CAMKII and Calcineurin regulate the lifespan of Caenorhabditis elegans through the FOXO transcription factor DAF-16
Figures
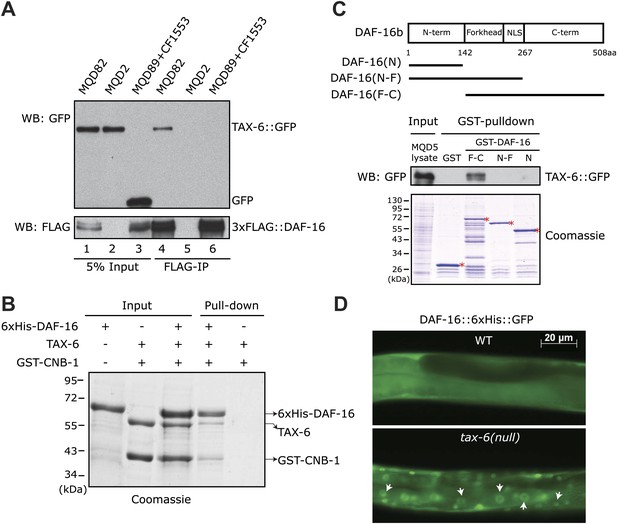
C. elegans Calcineurin TAX-6•CNB-1 directly binds to DAF-16 and negatively regulates DAF-16 nuclear localization.
(A) DAF-16 and TAX-6 form a complex in vivo. Immunoprecipitation of 3xFLAG::DAF-16 expressed under the daf-16 promoter in WT C. elegans pulled down TAX-6::GFP expressed under the tax-6 promoter. The lysates were obtained from transgenic strains MQD82 (co-expressing 3xFLAG::DAF-16 and TAX-6::GFP), MQD2 (expressing TAX-6::GFP), MQD89 (expressing 3xFLAG::DAF-16), and CF1553 (expressing GFP under a sod-3 promoter). (B) Calcineurin directly binds to DAF-16. Purified recombinant TAX-6•GST-CNB-1 was pulled down with Ni-NTA beads through its interaction with purified His-tagged DAF-16. (C) The C-terminal region of DAF-16 most likely mediates the interaction with Calcineurin. GST-DAF-16(F-C), but not GST, GST-DAF-16(N) or GST-DAF-16(N-F), pulled down TAX-6::GFP expressed in C. elegans (strain MQD5). The DAF-16 C-terminal region alone was not stable. Asterisk indicates full-length GST or GST fusion proteins. (D) DAF-16::6xHis::GFP is diffusely distributed in the WT animals but concentrated in the nucleus in tax-6(ok2065) animals. All GFP images shown in this paper are of L4 animals at 20°C unless otherwise indicated.
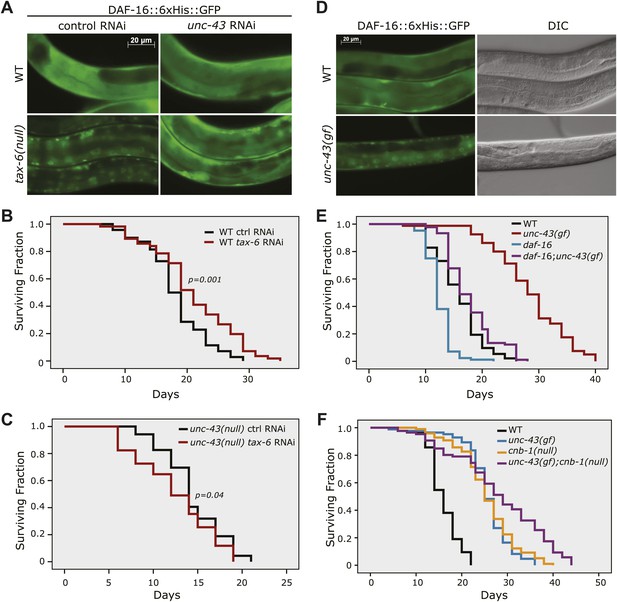
The effect of tax-6(lf) mutations on DAF-16 localization and lifespan requires unc-43, the CAMKII gene whose effect opposes that of tax-6.
(A) unc-43 RNAi abolished DAF-16 nuclear accumulation in tax-6(ok2065). Adult stage RNAi knockdown of tax-6 extended the WT lifespan (p=0.001) (B) but may have slightly shortened the lifespan of unc-43(n498n1186), a putative null mutant (p=0.04) (C). A constitutively active gain-of-function mutation, unc-43(n498), caused DAF-16::6xHis::GFP to accumulate in the nucleus (D) and extended lifespan in a largely daf-16-dependent manner (E). p<0.001 for daf-16(mu86);unc-43(gf) vs daf-16(mu86) or unc-43(gf). The log-rank p values are reported for all lifespan data in this study. (F) The unc-43(gf);cnb-1(null) double mutant has a longer lifespan than both the unc-43(gf) and cnb-1(null) mutants, and all three mutant strains live longer than WT animals. p<0.001 for WT vs any mutant, and p<0.001 for unc-43(gf);cnb-1(null) vs unc-43(gf) and cnb-1(null).
-
Figure 2—source data 1
The unc-43(null) mutant showed a WT-like lifespan that was epistatic to the longevity effect of tax-6(RNAi), while the unc-43(gf) mutant was long-lived.
(A) The lifespan extension by tax-6 RNAi required unc-43. (B) The longevity of unc-43(n498) was largely dependent on daf-16. (C) The unc-43(n498);cnb-1(ok276) double mutant lived longer than either single mutant.
