Cdc48/p97 promotes degradation of aberrant nascent polypeptides bound to the ribosome
Figures
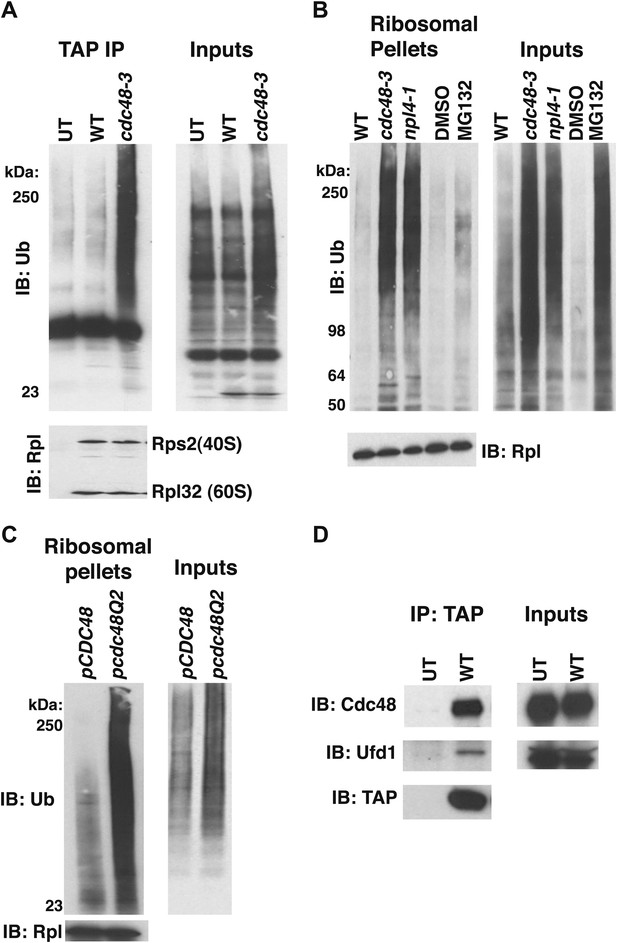
Ub conjugates accumulate on ribosomes isolated from cdc48 and npl4-1 mutants.
(A) Ribosome assembly is unimpaired in cdc48-3 mutant cells and Ub conjugates accumulate on cdc48-3 ribosomes. Ribosomes were immunoprecipitated (IP) from cdc48-3 (Untagged, UT), RPL18BTAP (WT), and cdc48-3 RPL18BTAP cells shifted to the non-permissive temperature (37°C) for 90 min. Purified ribosomes were evaluated by Coomassie blue staining (Figure 1—figure supplement 2A) and immunoblotting (IB) with Ub and anti-Rpl32 antibodies. Anti-Rpl32 also detects Rps2. (B) WT and mutant cells grown at 24°C were shifted to 30°C for two generations and pdr5Δ cells were either mock-treated with DMSO, or 30 μM MG132 for 30 min. Ribosomes were isolated (input cell lysates, right panel) by sedimentation through sucrose cushions. Ribosome pellets were evaluated by IB with Ub and Rpl32 antibodies (left panels). (C) The ATPase activity of Cdc48 promotes clearance of Ub conjugates from ribosomes. Mutant cdc48-3 cells containing plasmid-borne WT GAL-CDC48His6 or the Q2 mutant were grown in galactose for 2 hr to induce expression of ectopic Cdc48. Induced cells were shifted to 35°C for 1 hr to inactivate cdc48-3 before being harvested. Ribosomes were isolated by sedimentation through sucrose cushions and input cell lysates and ribosome pellets were evaluated as in (A). (D) Cdc48 binds to purified ribosomes. Ribosomes were affinity-purified from untagged or RPL18BTAP-tagged cells and the levels of bound Cdc48 and Ufd1 were evaluated by IB with the respective antibodies.
