Identification of Redeye, a new sleep-regulating protein whose expression is modulated by sleep amount
Figures
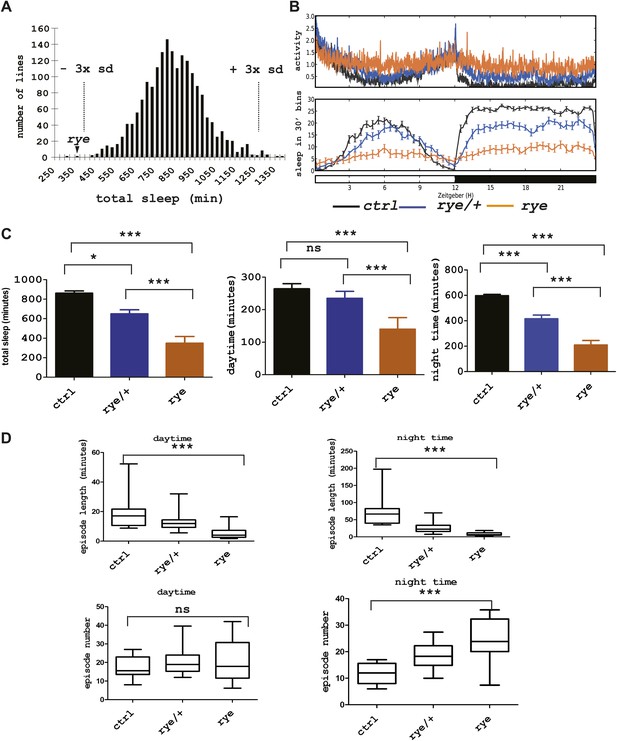
Identification of a short sleep mutant, redeye (rye), through a chemical mutagenesis (EMS) screen.
(A) Histogram depicting average sleep levels in females from homozygous EMS-mutagenized lines (n = 1857). The mean sleep value for each mutant was calculated by assaying sleep in 4–8 individual flies in the presence of 12 hr L-12 hr D cycles. Average sleep for all lines is indicated on the X axis in increments of 25 min. The Y axis depicts the number of mutant lines within each group. The dashed lines mark the sleep values that correspond to plus/minus 3× standard deviation of the mean. The arrowhead indicates the redeye mutant. (B) Top: average activity pattern of females from control lines and from rye mutants recorded in 12hr L-D cycles (n = 14–16 in 5 days). Bottom: sleep profiles with standard error bars. Black: lines isogenized on the 3rd chromosome for chemical mutagenesis; Blue: flies heterozygous for rye and the isogenized control chromosome; Orange: rye homozygotes. (C) Mean values of total sleep, daytime sleep and night-time sleep for rye mutants (Figure 1—source data 1). For each genotype 14–16 flies were assayed over a 5 day period. Bars represent standard error. One-way ANOVA was performed followed by Tukey post hoc analysis. * represents p<0.05, **p<0.01 and ***p<0.001. (D) Sleep quality of rye mutants: Daytime and night-time sleep episode length and episode number are plotted in the box-and-whisker diagram (Figure 1—source data 1). The middle line represents the median value; Bottom and top line of each box represent 25% and 75% respectively; Bottom bar and top bar represent 5% and 95% respectively. ns: not significant, ***p<0.001.
-
Figure 1—Source data 1
Measurement of sleep duration for all EMS mutants and sleep analysis of rye mutants.
