CO2 directly modulates connexin 26 by formation of carbamate bridges between subunits
Figures
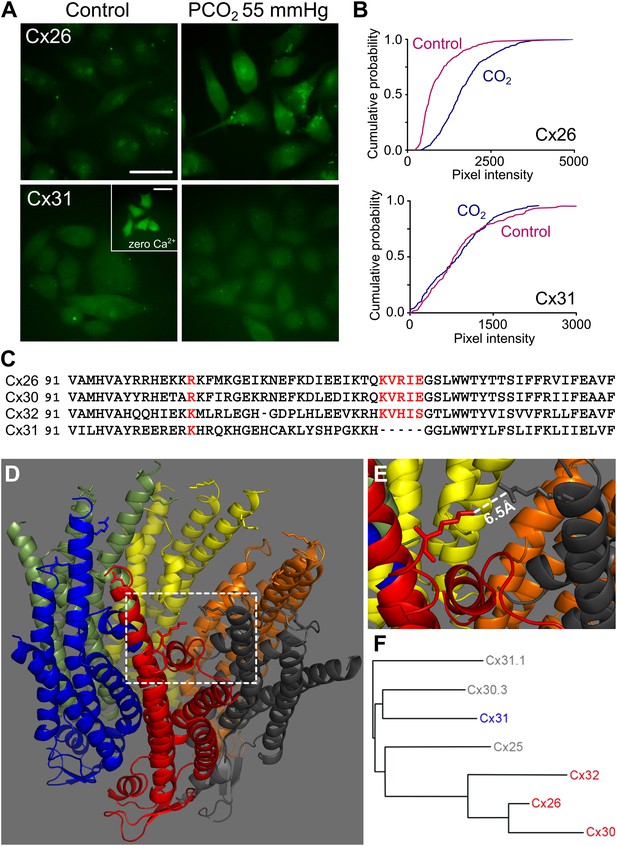
Identification of the motif in Cx26 that imparts CO2 sensitivity.
(A) Dye loading assay demonstrates CO2-dependent loading of carboxyfluorescein into HeLa cells expressing Cx26, but not into those expressing Cx31. The inset in Cx31 shows that these hemichannels are expressed and functional in the membrane by utilizing a zero Ca2+ stimulus to open them and allow dye loading. Scale bars 40 µm. (B) Cumulative probability plots of pixel intensity in the control and following exposure to PCO2 of 55 mmHg. Each curve is comprises the measurements of mean pixel intensity for at least 40 cells. (C) Sequences (from mouse) for Cx26, 30, 32 and 31 to show K125 and four following amino acids that are present in Cx26, Cx30 and Cx32, but absent from Cx31. R104 in Cx26 and 30, and K104 in Cx32 and Cx31 are also highlighted. Accession numbers: Cx26, NP_032151; Cx30, AAH13811; Cx32, AAH26833; and Cx31, NP_001153484. (D) The structure of Cx26 drawn from the 2zw3 PDB file, cytoplasmic face of the channel upwards. On each subunit K125 and R104 are drawn. (E) Detail from the structure of Cx26 (dashed square) showing the orientation of K125 (red) towards R104 (dark grey). The short distance between the two amino acid side chains suggests that this gap could be bridged by carbamylation by CO2 of K125 and a subsequent salt bridge with R104. (F) Phylogenetic tree showing relationship between Cx26 and other β connexins. The three CO2 sensitive connexins are very closely related to each other while Cx31 is more distant.
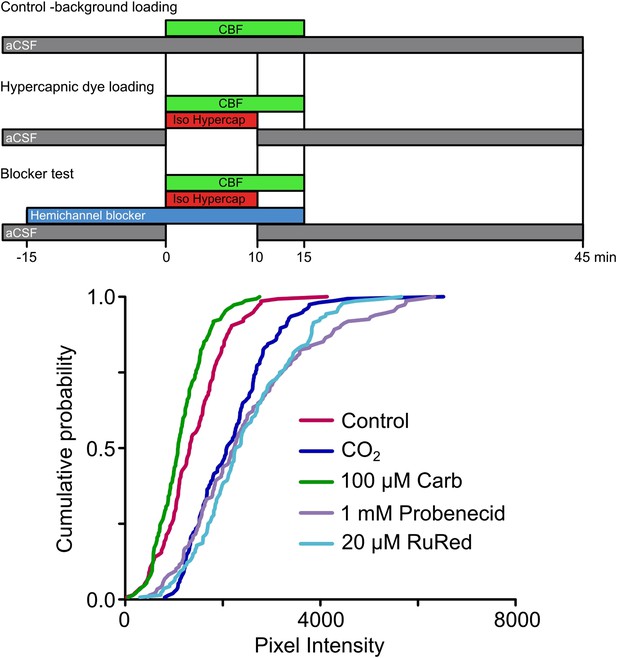
Expression of Cx26 in HeLa cells imparts sensitivity to CO2.
