PTRN-1, a microtubule minus end-binding CAMSAP homolog, promotes microtubule function in Caenorhabditis elegans neurons
Figures
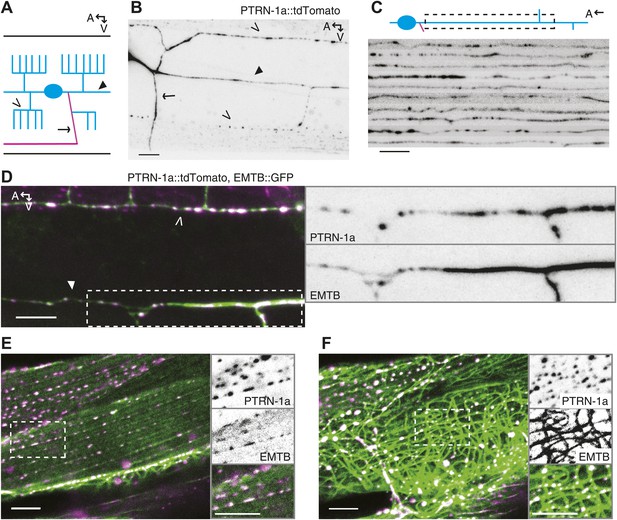
PTRN-1 localizes to puncta throughout neurites and colocalizes with MTs.
(A) Schematic diagram of the central region of the PVD neuron. The cell body (blue oval) is in the posterior half of the animal. An elaborate dendritic arbor (blue lines) extends from the base of the head to the posterior of the animal, and the single axon (magenta) is extended into the ventral nerve chord (VNC). Black lines represent the outline of the animal. (B) PTRN-1a::tdTomato localization in the PVD neuron. The cell body is outside of the image, close to the left edge. (C) Confocal micrographs from 10 animals showing PTRN-1a::tdTomato localization in the PVD primary dendrite directly posterior to the cell body. (D–F). Colocalization of PTRN 1a::tdTomato (magenta) and EMTB::GFP (green) in the PVD neurites (D), at the sarcolemma of the body wall muscle cells (E), and in the cell interior of the body wall muscle cells (F). Data were acquired from wyEx5968 and wyEx6022 transgenes coexpressed in the ptrn-1(tm5597) mutant. Closed arrowhead indicates the primary dendrite, the open arrowheads indicate tertiary dendrite, and arrow points to axon of the PVD neuron (A, B, D). A, anterior; V, ventral. Scale bar: 5 μm.
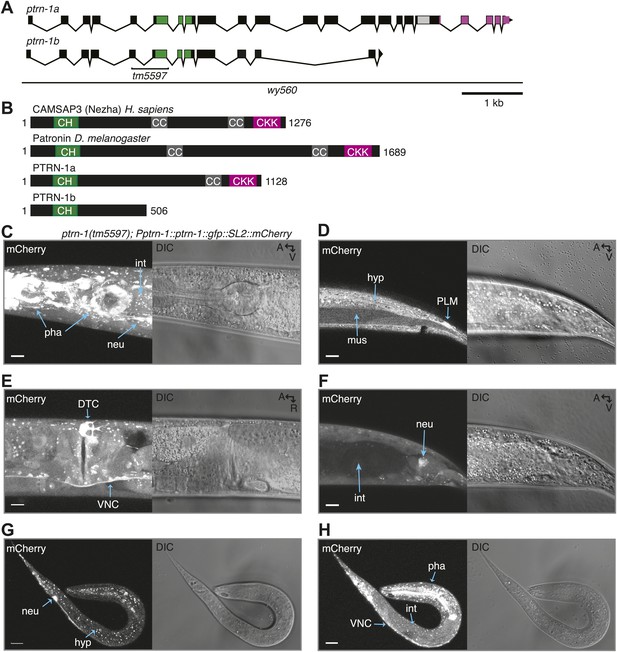
PTRN-1 is broadly expressed.
(A) The ptrn-1(F35B3.5) open reading frame, which encodes ptrn-1a and ptrn-1b. The tm5597 allele contains a 604 nt deletion, resulting in Met136>Thr, Ala137>STOP. The wy560 allele contains a 65.3-kb deletion spanning nucleotide 16,983,396-17,048,700 of LGX. (B) PTRN-1a has a conserved domain structure with CAMSAP proteins. CH, calponin homology; CC, coiled-coil; CKK, calmodulin-regulated spectrin-associated CKK domain. The PTRN-1a CKK domain is the most well-conserved portion of the protein, with 64% similarity to Patronin CKK. PTRN-1b lacks the CKK domain. (C–H) ptrn-1 is expressed in many tissues throughout development. Fluorescence of mCherry expressed from a fosmid encoding ptrn-1::GFP::SL2::mCherry in the ptrn-1(tm5597) mutant (SL2: trans-splice leader 2, which causes mCherry to be transcribed as part of the ptrn-1 transcript but translated independently). The head (C) and mid-body (E) of an adult, the posterior region of an L4 animal (D and F), and the whole body of an L1 animal (G and H) are shown. A, anterior; R, right; V, ventral; neu, neuron; int, intestine; pha, pharynx; DTC, distal tip cell; VNC, ventral nerve chord; PLM, the PLM touch receptor neuron; hyp, hypodermis; mus, muscle. Scale bar: 10 μm.
