Author response:
The following is the authors’ response to the previous reviews
Public Reviews:
Reviewer #2 (Public Review):
Strengths
(1) The definition of highly variable yet highly reproducible sulci such as the slocs-v feeds the community with new anatomo-functional landmarks (which is emphasized by the provision of a probability map in supp. mat., which in my opinion should be proposed in the main body).
We agree with Reviewer 2 that there is merit to including the probability maps as a main text Figure rather than Supplementary Figure. We have now added it to the main text.
Weaknesses
(1) While the identification of the sulci has been done thoroughly with expert validation, the sulci have not been labeled in a way that enables the demonstration of the reproducibility of the labeling.
Our group was unable to use an approach amenable to calculating inter-rater agreements to expedite the process of defining thousands of sulci at the individual level in multiple regions as this was our first study comprehensively documenting the sulcal organization of this region. Nevertheless, our method followed a rigorous, three-tiered procedure to ensure accurate sulcal definitions were identified in all participants. In the case of this study, authors YT and TG first defined sulci. These sulci were then checked by a trained expert (EHW). Finally, sulcal definitions were finalized by the senior author, an expert neuroanatomist (KSW). We emphasize that this process has produced reproducible anatomical results when charting other regions such as posteromedial cortex (Willbrand et al., 2023 Science Advances; Willbrand et al., 2023 Communications Biology; Maboudian et al., 2024 The Journal of Neuroscience; Ramos Benitez et al., 2024 Neuropsychologia), ventral temporal cortex (Miller et al., 2020 Scientific Reports; Parker et al., 2023 Brain Structure and Function), and lateral prefrontal cortex (Miller et al., 2021 The Journal of Neuroscience; Voorhies et al., 2021 Nature Communications; Yao et al., 2022 Cerebral Cortex; Willbrand et al., 2022 Brain Structure and Function; Willbrand et al., 2023 The Journal of Neuroscience; Willbrand et al., 2024 Brain Structure and Function) across age groups, species, and clinical populations. For the present study, by the time the final tier of our method was reached, we emphasize that a very small percentage (~2%) of sulcal definitions were actually modified. We will include an exact percentage in future publications in LPC/LOPJ.
Our Methods have been edited to describe these features (Pages 21-22):
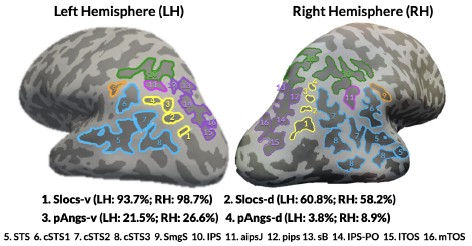
“As this is the first time the sulcal expanse of LPC/LOPJ was comprehensively charted with a focus on pTS, the location of each sulcus was confirmed through a three-tiered procedure for each participant in each hemisphere. First, trained independent raters (Y.T. and T.G.) identified sulci. Second, these definitions were checked by a trained expert (E.H.W.). Third, these labels were finalized by a neuroanatomist (K.S.W.). We emphasize that this procedure has produced reproducible results in our prior work across the cortex (Miller et al. 2021; Voorhies et al. 2021; Yao et al. 2022; Willbrand et al. 2023; Willbrand et al. 2022; Willbrand et al. 2024; Parker et al. 2023; Miller et al. 2020; Willbrand et al. 2022; Willbrand et al. 2023; Maboudian et al. 2024; Ramos Benitez et al. 2024). All LPC sulci were then manually defined and saved as .label files in FreeSurfer using tksurfer tools, from which morphological and anatomical features were extracted. We defined LPC/LPOJ sulci for each participant based on the most recent schematics of sulcal patterning by Petrides (2019) as well as pial, inflated, and smoothed white matter (smoothwm) FreeSurfer cortical surface reconstructions of each individual. In some cases, the precise start or end point of a sulcus can be difficult to determine on a surface (Borne et al., 2020); however, examining consensus across multiple surfaces allowed us to clearly determine each sulcal boundary in each individual. For four example hemispheres with these 13-17 sulci identified, see Fig. 1a (Supplementary Fig. 5 for all hemispheres). The specific criteria to identify the slocs and pAngs are outlined in Fig. 1b.”