- https://doi.org/10.7554/eLife.01473.004
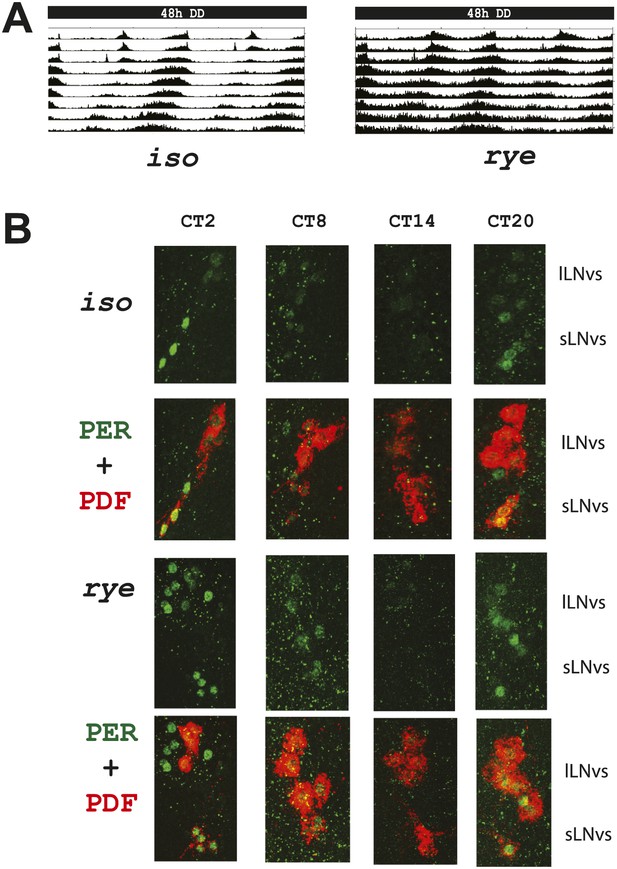
rye homozygotes display normal circadian rhythms and clock protein cycling.
(A) Double plotted activity records of iso controls and rye homozygotes in DD. Rhythmic, but prolonged cross-beam activity was recorded in rye homozygotes. (B) Immunostaining of PERIOD in ventral lateral neurons (LNvs) of iso controls and rye homozygotes at CT time points on the second day of DD following LD entrainment. PER oscillates in these clock neurons in iso as well as in rye homozygotes. PDF labels the small and large LNvs.
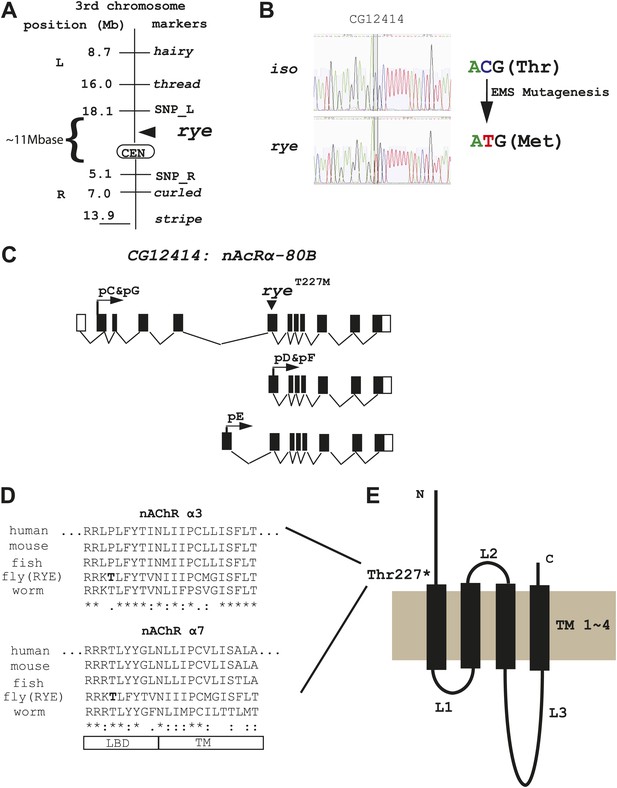
Genetic mapping and genome-wide deep-sequencing reveal a missense mutation in a nicotinic acetylcholine receptor α subunit gene in rye mutants.
