Apoptotic cells can induce non-autonomous apoptosis through the TNF pathway
Figures
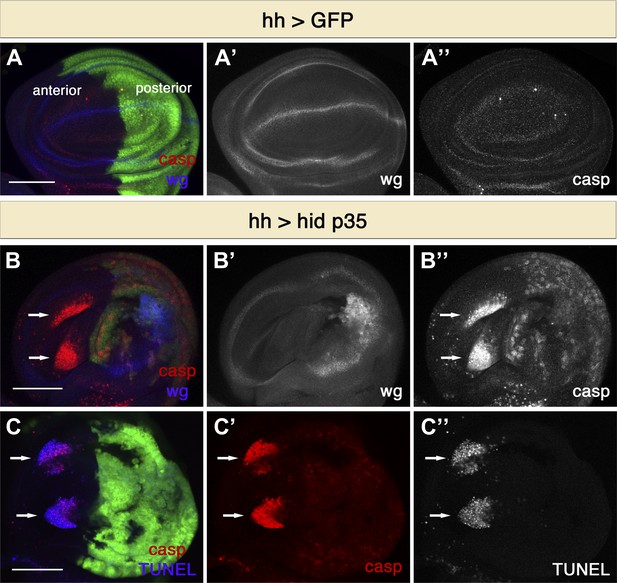
Undead cells promote apoptosis in neighboring cells.
(A) Wing disc of the genotype hh-Gal4>UAS-GFP showing wild-type wg expression (blue in A, white in A′) and normal levels of apoptosis, visualized by cleaved-caspase-3 (casp) staining (red in A, white in A′′). (B) Wing discs of the genotype hh-Gal4>UAS-GFP UAS-hid UAS-p35. Undead cells in the posterior compartment show ectopic wg activation (blue in B, white in B′) and are labeled by diffuse cleaved-caspase-3 staining (red in B, white in B′′). Non-autonomous apoptosis was observed in the anterior compartment (arrows). (C) Apoptotic cells in the anterior compartment (arrows) show cleaved-caspase-3 staining (red) and TUNEL staining (blue in C, white in C′′). Scale bars: 100 μm.
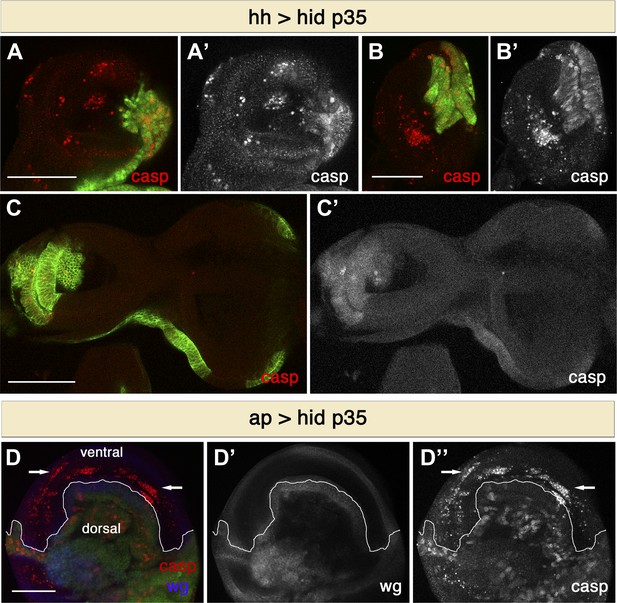
Non-autonomous apoptosis induced in different imaginal discs and with different drivers.
(A and B) Haltere (A) and leg (B) discs of the genotype hh-Gal4>UAS-GFP UAS-hid UAS-p35 also show non-autonomous apoptosis in the anterior compartment. (C) Eye-antennal discs of the genotype hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 (labeling with UAS-myr-mRFP is shown in green). In this case, non-autonomous apoptosis is not observed. (D) Wing disc of the genotype ap-Gal4>UAS-GFP UAS-hid UAS-p35. Undead cells in the dorsal compartment can be visualized by diffuse cleaved-caspase-3 staining (red in D, white in D′′). Non-autonomous apoptosis was observed in the ventral compartment (arrows). Scale bars: 100 μm.
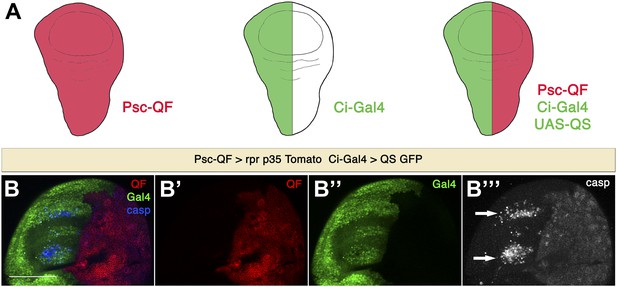
Apoptosis in the anterior compartment is non-autonomous.