Reviewer #3 (Public Review):
Weaknesses
(1) The numbers of subjects are inherently limited both in number as well as in being typically developing young adults.
First, although the sample size of the present study is small in number in comparison to large N, group-level neuroimaging analyses, it is comparable to precision neuroimaging studies examining sulcal features in individual participants (for example, Cachia et al., 2021 Frontiers in Neuroanatomy; Garrison et al., 2015 Nature Communications; Lopez-Persem et al., 2019 The Journal of Neuroscience; Miller et al., 2021 The Journal of Neuroscience; Roell et al., 2021 Developmental Cognitive Neuroscience; Voorhies et al., 2021 Nature Communications; Weiner, 2019 The Anatomical Record; Willbrand, et al., 2022 Science Advances; Willbrand, et al., 2022 Brain Structure & Function; Yao et al., 2022 Cerebral Cortex). We discuss this point in detail in the Limitations subsection of the Discussion (Page 17):
“This manual method is also arduous and time-consuming, which, on the one hand, limits the sample size in terms of number of participants, while on the other, results in thousands of precisely defined sulci. This push-pull relationship reflects a broader conversation in the human brain mapping and cognitive neuroscience fields between a balance of large N studies and “precision imaging” studies in individual participants (Gratton et al., 2022; Naselaris et al., 2021; Rosenberg and Finn, 2022). Though our sample size is comparable to other studies that produced reliable results relating sulcal morphology to brain function and cognition (for example, Cachia et al., 2021; Garrison et al., 2015; Lopez-Persem et al., 2019; Miller et al., 2021; Roell et al., 2021; Voorhies et al., 2021; Weiner, 2019; Willbrand et al., 2022a, 2022b; Yao et al., 2022), ongoing work that uses deep learning algorithms to automatically define sulci should result in much larger sample sizes in future studies (Borne et al., 2020; Lee et al., 2024, 2025; Lyu et al., 2021). The time-consuming manual definitions of primary, secondary, and PTS also limit the cortical expanse explored in each study, thus restricting the present study to LPC/LPOJ.”
Second, we utilized a young adult sample as this is what is the standard of the field when charting features of sulci for the first time (for example, Paus et al., 1996 Cerebral Cortex; Chiavaras & Petrides, 2000 Journal of Comparative Neurology; Segal & Petrides, 2012 European Journal of Neuroscience; Zlatkina & Petrides, 2014 Proceedings of the Royal Society B Biological Science; Sprung-Much & Petrides, 2018 Brain Structure & Function; Miller et al., 2021 The Journal of Neuroscience; Willbrand et al., 2022 Science Advances; Willbrand et al., 2023 Communications Biology; Drudik et al., 2023 Cerebral Cortex). Nevertheless, it is indeed crucial to confirm that this schematic is translatable to other age groups; however this exploration is beyond the scope of the present project and is for future investigation. We have added text to the Limitations subsection of the Discussion to emphasize the points (Pages 17-18):
“Additionally, the scope of the present study is limited in that the sample was only in young adults. This sample was selected as it is the standard of the field when charting features of sulci for the first time (for example, Paus et al. 1996; Chiavaras and Petrides 2000; Segal and Petrides 2012; Zlatkina and Petrides 2014; Sprung-Much and Petrides 2018; Miller et al. 2021; Willbrand et al. 2022; Willbrand et al. 2023; Drudik et al. 2023). Nevertheless, it is necessary to explore how well this updated schematic translates to different age groups, species, and clinical populations.”
Finally, it is worth mentioning that we have begun preliminary analyses on the translatability of this schematic, and have shown that it does hold in a pediatric sample (ages 6-18 years old; Author response image 1).