Top, diagram of dye loading protocol, showing the methods for assessing background dye loading under control conditions (no additional CO2), dye loading during a hypercapnic challenge (PCO2 55 mmHg) and the use of the hemichannel blockers to probe the identity of the hemichannel required for CO2 sensitive dye loading. Bottom, cumulative probability plots demonstrating that exposure of Cx26-expressing HeLa cells to elevated PCO2 caused dye loading compared to the background control. This CO2-dependent loading was blocked by 100 µM carbenoxolone (Carb), but unaffected by probenecid or ruthenium red (RuRed), demonstrating that the heterologously expressed Cx26 rather than any potential endogenous pannexin-1 or calhm1 was responsible for the CO2 sensitivity. Minimum of 40 cells measured from three independent repetitions of each treatment. The slight reduction in dye loading caused by carbenoxolone compared to the background control loading is to be expected, as Cx26 will partially gate in response to control aCSF which has a PCO2 of 35 mmHg. Parental HeLa cells do not exhibit CO2-dependent dye loading (Figure 3—figure supplement 1).
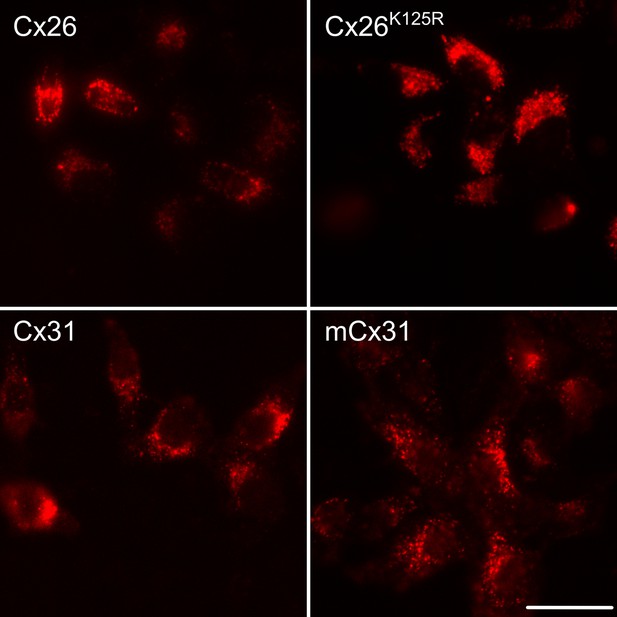
Expression of connexin variants in HeLa cells.
The connexin variants were tagged with mCherry, allowing verification of successful of expression. Examples of wild type Cx26 and Cx31 are shown along with two mutant variants. Scale bar 40 µm.
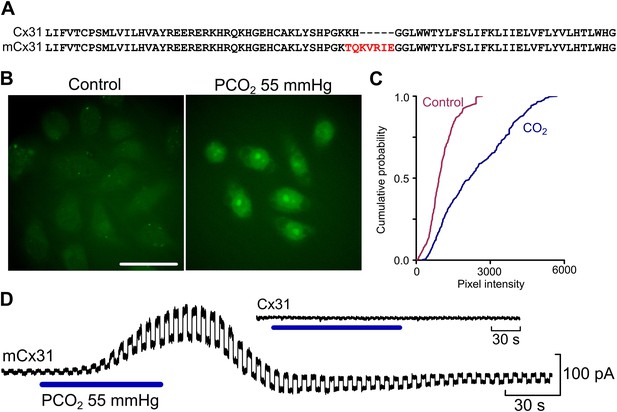
Insertion of the identified motif into Cx31 creates a CO2-sensitive hemichannel.
(A) Comparison of the WT and mutated Cx31 amino acid sequence to show the insertion of the K125 and surrounding residues. (B) The dye loading assay demonstrates gain of CO2-sensitivity in mCx31. Scale bar 40 µm. (C) Cumulative probability of mean pixel density of 40 cells in five independent replications. (D) Whole cell patch clamp recordings from HeLa cells expressing mCx31 and Cx31. Recordings were performed under voltage clamp at a holding potential of −50 mV with a constant 10 mV step to assess whole cell conductance. The cells expressing mCx31 show a clear conductance change on exposure to a change in PCO2, whereas cells expressing wild type Cx31 showed no such change (inset).
