ATP-dependent chromatin assembly is functionally distinct from chromatin remodeling
Figures
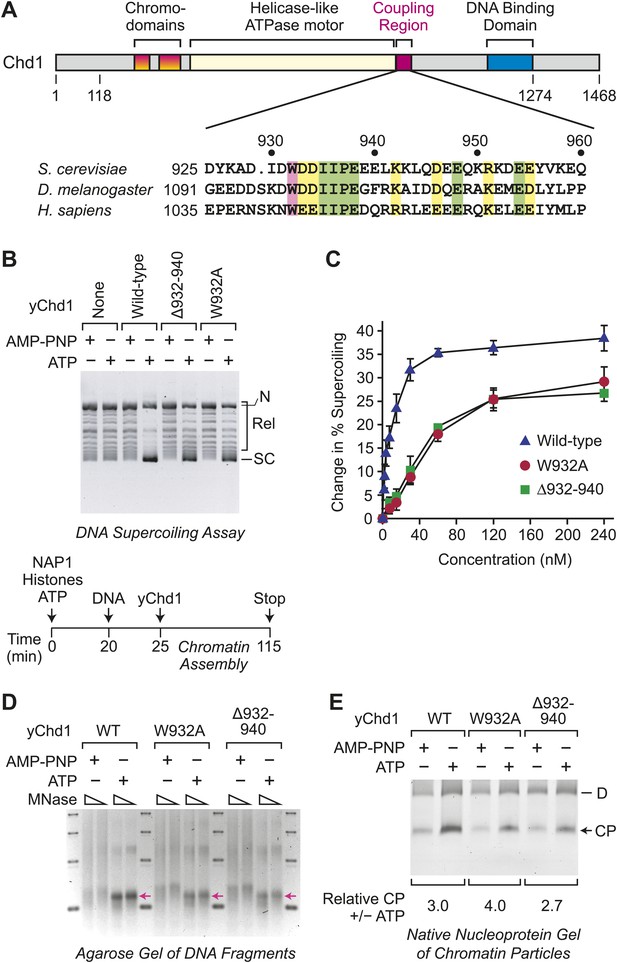
ATP-dependent nucleosome assembly is functionally distinct from chromatin remodeling.
(A) Diagram of Chd1 and the conserved coupling region. The numbers below the schematic diagram and above the amino acid sequences indicate positions in S. cerevisiae Chd1 (yChd1). (B) Chromatin remodeling-defective mutant yChd1 proteins (yChd1Δ932–940; yChd1W932A) can assemble nucleosomes in an ATP-dependent manner. Chromatin assembly reactions were performed with wild-type or mutant yChd1 proteins (120 nM) in the presence of either adenylyl-imidodiphosphate (AMP-PNP) or ATP. The efficiency of nucleosome assembly was monitored by the DNA supercoiling assay. The positions of supercoiled (SC), relaxed (Rel), and nicked open circular (N) DNAs are indicated. (C) Quantitative analysis of the efficiency of nucleosome assembly by mutant vs wild-type yChd1 proteins. Chromatin assembly reactions with yChd1 proteins were analyzed by the DNA supercoiling assay. The change in % supercoiling ([Δ supercoiled DNA/total DNA) × 100%) vs concentration of yChd1 (nM) is shown. The results are presented as the mean ± standard deviation (N ≥ 3). (D) Agarose gel electrophoresis of DNA fragments derived from chromatin assembled with wild-type or mutant yChd1 proteins. Chromatin assembly reactions were carried out as in (B), except that the concentration of Chd1 proteins was 60 nM. The reaction products were digested extensively with micrococcal nuclease (MNase) and subsequently deproteinized. The resulting DNA fragments were resolved on a 3% agarose gel and visualized by staining with ethidium bromide. The arrows indicate the position of DNA fragments derived from core particles. (E) Native nucleoprotein gel analysis of nucleosomes assembled with wild-type or mutant yChd1 proteins. Chromatin assembly reactions were carried out as in (D). The reaction products were digested extensively with MNase; the resulting nucleoprotein complexes were subjected to electrophoresis on a nondenaturing 5% polyacrylamide gel; and the DNA was stained with Sybr Green I (Invitrogen). The positions of core particles (CP) and dinucleosomes (D) are indicated.
