A new topology of the HK97-like fold revealed in Bordetella bacteriophage by cryoEM at 3.5 Å resolution
Figures

Identification of BPP-1 capsid proteins.
(A) SDS-PAGE of BPP-1 virion proteins stained with Coomassie blue; lane 1: molecular mass standards; lane 2: BPP-1 virion proteins. The two most abundant proteins of the BPP-1 virion are identified by mass spectrometry (B) to be Bbp17 (MCP) and Bbp16 (CP) (indicated by arrows). (B) Mass spectrometry result of Bbp16 (CP). The sequence is shown with individual peptides identified by mass spectrometry drawn as lines below their corresponding sequences, with line thickness and darkness representing relative abundance in the mass spectrometry profiles (thicker lines mean more abundant). Arrows indicate the two peptide fragments that run past the end of the rows.
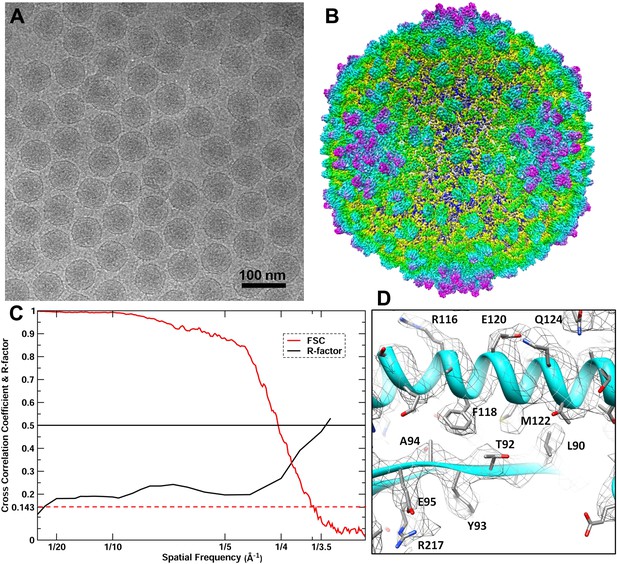
CryoEM reconstruction of the BPP-1 head at 3.5 Å resolution.
(A) Representative cryoEM image (defocus–1.6 μm) of the BPP-1. (B) CryoEM density of the BPP-1 head shown as radially-colored surface representation. See also Video 1. (C) R-factors (red) and Fourier shell correlation coefficients (FSC) (black) as a function of spatial frequency between maps from half datasets. (D) Close-up view of a local region of MCP, with densities of many amino acid side chains clearly resolved in both the helix and the loop. The atomic model is shown as ribbons and sticks with amino acid residues labeled.
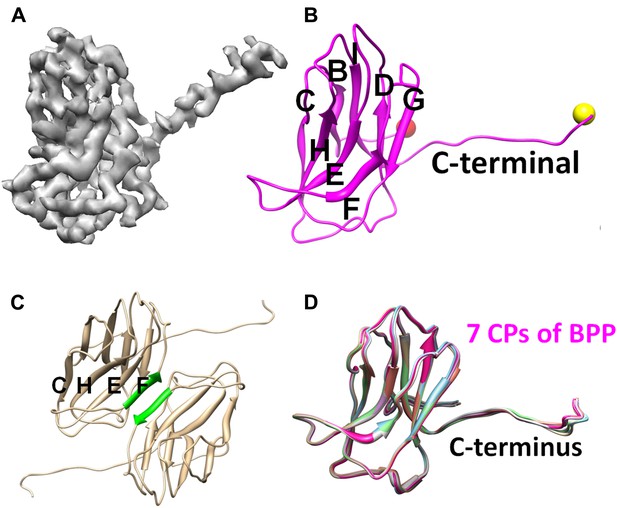
Structure of CP.
(A) CryoEM density map of CP (3.4 Å resolution, average of all seven conformers in an asymmetric unit). See also Video 2. (B) Ribbon model of CP, showing its jellyroll fold. Eight β-strands (B, C, D, E, F, G, H, I) fold into the two characteristic β-sheets (BIDG and CHEF), forming a ‘jellyroll’ (Harrison et al., 1978; Abad-Zapatero et al., 1980; Hogle et al., 1985; Rossmann et al., 1985). The N- and C-termini are marked by red and yellow balls, respectively. (C) The two F strands (green) of two neighboring CP monomers form hydrogen bonds in an antiparallel fashion, creating an augmented, 8-stranded β-sheet and a CP dimer. (D) Ribbon diagrams of the atomic models of the seven CP conformers of BPP-1.