- https://doi.org/10.7554/eLife.00518.005
A screen for the kinase(s) required for the nuclear accumulation of DAF-16::GFP induced by tax-6(null).
https://doi.org/10.7554/eLife.00518.006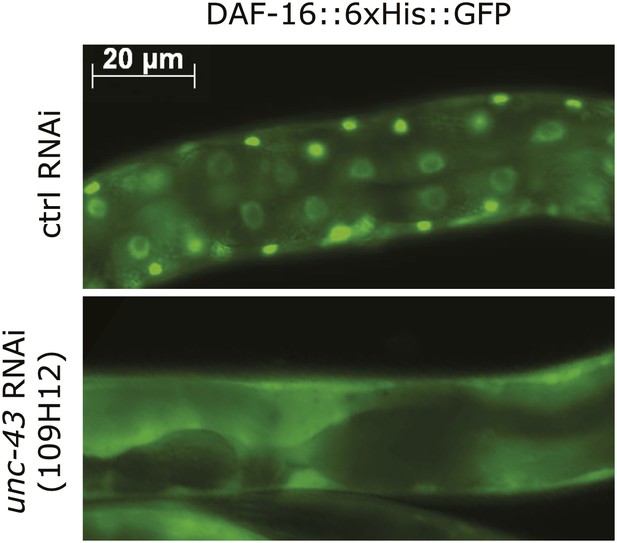
An unc-43 RNAi clone from the Ahringer library suppressed the nuclear accumulation of DAF-16::GFP induced by tax-6(null).
https://doi.org/10.7554/eLife.00518.007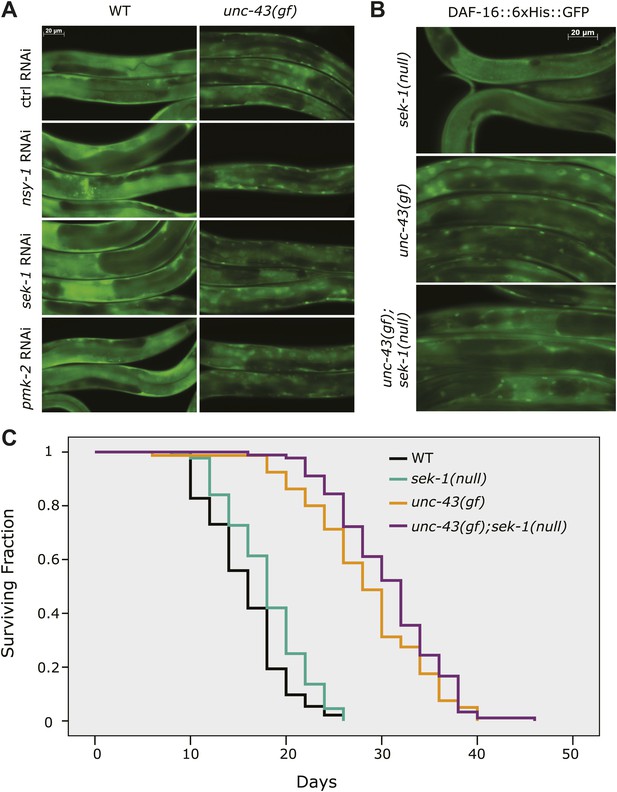
UNC-43 does not regulate DAF-16 localization through the NSY-1/SEK-1 MAPK kinase pathway.
(A) RNAi knockdown of nsy-1, sek-1, and pmk-2 failed to block the nuclear accumulation of DAF-16::6xHis::GFP in unc-43(gf) mutants. Worms were fed with the indicated RNAi bacteria from hatching and imaged at the L4 stage. (B) Similar to sek-1 RNAi, the sek-1(km4) mutation did not eliminate DAF-16 nuclear localization in unc-43(gf) worms. (C) km4, a null allele of sek-1, did not shorten the lifespan of WT or unc-43(gf) animals. In contrast, km4 may have slightly extended their lifespan. p=0.001 for sek-1(null) vs WT and p=0.047 for unc-43(gf); sek-1(null) vs unc-43(gf).
-
Figure 3—source data 1
km4, the null allele of sek-1, did not shorten the lifespan of unc-43(gf) worms.
- https://doi.org/10.7554/eLife.00518.009
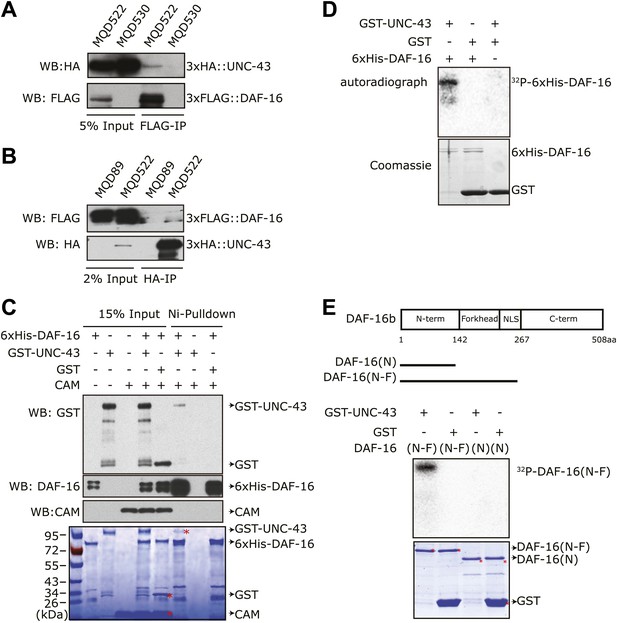
UNC-43 directly binds to and phosphorylates DAF-16.