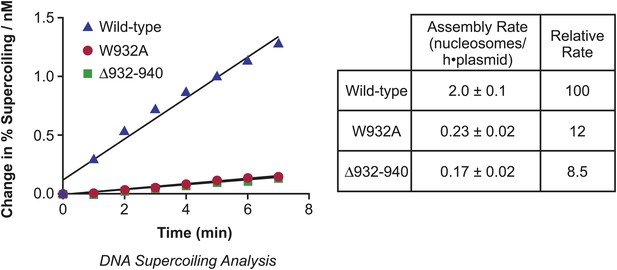
Analysis of the initial rates of nucleosome assembly by the wild-type, W932A, and Δ932–940 yChd1 proteins.
The initial rates were measured as change in % supercoiling ([Δ supercoiled DNA/total DNA] × 100%)/(nM protein) vs time (min). The table summarizes the nucleosome assembly rates as mean ± standard deviation (N = 3). The relative rates are given with respect to that of the wild-type protein.
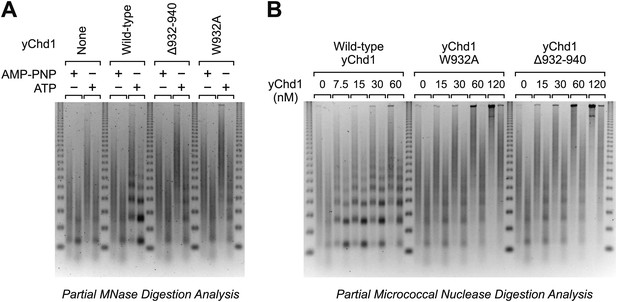
Chromatin remodeling-defective yChd1 proteins are unable to generate arrays of evenly-spaced nucleosomes during chromatin assembly.
(A) Chromatin assembly reactions were performed with wild-type or mutant yChd1 proteins (30 nM) in the presence of AMP-PNP or ATP. The reaction products were subjected to partial MNase digestion analysis. (B) Chromatin assembly reactions were performed with the indicated concentrations of yChd1 proteins and then subjected to partial MNase digestion analysis.
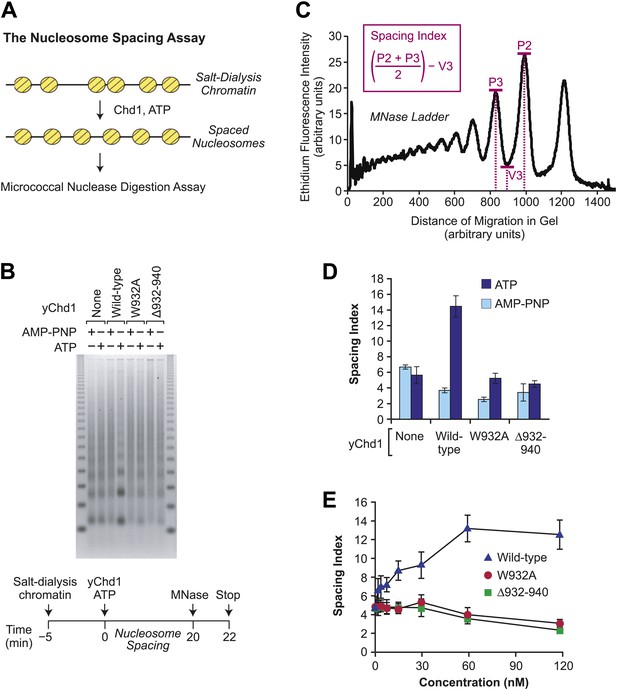
The chromatin remodeling-defective yChd1 proteins are not able to catalyze the formation of regularly-spaced nucleosomes.
