of mitochondria for metabolite profiling and absolute quantification of matrix metabolitesNat. Protoc 12:2215Google Scholar
Article and author information
Author information
Version history
- Sent for peer review:
- Preprint posted:
- Reviewed Preprint version 1:
- Reviewed Preprint version 2:
- Reviewed Preprint version 3:
- Version of Record published:
Cite all versions
You can cite all versions using the DOI https://doi.org/10.7554/eLife.90293. This DOI represents all versions, and will always resolve to the latest one.
Copyright
© 2023, Vengayil et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.