Pharmacological brake-release of mRNA translation enhances cognitive memory
Figures
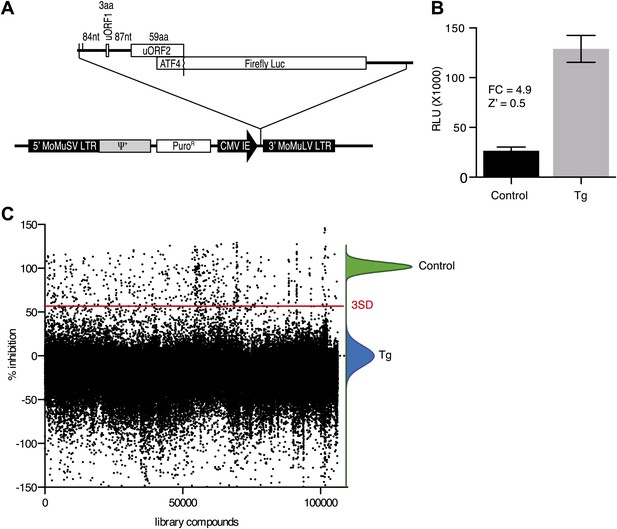
High-throughput cell-based screen for inhibitors of PERK signaling.
(A) Schematic representation of the ATF4 luciferase reporter used in the primary screen. The 5′ UTR of human ATF4 containing the uORFs 1 and 2 was fused to firefly luciferase and inserted into a retroviral expression system. (B) Primary screen optimization. HEK293T stably expressing the ATF4 luciferase reporter were plated in 384-well plates and treated for 6 hr with 100 nM thapsigargin (Tg) or DMSO as a no ER stress control. Luciferase production was measured at the end point after 6 hr (mean ± SD). The Z′ was calculated as 1−(3 [σ Tg + σ DMSO]/[μ Tg–μ DMSO]). (C) Primary screen results. The ATF4 luciferase reporter cell line was treated for 6 hr with 100 nM thapsigargin and library compounds (10 µM). Inhibition of the luciferase activity reporter was calculated as the percent reduction in relative luminescence normalized to thapsigargin treatment (0% inhibition) and the no-ER stress control (100% inhibition). Compounds were considered hits if they lied beyond three standard deviations (SD) from the thapsigargin treatment mean (red line).
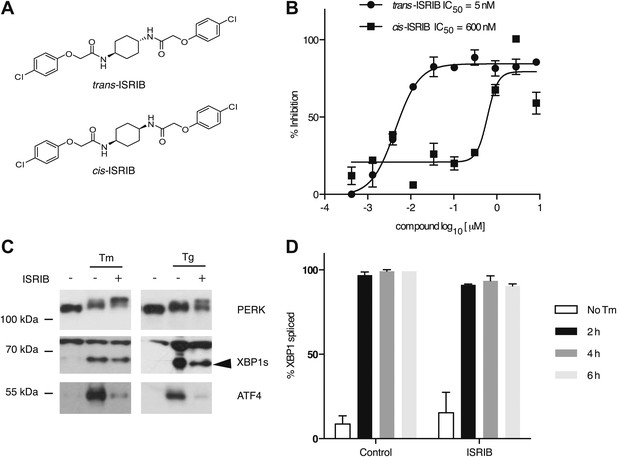
Identification of ISRIB as a potent cell-based inhibitor of PERK signaling.
