Peer review process
Not revised: This Reviewed Preprint includes the authors’ original preprint (without revision), an eLife assessment, public reviews, and a provisional response from the authors.
Read more about eLife’s peer review process.Editors
- Reviewing EditorCarla RothlinYale University, New Haven, United States of America
- Senior EditorCarla RothlinYale University, New Haven, United States of America
Reviewer #1 (Public Review):
Summary:
This manuscript by Aybar-Torres et al investigated the effect of common human STING1 variants on STING-mediated T cell phenotypes in mice. The authors previously made knock-in mice expressing human STING1 alleles HAQ or AQ, and here they established a new knock-in line Q293. The authors stimulated cells isolated from these mice with STING agonists and found that all three human mutant alleles resist cell death, leading to the conclusion that R293 residue is essential for STING-mediated cell death (there are several caveats with this conclusion, more below). The authors also bred HAQ and AQ alleles to the mouse Sting1-N153S SAVI mouse and observed varying levels of rescue of disease phenotypes with the AQ allele showing more complete rescue than the HAQ allele. The Q293 allele was not tested in the SAVI model. They conclude that the human common variants such as HAQ and AQ have a dominant negative effect over the gain-of-function SAVI mutants.
Strengths:
The authors and Dr. Jin's group previously made important observations of common human STING1 variants, and these knock-in mouse models are essential for understanding the physiological function of these alleles.
Weaknesses:
However, although some of the observations reported here are interesting, the data collectively does not support a unified model. The authors seem to be drawing two sets of conclusions from in vitro and in vivo experiments, and neither mechanism is clear. Several experiments need better controls, and these knock-in mice need more comprehensive functional characterization.
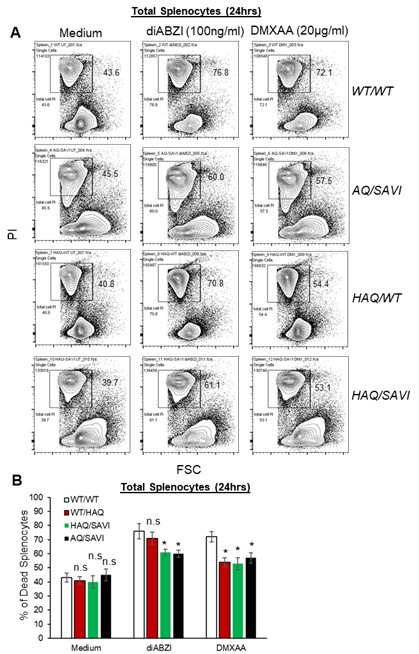
(1) In Figure 1, the authors are trying to show that STING agonist-induced splenocytes cell death is blocked by HAQ, AQ and Q alleles. The conclusion at line 134 should be splenocytes, not lymphocytes. Most experiments in this figure were done with mixed population that may involve cell-to-cell communication. Although TBK1-dependence is likely, a single inhibitor treatment of a mixed population is not sufficient to reach this conclusion.
(2) Q293 knock-in mouse needs to be characterized and compared to HAQ and AQ. Is this mutant expressed in tissues? Does this mutant still produce IFN and other STING activities? Does the protein expression level altered on Western blot? Is the mutant protein trafficking affected? In the authors' previous publications and some of the Western blot here, expression levels of each of these human STING1 protein in mice are drastically different. HAQ and AQ also have different effects on metabolism (pmid: 36261171), which could complicate interoperation of the T cell phenotypes.
(3) HAQ/WT and AQ/WT splenocytes are protected from STING agonist-induced cell death equally well (Figure 1G). HAQ/SAVI shows less rescue compared to AQ/SAVI. These are interesting observations, but mechanism is unclear and not clearly discussed. E.g., how does AQ protect disease pathology better than HAQ (that contains AQ)? Does Q293 allele also fully rescue SAVI?