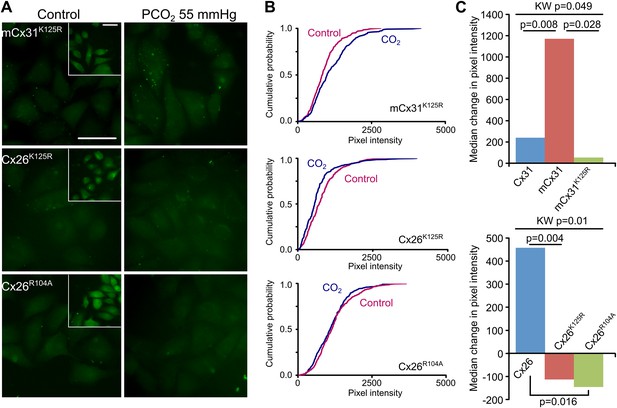
K125 and R104 are essential residues for CO2 sensitivity.
(A) Insertion of the motif (Figure 2A) from Cx26 but with the mutation K125R into Cx31 (mCx31K125R) does not give a gain of CO2 sensitivity indicating that this is an essential residue. Introducing the mutations K125R or R104A into Cx26 destroys the CO2-sensitivity of Cx26. Insets show the zero Ca2+ positive controls to demonstrate the presence of functional hemichannels in the cells. Scale bars, 40 µm. (B) Cumulative probability distributions demonstrate that none of these mutant channels are sensitive to CO2. (C) Summary data demonstrating: gain of function in the mCx31 hemichannel and subsequent loss in mCx31K125R; and loss of function in the Cx26K125R and Cx26R104A mutants. The graphs shown the median of the median change in pixel intensity from five independent replications for each type of hemichannel. KW: Kruskal-Wallis ANOVA, pairwise comparisons by the Mann-Whitney U-test.
-
Figure 3—source data 1
Median differences in pixel intensity between CO2 and control dye loading experiments for the various connexin hemichannel variants in the histograms of Figure 3C and statistical analysis: Kruskal-Wallis anova, pairwise Mann-Whitney tests and false discovery rate procedure.
- https://doi.org/10.7554/eLife.01213.008
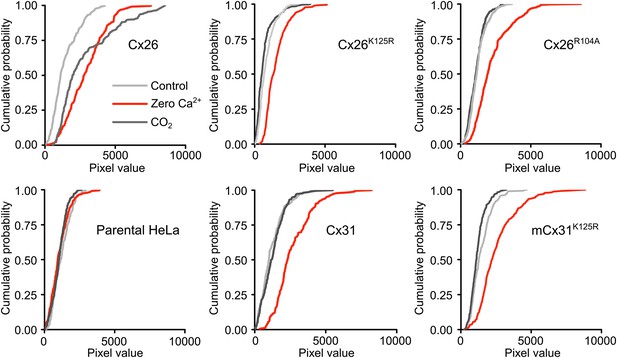
All connexin variants form functional hemichannels capable of opening in response to zero Ca2+.
Dye loading of wild type HeLa cells and HeLa cells expressing Cx26, Cx31 and the mutated versions used in this study. In all cases zero Ca2+ (red trace) increases the amount of dye loading in connexin expressing cells over the control (light grey trace). For reference the dye loading caused by CO2 is also shown (dark grey trace). Neither CO2 nor zero Ca2+ altered dye loading in parental HeLa cells devoid of heterologous connexin expression, demonstrating that these cells have virtually no endogenous hemichannels. For Cx26, the amount of dye loading caused by CO2 and zero Ca2+ was similar. In all other connexin variants zero Ca2+ caused dye loading but CO2 had no effect. The cumulative probability distributions comprise all measurements from five independent replications of every set of hemichannel variants that is the entire dataset arising from the image analysis of these connexin variants.