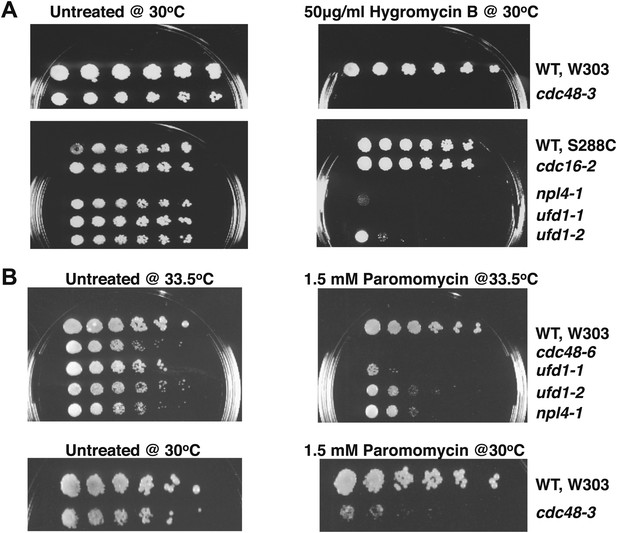
Cdc48 pathway mutants are sensitive to hygromycin B (A) and paromomycin (B).
Serial fivefold dilutions of wildtype and mutant cells were spotted on YPD plates either lacking (untreated) or containing the indicated amount of drug and incubated at the indicated temperature for 3 days.
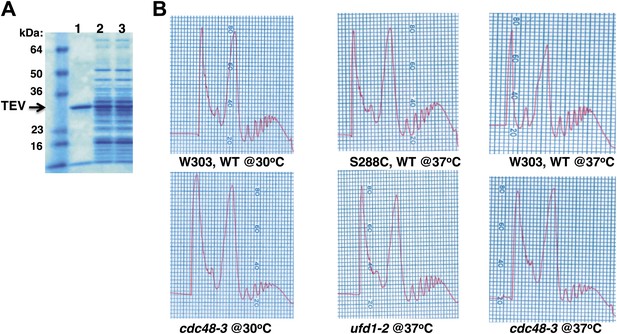
Ribosome assembly is unimpaired in Cdc48 pathway mutants.
(A) SDS-PAGE and Coomassie blue analysis of affinity-purified ribosomes from untagged (UT, lane 1), WT (lane 2) and cdc48-3 (lane 3) strains used in Figure 1A. (B) Absorbance profile (254 nm) of wildtype and mutant lysates fractionated on 10–50% sucrose gradients.
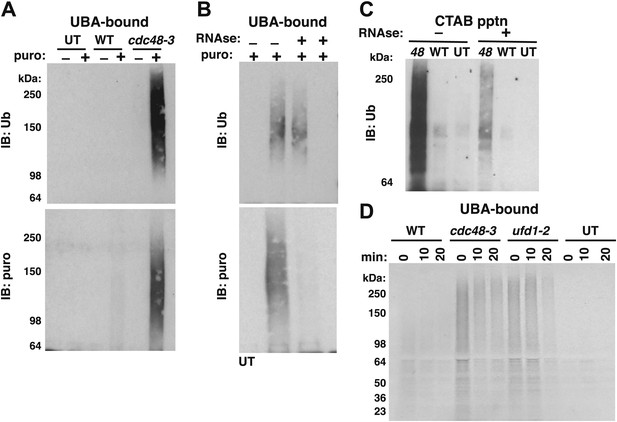
Ubiquitinated nascent peptides linked to tRNA accumulate on ribosomes in cdc48-3 and ufd1-2 mutants.