(A) Structures of ISRIB isosteromers. (B) Inhibition of the ATF4 luciferase reporter in HEK293T cells by ISRIB stereoisomers. Inhibition is plotted in relation to the concentration of either the cis or trans isomer of ISRIB. Cells were treated with 2 µg/ml of tunicamycin to induce ER stress and different concentrations of the inhibitors for 7 hr (N = 2, mean ± SD). (C) Effect of ISRIB on production of endogenous ATF4, PERK phosphorylation, and XBP1s production. An immunoblot analysis of PERK, ATF4 and XBP1s in HEK293T cells treated with different ER stress inducers (2.5 µg/ml tunicamycin [Tm] or 100 nM thapsigargin [Tg]) with or without 200 nM ISRIB for 3 hr is shown. The arrowhead marks the XBP1s specific band. (D) Effect of ISRIB on XBP1 mRNA splicing. Taqman assays for XBP1unspliced (XBP1u) and XBP1spliced (XBP1s) on cDNA synthesized from total RNA extracted from U2OS cells treated with 2 µg/ml of tunicamycin in the presence or absence of 200 nM ISRIB for the indicated times are shown. Percent splicing was calculated as the ratio of XBP1s over total XBP1 mRNA (XBP1u + XBP1s) (mean ± SD).
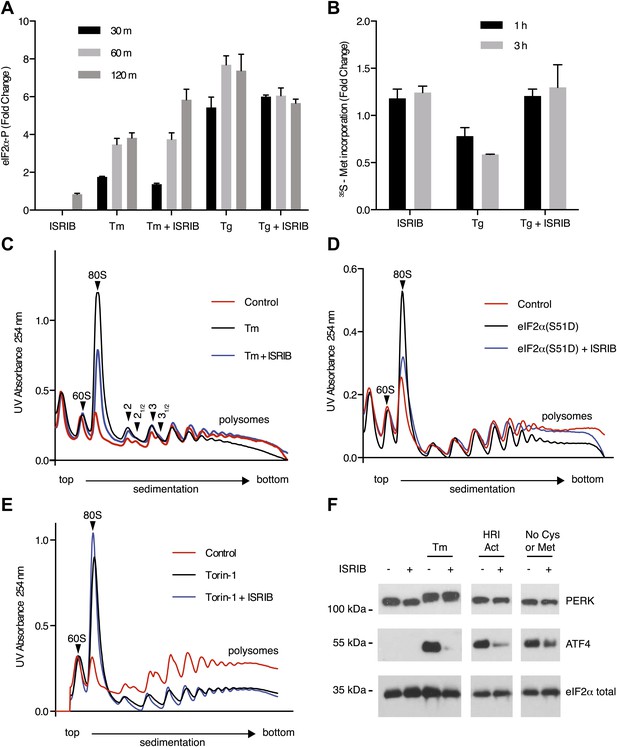
ISRIB makes cells resistant to eIF2α phosphorylation.
(A) ISRIB does not block eIF2α phosphorylation upon ER stress. eIF2α phosphorylation was measured using an alpha-screen Surefire eIF2α p-S51 assay (see ‘Materials and methods’). U2OS cells were plated in 96-well plates and treated with 2 µg/ml tunicamycin or 100 nM thapsigargin in the presence or absence of 100 nM ISRIB for the indicated times or with ISRIB alone for 120 m (N = 4, mean ± SD). See Figure 3—figure supplement 1 for supporting Western blot analysis of eIF2α phosphorylation. (B) ISRIB blocks global translational attenuation observed after eIF2α phosphorylation during ER stress. HEK293T cells were treated with 100 nM thapsigargin and 200 nM ISRIB for either 1 or 3 hr prior to a 20 min pulse with 35S methionine before lysis. Equal amounts of lysate were loaded on an SDS-PAGE gel and quantification of radiolabeled methionine incorporation of lysates was done by gel densitometry (N = 2, SD) using ImageJ. (see Figure 3—figure supplement 2 for SDS-PAGE). (C) Polysome gradient analysis showing the block in global translational attenuation upon addition of ISRIB on ER-stressed cells. MEFs were grown in the presence or absence of 2 µg/ml of tunicamycin with or without 200 nM ISRIB for 1 hr. Cell lysates were loaded on a 10–50% sucrose gradient, centrifuged at 