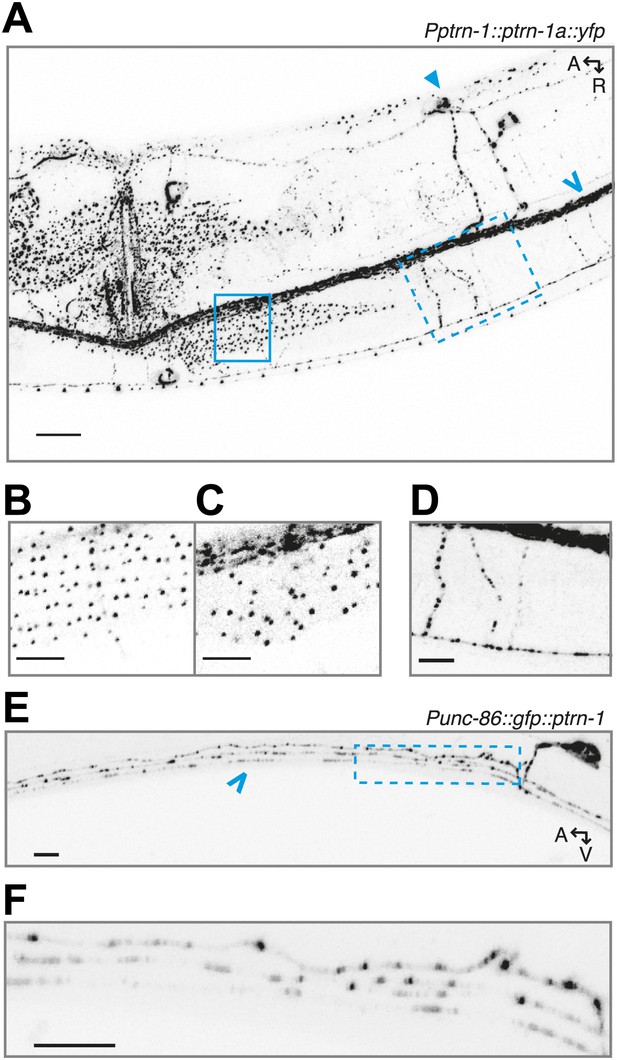
PTRN-1 exhibits punctate localization in neuronal processes and the body wall muscle cells.
(A) PTRN-1a::YFP expressed from the Pptrn-1 promoter in the ptrn-1(tm5597) mutant. Pictured is a young adult animal. (B-D) Expanded view with a subset of confocal slices of region in the solid box (B and C) and dashed box (D) from A. (B) A single confocal slice at the sarcolemma of the body wall muscle cell. (C) A single confocal slice of the interior of the same body wall muscle cell as B. (D) Commissures from the ventral nerve chord (VNC) intersecting a sublateral neuronal process; a maximum projection of ∼6 μm is shown. (E) GFP::PTRN-1 expressed from the Punc-86 promoter in the ptrn-1(tm5597) mutant exhibits similar localization to PTRN-1a::YFP in neurites in the ventral nerve cord. The cell body is the HSN neuron. As PTRN-1b is produced by removing an intron for which the splice sites are within two ptrn-1a exons, the gfp::ptrn-1a(cDNA) construct is expected to produce both GFP::PTRN-1a and GFP::PTRN1b. The comparable localization between these two constructs indicates that the ptrn-1a isoform is sufficient to achieve punctate localization in neurites. For comparison, Chalfie and Thomson used electron microscopy of MTs in the VNC to show that the average distance between MT minus ends is approximately 1.7 μm. (F) Expanded view of boxed region from E. Solid arrow head: PVD cell body, open arrow: VNC. A: anterior, R: right, V: ventral. Scale bar: 5 μm.
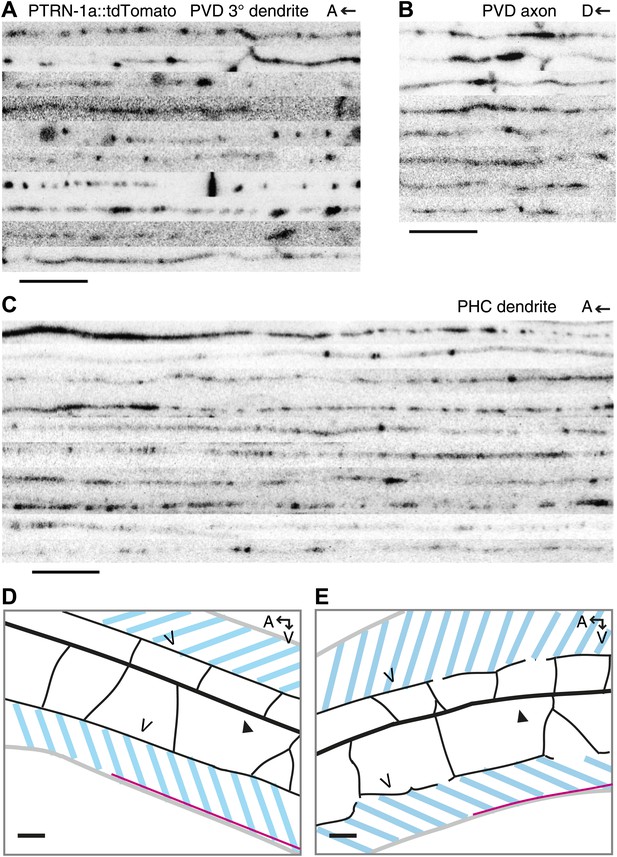
PTRN-1 localizes to puncta throughout the neurites in the PVD and PHC neurons.
(A–C) Confocal images of 8–10 ptrn-1(tm5597) mutants expressing PTRN-1a::tdTomato in a subset of neurons, including PVD and PHC, were straightened and aligned. Regions shown are the tertiary dendrite (posterior the cell body) (A) and the axon (B) of the PVD neuron, as well as the dendrite of the PHC neuron (C). The PHC neuron with indicated approximate region of interest on the dendrite is diagramed in Figure 3A. A, anterior; D, dorsal. Scale bar: 5 μm. (D and E) Schematic diagrams of animals shown in Video 1 (D) and Video 2 (E). Gray lines outline of the animal, black lines show PVD dendrites: the filled arrowhead indicates the PVD primary dendrite, the open arrowheads point to tertiary dendrites. Pink lines are PVD axons. The PVD cell body is out of frame to the right in both images. PTRN-1::tdTomato is also expressed in the body wall muscle of these animals, marked with the dashed blue lines. A, anterior; V, ventral; scale bar: 10 μm. We could not resolve the ends of the tertiary dendrites, but there are usually gaps between the tertiary branches from each secondary branch (see Figure 1).