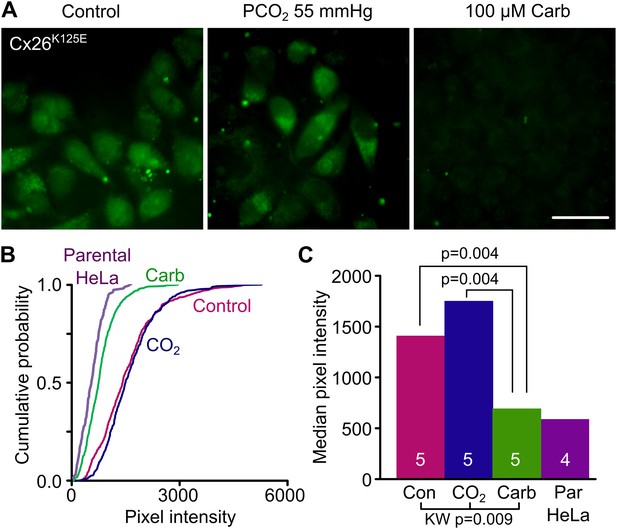
Engineering an analogue of the carbamylated lysine residue, Cx26K125E, creates a constitutively open hemichannel that no longer responds to CO2.
(A) HeLa cells expressing Cx26K125E readily load with dye under control conditions. Increasing the PCO2 does not give a further increase in dye loading. This dye loading was blocked by 100 µM carbenoxolone (Carb), indicating that it occurred through the heterologously expressed connexin. Scale bar 40 µm. (B) Cumulative probability plots comparing the median pixel intensities of at least 40 cells per experiment and five independent repetitions for the control, hypercapnic and carbenoxolone treatments with that of parental HeLa cells (four independent repetitions). (C) Summary data showing the median of the median pixel intensity for the three conditions for Cx26K125E and the background loading for parental HeLa cells. Pairwise comparisons by the Mann-Whitney U-test; KW Kruskall-Wallis Anova. Neither the difference between control and CO2 nor the difference between Cx26K125E treated with carbenoxolone and parental HeLa cells is significant.
-
Figure 4—source data 1
Median pixel intensity values for histogram in Figure 4C and statistical analysis: Kruskal-Wallis anova and pairwise Mann-Whitney tests.
- https://doi.org/10.7554/eLife.01213.011
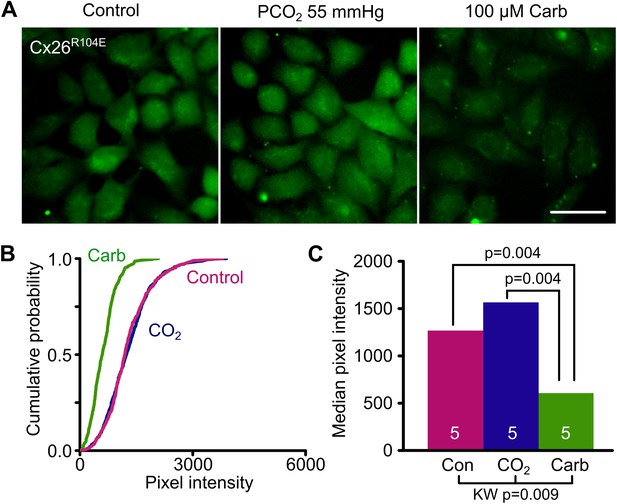
Bridging in the reverse direction: the mutation R104E forms a salt bridge with K125 in Cx26R104E to create a constitutively open hemichannel that no longer responds to CO2.
(A) HeLa cells expressing Cx26R104E readily load with dye under control conditions. Increasing the PCO2 does not give a further increase in dye loading. This dye loading was blocked by 100 µM carbenoxolone (Carb), indicating that it occurred through the heterologously expressed connexin. Scale bar 40 µm. (B) Cumulative probability plots comparing the median pixel intensities of at least 40 cells per experiment and five independent repetition for the control, hypercapnic and carbenoxolone treatments. (C) Summary data showing the median of the median pixel intensity for the three conditions for Cx26R104E. Pairwise comparisons by the Mann-Whitney U-test; KW Kruskall-Wallis Anova. The difference between control and CO2 is not significant.
-
Figure 5—source data 1
Median pixel intensity values for histogram in Figure 5C and statistical analysis: Kruskal-Wallis anova and pairwise Mann-Whitney tests.