(4) Figure 2 feels out of place. First of all, why are the authors using human explant lung tissues? PBMCs should be a better source for lymphocytes. In untreated conditions, both CD4 and B cells show ~30% dying cells, but CD8 cells show 0% dying cells. This calls for technical concerns on the CD8 T cell property or gating strategy because in the mouse experiment (Figure 1A) all primary lymphocytes show ~30% cell death at steady-state. Second, Figure 2C, these type of partial effect needs multiple human donors to confirm. Three, the reconstitution of THP1 cells seems out of place. STING-mediated cell death mechanism in myeloid and lymphoid cells are likely different. If the authors want to demonstrate cell death in myeloid cells using THP1, then these reconstituted cell lines need to be better validated. Expression, IFN signaling, etc. The parental THP1 cells is HAQ/HAQ, how does that compare to the reconstitutions? There are published studies showing THP1-STING-KO cells reconstituted with human variants do not respond to STING agonists as expected. The authors need to be scientifically rigorous on validation and caution on their interpretations.
(5) Figure 2G, H, I are confusing. AQ is more active in producing IFN signaling than HAQ and Q is the least active. How to explain this?
(6) The overall model is unclear. If HAQ, AQ and Q are loss-of-function alleles and Q is the key residue for STING-mediated cell death, then why AQ is the most active in producing IFN signaling and AQ/SAVI rescues disease most completely? If these human variants act as dominant negatives, which would be consistent with the WT/het data, then how do you explain AQ is more dominant negative than HAQ?
(7) As a general note, SAVI disease phenotypes involve multiple cell types. Lymphocyte cell death is only one of them. The authors' characterization of SAVI pathology is limited and did not analyze immunopathology of the lung.
(8) Line 281, the discussion on HIV T cell death mechanism is not relevant and over-stretching. This study did not evaluate viral infection in T cells at all. The original finding of HAQ/HAQ enrichment in HIV/AIDS was 2/11 in LTNP vs 0/11 in control, arguably not the strongest statistics.
Reviewer #2 (Public Review):
Aybar-Torres and colleagues utilize common human STING alleles to dissect the mechanism of SAVI inflammatory disease. The authors demonstrate that these common alleles alleviate SAVI pathology in mice, and perhaps more importantly use the differing functionality of these alleles to provide insight into requirements of SAVI disease induction. Their findings suggest that it is residue A230 and/or Q293 that are required for SAVI induction, while the ability to induce an interferon-dependent inflammatory response is not. This is nicely exemplified by the AQ/SAVI mice that have an intact inflammatory response to STING activation, yet minimal disease progression. As both mutants seem to be resistant STING-dependent cell death, this manuscript also alludes to the importance of STING-dependent cell death, rather than STING-dependent inflammation, in the progression of SAVI pathology. While I have some concerns, I believe this manuscript makes some important connections between STING pathology mouse models and human genetics that would contribute to the field.
Some points to consider:
(1) While the CD4+ T cell counts from HAQ/SAVI and AQ/SAVI mice suggest that these T cells are protected from STING-dependent cell death, an assay that explores this more directly would strengthen the manuscript. This is also supported by Fig 2C, but I believe a strength of this manuscript is the comparison between the two alleles. Therefore, if possible, I would recommend the isolation of T cells from these mice and direct stimulation with diABZI or other STING agonist with a cell death readout.
(2) Related to the above point - further exemplifying that the Q293 locus is essential to disease, even in human cells, would also strengthen the paper. It seems that CD4 T cell loss is a major component of human SAVI. While not completely necessary, repeating the THP1 cell death experiments from Fig 2 with a human T cell line would round out the study nicely.
(3) While I found the myeloid cell counts and BMDM data interesting, I think some more context is needed to fully loop this data into the story. Is myeloid cell expansion exemplified by SAVI patients? Do we know if myeloid cells are the major contributors to the inflammation these patients experience? Why should the SAVI community care about the Q293 locus in myeloid cells?
(4) The functional assays in Figure 4 are exciting and really connect the alleles to disease progression. To strengthen the manuscript and connect all the data, I would recommend additional readouts from these mice that address the inflammatory phenotype shown in vitro in Figure 5. For example, measuring cytokines from these mice via ELISA or perhaps even Western blots looking for NFkB or STING activation would be supportive of the story. This would also allow for some tissue specificity. I believe looking for evidence of inflammation and STING activation in the lungs of these mice, for example, would further connect the data to human SAVI pathology.