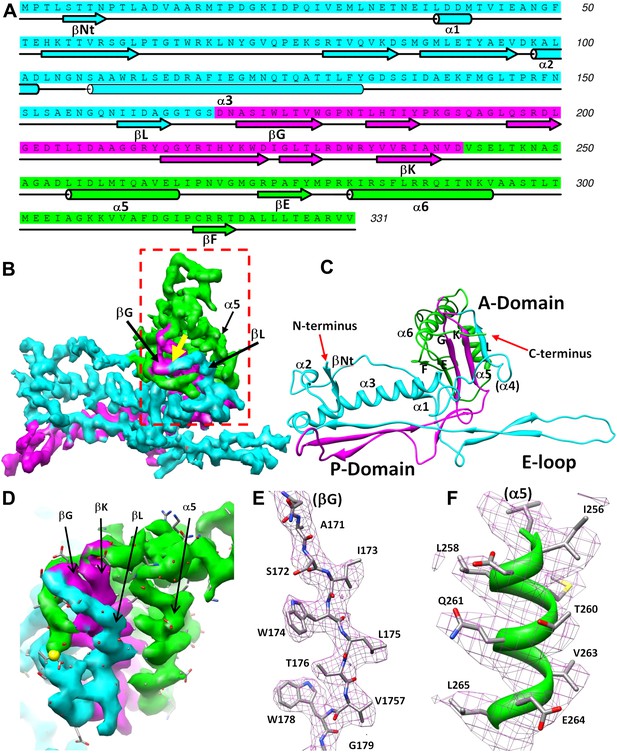
Structure of MCP.
(A) Sequence and secondary structure assignment of MCP. α-helices are marked by cylinders, β-strands by arrows, and loops by thin lines. The three structural elements of the Johnson fold, including N-, β- and α-elements, are shown in cyan, purple and green, respectively. (B) CryoEM density map of MCP (3.4 Å resolution, average of the six hexon MCP subunits in an asymmetric unit) shown as shaded surface using the same color coding of (A). The dashed box is shown in stereo in (D) contains the point of permutation (yellow arrow). See also Videos 3 and 4. (C) Ribbon diagram of the MCP atomic model with the three structural elements of the Johnson fold colored as in (A). (D) BPP-1 MCP density within the dash-box drawn in (B), showing the positions of the permutation (indicated by a yellow arrow in [B]) through which the N-element (cyan) is connected to the β-element (purple), instead of to helix α5 (green, far away), as in HK97 gp5. (E) CryoEM density (mesh) of the βG strand in (D) superimposed with its atomic model (sticks). (F) CryoEM density (mesh) of the α5 helix in (D) superimposed with its atomic model (ribbon with sticks).
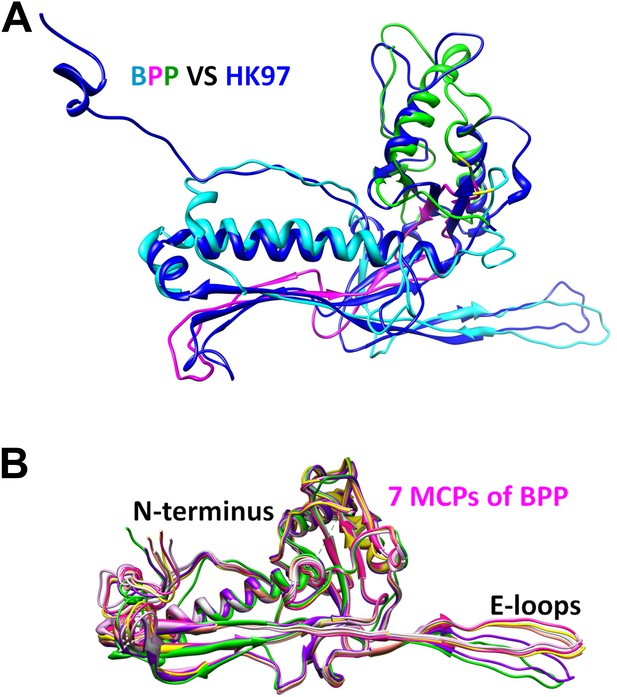
Structural comparisons between one hexameric MCP of BPP-1 and gp5 of HK97.