(A) Schematic representation of the nucleosome spacing reaction. Randomly-distributed nucleosomes were generated by salt dialysis reconstitution of chromatin. Wild-type or mutant Chd1 protein was added along with ATP, and the reaction products were characterized by partial MNase digestion analysis. (B) Mutant yChd1 proteins do not reposition nucleosomes into periodic arrays. Spacing reactions were performed by incubating salt-dialysis chromatin with wild-type or mutant yChd1 proteins (30 nM) in the presence of either AMP-PNP or ATP. The reaction products were characterized by partial MNase digestion analysis. (C) Determination of the spacing index. Agarose gels from nucleosome spacing reactions were stained by ethidium bromide and then subjected to imaging and analysis on ImageQuantTL (GE) to obtain densiometry scans. The spacing index is the average height of the di- and tri-nucleosome peaks ([P2 + P3)/2) minus the height of the valley (V3) between the peaks, as indicated by the formula shown in the figure. (D) Quantitative analysis of nucleosome spacing by wild-type and mutant yChd1 proteins. The spacing indices were determined for the products of reactions such as those shown in (B). The results are presented as the mean ± standard deviation (N = 6). (E) The chromatin remodeling-defective yChd1 proteins exhibit little to no activity in the nucleosome spacing assay at different concentrations. Nucleosome spacing reactions were performed with yChd1 proteins and subjected to partial MNase digestion analysis. The graph depicts the average spacing index ± standard deviation (N = 4) vs concentration (nM) for each of the indicated proteins.
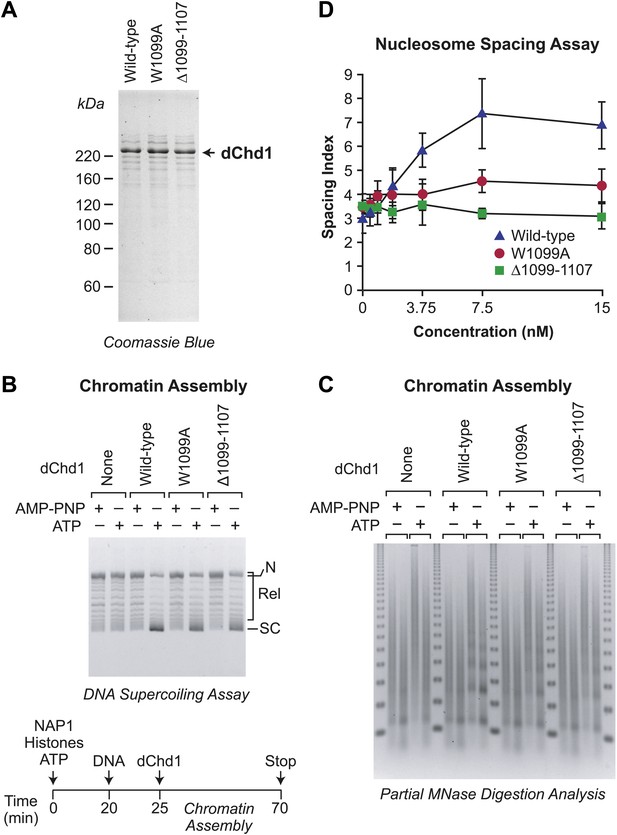
ATP-dependent nucleosome assembly is observed with chromatin remodeling-defective Drosophila Chd1 (dChd1) proteins.
(A) Purification of recombinant wild-type and mutant dChd1. The purified proteins were analyzed by polyacrylamide-SDS gel electrophoresis and visualized by staining with Coomassie Blue. (B) Chromatin remodeling-defective dChd1 proteins can assemble nucleosomes in an ATP-dependent manner. Chromatin assembly reactions were performed with wild-type or mutant dChd1 proteins (60 nM) in the presence of either AMP-PNP or ATP. The reaction products were analyzed by the DNA supercoiling assay. The positions of supercoiled (SC), relaxed (Rel), and nicked open circular (N) DNAs are indicated. (C) Chromatin assembly with remodeling-defective dChd1 proteins does not yield regularly-spaced nucleosomes. Chromatin assembly reactions were performed with wild-type or mutant dChd1 proteins (60 nM) in the presence of either AMP-PNP or ATP. Reaction products were subjected to partial MNase digestion analysis. (D) The chromatin remodeling-defective dChd1 proteins exhibit reduced nucleosome spacing activity. Spacing assays were performed with dChd1 proteins and subjected to partial MNase digestion analysis. This graph depicts the mean spacing index ± standard deviation (N = 3) vs concentration (nM).