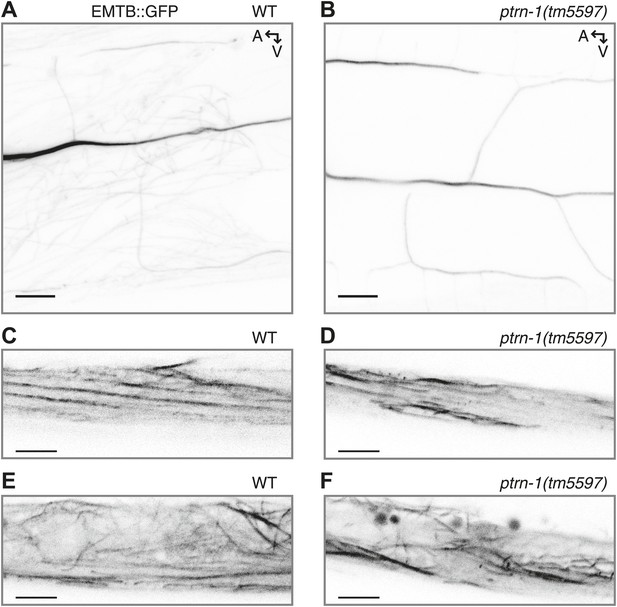
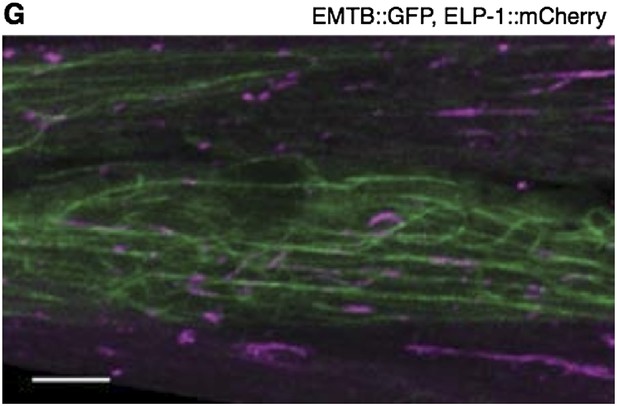
EMTB::GFP binds MTs in the PVD neuron and the body wall muscles.
(A and B) EMTB::GFP in the PVD neuron of wild-type (A) and ptrn-1(tm5597) mutant (B) animals. Note that fluorescence intensity becomes progressively dimmer from the primary to the quaternary dendritic processes, as compared to the cytosolic GFP shown in Figure 4—figure supplement 1. (C–F) EMTB::GFP fluorescence in the body wall muscle cells of wild-type (C and E) and ptrn-1(tm5597) mutant (D and F) animals. (C and D) Confocal slice at the sarcolemma of the body wall muscle; (E and F) the interior of the body wall muscle cell.
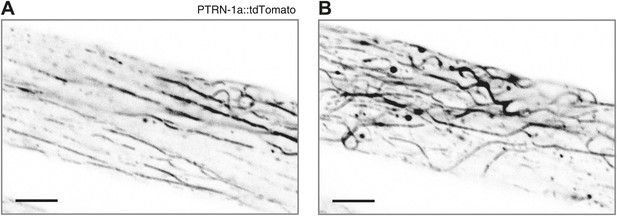
Highly expressed PTRN-1a::tdTomato binds along MT filaments.
PTRN-1a::tdTomato near the membrane (A) and in the interior (B) of a body wall muscle cell in an animal carrying a highly expressing ptrn-1a::tdTomato transgene. Compare to MT localization in Figure 1—figure supplement 4. Scale bar: 5 μm.
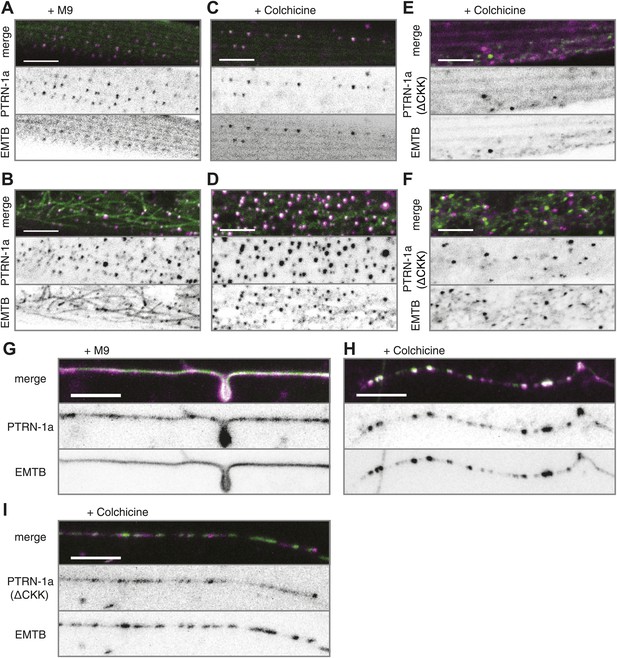
PTRN-1 stabilizes MT foci in neurons and muscles.
(A–D) PTRN-1a::tdTomato and EMTB::GFP at the sarcolemma (A and C) and cell interior (B and D) of body wall muscle cells after acute colchicine exposure (C and D) or M9 control (A and B). (E and F) PTRN-1a(ΔCKK)::tdTomato and EMTB::GFP at the sarcolemma (E) and cell interior (F) of body wall muscle cells after acute colchicine exposure. (G–H) Localization of PTRN-1a::tdTomato and EMTB::GFP in the PVD dendrite after acute colchicine exposure (H) or M9 control (G). (I) PTRN-1a(ΔCKK)::tdTomato and EMTB::GFP in the PVD primary dendrite after acute colchicine exposure. All data acquired from wyEx5968 with either wyEx6022 (A–D and G and H), wyEx6092 (I), or wyEx6165 (E and F) co-expressed in bus-17(e2800) mutant animals. Scale bar: 5 μm.
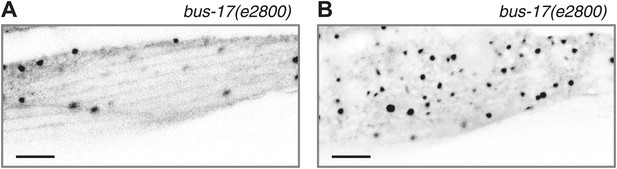
Acute colchicine exposure changes EMTB::GFP localization in body wall muscles.
(A and B) EMTB::GFP localization in the body wall muscle after acute colchicine treatment in the bus-17(e2800) genetic background. (A) Confocal slice at the sarcolemma; (B) the interior of the body wall muscle cell. Note that this animal, unlike those shown in Figure 2, does not have the wyEx6022 ptrn-1::tdTomato transgene, so the EMTB::GFP puncta from wyEx5968 that remain after the acute colchicine treatment are not caused by PTRN-1::tdTomato overexpression.