(A) Superposition of the structure of HK97 gp5 (blue) with that of BPP-1 MCP (cyan, purple, and green, highlighting its three structural elements). (B) Ribbon diagrams of the atomic models of the seven MCP conformers in each asymmetric unit of BPP-1.
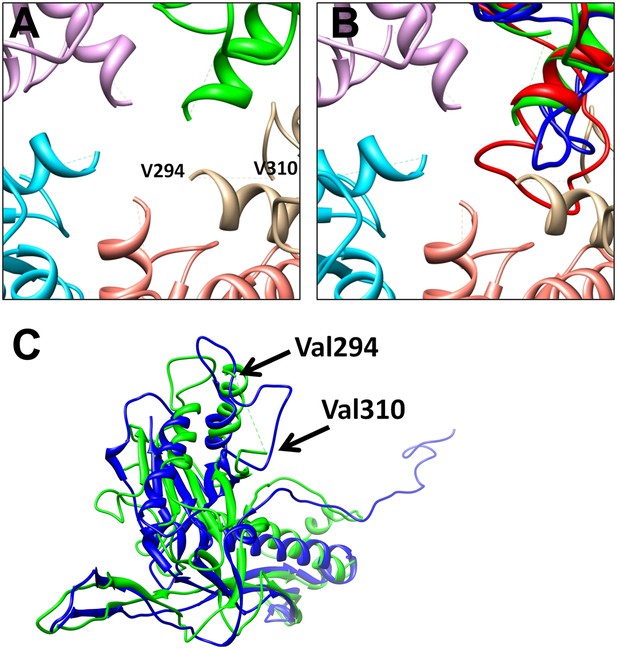
Differences between BPP-1 pentameric MCP and HK97 gp5.
(A) The central pore in a MCP pentamer of BPP-1. The loop A295-V309 in BPP-1 MCP is disordered while the counterpart in HK97 gp5 (blue in [B]) is ordered. (B) The same as (A) with the addition of a pentameric gp5 of HK97 (blue) and a hexameric MCP of BPP-1 (red) superimposed on one of the green pentameric MCP, showing the ordered loops of the pentameric gp5 and the hexameric BPP-1 MCP. (C) Superposition of the pentameric MCP of BPP-1 (green) and the pentameric gp5 of HK97 (blue).
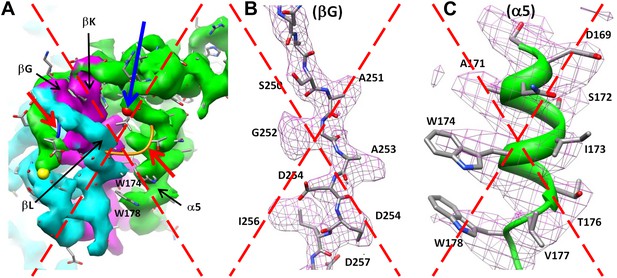
Incorrect MCP model obtained by enforcing the HK97 topology into the BPP-1 MCP cryoEM density, followed by five cycles of model refinement (‘interchanged model’).
We draw the large dashed crosses on the three panels to indicate that the model shown here is incorrect. (A) Superposition of the cryoEM density and the interchanged model for the region shown in Figure 4D. The two linking loops (indicated by the two red arrows) do not have matching cryoEM density. (B and C) Superposition of the cryoEM density (mesh) and the interchanged model (sticks and ribbons) for two regions indicated in (A): the βG strand (B) and α5 helix (C). Note the mismatches of side chain features here, as compared the nice match shown in Figure 4E,F.
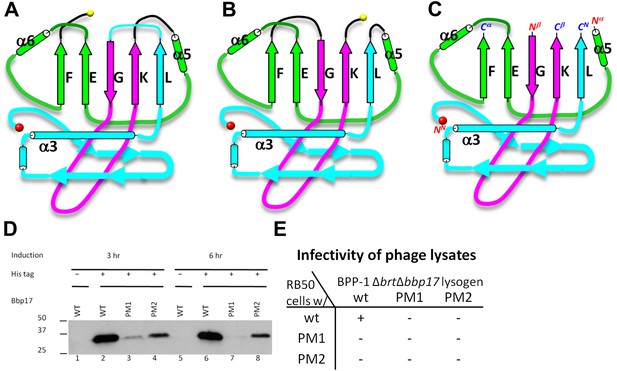
Two different topologies of the Johnson fold and structure-based mutagenesis of MCP.