(A) 3xHA::UNC-43 co-immunoprecipitated with 3xFLAG::DAF-16 and (B) vice versa from lysates of transgenic C. elegans expressing both proteins. The transgenic strains are MQD522 (co-expressing 3xHA::UNC-43 and 3xFLAG::DAF-16), MQD530 (expressing 3xHA::UNC-43), and MQD89 (expressing 3xFLAG::DAF-16). (C) UNC-43 can directly bind to DAF-16. Purified GST-UNC-43 but not CAM or the GST control was pulled down by Ni-NTA beads through its interaction with 6xHis-DAF-16. A Coomassie gel is shown at the bottom. (D)–(E) In vitro kinase assays in the presence of [32P]-γ-ATP, in which purified GST-UNC-43 directly phosphorylated His-tagged DAF-16 (D) or DAF-16 (N-F) fragments (E). The Coomassie-stained gel is shown below the autoradiograph.
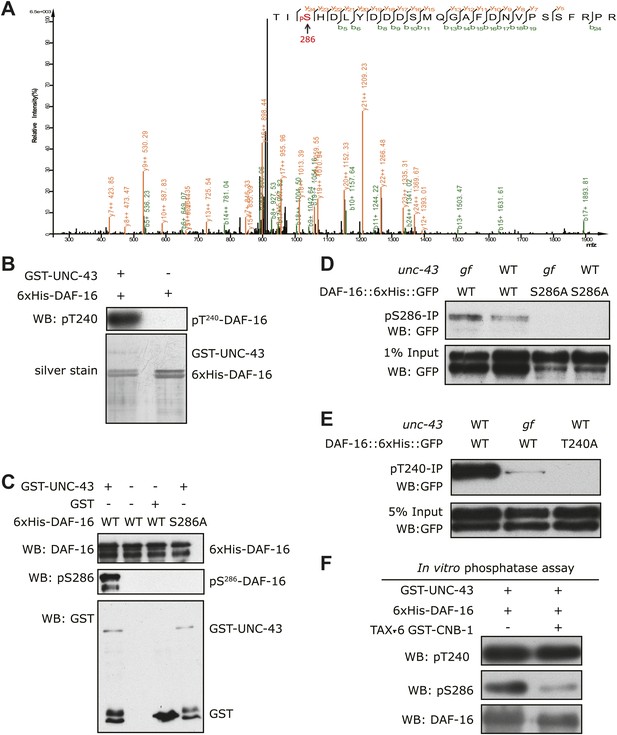
UNC-43 phosphorylates S286 of DAF-16, and TAX-6•CNB-1 removes this modification.
(A) A mass spectrum (neutral loss-triggered MS3) of a DAF-16 peptide phosphorylated at S286 by UNC-43 in vitro. (B and C) UNC-43 in vitro kinase assays with purified WT or S286A 6xHis-DAF-16 as substrate. DAF-16 phosphorylation was detected with antibodies specific for either phospho-T240 (B) or phospho-S286 (C). (D and E) UNC-43 phosphorylates DAF-16 at S286 but not T240 in vivo. Phosphorylated DAF-16::6xHis::GFP was immunoprecipitated from unc-43(wt) or unc-43(gf) animals using antibodies specific for either phospho-S286 (D) or phospho-T240 (E) and visualized by blotting with an anti-GFP antibody. Transgenic strains expressing S286A or T240A DAF-16::6xHis::GFP served as negative controls. All strains carry the daf-16 null allele mu86 in the background. (F) TAX-6•CNB-1 dephosphorylates DAF-16 specifically at S286. Purified 6xHis-DAF-16 was phosphorylated by UNC-43 in vitro and then incubated with purified TAX-6•GST-CNB-1 after heat inactivation of UNC-43. Phospho-T240, phospho-S286, and total DAF-16 levels were assayed by western blotting.
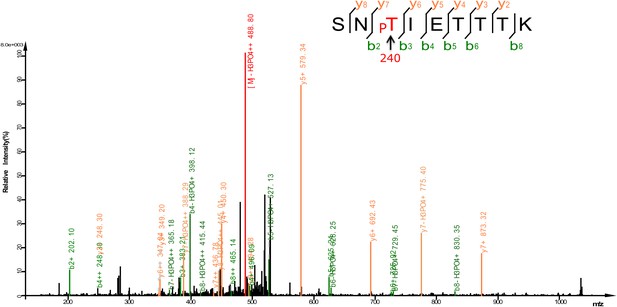
Mass spectrum showing a DAF-16 peptide phosphorylated at T240, one of the sites phosphorylated by UNC-43 in vitro.
https://doi.org/10.7554/eLife.00518.012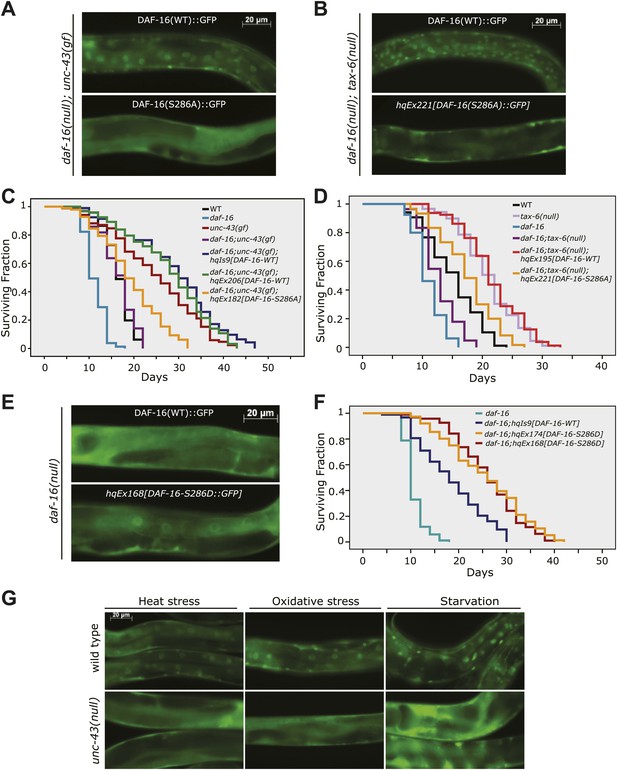
Phosphorylation of DAF-16 at S286 induces the nuclear accumulation of DAF-16 and extends lifespan.