(A) Through genetic mapping, using classical phenotypic markers and newly identified SNP markers, rye was mapped between two SNP markers (SNP_L and SNP_R) near the centromere on the 3rd chromosome. (B) Paired-end genomic deep-sequencing of rye mutants identified a missense mutation in CG12414. We confirmed this through Sanger sequencing. This C-T/G-A transition that generated the ryeT227M allele is typical of EMS-induced mutations. (C) Schematic representation of the rye candidate gene, a nicotinic acetylcholine receptor (CG12414: nAcRα-80B). The gene spans ∼100 kb with alternative splicing predicted to produce five transcripts (RC-RG). The spliced forms are predicted to translate into three proteins isoforms (pC-pG). pC(pG) produces the largest protein with the longest ligand binding domain (LBD). (D) Alignment of the partial sequence of Drosophila RYE with nAChR α3 and α7 subunits in other animals using ClustalW2. The region shown is at the boundary of the ligand binding domain (LBD) and transmembrane domain (TM), and is evolutionarily conserved. T227 in RYE is marked in bold form. ‘*’ identity; ‘:’ high similarity; ‘.’ similarity. (E) Protein sequence analysis predicts four transmembrane domains (TM) in RYE, which is typical of most nAChR proteins. RYE appears to contain a single ligand binding domain (LBD) in the extracellular region, and four TMs with three loop regions (L1-L3). The mutated threonine227 is in the LBD, close to the beginning of the TM.
-
Figure 3—Source data 1
Recombination mapping of the rye mutation using a chromosome marked with visible markers h,th,cu,sr,e, and using SNP markers.
- https://doi.org/10.7554/eLife.01473.008
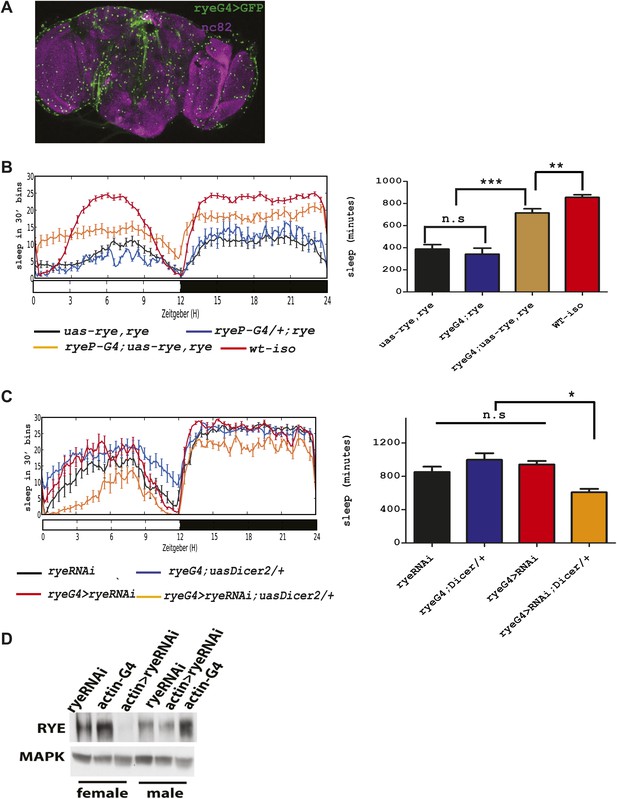
An alpha subunit of the nicotinic acetylcholine receptor accounts for the rye mutant phenotype.
(A) Expression pattern of ryeP-GAL4. Rye-GAL4 was used to express nGFP, which is visualized with an anti-GFP antibody. The anti-nc82 staining marks the neuropil. (B) Expression of the alpha subunit, as described in Figure 3, increases sleep duration in rye mutants (Figure 4—source data 1). Left: sleep profiles, with standard error, of rye mutants and mutants expressing a UAS construct of the putative rye cDNA under control of its own promoter (ryeP-Gal4) in a 12:12 LD cycle. Right: quantification of sleep length. **p<0.01, ***p<0.001. (C) Reduction of rye expression in rye neurons through the expression of an RNAi construct, together with Dicer2, diminishes sleep length (Figure 4—source data 1). Left: sleep profile in a 12:12 LD cycle. Right: quantification of sleep length. *p<0.05. (D) Western blot analysis shows reduced expression of RYE when actin-GAL4 is used to drive rye RNAi (VDRC#11392) with Uas-Dicer2 in female and male flies.