(A) Puromycin-dependent binding to UBA resin of Ub conjugates from cdc48-3 ribosomes. Ribosomes were affinity-purified from untagged or wildtype and mutant RPL18BTAP cells grown at 37°C for 90 min and eluted with TEV protease in the presence or absence of puromycin (puro), followed by incubation with UBA resin. Bound fractions were resolved by SDS-PAGE and immunoblotted with antibodies to Ub (top panel) or puromycin (lower panel). (B) Transfer of Ub conjugates to puromycin was RNAse A-sensitive. Ribosomes were affinity-purified from cdc48-3 (UT) or cdc48-3RPL18BTAP cells as described above in the absence (lanes 1, 2 and 3) or presence of 200 μg/ml RNAse A. Following elution with TEV protease, samples from the tagged cells (lane 3) were treated with 200 μg/ml RNAse A at 30°C for 10 min before incubation of all samples with puromycin and binding to UBA resin. The bound fractions were evaluated as in (A). (C) CTAB-precipitable Ub conjugates accumulate on cdc48-3 ribosomes. Ribosomes affinity-purified from the same strains used in panel (A) were treated with RNAse A (or not) and subjected to precipitation (pptn) with CTAB. Precipitates were resolved by SDS-PAGE and immunoblotted with anti-Ub. 48 Refers to cdc48-3. (D) Ubiquitinated newly-synthesized proteins accumulate on ribosomes isolated from cdc48-3 and ufd1-2 mutants. Cells were pulse-labeled for 90 s with 35S methionine and chased with cold methionine and cycloheximide for the indicated times. Ribosomes were affinity-purified, eluted with TEV protease in the presence of puromycin, and loaded onto UBA resin. The bound fraction was evaluated by SDS-PAGE followed by autoradiography. Densitometry indicated that <5% of the high MW material was released from ufd1-2 ribosomes between the 0- and 10-min time points.
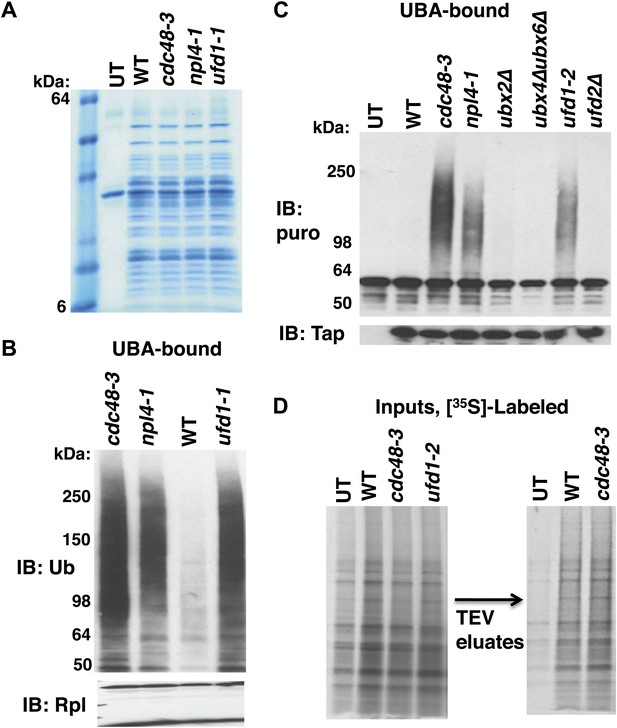
Cdc48-Ufd1-Npl4 complex is required to clear ubiquitinated nascent peptides from ribosomes.
(A) SDS-PAGE/Coomassie blue profiles of ribosomes affinity-purified from the indicated strains carrying the RPL18BTAP allele. UT: wildtype cells lacking the RPL18BTAP allele. (B) Ribosomes from the indicated strains grown at 30°C for two generations were pelleted through sucrose cushions and analyzed by immunoblotting (IB) for anti-Rpl32 (bottom panel). Resuspended ribosomes were treated with puromycin in high salt and then incubated with UBA resin. The bound fraction was evaluated by SDS-PAGE and IB with anti-ub (top panel). (C) Ribosomes were affinity-purified from wildtype and mutant strains carrying the RPL18BTAP allele and eluted from the magnetic beads with TEV protease in the presence of puromycin. The eluate was either evaluated directly by IB with anti-TAP (bottom panel) or was incubated with UBA beads. The bound material was fractionated by SDS-PAGE and analyzed by IB with anti-puromycin antibody (top panel). (D) Left panel: Total cell lysates for [35S]-labeled cells were fractionated by SDS-PAGE and subjected to autoradiography to show incorporation of label during the 90 sec pulse. Right panel: Ribosomes were affinity-purified from lysates depicted on left. Ribosomes eluted from the magnetic beads with TEV protease were analyzed by SDS-PAGE and autoradiography to reveal newly-synthesized proteins contained in the preparations. UT: wildtype cells lacking the RPL18BTAP allele.