The S286A mutation prevented DAF-16::6xHis::GFP from accumulating in the nucleus in unc-43(n498gf) (A) or tax-6(ok2065) animals (B). Unlike the DAF-16::6xHis::GFP transgene, DAF-16(S286A)::6xHis::GFP failed to restore the long lifespan in daf-16(mu86);unc-43(n498gf) (C) or daf-16(mu86);tax-6(ok2065) mutants (D). p<0.001 for unc-43(gf) vs daf-16;unc-43(gf);hqEx182 in (C) and tax-6(null) vs. daf-16;tax-6(null);hqEx221 in (D). The S286D mutation caused DAF-16::6xHis::GFP to accumulate in the nucleus (E) and extended lifespan in the daf-16(mu86) background (F). p<0.001, daf-16;hqEx174/168 vs. daf-16;hqIs9 in (F). 2 hr of heat stress at 28°C, 5 min of paraquat treatment, or 20 hr of food deprivation induced nuclear accumulation of DAF-16::6xHis::GFP in the WT animals but not the unc-43(n498n1186) mutants (G).
-
Figure 6—source data 1
The DAF-16(S286A) mutation largely suppressed the longevity induced by either unc-43(gf) or tax-6(null), while the DAF-16(S286D) mutation extended lifespan.
(A) Mutation of DAF-16 S286 to A partially suppressed the unc-43(gf) induced longevity. (B) Mutation of DAF-16 S286 to A partially suppressed the longevity of tax-6(null). (C) daf-16(mu86) animals expressing DAF-16 (S286D)::GFP had a longevity phenotype.
- https://doi.org/10.7554/eLife.00518.014
UNC-43 does not phosphorylate T240 in vivo to promote DAF-16 nuclear accumulation.
(A) DAF-16(T240A)::6xHis::GFP accumulated in the intestinal nuclei even in the absence unc-43(gf). This is incompatible with T240 being the UNC-43 phosphorylation site because mutating such a site to alanine was expected to inhibit DAF-16 nuclear accumulation. Rather, this result confirms that T240 is an AKT phosphorylation site (Lin et al., 2001). (B) 80% of the worms expressing a GFP-tagged DAF-16(T240A-S286A) mutant protein under a daf-16 promoter in the unc-43(n498);daf-16(mu86) background arrested as dauers, and the GFP signal was predominantly in the head neurons. A few individuals escaped dauer arrest and grew into adults. These animals displayed a strong nuclear accumulation of DAF-16(T240A-S286A) in body-wall muscles and hypodermis but a weakened nuclear accumulation in the intestine compared to those expressing WT DAF-16. In comparison, the S286A mutation prevented DAF-16 from accumulating in the nucleus when expressed in the same background (Figure 6A).
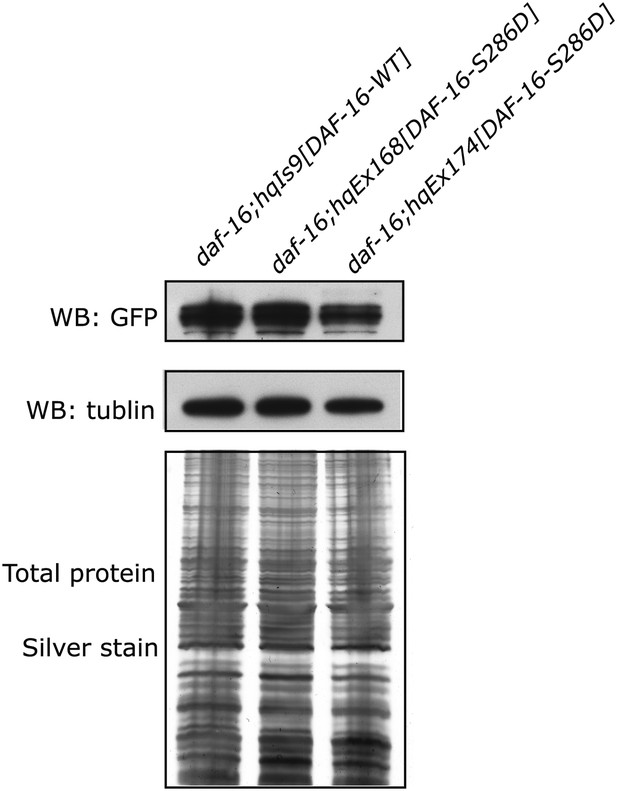
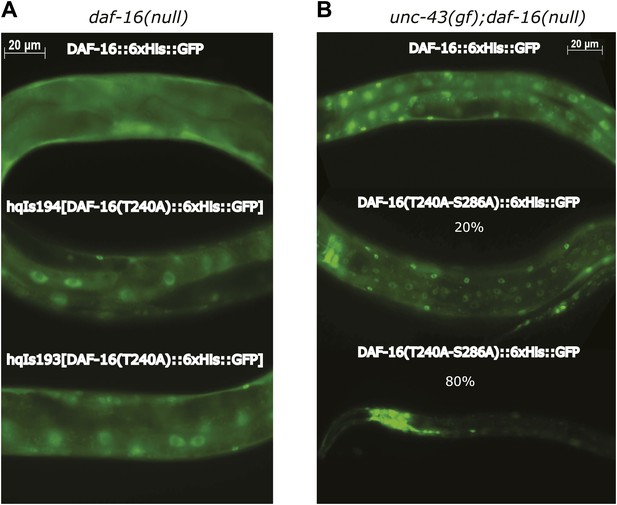
Similar expression levels of DAF-16(S286D)::6xHis::GFP and DAF-16::6xHis::GFP in three strains used in Figure 6F.