- https://doi.org/10.7554/eLife.01213.013
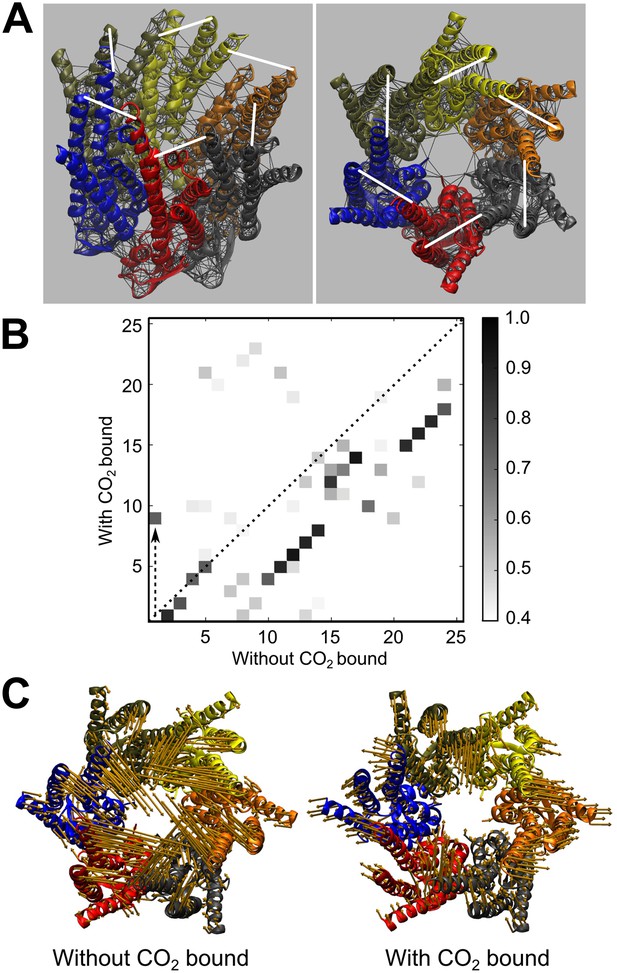
Elastic network model (ENM) of Cx26 demonstrating that CO2 binding restricts the motion of the hemichannel and biases it to the open state.
(A) Left, diagram of Cx26 from the 2zw3 structure, indicating the ENM (black lines) superimposed on the tertiary structure of Cx26 and showing the position of the hookean springs (white lines) introduced to simulate binding of CO2 to K125 and bridging to R104. Right, ENM of Cx26 seen end on from the cytoplasmic side of the channel showing the six springs (white lines) that represent CO2 binding. (B) Frequency modes of channel motion plotted for CO2 bound against those without CO2 bound. The grey scale on the right indicates the similarity of the modes between the CO2 bound and unbound states. Note that there is a high degree of similarity between most modes in the bound and unbound state, indicating that binding of CO2 reorders the modes of motion. In the graph, the modes that fall on the dotted line (x = y) have not changed between the two states. Mode 1 without CO2 bound (closing of hemichannel) moves to Mode 9 with CO2 bound (dashed upward arrow) indicating that it contributes much less to the total channel motions when CO2 is bound. Most of the other modes fall below the dotted line, indicating that they become more frequent when CO2 is bound. (C) Vectors indicating the Mode 1 motions of the α helices without CO2 bound (left) and with CO2 bound (right). The binding of CO2 and creation of the carbamylation bridge between subunits greatly restricts hemichannel motion.
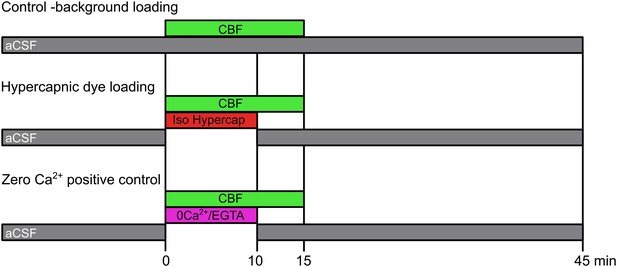
Dye loading protocols.
The control background loading tests for any potential CO2-insensitive pathways of dye loading that are constitutively active in the HeLa cells. Hypercapnic dye loading uses the 50 mM HCO3− aCSF to test CO2-sensitive loading under conditions of isohydric hypercapnia (PCO2 55 mmHg). The zero Ca2+ positive control tests for the presence of functional hemichannels in those cases where the misexpressed hemichannels exhibit no sensitivity to CO2.
Videos
Hemichannel mode 1 motions in absence of CO2.
https://doi.org/10.7554/eLife.01213.015Hemichannel mode 1 motions in presence of CO2.
https://doi.org/10.7554/eLife.01213.016Additional files
-
Supplementary file 1
Conductance changes source data.
Raw values for whole cell conductance changes (nS) in response to an isohydric CO2 challenge (PCO2 55 mmHg) in Cx31 and mCx31 expressing HeLa cells.
- https://doi.org/10.7554/eLife.01213.018