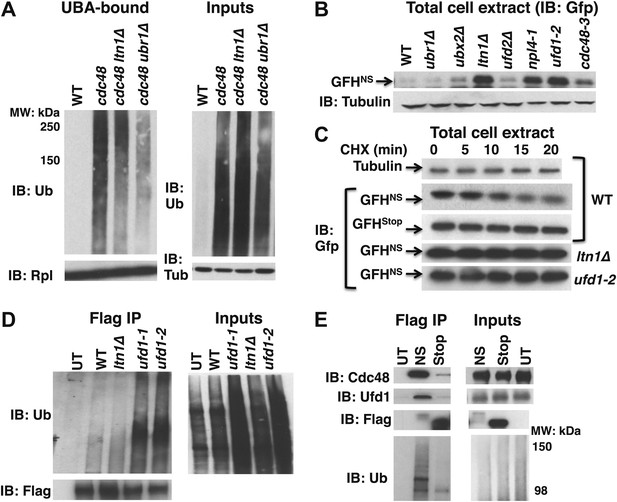
Aberrant proteins derived from non-stop mRNA accumulate in cells deficient in Cdc48–Ufd1–Npl4 function.
(A) Ltn1 and Ubr1 contribute to accumulation of Ub conjugates on cdc48-3 ribosomes. Ribosomes from the indicated strains grown at 30°C were isolated from input cell lysates (right panels) by pelleting through sucrose cushions, treated with puromycin, and incubated with UBA resin. The bound fraction (left panels) and inputs were evaluated by SDS-PAGE and immunoblotting with Ub, tubulin (Tub), and Rpl32 antibodies as indicated. The -3 allele of cdc48 was used. (B) The non-stop reporter GFHNS accumulates in Cdc48 pathway mutants. Glass bead/SDS extracts from exponential cultures (grown at 30°C) of the indicated mutants harboring a plasmid that expresses GFHNS were analyzed by SDS-PAGE and IB with anti-Gfp. Tubulin served as the loading control. (C) Cycloheximide chase analysis of cells expressing either the non-stop (GFHNS) or stop codon-containing (GFHStop) reporters. Glass bead/SDS extracts prepared from aliquots harvested at the indicated times from wildtype and mutant cultures were analyzed by SDS-PAGE and IB with anti-Gfp. Samples from WT expressing GFHNS were also evaluated by IB with anti-tubulin to confirm equal loading. Note that extracts prepared from cells expressing GFHStop were loaded at one-fifth the amount of GFHNS. (D) Non-stop protein accumulates in the ubiquitinated state in ufd1 mutants. Glass bead/SDS extracts of wildtype and mutant cells expressing plasmid-borne GFHNS and grown at 37°C for 90 min were evaluated directly (inputs) or immunoprecipitated with anti-Flag antibodies after 10-fold dilution with buffer containing Triton X-100. Bound proteins and inputs were evaluated by SDS-PAGE and IB with anti-Ub and Flag antibodies. UT corresponds to WT cells not expressing GFHNS. (E) Cdc48 and Ufd1 interact selectively with non-stop protein. Lysates prepared from cells expressing Flag-tagged GFHNS (NS) or GFHStop (Stop) reporters as well as cells lacking a plasmid (UT) were immunoprecipitated with anti-Flag antibodies. Total cell extracts (inputs; right panels) and bound proteins (left panels) were evaluated by SDS-PAGE and IB with anti-Cdc48, Ufd1, Flag and Ub antibodies.