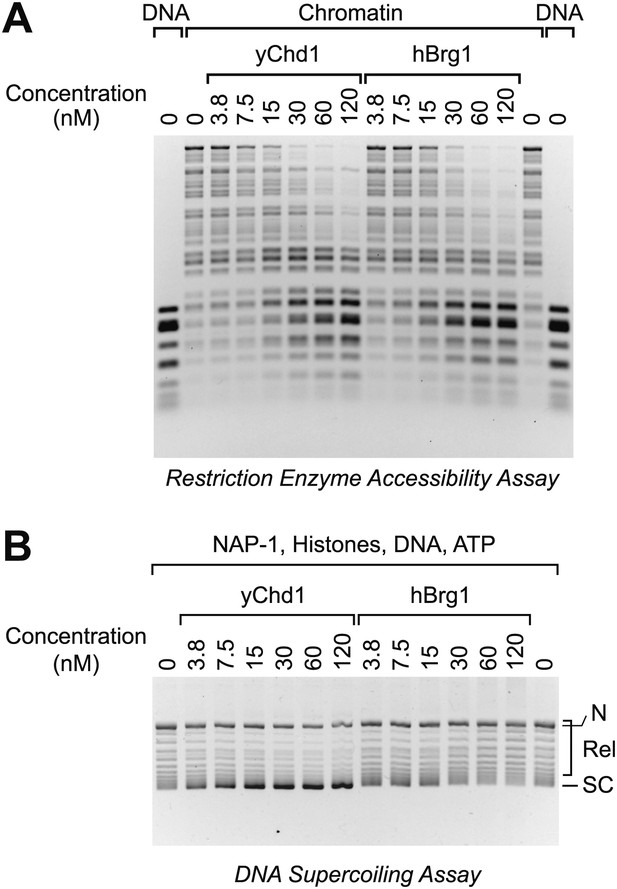
The Brg1 chromatin remodeling factor does not catalyze chromatin assembly.
(A) Purified human Brg1 (hBrg1) has a specific activity for chromatin remodeling that is similar to that of wild-type yChd1. Restriction accessibility assays were performed with salt dialysis-reconstituted chromatin with Hae III restriction enzyme and the indicated concentrations of either yChd1 or hBrg1. Naked DNA was used as a reference. After digestion with Hae III, the nucleic acids were deproteinized, subjected to agarose gel electrophoresis, and then visualized by staining with ethidium bromide. (B) Brg1 does not assemble chromatin. Chromatin assembly reactions were performed with the indicated concentrations of yChd1 or hBrg1, and the extent of chromatin assembly was monitored by the DNA supercoiling assay.
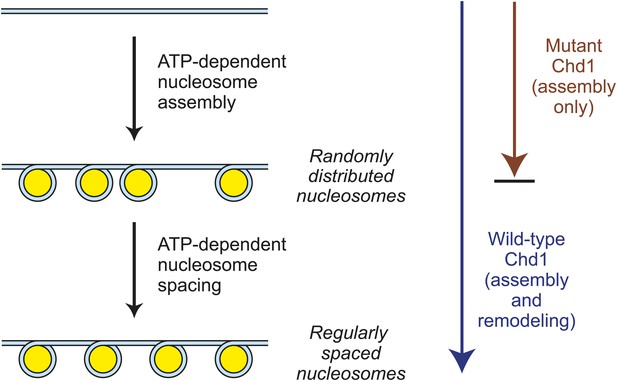
A model for the functions of ATP-driven nucleosome assembly and remodeling activities in the assembly of chromatin.