-
Figure 4—Source data 1
Sleep behaviour of transgenically rescued rye mutants and rye RNAi lines.
- https://doi.org/10.7554/eLife.01473.010
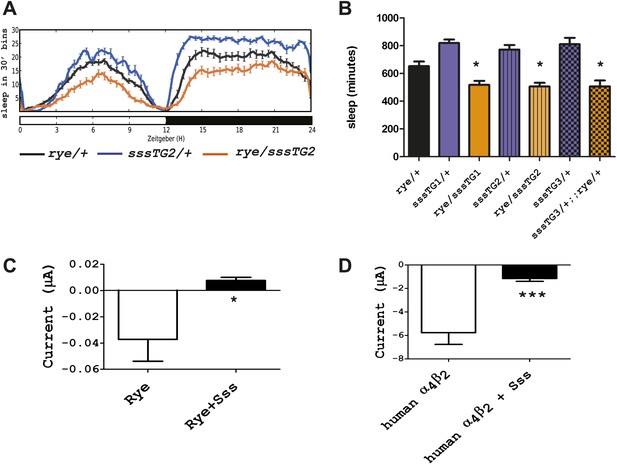
Overexpression of sss exacerbates the rye phenotype by repressing RYE activity.
(A) An extra copy of SSS reduces sleep in rye heterozygotes. Sleep profile of rye heterozygotes with or without a genomic sss transgenic line inserted on the third chromosome (sssTG2) and of sss transgenics alone. (B) Quantification of sleep in the genotypes as indicated (Figure 5—source data 1). TG1 and TG3 are additional independent genomic sss transgenes inserted on the 3rd and X chromosomes respectively. Sleep reduction was observed in rye transheterozygotes with all three sss transgenic lines. *p<0.05. (C) Heterologous expression of SSS reduces current following ACh application in Xenopus oocytes expressing Drosophila RYE and human nAChR β2 (Figure 5—source data 1). *p<0.05. (D) Heterologous expression of SSS reduces current following ACh application in Xenopus oocytes expressing human nAChR α4β2 (Figure 5—source data 1). ***p<0.001.
-
Figure 5—Source data 1
Sleep analysis of rye mutants overexpressing sss, and whole-cell membrane current recording of Xenopus oocytes.
- https://doi.org/10.7554/eLife.01473.012
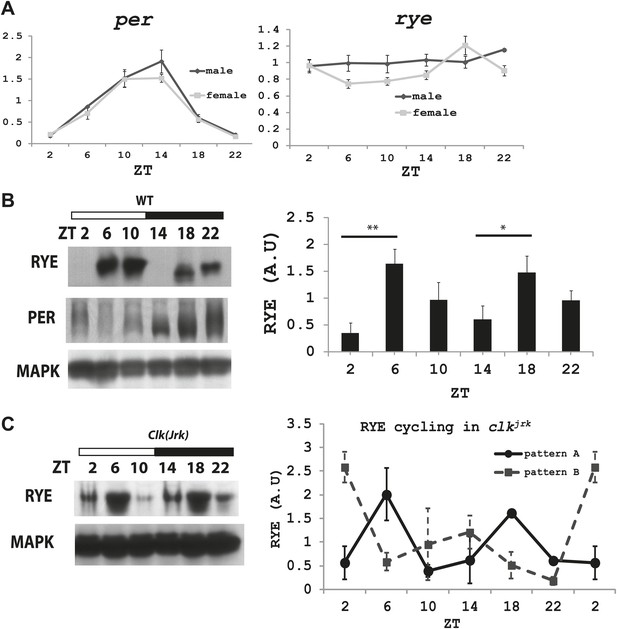
The RYE protein is expressed cyclically in association with the sleep state, but independently of the circadian clock.