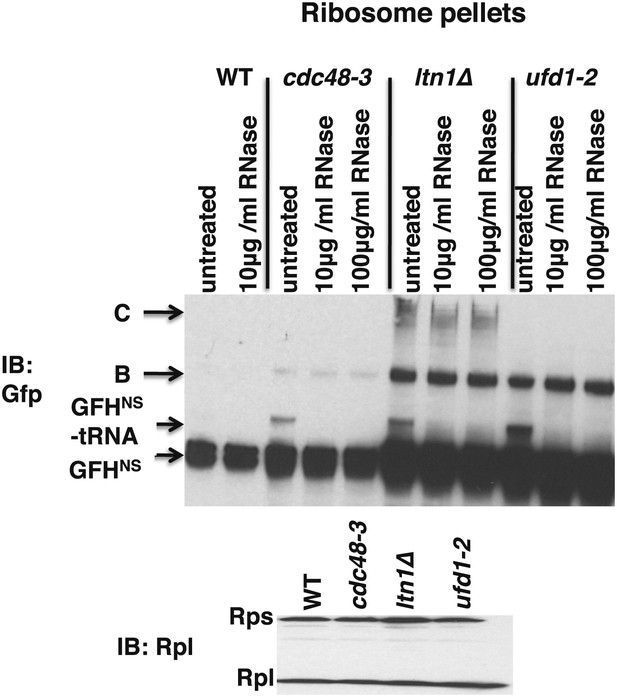
Ribosomes from wildtype and mutant cells grown at 37°C for 90 min were sedimented through sucrose cushions.
Pellets were resuspended and either evaluated by SDS-PAGE and immunoblotting (IB) with anti-Rpl32 (bottom panel) or incubated with the indicated amount of RNAse for 10 min at 30°C. Aliquots of the RNAse reactions were evaluated by separation on NuPAGE gels and IB with anti-Gfp (top panel). Species B and C are RNAse-insensitive species whose physical nature is unknown.
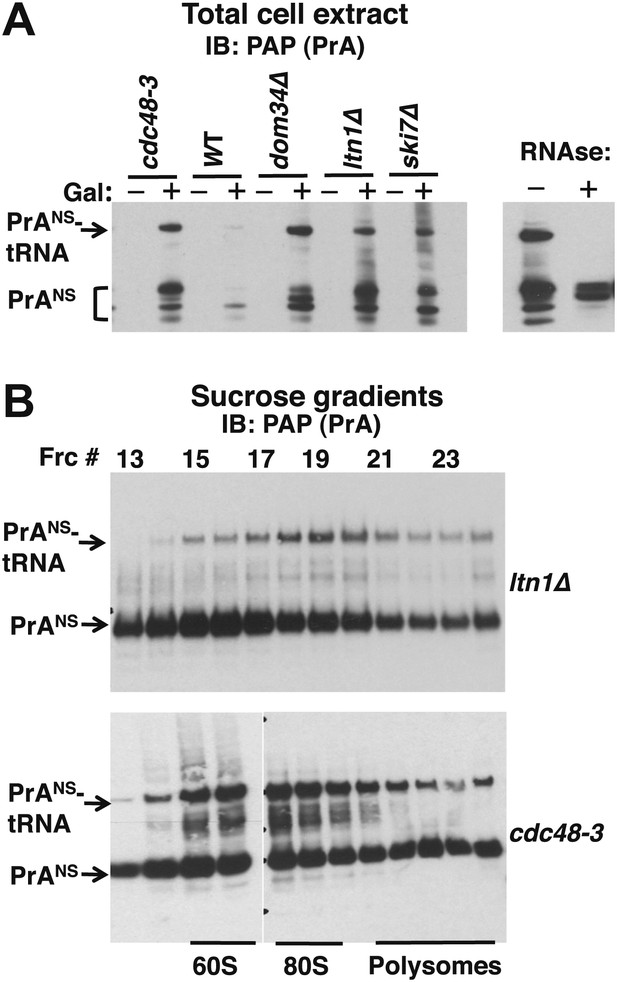
Non-stop reporter PrANS accumulates on 60S and 80S ribosomes in a tRNA-linked form in cdc48-3 cells.
(A) NuPAGE gel analysis reveals accumulation of tRNA-linked PrANS in cdc48-3 and ltn1Δ cells. Wildtype and mutant cells containing reporter plasmid were grown at 30°C for one generation and expression of reporter was induced (or not) by the addition of 2% galactose (±Gal) for 2 hr. Glass bead/SDS extracts were fractionated on a NuPAGE gel to preserve tRNA-linked species, and analyzed by IB with PAP to detect protein A. Equal loading was confirmed by Ponceau S staining and IB for tubulin (Figure 4—figure supplement 1A). The right panel shows collapse of the tRNA-linked species in cdc48-3 extract following RNAse treatment. (B) tRNA-linked PrANS accumulates on 60S and 80S ribosomes in ltn1Δ and cdc48-3 mutants. Sucrose gradient (10–30%) fractions from cells expressing plasmid-borne PrANS and grown at 30°C for two generations were concentrated by TCA precipitation and evaluated by fractionation on a NuPAGE gel and IB with anti-PAP or anti-Rpl32 (Figure 4—figure supplement 1B). Fractions 15 and 16 (enriched for 60S) and 17–19 (enriched for 80S ribosomes) are indicated.