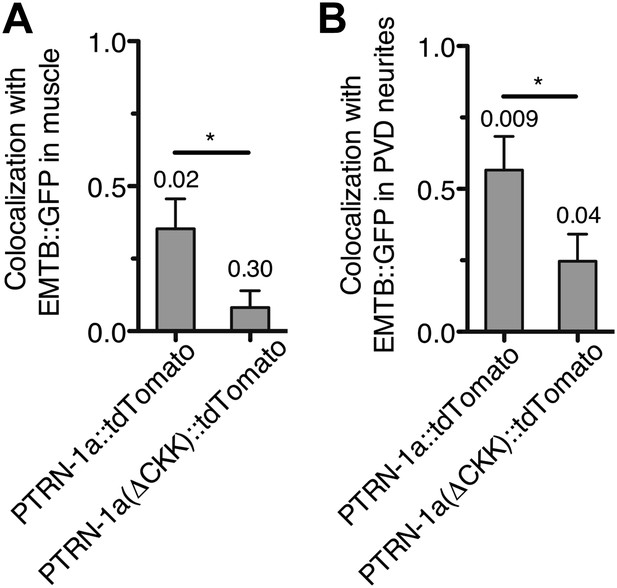
PTRN-1::tdTomato colocalizes with EMTB::GFP puncta after MT depolymerization by colchicine.
bus-17(e2800) mutant animals co-expressing EMTB::GFP with either PTRN-1::tdTomato or PTRN-1a(ΔCKK)::tdTomato were imaged after acute colchicine treatment (example images shown in Figure 2), and the Pearson's colocalization coefficient (PCC) between the two fluorescent proteins was calculated for body wall muscle cells (A) and PVD dendrites (B). In A, plotted is the average PCC calculated from maximum projection images of confocal stacks from four (PTRN-1a::tdTomato) or three (PTRN-1a(ΔCKK)::tdTomato) body wall muscle cells. In B, plotted is the average PCC calculated from linescans of PVD dendrites from four animals each for both PTRN-1a::tdTomato and PTRN-1a(ΔCKK)::tdTomato. (*p<0.05, one-tailed students t test). The number above each bar indicates the p value from a one-tailed, one-sample t test comparing the calculated PCC against 0 (no colocalization) (Mcdonald and Dunn, 2013). In the PVD neurites, PTRN-1a(ΔCKK)::tdTomato exhibited a small but significant colocalization with EMTB::GFP, which was contrary to the hypothesis that the CKK domain is necessary for MT binding and stabilization. This colocalization might be due to the presence of other MT-protecting proteins or endogenous PTRN-1.
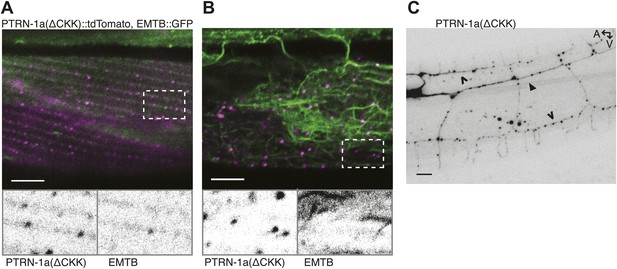
PTRN-1a(ΔCKK) exhibits punctate localization in body wall muscle cells and neurons.
(A and B) PTRN-1a(Δ CKK)::tdTomato, in which the MT-binding CKK domain has been deleted, co-expressed with EMTB::GFP in ptrn-1(tm5597) mutant animals. Images show the body wall muscle cells at the sarcolemma (A) and in the cell interior (B). Expanded boxes highlight regions where PTRN-1(ΔCKK)::tdTomato and EMTB::GFP do not colocalize. (C) PTRN-1a(ΔCKK)::tdTomato expressed in the ptrn-1(tm5597) mutant exhibits punctate localization in the processes of the PVD neuron. A, anterior; V, ventral. Filled arrowheads point to PVD primary dendrite, open arrowheads point to PVD tertiary dendrites. Scale bar: 5 μm.
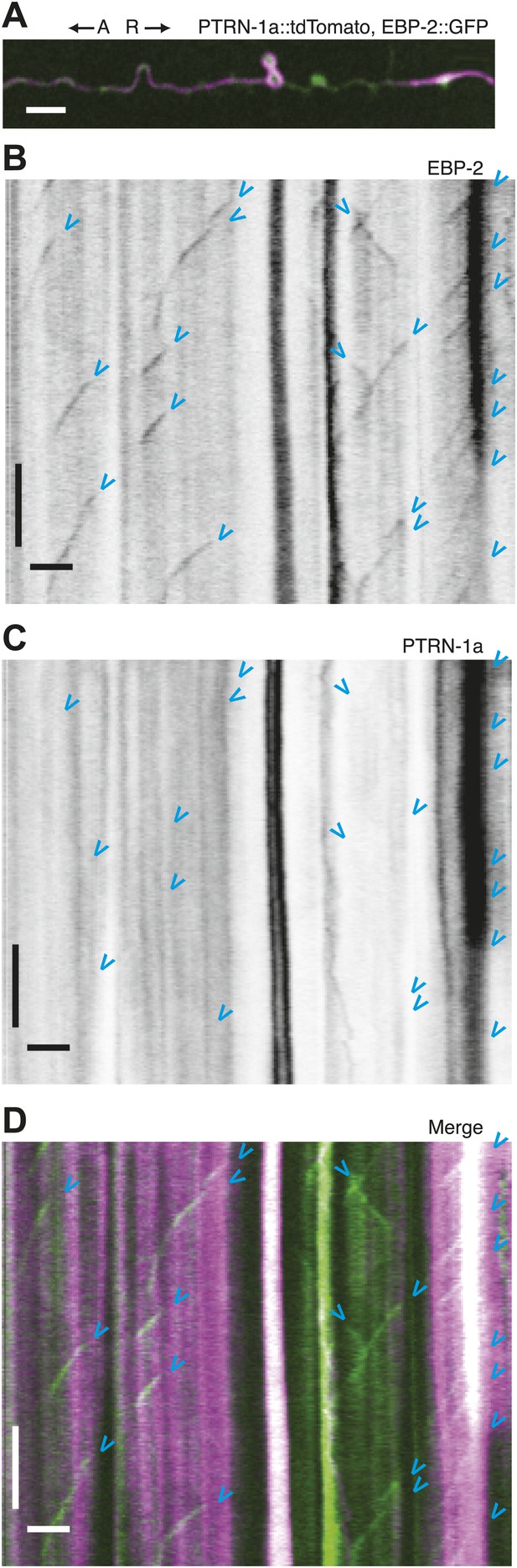
Immobility of PTRN-1a::tdTomato puncta contrasts with EBP-2::GFP movements in the PVD dendrite.
