The Caenorhabditis elegans microtubule minus-end binding homolog PTRN-1 stabilizes synapses and neurites
Figures
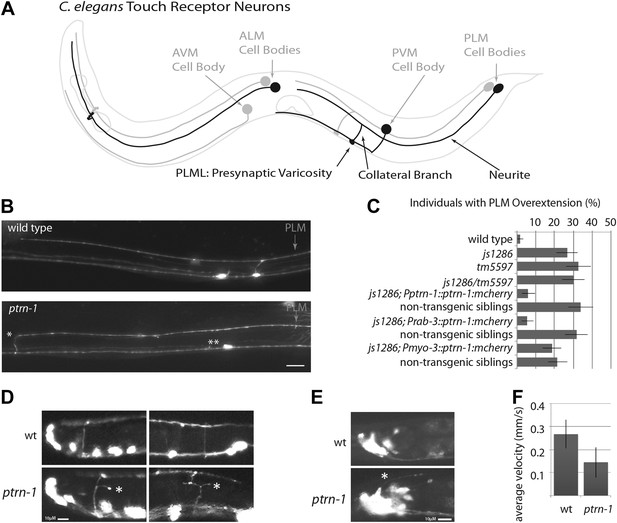
ptrn-1 mutant neurite overextension and collateral branch retraction phenotypes.
(A) Schematic diagram of an L4 C. elegans hermaphrodite showing the morphology of the touch receptor neurons. Neurons on the left side are in black, and those on the right side are in gray. (B) Morphology of mechanosensory neurons in wild-type and ptrn-1 mutants, as seen by expression of mRFP under the control of the mec-7 promoter. Defects in ptrn-1 animals include PLM neurite overextension and ectopic formation of presynaptic varicosities (*), retraction of PLM collateral presynaptic varicosities and branches (**). Scale bar = 10 µm. Additional examples and variance in Ptrn-1 phenotypes are presented in Figure 1—figure supplement 1. (C) Quantification of the PLM neurite overextension phenotype at 22°C. Both ptrn-1 alleles (js1286 and tm5597) show similar defects, as do trans-heterozygotes. Also shown is the rescue of the PLM neurite overextension phenotype by expression of PTRN-1a-mCherry under a native promoter and in neurons under a rab-3 promoter, but not under a myo-3 promoter. Non-transgenic siblings were identified as individuals from mothers segregating the transgene, but that lacked detectable mCherry fluorescence. n = 50–60; error bars = SEP. (D) Aberrant formation and extension of neurites (*) was found in D-type motor neurons. Quantification is in Figure 1—figure supplement 2. (E) Aberrant formation and extension of neurites (*) was found in command interneurons. Quantification is in Figure 1—figure supplement 2. (F) Quantification of locomotory defects using L4 animals. Error bars represent standard deviation. N = 6.
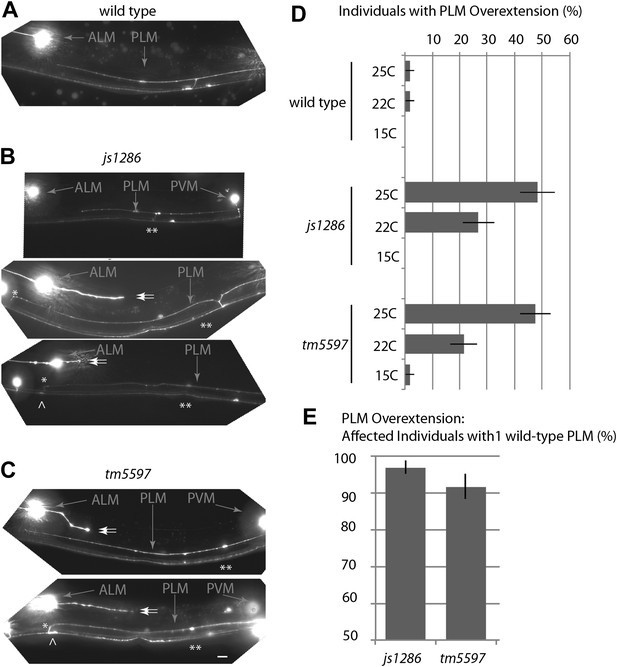
Variance in the ptrn-1 mutant TRN phenotypes.