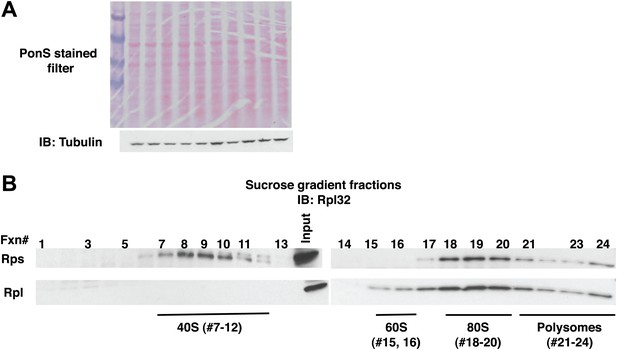
Fractionation of ribosome subunits on sucrose gradients.
(A) Ponceau S staining (top panel) and anti-tubulin IB (bottom panel) of nitrocellulose filter used for Figure 4A. The lanes in Figure 4A with no signal contain lysate from uninduced cells. (B) Lysates from wildtype cells containing the PrANS reporter were fractionated on 10–30% sucrose gradients in parallel with lysates from ltn1Δ and cdc48-3 grown at 30°C for two generations (Figure 4B). 0.2 ml fractions were collected and aliquots were resolved by SDS-PAGE followed by IB with anti-Rpl32.
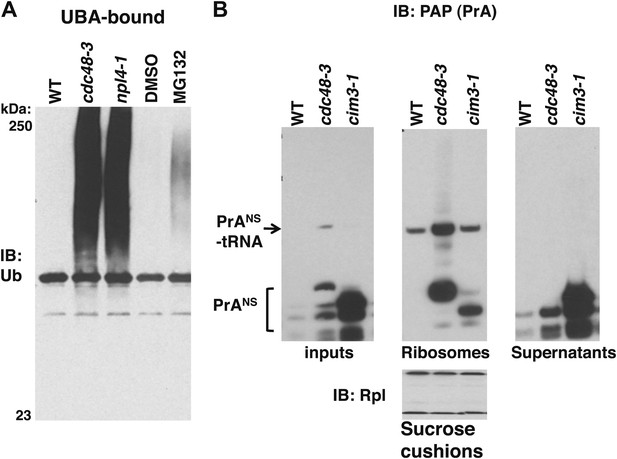
Non-stop reporter PrANS accumulates on ribosomes in cdc48-3 cells but is released from ribosomes in cells deficient in proteasome activity.
(A) Ub conjugates accumulate to a greater extent on ribosomes isolated from cdc48-3 and npl4-1 mutants than on ribosomes isolated from cells treated with proteasome inhibitor MG132. Puromycin-treated ribosomes from Figure 1B were incubated with UBA resin. The bound fraction was evaluated by SDS-PAGE and immunoblotting with anti-Ub. (B) PrANS preferentially accumulates on the ribosome in cdc48-3 cells and in the post-ribosome supernatant in a proteasome mutant. Ribosomes were isolated from cells grown at 37°C for 90 min by pelleting through sucrose cushions. Total cell lysate inputs (left panel), ribosome pellets (middle panel) and post-ribosome supernatants (right panel) were evaluated by separation on a NuPAGE gel and IB with PAP. Equivalent recovery of ribosomes in the pellet fractions was confirmed by IB with anti-Rpl32 (bottom panel). The same cell cultures were also evaluated by preparing extracts in SDS and resolving aliquots on a Tris-Glycine (Figure 5—figure supplement 1B, left panel) or NuPAGE (Figure 5—figure supplement 1B, right panel) gel.