150,000×g for 2.4 hr and absorbance at 254 nm was measured across the gradient (see Figure 3—figure supplement 3 for quantitation of polysome profile). A representative experiment is shown (N = 3). See Figure 3—figure supplement 4 for a close-up of the disome and trisome peaks. (D) Cells treated with ISRIB are resistant to the global translational attenuation exerted by forced expression of eIF2α(S51D). HEK293Trex cells were transduced with a tetracycline inducible phospho-mimetic (S51D) allele of eIF2α. Transgene expression was induced by addition of 25 nM doxycycline for 14 hr in the presence or absence of 200 nM ISRIB. Lysates were collected and analyzed as described in panel (C) (see Figure 3—figure supplement 6 for quantitation of polysome profile). A representative experiment is shown (N = 2). (E) ISRIB does not reverse global translational attenuation exerted through inhibition of CAP-dependent initiation. Wild-type MEFs were treated with 750 nM Torin-1 in the presence or absence of 200 nM ISRIB for 2 hr. Lysates were collected and analyzed as described in panel (C). A representative experiment is shown (N = 2). (F) ISRIB blocks production of ATF4 upon GCN2 or HRI activation. An immunoblot analysis of PERK, ATF4 and total eIF2α in HEK293T cells starved for cysteine and methionine or treated with an HRI activator (6 µM) for 5 hr in the presence or absence of 200 nM ISRIB is shown. Tunicamycin was used as a positive control for induction of ATF4 and the shift in PERK mobility. Under amino acid starvation we consistently observe a partial reduction of ATF4 production by ISRIB by Western blot analysis but observe a complete block in induction of the ATF4 luciferase reporter (see Figure 3—figure supplement 7).
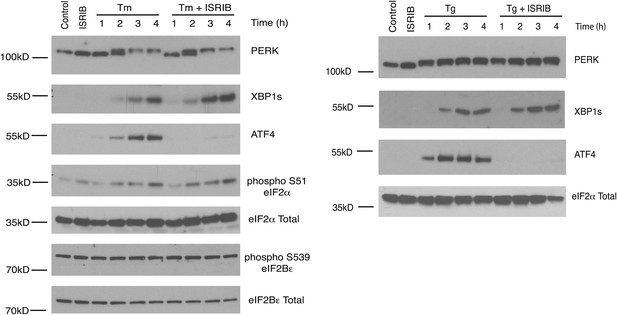
ISRIB does not inhibit eIF2α phosphorylation or XBP1s production.
Western blot analysis of PERK, ATF4, XBP1s, phospho S51-eIF2α, total eIF2α, phospho S539-eIF2Bε and total eIF2Bε in HEK293T cells treated with or without 2 µg/ml of tunicamycin or 100 nM thapsigargin in the presence or absence of 200 nM ISRIB for the indicated times.
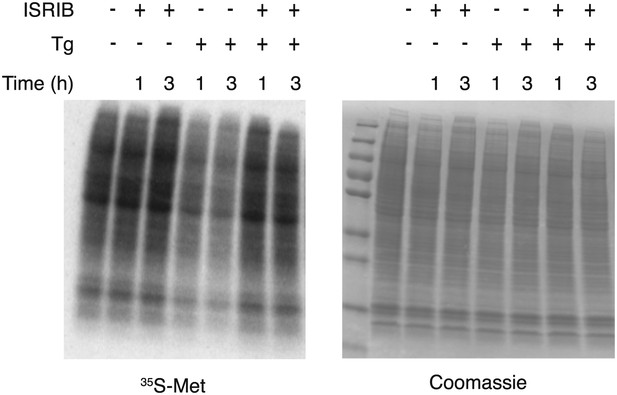
ISRIB blocks translational attenuation upon ER stress.
Autoradiogram (left) and total protein (right) obtained from HEK293T cells that were treated with 100 nM thapsigargin with or without 200 nM ISRIB for either 1 or 3 hr prior to a 20 min pulse with 35S-methionine before lysis. Equal amounts of lysate were loaded on an SDS-PAGE gel.