Midbody of wild-type and mutant L4 animals expressing mRFP in TRNs under the mec-7 promoter (jsIs973) showing the wild-type and ptrn-1 mutant morphology of PLM and PVM, as well as the ALM soma. Scale bar = 10 µm. (A) The wild type. (B) ptrn-1(js1286) animals showing PLM neurite overextensions (*), ectopic synaptic varicosities (^), loss of collateral branches and associated PLM synaptic varicosities (**), and secondary posterior-directed ALM processes (double arrows) are noted. (C) ptrn-1(tm5597) animals. (D) Temperature-dependence of PLM overextension in ptrn-1 mutants. n = 60–80 animals. Error-bars = SEP. (E) Variance in penetrance of the overextension defect is less than 10% in ptrn-1 mutants. n=71–94, error bars = SEP.
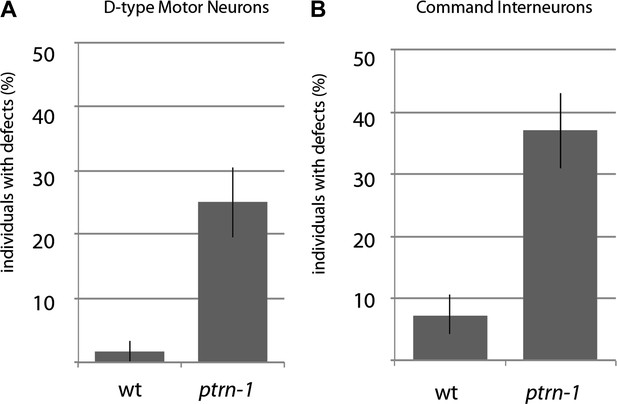
Quantification of other morphology phenotypes in ptrn-1 mutants.
(A) Quantification of D-type motor neuron defects visualized using an oxIs12 [Punc-47::GFP] transgene. Error bars represent SEP. N=60. (B) Quantification of command interneuron defects visualized using nuIs25 [Pglr-1::GFP] transgene. Error bars represent SEP. N = 65–68.
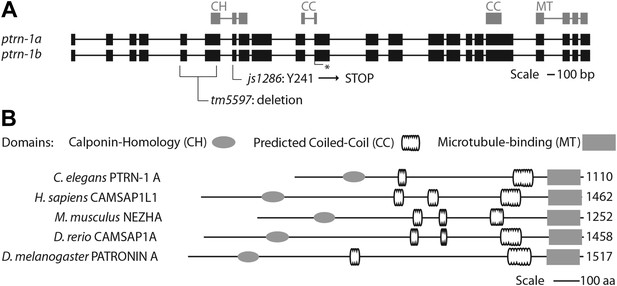
Gene and protein structure of C. elegans ptrn-1.
(A) Exon (thick regions) and intron (thin regions) organization of the ptrn-1 locus. The position of structural domains is demarcated above and the position of the genetic lesion for each of the ptrn-1 alleles is shown below. ptrn-1b differs from ptrn-1a by the inclusion of 6 bp (2 amino acids) at the exon 10–11 boundary (*). Nucleotide sequences for ptrn-1a and ptrn-1b are listed in Figure 2—figure supplements 1 and 2, respectively. Omitted from the figure is a distinct ptrn-1 isoform lacking the C-terminal half of the protein, which is not represented in RNA-seq data for which the sole support is a single compound EST yk1268c03. (B) The CAMSAP protein family consists of a calponin-homology domain followed by one or two 30–40 amino acid coiled-coil regions, a 70–85 amino acid coiled-coil region and a CKK microtubule-binding domain. CAMSAP sequences from C. elegans (this paper), D. melanogaster (AAO41362.1), D. rerio (NP_001159727.1), and H. sapiens (AAI25231.1) were used for homology analysis. Domains were annotated by running sequences through the NCBI Conserved Domain Database. Coiled-coil regions were defined as those predicted by both the Paircoil2 and COILS programs.