Author response image 1.
Example pediatric participant with all LPC/LOPJ sulci identified in both hemispheres. Incidence rates for the variable pTS identified in the present work in a pediatric sample are included below (N = 79 participants)
(2) While the paper begins by describing four new sulci, only one is explored further in greater detail.
We focused on the slocs-v as it has a high incidence rate, making it amenable to our analytic pipelines relating sulci to cortical morphology, architecture, and function, as well as cognition (Miller et al., 2021 The Journal of Neuroscience; Voorhies et al., 2021 Nature Communications; Yao et al., 2022 Cerebral Cortex; Willbrand et al., 2022 Science Advances; Willbrand et al., 2023 The Journal of Neuroscience; Maboudian et al., 2024 The Journal of Neuroscience). However, we want to emphasize that throughout the paper there are multiple analyses that further describe the three more variable sulci: 1) detailing their sulcal patterning (Supplementary Tables 1-4) and 2) detailing their morphology and architecture (Supplementary Fig. 6). We do agree though that it is a worthwhile endeavor to further describe these sulci—especially if the data is readily available. As such, to complement our behavioral analysis identifying a relationship between the morphology of the consistent sulci and spatial orientation and considering the well-documented relationship between sulcal incidence and cognition (for review see Cachia et al., 2021 Frontiers in Neuroanatomy), we tested whether the number of variable sulci and the incidence of each variable sulcus specifically were related to spatial orientation. This procedure produced null results on all neuroanatomical variables, which we now mention in the Results (Page 11):
“Finally, as in prior work examining variably-present PTS in other cortical expanses (for example, (Amiez et al., 2018; Cachia et al., 2014; Fornito et al., 2004; Willbrand et al., 2024b), we assessed whether the presence/absence of the more variable PTS identified in the present work (slocs-d, pAngs-v, and pAngs-d) was related to spatial orientation, reasoning, and processing speed task performance. We identified no significant associations between the presence/absence of these sulci in either hemisphere with performance on these tests (ps > .05).”
(3) There is some tension between calling the discovered sulci new vs acknowledging they have already been reported, but not named.
To resolve this tension, we have revised the text to 1) ensure proper acknowledgment that sulci have been noticed in this region, 2) point out that these sulci were left unnamed and undescribed, and 3) emphasize that one of the primary goals of this project was to comprehensively detail the sulcal organization of this region at a precise, individual-level considering these often-overlooked sulci.
This is primarily done at the beginning of the Results (Pages 4-5), where we now write:
“Four previously undescribed small and shallow sulci in the lateral parieto-occipital junction (LPOJ)
In previous research in small sample sizes, neuroanatomists noticed shallow sulci in this cortical expanse, but did not describe them beyond including an unlabeled sulcus in their schematic at best (Supplementary Methods and Supplementary Figs. 1-4 for historical details). In the present study, we fully update this sulcal landscape considering these overlooked indentations. In addition to defining the 13 sulci previously described within the LPC/LPOJ, as well as the posterior superior temporal cortex in individual participants (Methods) (Petrides, 2019), we could also identify as many as four small and shallow PTS situated within the LPC/LPOJ that were highly variable across individuals and left undescribed until now (Supplementary Methods and Supplementary Figs. 1-4). Though we officially name and characterize features of these sulci in this paper for the first time, it is necessary to note that the location of these four sulci is consistent with the presence of variable “accessory sulci” in this cortical expanse mentioned in prior modern and classic studies (Supplementary Methods). For four example hemispheres with these 13-17 sulci identified, see Fig. 1a (Supplementary Fig. 5 for all hemispheres).”
(4) The anatomy of the sulci, as opposed to their relation to other sulci, could be described in greater detail.
To detail these sulci above and beyond their relation to other sulci, we document the anatomical metrics of all sulci in Supplemental Figure 6:
Results (Page 8):
The morphological and architectural features of all LPC/LPOJ sulci are described in Supplementary Fig. 6.