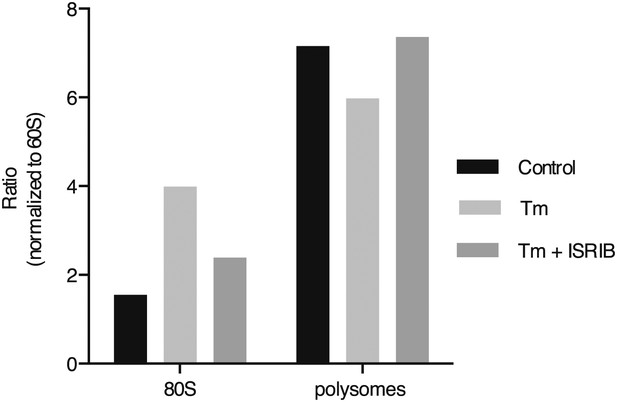
ISRIB blocks translational attenuation upon ER stress.
The polysome profile in Figure 3C was quantitated by calculating the area under the curve corresponding to the monosome peak (80S), or the area under the curve corresponding to the trace covering the polysome region and then plotted as a ratio over the area under the curve corresponding to the peak of the 60S subunit.
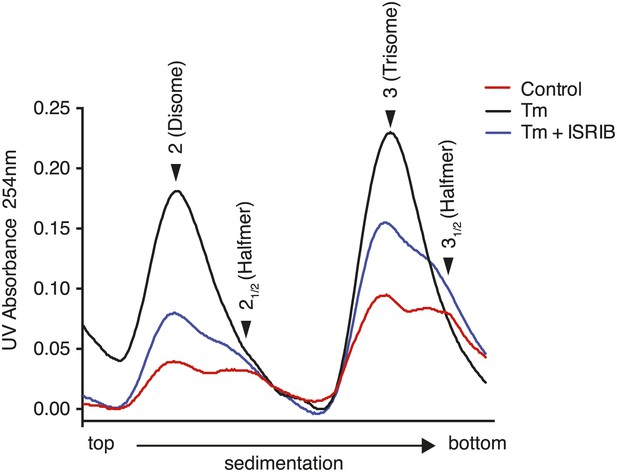
ISRIB partially restores the halfmer population in ER stressed cells.
Wildtype MEFs were grown in the presence or absence of 2 µg/ml of tunicamycin with or without 200 nM ISRIB for 1 hr. This graph is a close up of the disome and trisome peaks of the polysome gradients in Figure 3C.
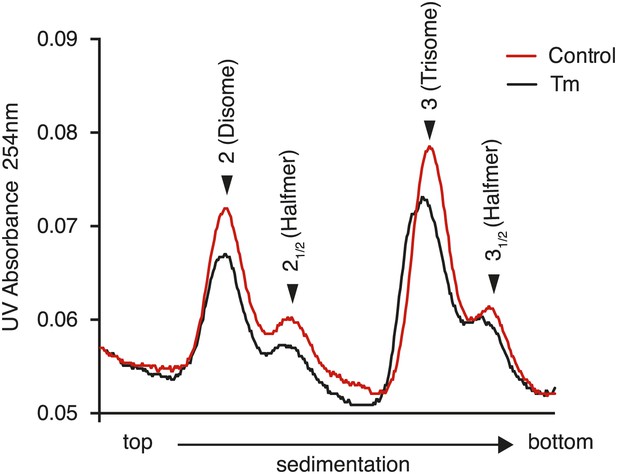
Disappearance of the halfmer peaks upon ER-stress is dependent on eIF2α phosphorylation.
eIF2αS51A/S51A (Eif2s1S51A/S51A)MEFs were grown in the presence or absence of 2 µg/ml of tunicamycin with or without 200 nM ISRIB for 1 hr and polysomes gradients were processed and analyzed as described in Figure 3C.