(A) Combination of Q and Gal4 systems to independently control expression in the anterior and posterior compartments. We make use of the Psc-QF driver, which is expressed ubiquitously in the wing disc (red), and the Ci-Gal4 driver, which is expressed in the anterior compartment (green). By using Ci-Gal4 to drive expression of the QS suppressor (UAS-QS), QF expression can be restricted to the posterior compartment. In this way, we can control transgene expression independently in the anterior compartment (with the Gal4 system, green) and in the posterior compartment (with the Q system, red). (B) Wing discs of the genotype Psc-QF>QUAS-Tomato QUAS-rpr QUAS-p35 Ci-Gal4>UAS-GFP UAS-QS. QF expression is restricted to the posterior compartment (red in B and B′), while Gal4 expression can be visualized in the anterior compartment (green in B and B′′). Expression of rpr and p35 in the posterior compartment using the Q system leads to generation of undead cells in the posterior compartment and the induction of non-autonomous apoptosis in the anterior compartment, as shown by cleaved caspase-3 staining (blue in B, white in B′′′). Dying cells in the anterior compartment (arrows) are of anterior origin, as shown by the expression of Ci-Gal4>UAS-GFP. Scale bar: 100 μm.
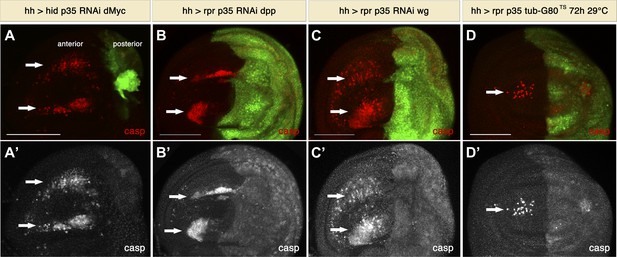
Signaling from apoptotic cells induces non-autonomous apoptosis.
(A) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-hid UAS-p35 UAS-RNAi dMyc. The posterior compartment is very reduced in size, but non-autonomous apoptosis is still present in the anterior compartment (arrows), as shown by staining with cleaved caspase-3 (red in A, white in A′). (B and C) Wing discs of the genotype hh-Gal4>UAS-GFP UAS-rpr UAS-p35 UAS-RNAi-dpp (B) and hh-Gal4>UAS-GFP UAS-rpr UAS-p35 UAS-RNAi-wg (C). Downregulation of the mitogenic signals Wg and Dpp produced by undead cells does not affect the amount of non-autonomous apoptosis, which is labeled with cleaved caspase-3 antibody (red in B and C; white in B′ and C′). (D) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-rpr UAS-p35 tub-Gal80TS grown at 29°C for the last 72 hr of larval development. Some apoptosis was still observed in the anterior compartment (arrows). Scale bars: 100 μm.
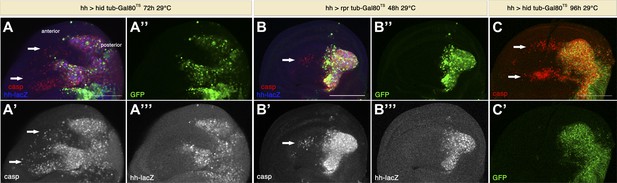
Genuine apoptosis also induces non-autonomous apoptosis.
(A) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-hid tub-Gal80 TS grown at 29°C for 72 hr. (B) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-rpr tub-Gal80TS grown at 29°C for 48 hr. (C) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-hid tub-Gal80TS grown at 29°C for 96 hr. In all cases, the posterior compartment is labeled by GFP expression (green). In A and B, the posterior compartment is also labeled with the hh-lacZ construct (blue in A and B, white in A′′′ and B′′′). Genuine apoptosis can be visualized in the posterior compartment by punctate staining with cleaved caspase-3 (red in A–C, white in A′ and B′). Non-autonomous apoptosis was also induced in the anterior compartment (arrows). Scale bars: 100 μm.
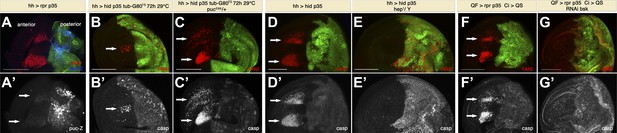
Apoptosis-induced apoptosis depends on JNK signaling.