(A) Diagram of the BPP topology of the Johnson fold: N- , β- , α-elements. (B) Diagram of the HK97 topology of the Johnson fold: N- , α- , β-elements. (C) Diagram of the three structural elements of the Johnson fold with free N- and C-ends. (D) Expression of BPP-1 MCP mutants with the β- and α-elements swapped, thus adopting the HK97 topology. Wild type (WT) and mutant (PM1 and PM2) BPP-1 MCPs with a 6xhistidine tag at the C-terminus were induced for expression from an fhaB promoter in RB50 cells for 3 (lanes 1–4) and 6 (lanes 5–8) hr, respectively. The expressions levels were determined by Western blot with a mouse anti-6xhistidine monoclonal antibody. Lanes 1 and 5 are negative controls with wild type Bbp17 that does not contain a 6xhistidine tag. (E) Infectivity of phage lysates as measured by their ability to form plaques on transformed RB50 cells. ‘+’: plaque observed; ‘−’: no plaque observed. The color scheme in (A–C) is the same as in Figure 4A–D.
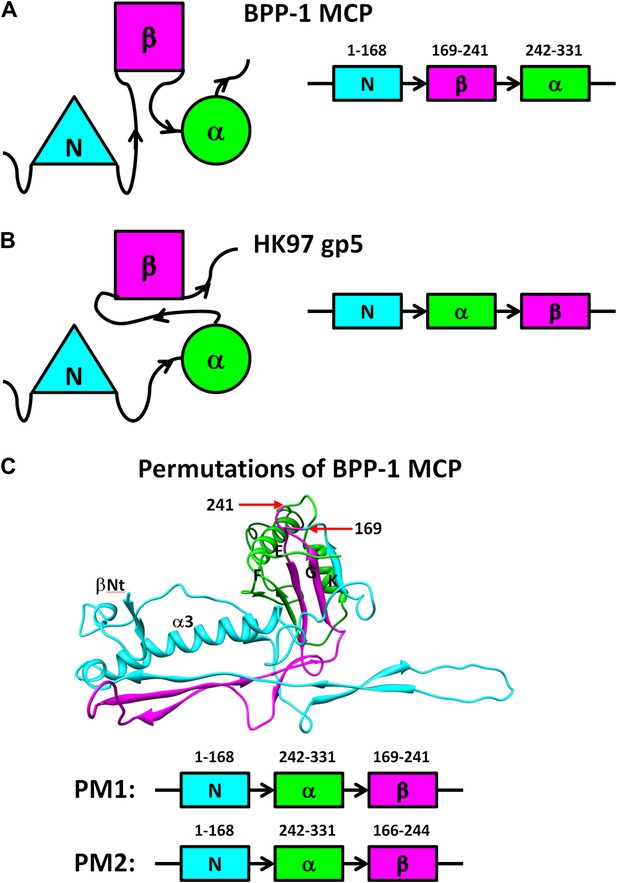
Diagram of non-circular permutation of the three structural elements of the Johnson fold in BPP-1 MCP and HK97 gp5.
(A) Folding topology of the three elements in the BPP-1 MCP. (B) Fold topology of the three corresponding elements in the HK97 gp5. (C) Illustration of the two BPP-1 MCP non-circular permutation mutations, PM1 and PM2, obtained by swapping the order of the β-element and the α-element to that of HK97 gp5. The three structural elements are shown in cyan, purple and green as in Figure 4 and the exact amino acid residue numbers at the points of swapping are indicated.
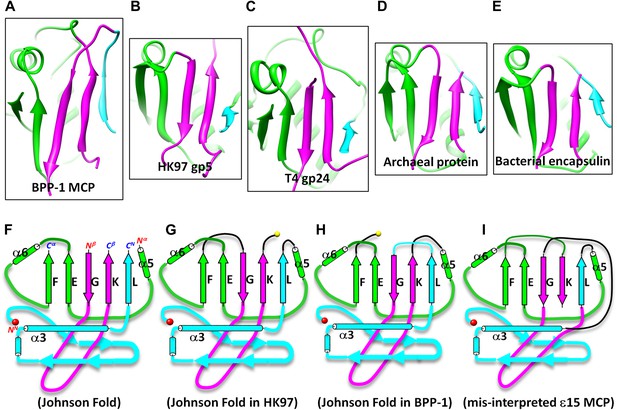
Johnson fold in HK97-like proteins.