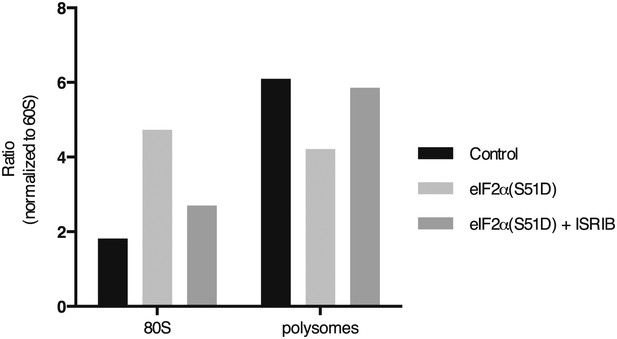
ISRIB sustains translation upon expression of eIF2α(S51D).
The polysome profile in Figure 3D was quantitated as described in Figure 3—figure supplement 3.
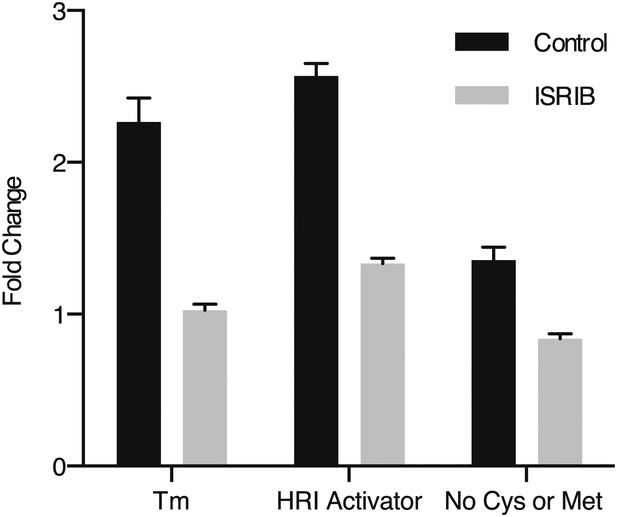
ISRIB blocks induction of the ATF4 luciferase translational reporter upon HRI and GCN2 activation.
HEK293T carrying the ATF4 luciferase reporter were treated with 2 µg/ml of tunicamycin to induce ER stress, 6 µM of the HRI activator or grown in media lacking cysteine and methionine for 7 hr in the presence or absence of 200 nM ISRIB (N = 4). The relative luciferase units are normalized to the no treatment control. Using this reporter we observe a smaller fold change in production of luciferase by amino acid starvation that is fully blocked by addition of ISRIB.
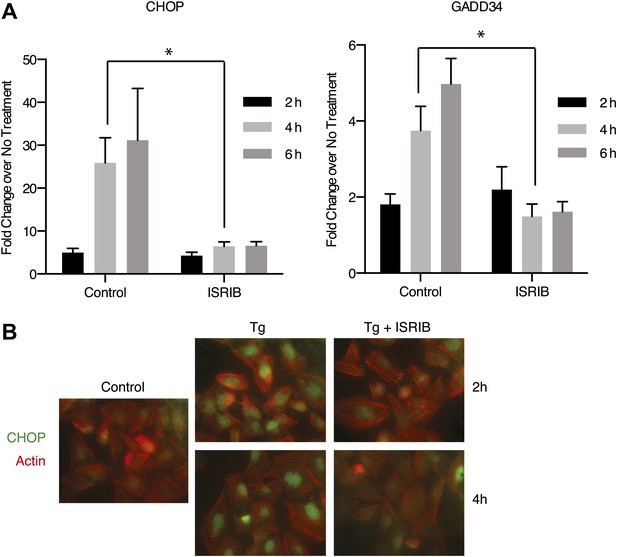
ISRIB impairs induction of the transcriptional network controlled by ATF4.