(A) Wing disc of the genotype hh-Gal4>UAS-myr-mRFP UAS-rpr UAS-p35 pucE69-lacZ/+ grown at 25°C (labeling of the posterior compartment with UAS-myr-mRFP is shown in green). Non-autonomous apoptosis in the anterior compartment is visualized by staining with cleaved caspase-3 antibody (red). puc-lacZ expression (blue in A, white in A′) reveals very strong activation of JNK pathway in undead cells and modest activation in dying cells in the anterior compartment (arrows). (B) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-hid UAS-p35 tub-Gal80TS grown at 29°C for the last 72 hr of larval development. A reduced amount of non-autonomous apoptosis was observed in the anterior compartment (arrows), as shown by staining with cleaved caspase-3 antibody (red in B, white in B′). (C) Wing disc of the genotype hh-Gal4>UAS-GFP UAS-hid UAS-p35 tub-Gal80TS pucE69-lacZ/+ grown at 29°C during the same period of time. The amount of apoptosis in the anterior compartment was greatly increased (arrows). (D) Wing disc of the genotype hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 grown at 25°C (labeling of the posterior compartment with UAS-myr-mRFP is shown in green). Cleaved caspase-3 staining (red in D, white in D′) reveals a large amount of non-autonomous apoptosis in the anterior compartment (arrows). (E) Downregulation of the JNK pathway suppresses non-autonomous apoptosis, even though undead cells are still present in the posterior compartment, as shown by caspase-3 staining (red in E, white in E′). Genotype: hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 hep1/Y (mRFP is also shown in green). (F) Wing disc of the genotype Psc-QF>QUAS-Tomato QUAS-rpr QUAS-p35 Ci-Gal4>UAS-QS. Expression of the driver QF is restricted to the posterior compartment (labeling of the compartment with QUAS-Tomato is shown in green). Cleaved caspase-3 (red in F, white in F′) staining labels undead cells in the posterior compartment and apoptotic cells in the anterior compartment (arrows). (G) Inhibition of the JNK pathway specifically in the anterior compartment completely suppresses non-autonomous apoptosis. Genotype: Psc-QF>QUAS-Tomato QUAS-rpr QUAS-p35 Ci-Gal4>UAS-QS UAS-RNAi-bsk. Caspase-3 staining labels intact undead cells in the posterior compartment (red in G, white in G′). Labeling of the compartment with QUAS-Tomato is shown in green. Scale bars: 100 μm.
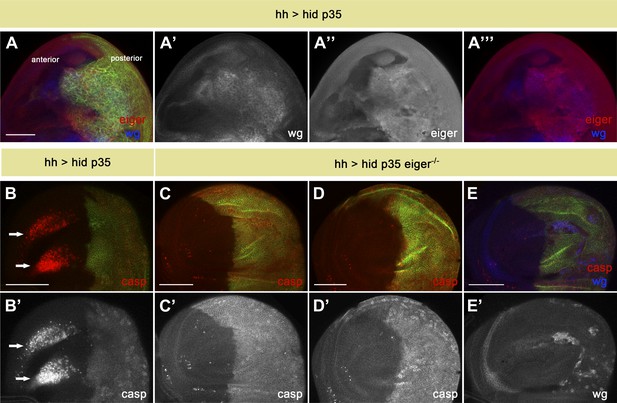
Eiger is responsible for apoptosis-induced apoptosis.
(A) Wing disc of the genotype hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35. The posterior compartment shows ectopic wg expression (blue in A and A′′′, white in A′). Eiger levels are also elevated in undead cells (red in A and A′′′, white in A′′). (B) Wing disc of the same genotype stained with anti-cleaved caspase-3 antibody (red in B, white in B′). Non-autonomous apoptosis in the anterior compartment is indicated by arrows. (C–E) Apoptosis-induced apoptosis is suppressed in an eiger mutant background, as shown by cleaved caspase-3 staining (red in C–E, white in C′ and D′). Ectopic expression of Wg is observed in the posterior compartment (blue in E, white in E′), suggesting that undead cells are not compromised in their signaling capacity under these conditions. Genotypes: hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 eiger1/eiger1 (C), hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 eiger1/eiger3 (D) and hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 eiger3/eiger3 (E). The posterior compartment is labeled with UAS-myr-mRFP (green) in all cases. Scale bars: 100 μm.