GFP fusion proteins in whole-worm lysates were quantified by anti-GFP WB. SDS-PAGE and anti-tubulin WB show equal loading of total proteins. For hqEx168 and hqEx174, because the transgene arrays were not chromosomally integrated, GFP positive worms were hand-picked under the microscope and pooled to make lysates.
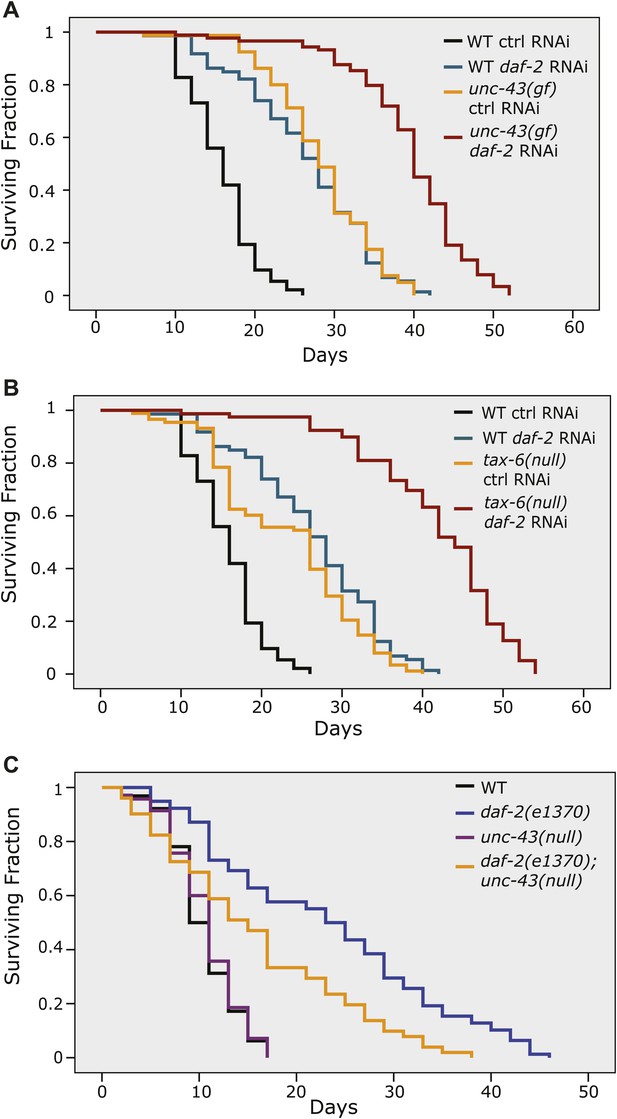
The longevity of daf-2 mutants was partially suppressed by unc-43(null) and further enhanced by unc-43(gf) or tax-6(null).
unc-43(n498), a gain-of-function allele (A), and tax-6(ok2065), a null allele (B), each extended lifespan in the WT background and further enhanced the longevity of daf-2(RNAi) animals. (C) The daf-2(e1370);unc-43(n498n1186) double mutant lived a significantly shorter life than daf-2(e1370), while the unc-43(n498n1186) mutant had a WT-like lifespan. In (A) and (B), p<0.001 for WT vs tax-6(null) or unc-43(gf), unc-43(gf);daf-2(RNAi) vs. daf-2(RNAi) or unc-43(gf), and tax-6(null);daf-2(RNAi) vs. daf-2(RNAi) or tax-6(null). In (C), p<0.001 for daf-2 vs. daf-2;unc-43(null), p=0.61 for unc-43(null) vs. WT in a lifespan assay at 25°C.
-
Figure 7—source data 1
The long lifespan of daf-2(RNAi) animals was further extended by either unc-43(gf) or tax-6(null), but shortened by unc-43(null).
(A) Both unc-43(gf) and tax-6(null) further increased the lifespan of daf-2 RNAi animals. (B) unc-43 was partially required for the long lifespan of daf-2 animals. (C) The DAF-16 S286A mutation appears not to affect the daf-2 longevity.