(A) ER-Stress dependent induction of CHOP and GADD34 mRNA is impaired in cells treated with ISRIB. qPCR analysis of total RNA extracted from U2OS cells treated with 2 µg/ml of tunicamycin in the presence or absence of 200 nM ISRIB for the indicated times. mRNA levels for each sample were normalized to GAPDH (N = 4, mean ± SD). p values are derived from a one-tail Student’s t-test for unpaired samples. Statistical significance: CHOP, *p=0.0006; GADD34, *p=0.0008. (B) ISRIB blocks CHOP production during ER stress. An immunofluorescence analysis of U2OS cells treated with 100 nM thapsigargin for 2 or 4 hr in the presence or absence of 200 nM ISRIB is shown. A secondary Alexa Dye 488 anti-mouse antibody and rhodamine-phalloidin were used to visualize CHOP and actin, respectively.
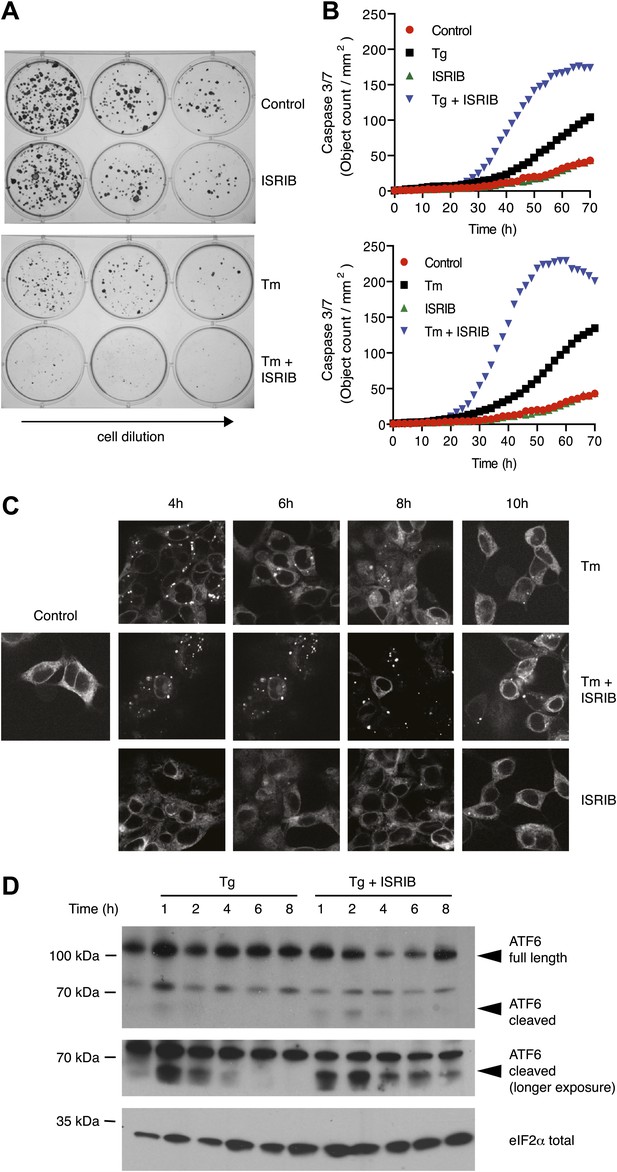
ISRIB impairs adaptation to ER-stress prolonging activation of the UPR sensors.