(A) BPP-1 MCP. (B) HK97 gp55. (C) gp24 of T4 phage35. (D) The archaeal ‘particle-forming’ protein32. (E) Bacterial encapsulin33. (F) Spatial organization of the three structural elements of the Johnson fold. (G–I) Topologies of HK97 gp5 (G), BPP-1 MCP (H) and the mis-interpreted Epsilon15 gp7 (Jiang et al., 2008) (I), showing the conservation of the structural elements and the topology difference among the known HK97-like proteins. The three structural elements are marked with cyan, purple and green as in the Figure 4A–D.
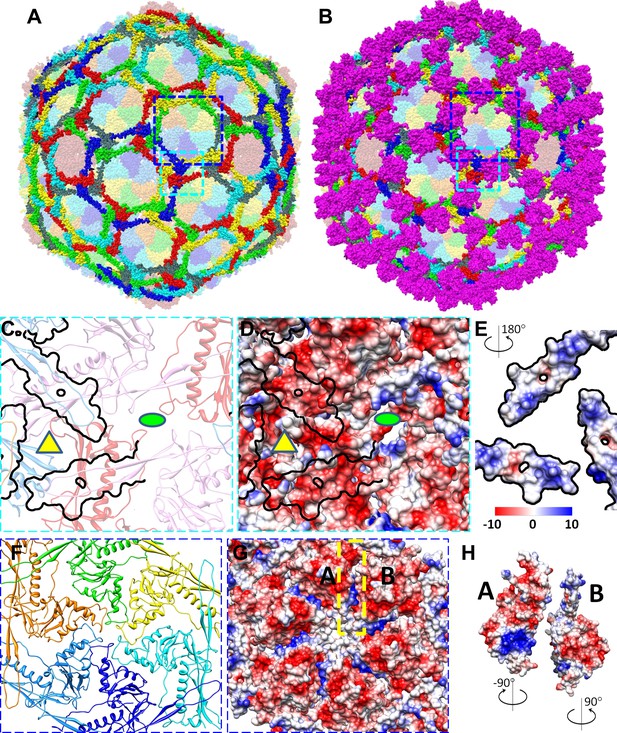
Non-covalent chainmail of the BPP-1 head.
See also Video 5. (A) Chainmail network formed by BPP-1 MCPs. The P-domain and E-loop of MCP in neighboring capsomers join head to tail to form rings (bright colors), which concatenate to form non-covalent chainmail. The P-domains and E-loops contributing to the formation of the same ring are shown in the same color. Other domains of MCP are dimmed. (B) Same view as in (A) but with CP dimers (purple) also shown. (C–E) Inter-capsomeric MCP-MCP interactions. The close-up view of all MCPs within the region within the cyan box in (A) illustrates the interaction interfaces (outlined by black lines) between the overlying E-loops of three MCP monomers and the underlying domains of other MCPs. These MCP monomers are shown as ribbons in different colors and belong to different hexon (or penton) capsomers. The local threefold and twofold axes are denoted by a yellow triangle and a green ellipse, respectively. Complementary electrostatic potentials are evident at the interaction interfaces, shown separately to reveal the surfaces of the underlying MCPs (D) and the 180°-rotated overlying E-loops (E), respectively. (F–H) Intra-capsomeric MCP–MCP interactions. The region within the blue box of (A) contains a hexon with its six MCP monomers shown either as ribbons in different colors (F) or as electrostatic potential surfaces (F). Adjacent MCP subunits within the hexon share one interaction interface (e.g., the yellow dashed rectangle for subunits A and B in F) and their complementary electrostatic potential surfaces are evident in their rotated views (H). The electrostatic potential scale is shown in the color bar in (E).