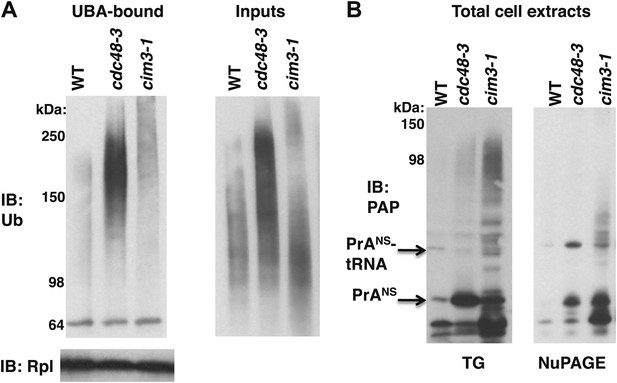
Maximal Ub conjugate accumulation on ribosomes isolated from cdc48-3 mutant cells.
(A) Ribosomes from wildtype and mutant cells grown at 37°C for 90 min were sedimented through sucrose cushions. Pellets were resuspended and either evaluated by SDS-PAGE and IB with anti-Rpl32 (lower left panel) or incubated with UBA resin following treatment with puromycin. The bound fraction (upper left panel) and the input cell lysates (right panel) were evaluated by SDS-PAGE and IB with anti-ub (top panel). (B) Extracts prepared by the glass bead/SDS method from the indicated strains grown at 37°C for 90 min were analyzed on a Tris-Glycine (TG; left panel) or NuPAGE (right panel) gel.
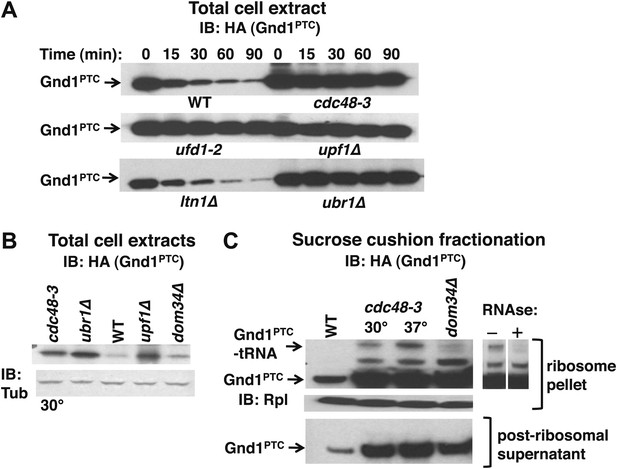
The Cdc48 pathway is required for degradation of prematurely terminated Gnd1PTC.
(A) A cycloheximide chase was performed with the indicated mutants grown at 30°C for two generations. Glass bead/SDS extracts were prepared at the indicated times after adding cycloheximide and analyzed by immunoblotting with anti-HA antibody to detect HA-tagged Gnd1PTC. (B) A tRNA-linked form of Gnd1PTC accumulates on ribosomes from cdc48-3 mutants. Total cell extracts prepared identically to those used for the ribosome analysis in panel C were immunoblotted with anti-HA to detect Gnd1PTC and anti-tubulin to evaluate loading. cdc48-3 mutant cells were incubated at a semi-permissive (30°C) temperature for 90 min prior to lysis whereas all other strains were grown at 30°C for two generations. (C) Ribosomes (top panels) and post-ribosomal supernatants (bottom panel) were isolated from the same strains in (B) by sedimentation through sucrose cushions and analyzed by immunoblotting with anti-HA to detect Gnd1PTC. The ribosome pellets were also immunoblotted with anti-Rpl32 (middle panel) to confirm equivalent recovery. The right panel shows collapse of the tRNA-linked species following RNAse treatment of the ribosomal pellet from cdc48-3. cdc48-3 cells were incubated at either the semi-permissive (30°C) or non-permissive (37°C) temperature prior to lysis, as described in (B).
Ponceau S staining of filters for Figure 6A.
Ponceau S staining of filters for Figure 6A. A tubulin loading control is shown for the top filter.
Additional files
-
Supplementary file 1
Yeast strains and plasmids used in this study.
- https://doi.org/10.7554/eLife.00308.016