- https://doi.org/10.7554/eLife.00518.018
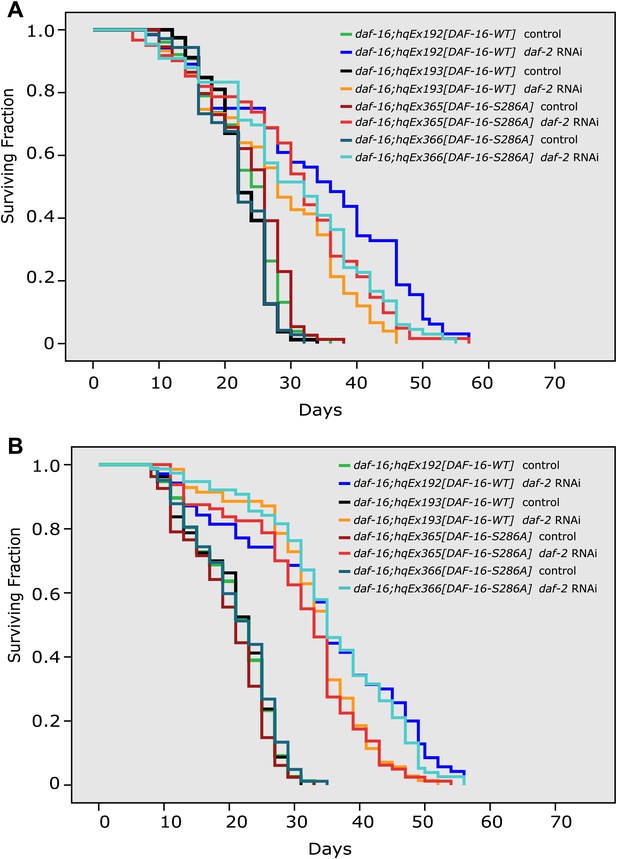
The DAF-16 S286A mutation appears not to affect daf-2 longevity.
(A)–(B) Two repeat experiments showing similar lifespan extension by daf-2 RNAi of daf-16(mu86) worms expressing DAF-16::6xHis::GFP (hqEx192 and hqEx193) or DAF-16(S286A)::6xHis::GFP (hqEx365 and hqEx366). n ≥ 61.
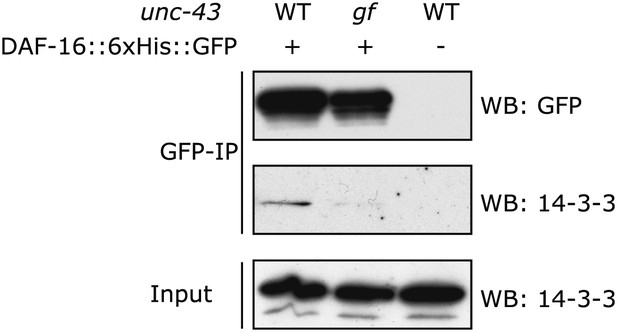
In the unc-43(n498gf) mutant, in which DAF-16 accumulates in the nucleus, the amount of DAF-16/14-3-3 complex is reduced.
DAF-16::6xHis::GFP was immunoprecipitated from worms carrying a WT or the gf allele of unc-43. The amount of 14-3-3 present in the lysate and that associated with DAF-16::6xHis::GFP were visualized by WB using an antibody that recognizes both 14-3-3 proteins (FTT-2 and PAR-5) in C. elegans.
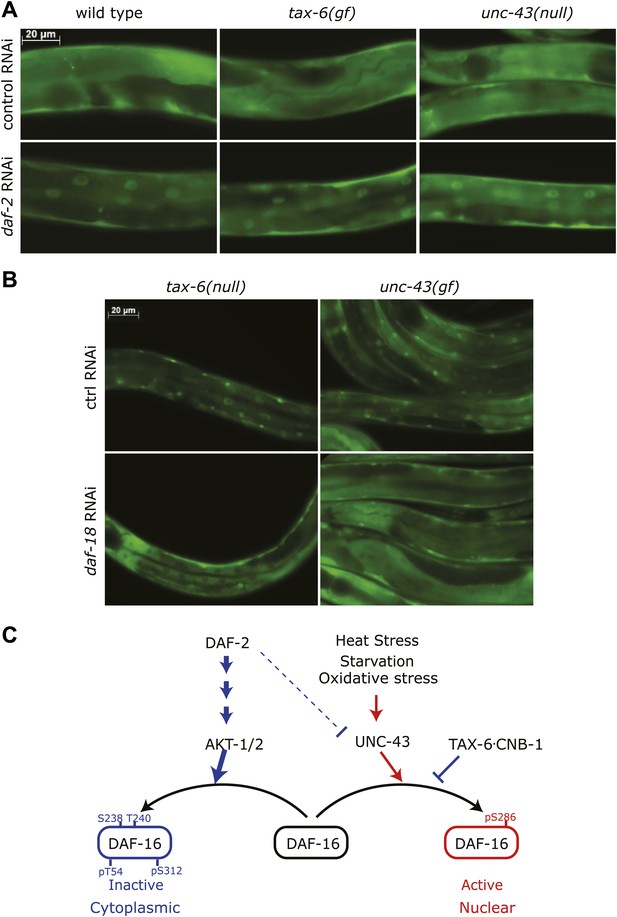
Insulin signaling overpowers CAMKII and Calcineurin in regulation of DAF-16 localization in C. elegans.
(A) tax-6(gf) or unc-43(null) failed to abolish DAF-16 nuclear accumulation induced by daf-2(RNAi). (B) tax-6(null) or unc-43(gf) failed to overcome the inhibition of DAF-16 nuclear localization by daf-18(RNAi). (C) A model showing the regulation of DAF-16 (FOXO) by insulin signaling, UNC-43 (CAMKII), and TAX-6•CNB-1 (Calcineurin).