In this model, nucleosomes can be assembled by an ATP-dependent motor protein in the essentially complete absence of chromatin remodeling activity, as seen with the mutant Chd1 proteins. The resulting nucleosomes are, however, randomly distributed throughout the DNA template. These randomly-distributed nucleosomes can be converted into periodic nucleosome arrays by an ATP-dependent nucleosome remodeling (spacing) activity that can be distinguished from the chromatin assembly activity. These processes may occur concurrently with the wild-type Chd1.
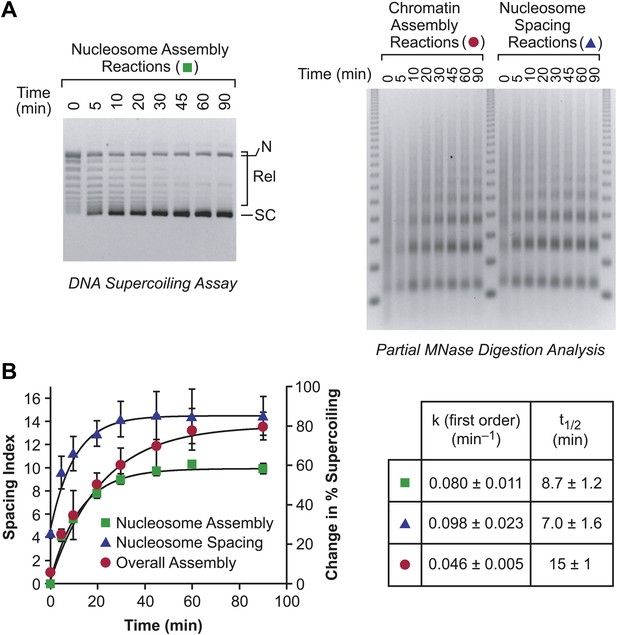
The individual rates of nucleosome formation and spacing are faster than the overall rate of assembly of periodic arrays of nucleosomes.
(A) Determination of the rates of nucleosome formation, nucleosome spacing, and assembly of regularly-spaced nucleosomes. Nucleosome formation in chromatin assembly reactions was monitored by using the DNA supercoiling assay. The positions of supercoiled (SC), relaxed (Rel), and nicked open circular (N) DNAs are indicated. Nucleosome spacing reactions were analyzed by partial MNase digestion analysis. The overall assembly of periodic arrays of nucleosomes was determined by performing chromatin assembly reactions and analyzing the reaction products by partial MNase digestion analysis. All reactions were performed with wild-type yChd1 at 30 nM. (B) Quantitation of the nucleosome formation and spacing assays, such as those shown in (A). The figure displays the change in % supercoiling ([Δ supercoiled DNA/total DNA] × 100%) and spacing indices vs reaction time (min). The reactions followed first-order kinetics, and the first-order rate constants (min−1) and half-times for reaction (t1/2, min) are given in the table. The data points are presented as mean ± standard deviation (N = 3), and are depicted with the plots of the first order curves (r2 > 0.95 for all graphs).
Tables
Nucleosome sliding and ATP hydrolysis activities of wild-type and mutant dChd1 proteins
| dChd1 protein | Nucleosome sliding activity | ATP hydrolysis activity (stimulated by DNA) | ATP hydrolysis activity (stimulated by nucleosomes) | |||
|---|---|---|---|---|---|---|
| kcat (min−1) | Relative rate (%) | kcat (min−1) | Relative rate (%) | kcat (min−1) | Relative rate (%) | |
| Wild-type | 2.2±0.2 | 100 | 61±2 | 100 | 115±3 | 100 |
| W1099A | 0.045±0.009 | 2.0±0.4 | 34±2 | 56±3 | 44±3 | 38±3 |
| Δ1099–1107 | 0.039±0.011 | 1.8±0.5 | 30±2 | 49±3 | 42±4 | 37±3 |