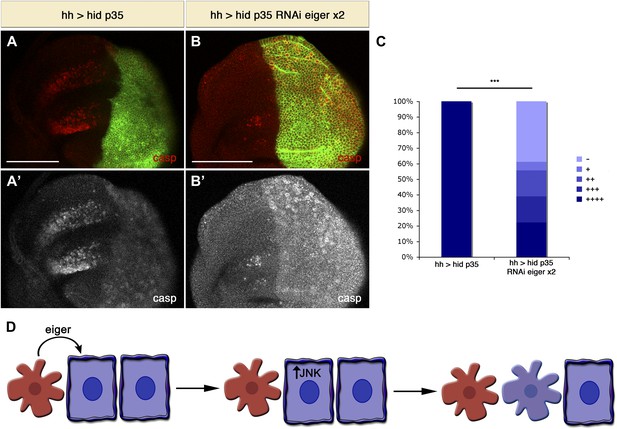
Downregulation of Eiger in undead cells significantly reduces apoptosis-induced apoptosis.
(A) Wing disc of the genotype hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 grown at 29°C. (B) Wing disc of the genotype hh-Gal4>UAS-myr-mRFP UAS-hid UAS-p35 UAS-RNAi-eiger KK108814 UAS-RNAi-eiger KK108814, also grown at 29°C. Apoptosis is shown in all cases by staining with cleaved caspase-3 antibody (red in A and B, white in A′ and B′). The posterior compartment is labeled with UAS-myr-mRFP (green). (C) Measurement of the levels of apoptosis-induced apoptosis in both conditions. The amount of non-autonomous apoptosis is graded in five different categories, from widespread apoptosis (++++) to no apoptosis (−). Downregulation of Eiger in the posterior compartment significantly decreases AiA. p<0.001. (D) Model for apoptosis-induced apoptosis. Apoptotic cells produce Eiger, the Drosophila TNF homolog, which activates the JNK pathway in neighboring cells, leading to cell death in a non-autonomous manner. Scale bars: 100 μm.
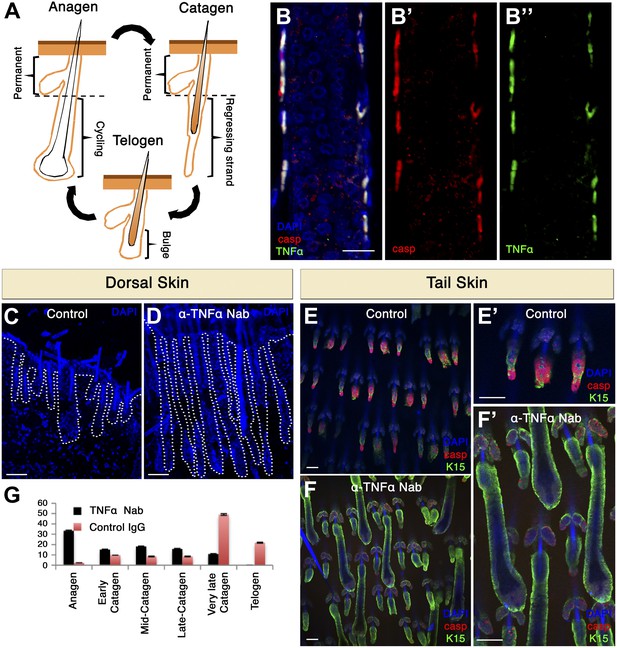
TNF-α is expressed in apoptotic HF cells during catagen and is essential for coordinated apoptosis and hair cycle progression in mice.
(A) Schematic diagram depicting the HF cycle. HFs cycle between phases of growth (anagen), destruction (catagen) and rest (telogen). During catagen, apoptosis leads to the degeneration of the lower two-thirds of the HF. Post-catagen, the HF enters the quiescent telogen phase, which later on enters a new cycle of hair growth (anagen). (B) Immunofluorescence staining indicating that apoptotic HF cells express TNF-α during catagen (P16) in wild-type conditions. Apoptotic cells are labeled with cleaved caspase-3 (red) and TNF-α staining is shown in green. (C–F) Neutralizing TNF-α in vivo impairs the HF cycle progression as a result of decreased apoptosis. (C and D) DAPI staining of dorsal skin demonstrates impaired HF cycle in response to TNF-α neutralization (P17.5). (E and F) Immunofluorescence staining of tail whole mounts indicates decreased cleaved caspase-3 staining (red) and desynchronization of the HF cycle in TNF-α neutralized mice (P17.5). (G) Statistical analysis of the HF phase in control and TNF-α inhibited mice. Scale bars: 20 μm in B, 100 μm in C–F.