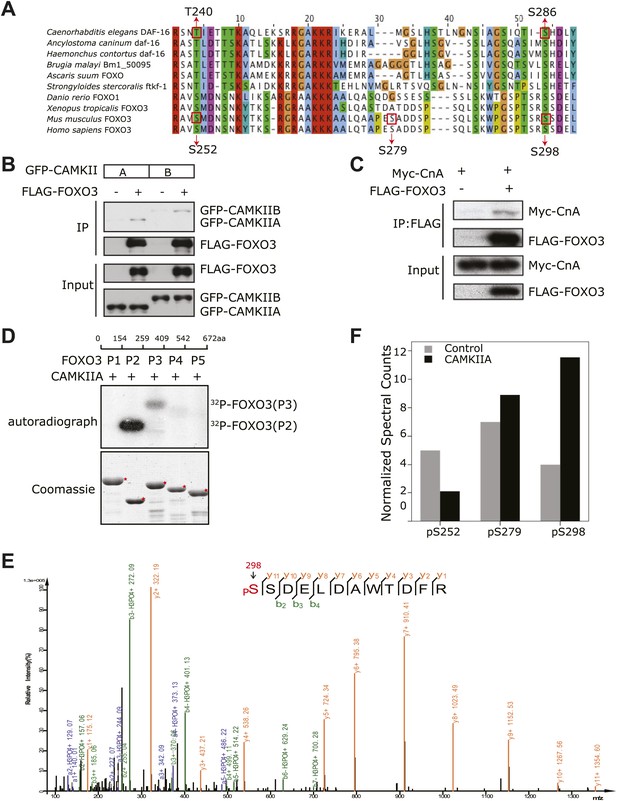
CAMKII and Calcineurin regulate phosphorylation of mouse FOXO3 at S298.
(A) Sequence alignment of selected FOXO homologs from nematode species to human. The region containing DAF-16 T240 and S286 is shown. T240 is conserved in all FOXO homologs. S286 is less conserved, but it is found in many nematode FOXOs and a number of vertebrate FOXOs. The DAF-16 or mouse FOXO3 residues phosphorylated in vitro by UNC-43 or CAMKIIA are boxed, with amino acid positions shown above or below. GFP-tagged mouse CAMKIIA or CAMKIIB, or Myc-tagged human Calcineurin A was transfected either alone or with FLAG-tagged mouse FOXO3 into HEK293T cells. Immunoprecipitation of FLAG-FOXO3 pulled down both CAMKII isoforms (B) and Calcineurin (C). (D) Two FOXO3 fragments P2 and P3, corresponding to amino acids 154–259 and 259–409, were phosphorylated in vitro by CAMKIIA in the presence of [32P]-γ-ATP. The input protein substrates are shown at the bottom. (E) Mass spectrum showing a FOXO3 peptide phosphorylated by CAMKIIA at S298. (F) CAMKIIA phosphorylates FOXO3 at S298 in vivo. FLAG-FOXO3 immunoprecipitated from HEK293T cells co-transfected or not with CAMKIIA was digested with trypsin and subjected to mass spectrometry analysis. Spectral counts (i.e., number of observations) of the indicated phospho-S/T are normalized to the total spectral counts of FLAG-FOXO3.
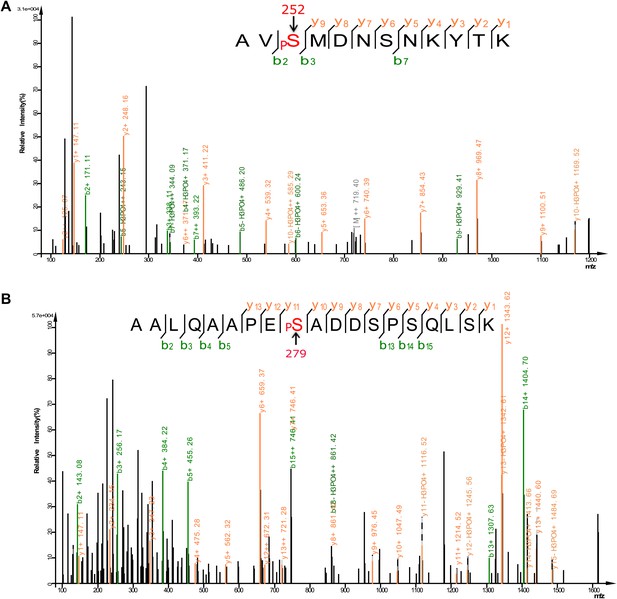
Analysis of FOXO3 phosphorylation sites in vivo.
Mass spectra showing two phosphorylation sites S252 (A) and S279 (B) identified from FLAG-mFOXO3 expressed in HEK293T cells. Mass spectrum identifying a third phosphorylation site S298 is shown in Figure 9E.
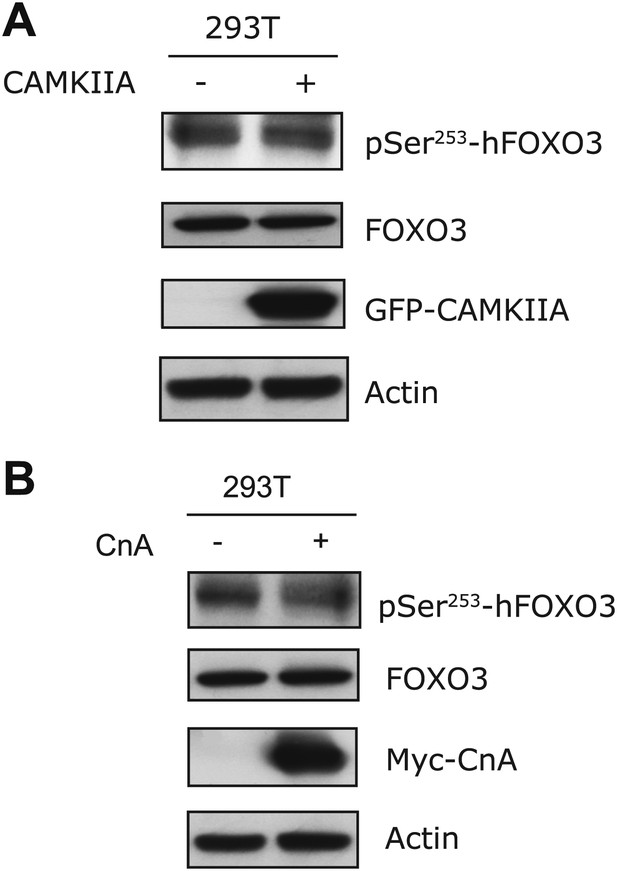
CAMIIA had no effect on S253 phosphorylation of human FOXO3 (corresponding to S252 of mouse FOXO3) in vivo.