(A) Quantitative PCR analyses show oscillations of per mRNA (left panel), but constant levels of rye mRNA (right panel). actin was used as a normalization control (Figure 6—source data 1). (B) Left: a representative western blot of head extracts from wild type flies shows cyclic expression of RYE with two daily peaks, in the middle of the day (ZT6-10) and in the middle of the night (ZT18-22), corresponding to the sleep state (Figure 1B). PER, in contrast, shows only one daily peak. MAPK was used to control for loading. Right: densitometry quantification of western blots with error bars representing standard error (n = 8). RYE value at ZT0 is set as 1 (Figure 6—source data 1). *p<0.05, **p<0.01. (C) RYE cycling is similar to wild type in Clkjrk flies, indicating that cycling per se does not require a functional clock. However, the phase is variable, so for quantification purposes five independent experiments were split into two groups of roughly similar phase (Figure 6—source data 1).
-
Figure 6—Source data 1
qPCR analysis of per and rye expression during LD cycle and densitometry quantification of RYE expression.
- https://doi.org/10.7554/eLife.01473.014
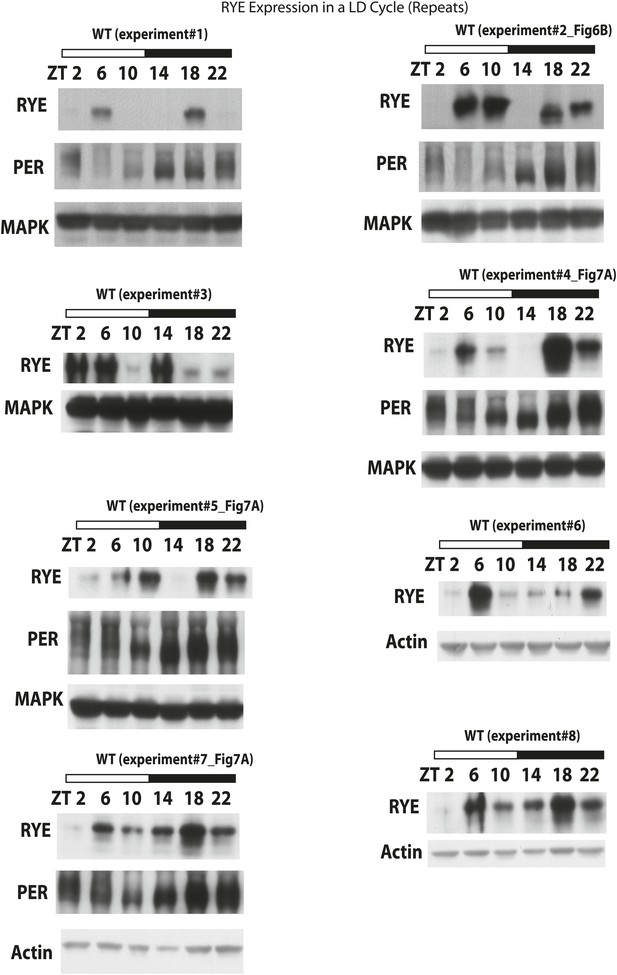
RYE expression in a LD cycle (repeats).