(A) Live imaging was performed on a ptrn-1(tm5597) mutant animal co-expressing EBP-2::GFP (green, from the wyEx4828 transgene), which labels growing plus-end of MTs, and PTRN-1a::tdTomato (magenta, from the wyEx6022 transgene) along a section of the tertiary dendrite of the PVD neuron. (B–D). Kymographs of EBP-2::GFP (B), PTRN-1a::tdTomato (C), and overlay of EBP-2::GFP (green) with PTRN-1a::dtTomato (magenta) (D) from a 110 s video acquired from the PVD process shown in A. Time runs top to bottom. Arrows point to start of EBP-2::GFP movements. A, anterograde; R, retrograde. Scale bar: 5 μm, ∼22 s.
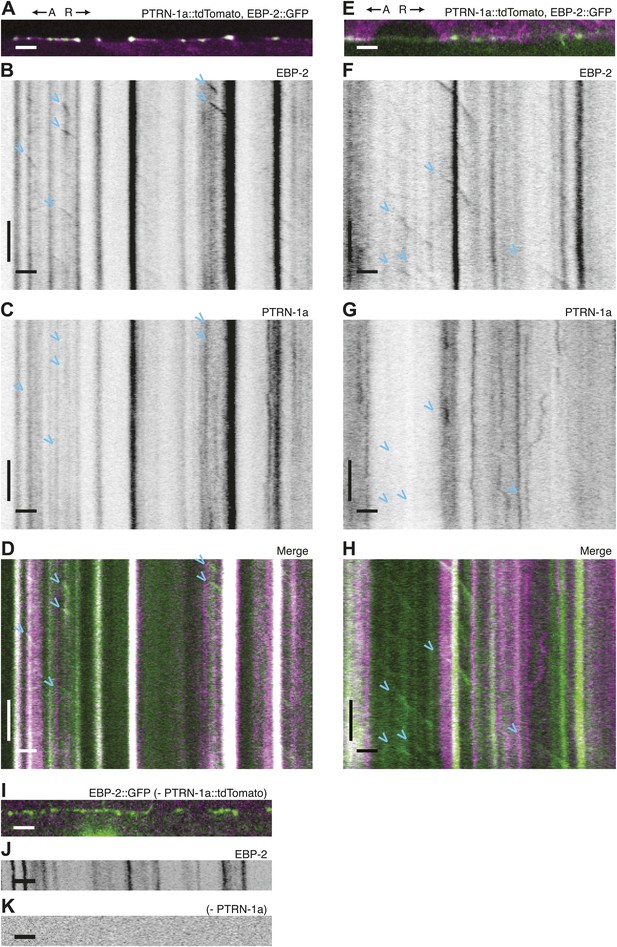
Immobility of PTRN-1a::tdTomato puncta contrasts with EBP-2::GFP movements in the PVD dendrite.
Replicate 2 (A–D) and replicate 3 (E–H) of experiment in Figure 2. (A and E) Live imaging was performed on ptrn-1(tm5597) mutant animals coexpressing EBP-2::GFP (green, from the wyEx4828 transgene), and PTRN-1a::tdTomato (magenta, from the wyEx6022 transgene) along a section of the tertiary dendrite of the PVD neuron. (B–D and F–H). Kymographs of EBP-2::GFP (B,F), PTRN- 1a::tdTomato (C and G), and overlay of EBP-2::GFP (green) with PTRN-1a::dtTomato (magenta) (D and H) from a 110 s video acquired from the PVD process shown in A and E. (I–K) Live imaging was performed as above on an animal lacking the ptrn-1a::tdTomato transgene. Time runs top to bottom. A, anterograde; R, retrograde. Scale bar: 5 μm, ∼22 s.
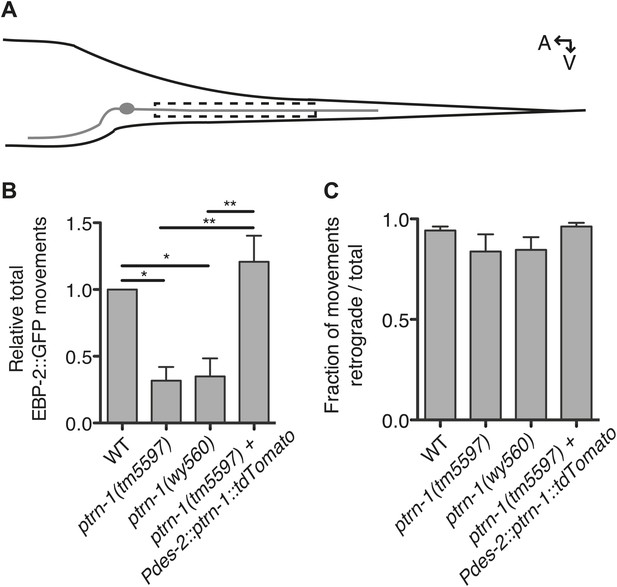
PTRN-1 promotes MT polymerization in neurites.
(A) Schematic diagram of the PHC neuron. The anterior process is the axon; the posterior process is the dendrite. Live imaging was used to monitor EBP-2::GFP movements in the boxed region of the PHC dendrite. A: anterior, V: ventral. (B and C) Quantification of EBP-2::GFP anterograde and retrograde movements in the PHC dendrite of wild-type (WT) vs ptrn-1(tm5597) and ptrn-1(wy560) mutant animals, and vs the ptrn-1(tm5597) mutant carrying the Pdes-2::ptrn-1::tdTomato transgene, which is expressed in a subset of neurons as well as the body wall muscle. (B) Total EBP-2::GFP movements in each strain normalized against the wild-type control. (C) Fraction of EBP-2::GFP movements in each strain that moved in the retrograde direction. Mean ± SEM. (n = 3 experiments, each with at least 10 animals/genotype, *p<0.05, **p<0.01, ANOVA with Bonferroni post test).