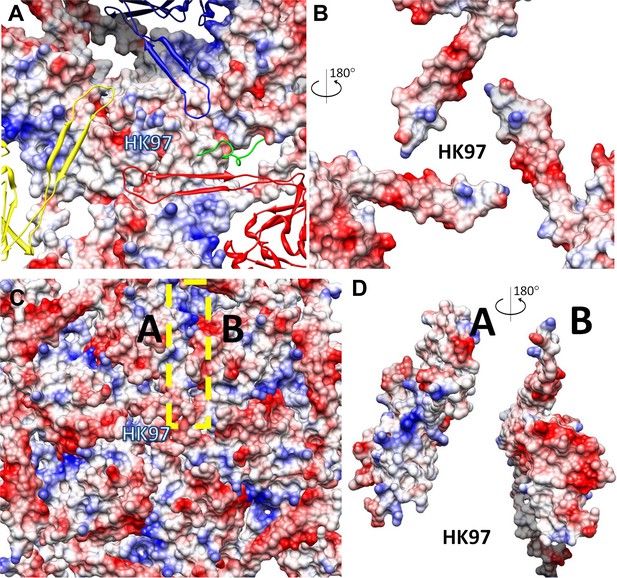
Inter-capsomer interactions in HK97 capsid.
(A) Electrostatic potential surface of the underlying HK97 gp5. Overlying molecules, including the E-loops of three gp5 (yellow, blue and red). The surface of MCP at this region has mainly positive charge (blue). The C-terminal loop (green, marked by the dashed cyan circle) of gp5 occupied a different region of the MCP shell and interacts with CP in BPP-1. (B) Electrostatic potential surface of the E-loops of the three overlying gp5 monomers, exhibiting mostly negative charge (red). The interacting surface of the three gp5 molecules has more positive charge, in contrast to the mostly negative charge as in BPP-1 MCP. The interacting surface of the gp5 E-loops has mostly negative charge, in contrast to the mostly positive charge as in BPP-1. (C and D) Interactions within a gp5 hexon of HK97. A region of HK97 capsid containing a single hexon with six gp5 monomers was displayed as electrostatic potential surfaces (C). Adjacent gp5 subunits within the hexon share one interaction interface (e.g., the yellow dashed rectangle for subunits A and B in C) and their complementary electrostatic potential surfaces are evident in their rotated views (D). The interacting interfaces are slightly different from those in BPP-1, but nonetheless, are also complementary to other another. The scale of electrostatic potential is the same as that in Figure 6H.
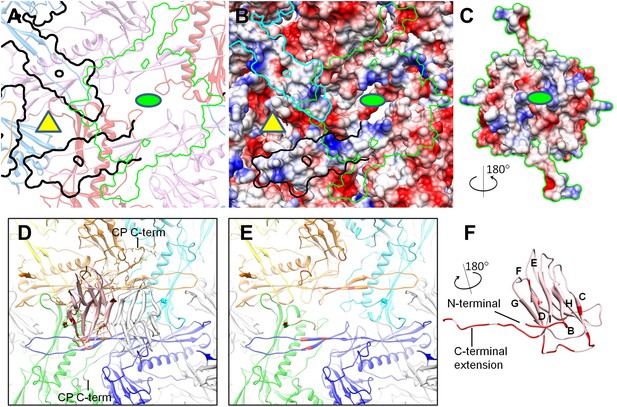
Interactions between CP and MCP.
(A–C) The same views as Figure 6C,D are shown in (A) and (B), respectively, except for the addition of a green outline depicting the interaction interface between a CP dimer and its underlying MCPs. At the interaction interface with MCPs, the electrostatic potential surface of the CP dimer (C) is complementary to that of its underlying MCPs (B). (D–F) Details of the interactions (segments highlighted in red) between CP and MCP. (D) The two monomers in the CP dimer are shown as pink and grey ribbons and the five underlying MCPs as yellow, orange, cyan, blue, and green ribbons. Side chains involved in interactions with the pink CP are shown displayed as sticks. Note the extensive interactions between the CP C-terminal extension and three different MCP monomers. (E) The same as (D) but without the overlying CP dimer and sticks of interacting side chains to better reveal MCP segments involved in the interactions (red). (F) The bottom view of the pink CP monomer to better reveal its segments (red) involved in CP–MCP interactions, which include its N-terminal loop, C-terminal loop, and the loop connecting strands C and D in the jellyroll.