(A) ISRIB sensitizes cells to acute ER stress. HEK293T cells were subjected with an acute dose of tunicamycin (2 µg/ml), ISRIB (200 nM) or a combination of both for 24 hr. The treated cells were equally diluted to a concentration that would allow single cell clonal expansion and re-seeded onto six-well plates in a threefold dilution series. Clonal colonies were visualized by Crystal Violet stain. (B) ISRIB synergizes with ER stress to activate caspase 3/7. Hela cells were plated in 96-well plates and treated with 5 µg/ml of tunicamycin or 500 nM thapsigargin with or without 25 nM ISRIB for the indicated times. Caspase3/7 activation was measured using Cellplayer kinetic caspase 3/7 reagent and cells were imaged in an IncuCyte system. Green object count/mm2 representing caspace-3/7 activation was measured at 2 hr intervals (See Figure 5—figure supplement 1 for endpoint quantitation of % cells with activated caspase 3/7). (C) IRE1 oligomers are sustained on ER-stressed cells treated with ISRIB. Confocal microscopy micrographs of HEK293Trex cells carrying an inducible GFP-tagged IRE1 allele were treated with 10 nM doxycycline for 24 hr to induce the transgene, followed by treatment with 5 µg/ml of tunicamycin in the presence or absence of 200 nM ISRIB for the indicated times. (See Figure 5—figure supplement 2 for corresponding XBP1 mRNA splicing data). (D) ATF6 cleavage is sustained in ER-stressed cells treated with ISRIB. Immunoblot analysis of ATF6 processing in HEK293Trex cells carrying an inducible FLAG epitope-tagged ATF6. Cells were treated with 50 nM doxycycline for 18 hr to induce the transgene followed by treatment with 100 nM thapsigargin in the presence or absence of 200 nM ISRIB for the indicated times. Total eIF2α is used as a loading control.
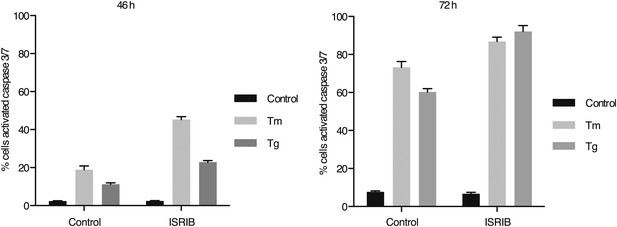
ISRIB synergizes with ER-stress to induce caspase 3/7.
Green object count/mm2 representing caspase-3/7 activation depicted in Figure 5A was normalized to the total number of cells at two different endpoints. In order to quantify the total number of cells, Vybrant DyeCycle Green staining solution (1 µM) was added directly to the well immediately after the caspase-3/7 scan and incubated for 1 hr prior to acquiring final images at both 46 and 72 hr. Data is presented as % cells with activated caspase 3/7 at these two endpoints. Note that by 72 hr the ER-stress inducing conditions used in this experiment are so detrimental that they diminish the synergistic effects observed by addition of ISRIB. The synergy was clearly seen at the 46 hr time-point.
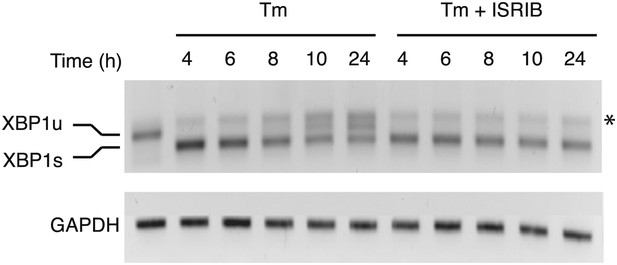
XBP1 splicing is sustained in ER-stressed cells upon addition of ISRIB.
HEK293T cells were treated with tunicamycin (2 µg/ml) for the indicated times in the presence or absence of 200 nM ISRIB. RNA was isolated from the cells and reverse transcribed followed by PCR with oligos that amplify both the unspliced and spliced versions of XBP1 mRNA or GAPDH. The DNA was electrophoresed in a 2.5% agarose gel. The asterix (*) denotes a hybrid PCR product.