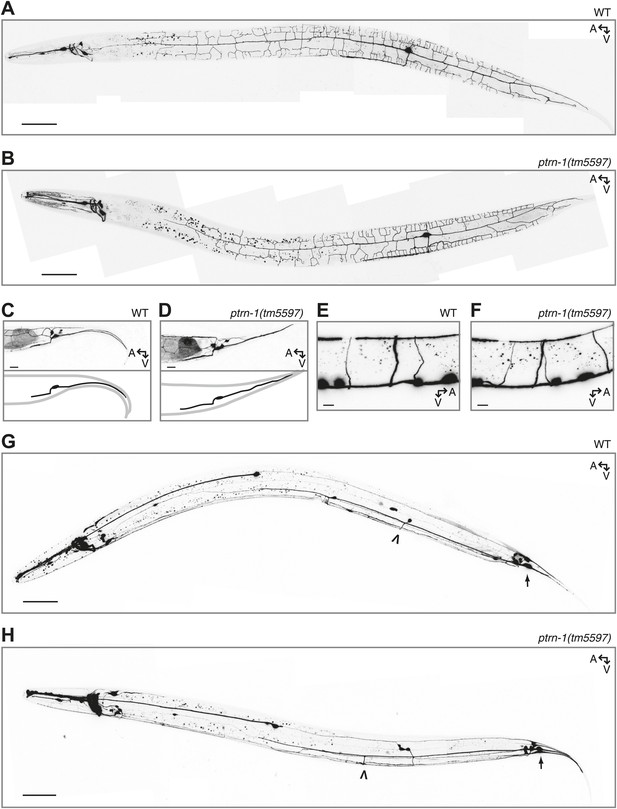
Neuronal morphology is grossly unaffected by loss of ptrn-1.
(A and B) The PVD neuron visualized by cytosolic GFP in wild-type (A) versus ptrn-1(tm5597) mutant (B) animals. (C and D) The PHC neuron visualized by cytosolic GFP in wild-type (C) versus ptrn-1(tm5597) mutant (D) animals. In the schematic diagrams, the PHC neuron is represented in black. (E and F) The DD/VD-type motorneurons visualized by cytosolic mCherry in wild-type (E) versus ptrn-1(tm5597) mutant (F) animals. (G and H) The PLM neuron, along with other touch receptor neurons, visualized by cytosolic GFP in wild-type (G) versus ptrn-1(tm5597) mutant (H) animals. Arrow points to PLM cell body, open arrowhead points to PLM commissure. Fluorescence in the head is from the co-injection marker. A, anterior; V, ventral. Scale bar: 50 μm (A, B, G, H), 5 μm (C–F).
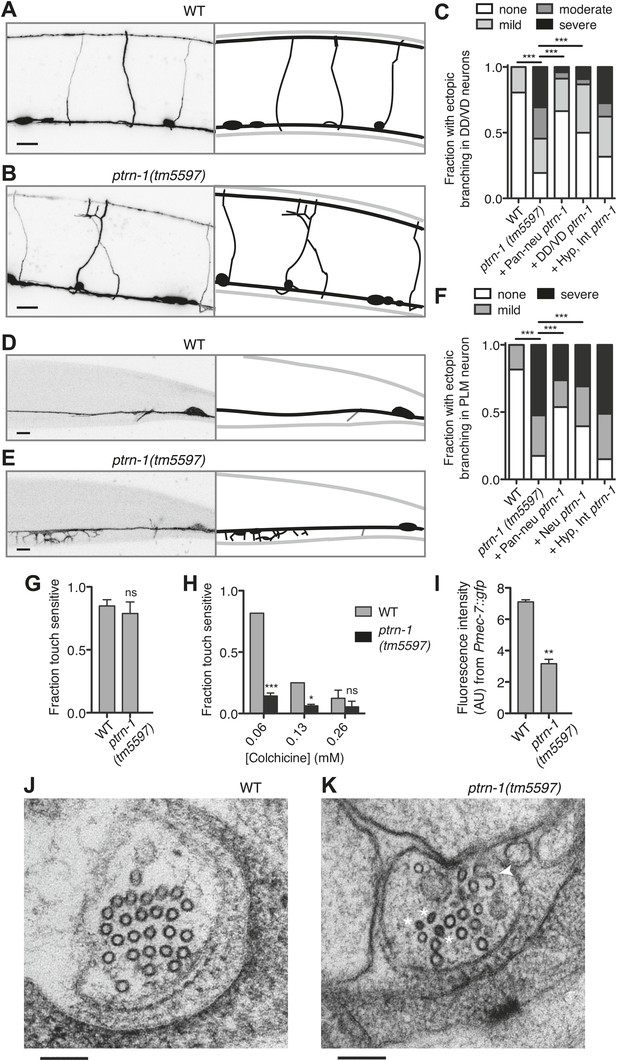
PTRN-1 supports MT stability in neurites.