https://doi.org/10.7554/eLife.01473.015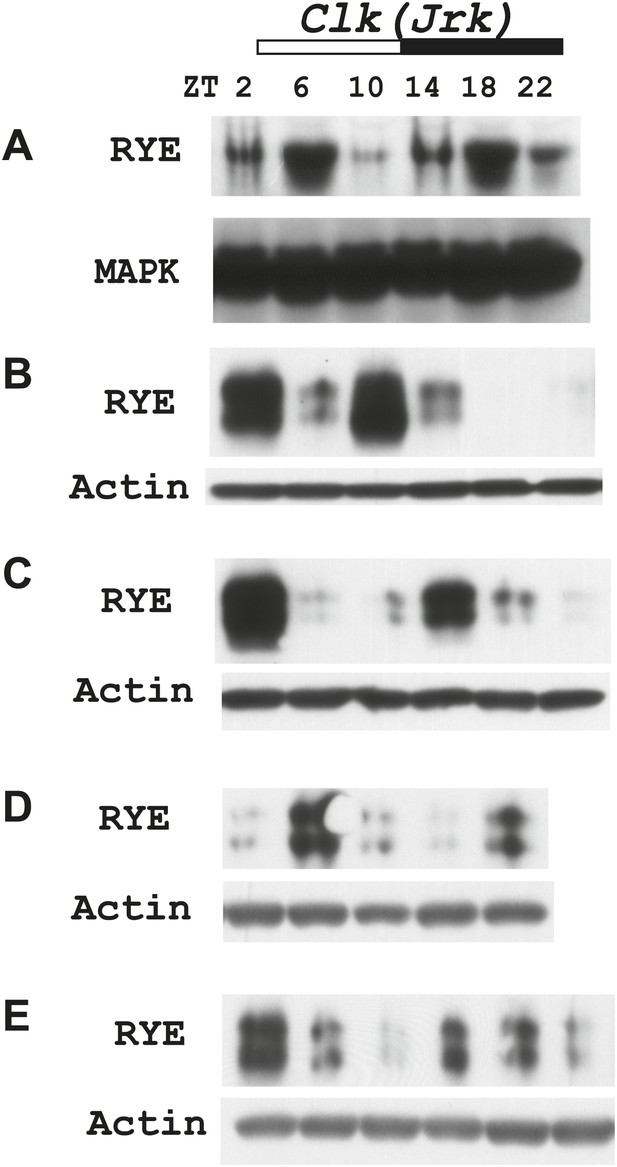
Supporting data for Figure 6C.
https://doi.org/10.7554/eLife.01473.016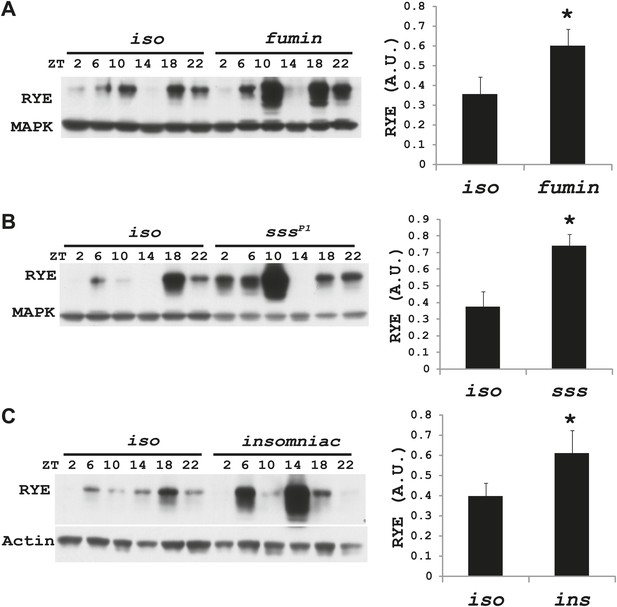
RYE levels are elevated in short-sleeping mutants.
(A) Western analyses of RYE expression over the course of a day in iso controls and fumin mutants. While RYE still cycles in fumin, the mean value of all six daily time points from three independent experiments (n = 3) shows that overall levels of RYE are higher than in wild type controls (Figure 7—source data 1). *p<0.05. (B) Western analyses of RYE expression over the course of a day in iso controls and sss mutants. As in A, the quantification reflects the mean across the day from three independent experiments (n = 3). *p<0.05. (C) Western analyses of RYE expression over the course of a day in iso controls and insomniac mutants. Quantification as above (n = 3). *p<0.05.
-
Figure 7—Source data 1
Densitometry quantification of RYE expression in short sleep mutants.