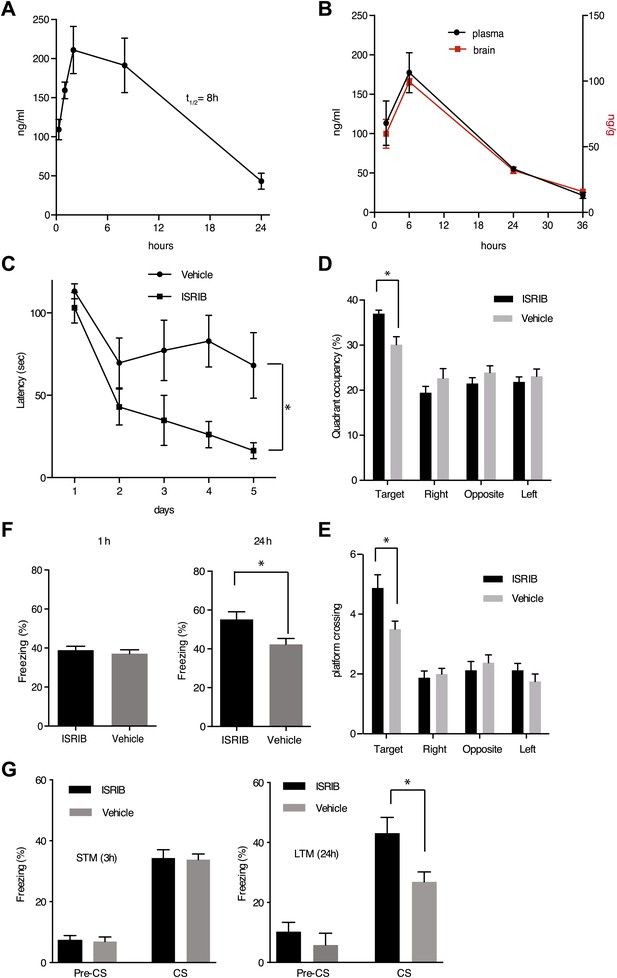
ISRIB enhances spatial and fear-associated learning in rodents.
(A) Plasma concentration (ng/ml) of ISRIB after a single intraperitoneal injection (5 mg/kg). Plasma was collected at the indicated times and the concentration was determined by mass spectrometry (mean ± SEM, N = 3). (B) Brain (ng/g tissue) and plasma concentrations (ng/ml) of ISRIB after a single intraperitoneal injection (2.5 mg/kg). Data (mean ± SEM, N = 3) were obtained at the indicated times. (C) Escape latencies are significantly shorter in mice treated with ISRIB. Data (mean ± SEM) were obtained in a weak 5 days-long training session in the hidden platform version of the Morris water maze (1 trial per day). Mean escape latencies were plotted as a function of training days in mice treated with ISRIB (closed squares, N = 8) or vehicle (open circles N = 8) (*p<0.05). Mice were injected daily with ISRIB immediately after training. (D) After completion of training in the study shown in panel (A), mice treated with ISRIB (black column) showed a significant preference for the target quadrant (*p<0.05). The probe test was performed 24 hr after the last training session. p values are derived from a two-tailed Student’s t test for unpaired samples. (E) After completion of training in the study shown in panel a, mice treated with ISRIB (black column) increased the number of times they crossed the platform location as compared to the vehicle-treated mice (grey column) (*p<0.05). p values are derived from a two-tailed Student’s t test for unpaired samples. (F) Systemic administration of ISRIB (intraperitoneally after training) enhances long-term contextual fear memory (right bars, 24 hr), while it does not affect short-term memory (left bars, 1 hr) (N = 10 per group, *p<0.05). Data are presented as mean ± SEM. (G) Auditory fear conditioning is enhanced in rats treated with ISRIB. Freezing in response to a tone was assessed 3 hr (short-term memory, STM, left panel) and 24 hr (long-term memory, LTM, right panel) after training (vehicle-treated N = 8, and ISRIB-treated N = 7) after tone presentation (CS) and before tone presentation (pre-CS). For these experiments vehicle or ISRIB was infused directly by cannula into the amygdala after training. ISRIB-infused rats show increase freezing at 24 hr (*p<0.05).