(A-C) Wild-type (A) and ptrn-1(tm5597) mutant (B) animals were grown in 0.13 mM colchicine to the L4 stage, and cytosolic RFP was used to visualize the DD/VD neurons. Scale bar: 10 μm. (C) Fraction of animals with ectopic sprouting from the DD/VD neurons, scored based on severity as described in ‘Materials and methods’ (n = at least 80 animals/genotype, ***p<0.001, Chi-squared test with Šidák correction). (D–F) Wild-type (D) and ptrn-1(tm5597) mutant (E) animals were grown in 0.035 mM colchicine to the L4 stage, and the PLM neuron was visualized with myr::GFP. Scale bar: 5 μm. (F) Fraction of animals exhibiting ectopic sprouting from the PLM neuron, scored based on severity (n = at least 60 animals/genotype, ***p<0.001, Chi-squared test with Šidák correction). In schematic diagrams, the light gray lines represent the outline of the animal, DD and PLM neurons are black, and other neurons (in D and E only, one short, unbranched process near the PLM cell body of each image) are dark gray. For tissue specific rescue, DD/VD (Punc-47L), Pan-neu: pan-neuronal (Prab-3), Hyp: hypodermal (Pdpy-7), Int: intestinal (Pvha-6), neu: a subset of neurons including PLM (Punc-86). (G) Touch sensitivity of wild-type vs ptrn-1(tm5597) mutant animals. Mean ± SEM. (n = 3 experiments, each with 8–12 animals/genotype, ns not significant (p=0.46), t test). (H) Touch sensitivity of wild-type vs ptrn-1(tm5597) mutant animals grown in the indicated concentrations of colchicine. Mean ± SEM. (n = 2 experiments, each with 10 animals/genotype, ***p<0.001, *p<0.05, t test for each drug concentration). (I) Average fluorescence of GFP expressed from the Pmec-7 (β-tubulin) promoter in the PLM cell body of wild-type vs ptrn-1 mutant animals. Mean ± SEM. (n = 2 experiments, each with at least 13 animals/genotype, **p<0.01, t test). (J and K) Transmission electron microscopy of the PLM neuron in wild-type (J) and ptrn-1(tm5597) mutant (K) young adult animals, sectioned near the rectum. Note MTs with abnormally smaller diameters (Asterisks), and a MT sheet shaped like an ‘S’ (Arrow head). Scale bar: 100 nm.
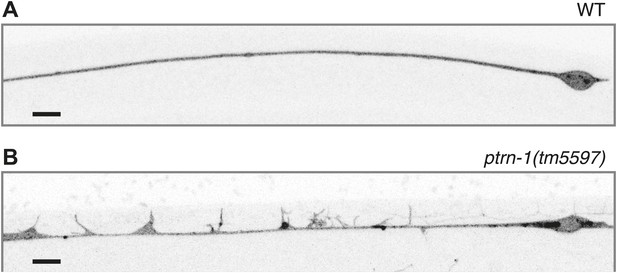
PTRN-1 protects the ALM touch receptor neuron against ectopic neurite sprouting during growth in colchicine.
Wild-type (A) and ptrn-1(tm5597) mutant (B) animals were grown in 0.035 mM colchicine to the L4 stage, and the ALM neuron was visualized with myr::GFP. Scale bar: 5 μm.
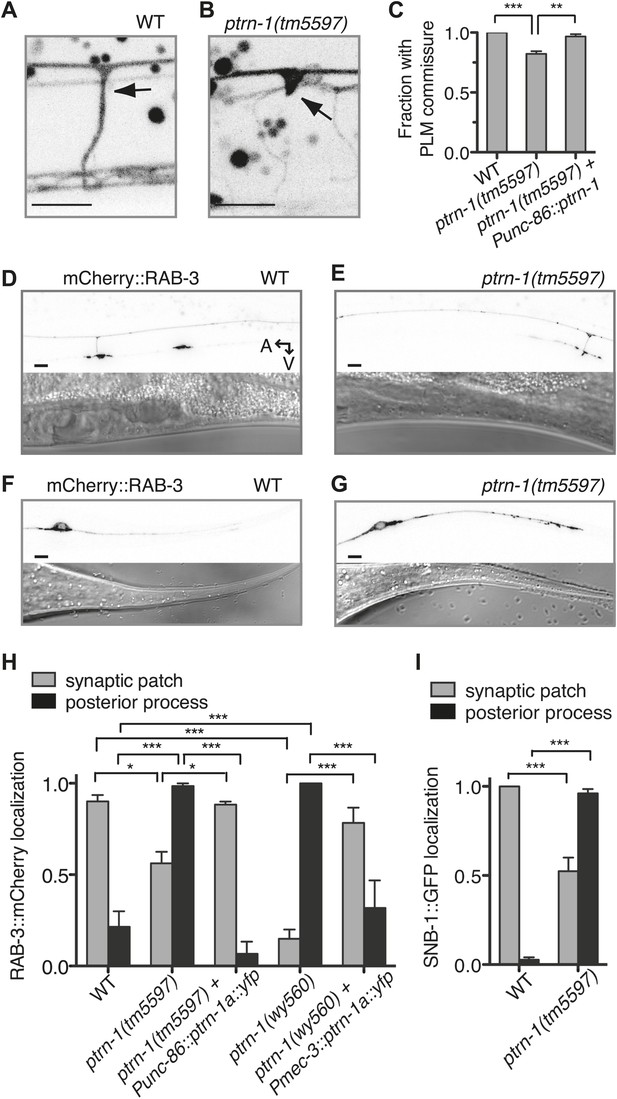
PTRN-1 promotes synapse localization and neurite morphology in the PLM neuron.
(A and B) myrGFP was used to visualize the PLM commissure in wild-type (A) and ptrn-1(tm5597) mutant (B) animals. Arrows point to commissure or commissure bud. (C) Fraction of animals with a PLM commissure connecting the axon to the ventral nerve cord. Mean ± SEM. (n = 3 experiments, each with at least 30 animals/genotype, **p<0.01, ***p<0.001, one-way ANOVA with Bonferroni post test). (D and E) mCherry::RAB-3 at the synaptic patch of the PLM neurons of wild-type (D) and ptrn-1(tm5597) mutant (E) animals. (F and G) mCherry::RAB-3 in the posterior process of the PLM neurons in wild-type (F) and ptrn-1(tm5597) mutant (G) animals. (H) Fraction of wild-type and ptrn-1 mutant animals with visible accumulation of mCherry:RAB-3 at the synaptic patch and the posterior process of the PLM neuron. The Punc-86 promoter is expressed in a subset of neurons including the TRNs; the Pmec-3 promoter is expressed in the TRNs. Animals with two visible mCherry:RAB-3 patches in the PLM synaptic region were counted as having synaptic accumulation, and animals with one or no visible mCherry::RAB-3 patches were considered to have loss of synaptic accumulation. Mean ± SEM. (n = 2 experiments, each with 30 animals/genotype, *p<0.05, ***p<0.001, two-way ANOVA with Bonferroni post test). (I) Fraction of wild-type and ptrn-1(tm5597) mutant animals with visible accumulation of SNB-1::GFP at the synaptic patch and the posterior process of the PLM neuron. Synaptic patch accumulation was scored as in H. Mean ± SEM. (n = 2 experiments, each with at least 20 animals/trial. ***p<0.001, two-way ANOVA with Bonferroni post test).