(A) Transfection of CAMKIIA into HEK293T cells failed to increase phosphorylation of hFOXO3 at S253. (B) Overexpression of Calcineurin A into HEK293T cells did not reduce phosphorylation of human FOXO3 at S253, which was recognized using an antibody specific for S253 phosphorylation.
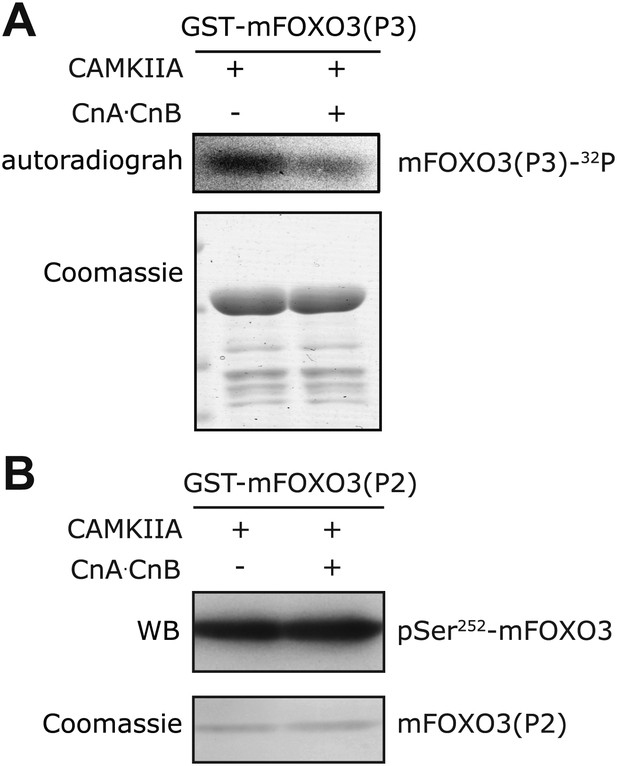
In vitro Calcineurin phosphatase assay on FOXO3.
(A) The S298-containing P3 fragment of mouse FOXO3 was first phosphorylated in vitro by CAMKIIA in the presence of [32P]-γ-ATP, then incubated with Calcineurin after the in vitro kinase reaction. Calcineurin markedly reduced the CAMKIIA-induced phosphorylation on P3. (B) The P2 fragment of mouse FOXO3 that was phosphorylated by CAMKIIA in vitro and then incubated with Calcineurin after inactivation of the kinase. Calcineurin had no effect on mFOXO3 S252 phosphorylation as visualized by immunoblotting with a phospho-S252 specific antibody.
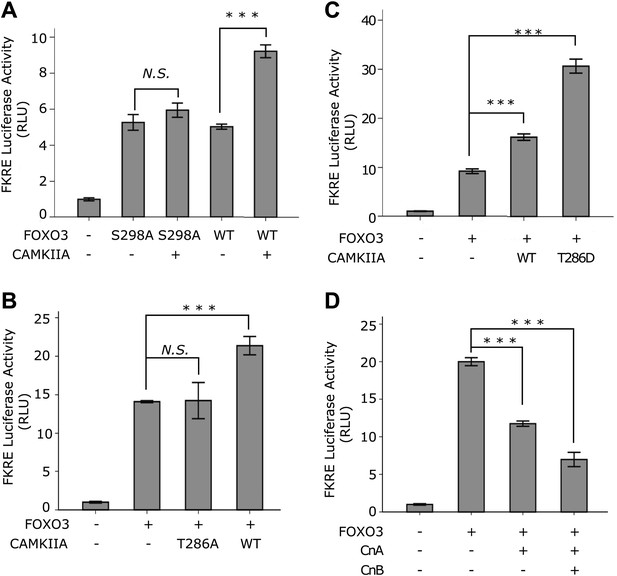
CAMKII and Calcineurin regulate the transcriptional activity of FOXO3.
(A)–(D) HEK293T cells were transfected with the indicated constructs together with a 3xIRS-firefly luciferase reporter and a TK-renilla luciferase reporter. Mean ± SD of firefly/renilla luciferase activity relative to the empty vector transfection is plotted (***p<0.0001, Student’s t-test, n = 3; N.S. for not significant). (A) Co-transfection of CAMKIIA stimulated the transcriptional activity of WT FOXO3 but not the FOXO3 (S298A) mutant. (B) WT but not an inactive T286A mutant CAMKIIA, transcriptionally activated FOXO3. (C) A constitutively active T286D mutant CAMKIIA further enhanced FOXO3 activity compared to WT CAMKIIA. (D) FOXO3 was transcriptionally inhibited by Calcineurin A, and further inhibited if Calcineurin A and Calcineurin B were both expressed.
Additional files
-
Supplementary file 1
(A) C. elegans strains used. (B) Oligos for genotyping.
- https://doi.org/10.7554/eLife.00518.027