- https://doi.org/10.7554/eLife.01473.018
Supporting data for Figure 7A.
https://doi.org/10.7554/eLife.01473.019
Supporting data for Figure 7B.
https://doi.org/10.7554/eLife.01473.020
Supporting data for Figure 7C.
https://doi.org/10.7554/eLife.01473.021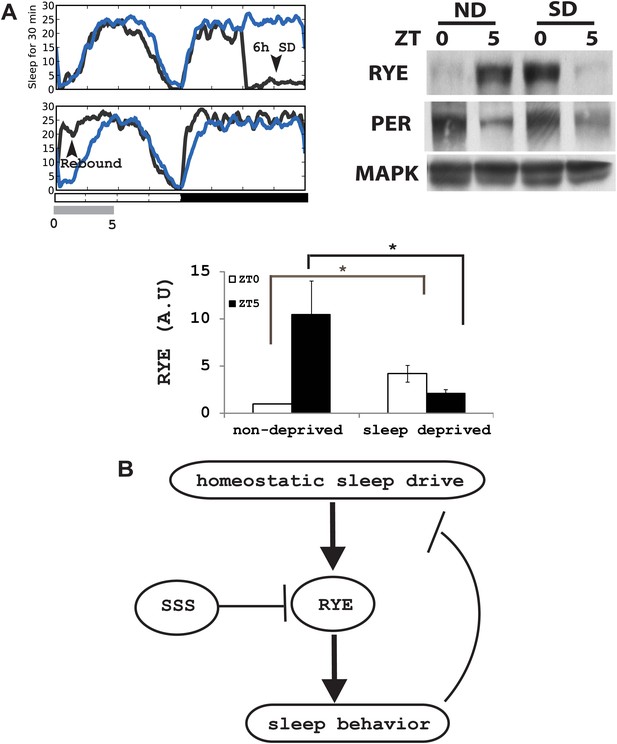
RYE expression is under control of the sleep homeostat.
(A) Left: sleep profile of sleep deprived flies. Blue line: non-deprived controls, Grey line: sleep deprived flies. Arrowheads point to the 6 hr sleep deprivation (SD) window (ZT18-24) and sleep rebound the following morning (ZT0-5). Right: a representative western blot of fly head samples at ZT0 and ZT5 in sleep-deprived (SD) animals and non-deprived (ND) controls. RYE levels are high immediately following sleep deprivation (SD0) and dissipate after sleep rebound (SD5). In contrast, the circadian clock is not affected by SD, since PER cycling is comparable in the SD group and the non-deprived control (ND). Densitometry quantification of the western data (n = 5) is shown below with error bars showing standard error (Figure 8—source data 1). *p<0.05. (B) Model for the role of RYE in the homeostatic regulation of sleep: We propose that RYE is required to maintain sleep, and posttranslational regulation of RYE reflects homeostatic sleep drive. Sleep need builds up during wakefulness and upregulates levels of RYE, which are essential to maintain sleep behavior. Homeostatic drive dissipates during sleep, and levels of RYE are reduced, leading to wakefulness. SSS represses RYE activity, thus acting as a wake-promoting factor in this particular context.
-
Figure 8—Source data 1
Densitometry quantification of RYE expression after SD.
- https://doi.org/10.7554/eLife.01473.023
Supporting data for Figure 8A.
https://doi.org/10.7554/eLife.01473.024Tables
Rhythmic behavior of rye homozygotes in constant darkness
| Genotype | Rhythmicity% (n)* | Period ± SEM (h)† | FFT ± SEM† |
|---|---|---|---|
| W1118 (iso) | 93.8% (30/32) | 23.21 ± 0.04 | 0.051 ± 0.005 |
| rye | 62.1% (18/29) | 23.66 ± 0.09 | 0.054 ± 0.005 |
-
*
Flies were entrained to a light–dark cycle (12 hr/12 hr) for 3 days before being moved into constant darkness (DD). Behavior was analyzed from day 3 to day 11 in DD, and flies with a fast fourier transform value (FFT) above 0.01 were considered rhythmic.
-
†
Period lengths and FFT values of rhythmic flies are listed as average value plus minus the standard error of mean (SEM).