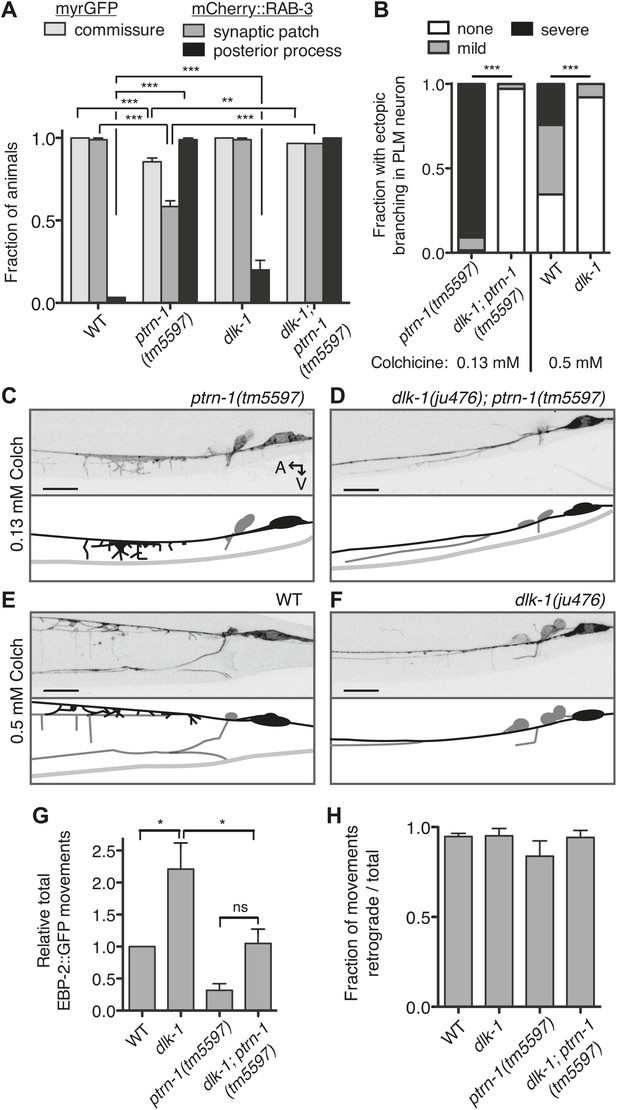
Aberrant phenotype of the PLM neuron in the ptrn-1 mutant is mediated partially by the DLK-1 pathway.
(A) The wyIs97(Punc-86::myrGFP, Punc-86::mCherry::rab-3) transgene was used to simultaneously visualize commissure formation and SV localization. Only animals with an intact PLM commissure were counted for mCherry:RAB-3 localization at the synaptic patch. Values represent mean ± SEM. (n = 3 experiments, each with at least 30 animals/genotype, **p<0.01, ***p<0.001, two-way ANOVA with Bonferroni post test). (B–F) Animals were grown in 0.13 mM colchicine (C and D) or 0.5 mM colchicine (E and F) to the L4 stage, and the PLM neuron was visualized with myr::GFP. In schematic diagrams, the PLM neurons are black, and other neurons in the image are dark gray. (B) Fraction of animals exhibiting ectopic sprouting from the PLM neuron, scored based on severity (n = at least 120 animals/genotype, ***p<0.001, Chi-squared test for each drug concentration). (G and H) Quantification of EBP-2::GFP anterograde and retrograde movements in the PHC dendrite. (G) Total EBP-2::GFP movements in each strain normalized against the wild-type control. (H) Fraction of EBP-2::GFP movements in each strain that moved in the retrograde direction. Mean ± SEM. (n = 3 experiments, each with at least 9 animals/genotype, *p<0.05, ns not significant, one-way ANOVA with Bonferroni post test. The data for the ptrn-1 single mutant is the same as that shown in Figure 4).
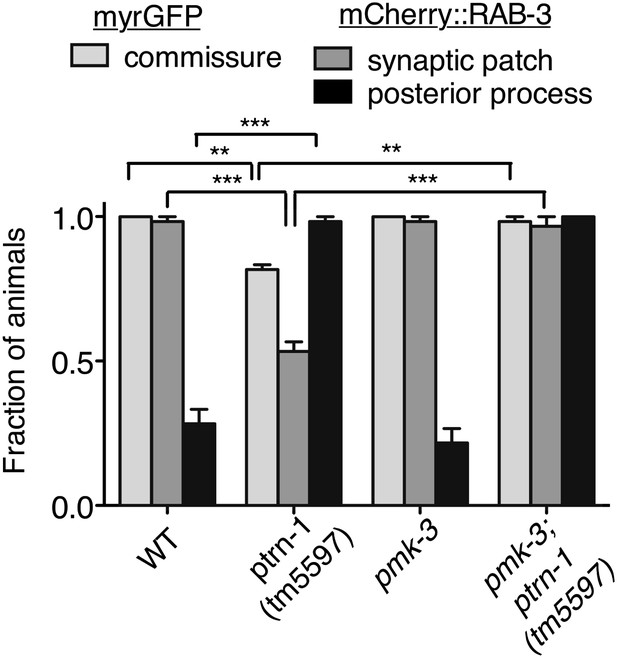
Loss of pmk-3 partially suppresses the aberrant phenotype of the PLM neuron in the ptrn-1 mutant.
wyIs97(Punc-86::myrGFP, Punc-86::mCherry::rab-3) transgene was used to simultaneously visualize commissure formation and SV localization. Only animals with an intact PLM commissure were counted for mCherry:RAB-3 localization at the synaptic patch. Values represent mean ± SEM. (n = 2 experiments, each with at least 30 animals/genotype, **p<0.01, ***p<0.001, two-way ANOVA with Bonferroni post test).
Videos
PTRN-1::tdTomato movements in the PVD neuron.
First example video of a L4 animal showing 40 min with 90 s/frame. See Figure 1—figure supplement 3D for diagram of neuron morphology.
PTRN-1::tdTomato movements in the PVD neuron.
Second example video of a L4 animal showing 40 min with 90 s/frame. See Figure 1—figure supplement 3E for diagram of neuron morphology.