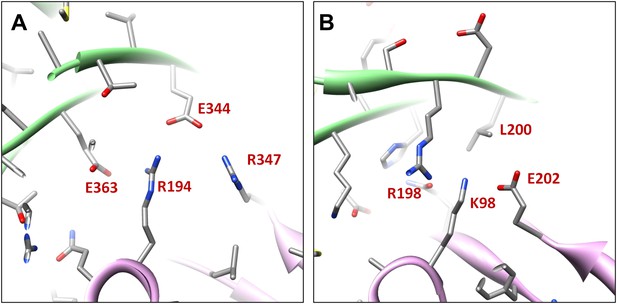
Salt bridges between adjacent HK97 gp5 molecules and lack of them in the corresponding positions in BPP-1 MCP molecules.
(A) Close-up view of two adjacent HK97 gp5 subunits, showing salt bridges between Arg194, Arg347 and Glu363 and Glu344. (B) There are no such salt bridges in the corresponding regions in the BPP-1 MCP molecules.
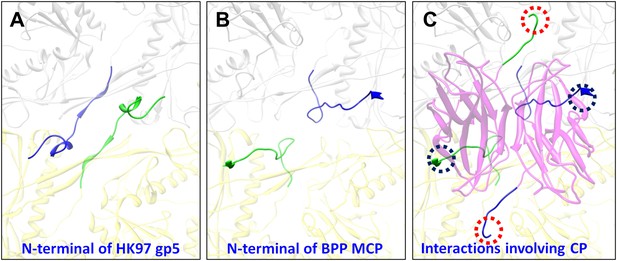
Interactions between CP and MCPs.
(A) Ribbon models of two adjacent HK97 gp5 subunits, showing the interaction of the N-terminal loop (bright colored) with an adjacent gp5 (dimmed). (B) BPP-1 regions corresponding to that in (A) showing the lack of the HK97 type of interactions between the MCP N-terminal loop (bright colored) and adjacent MCP molecules (dimmed). (C) The same region in (B) but with the CP dimer (purple), showing that the N-terminal loop of MCP hydrogen-bonds to β-strand C of CP (black circles) to form an augmented 10-stranded β sheet, and that the C-terminal loop (red circles) of each CP has extensive interactions with nearby MCP molecules.
Videos
Shaded surface view of the cryoEM density of the BPP-1 capsid at 3.5 Å resolution.
The map is color-coded according to the radius. Related to Figure 2.
Shaded surface view of the cryoEM density of the averaged CP at 3.4 Å resolution (gray) superimposed with the atomic model CP (ribbon and sticks).
Related to Figure 3.
Shaded surface view of the cryoEM density of the averaged MCP at 3.4 Å resolution.
The three structural elements are color coded (cyan, purple and green) according to the structural elements of the Johnson fold as illustrated in Figure 4. Related to Figure 4.
CryoEM density of MCP around the positions of permutation.
The three structural elements are color coded (cyan, purple, and green) according to the structural elements of the Johnson fold as illustrated in Figure 4. Related to Figure 4.
Non-covalent chainmail formed by MCPs on the BPP-1 capsid.
For clarity, only the P domain and the E-loop of MCP subunit are shown. These domains from different capsomers join in a head to tail fashion to form concatenated rings. Ball-and-stick models are produced from our atomic model of BPP-1 and those in the same ring are rendered in the same color. Related to Figure 6.
Tables
Statistics of atomic model refinement with Phenix
| CP (Bbp16) | MCP (Bbp17) | Asymmetric unit (7CPs+7MCPs) | |
|---|---|---|---|
| Residues resolved | 1–140 | 7–331 | 1–140 (CP); 5–331 (MCP) |
| Resolution (Å) | 3.4 | 3.4 | 3.5 |
| Rwork (overall: 40–3.4 Å) | 0.28 | 0.27 (0.31*) | 0.26 |
| Rfree (overall: 40–3.4 Å) | 0.28 | 0.28 (0.30*) | 0.26 |
| Rwork (best resolution zone) | 0.47 (1/3.4 Å) | 0.50 (0.52*) (1/3.4 Å) | 0.51 (1/3.5 Å) |
| Rfree (best resolution zone) | 0.45 (1/3.4 Å) | 0.45 (0.50*) (1/3.4 Å) | 0.47 (1/3.5 Å) |
| Ramachandran plot values | |||
| Most favored (%) | 85.5 | 86.7 | 88.0 |
| Generously allowed (%) | 12.3 | 11.8 | 10.3 |
| Disallowed regions (%) | 2.2 | 1.5 | 1.7 |
-
*
R-factors of the interchanged model by forcing the BPP-1 MCP to trace the HK97 topology.