Nucleotide sequence of ptrn-1a.
https://doi.org/10.7554/eLife.01637.007
Nucleotide sequence of ptrn-1b.
https://doi.org/10.7554/eLife.01637.008
C .elegans ptrn-1 is broadly expressed in neuronal tissue.
(A) ptrn-1 expression pattern from a transcriptional fusion between mCherry and a ptrn-1 promoter segment sufficient to rescue Ptrn-1 TRN cellular phenotypes (see Figure 1C for rescue). 0.8 kb was chosen as the promoter fragment because of the presence of a 1.2 kb Mariner transposable element at this position 5′ of the ptrn-1 ATG. Scale bars = 10 µm. (B–D) Views at higher magnification of the head neuronal ganglion (B), ventral nerve cord (C), and pre-anal and lumbar neuronal ganglia (D). mCherry is primarily nuclear localized.
Close-up view of the ptrn-1 expression pattern.
(A) ptrn-1 expression pattern from a transcriptional fusion between mCherry and a ptrn-1 promoter segment sufficient to rescue Ptrn-1 TRN cellular phenotypes (see Figure 1C for rescue). View of the head neuronal ganglion. (B–E) High-magnification views of individual neuronal cell bodies showing expression of the mCherry reporter construct. Scale bars = 10 µm.
PTRN-1-localization pattern.
Expression of PTRN-1-mcherry under a ptrn-1 0.8 kb promoter segment sufficient to rescue Ptrn-1 TRN cellular phenotypes (A–C) (see Figure 1C for rescue data). Enlarged views of neurites (A1), a head ganglion cell body (A2), and a tail neuron with isolated morphology (C1) are shown. Puncta (arrows), nuclear exclusion (*), and neurite (double arrow) are noted. Single-cell resolution showing a N-terminal GFP-PTRN-1 fusion protein under the control of the TRN mec-7 promoter in ALM and PLM neurons (D–G). Puncta (arrows) and neurites (double arrows) are noted. Scale bars = 10 µm. Whole-mount immunofluorescent staining with anti-GFP (H) and anti-MEC-7 (I) antibodies. Co-localization of GFP-PTRN-1 with MEC-7 in mechanosensory neurons is shown.
ptrn-1 mutants fail to stabilize mature PLM morphology and synaptic contacts.
(A) Schematic diagram illustrating wild-type PLM neuron developmental changes during the L1 larva from 0 to 12 hr after hatching, as well as the structure of PLM neurons in L4 larva—note that the figure is not drawn to scale. PLM neurons maintain their relative position within the animal from L1 to L4, but L4 are approximately four times longer than L1 animals. (B) Changes observed in PLM neuronal morphology in wild-type and ptrn-1 (js1286) mutants from a synchronized population of animals analyzed at L1 (10 hr) and L4 (36 hr) stages. Animals were grown at 22°C. PLM neurons show both overextension and ventral targeting of the anterior neurite in ptrn-1 mutants, but rarely in wild type. The ventral targeting posterior neurites display variable sized varicosities that are labeled with presynaptic components. ptrn-1 animals also often exhibit loss of collateral branch and associated presynaptic structures. N = 90–98 neurites. (C) Retraction of collateral branch synaptic varicosities and overextended ectopic ventral neurite targeted varicosities in animals imaged as both L1 and L4 larva. N = 11 and 68 neurites. (B and C) Black lines and circles represent neurites and presynaptic varicosities respectively. Gray lines and open circles represent variability in ptrn-1 animals.
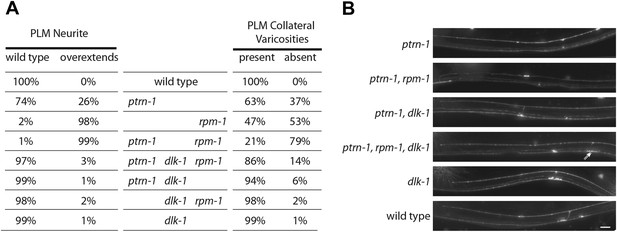
dlk-1 MAP kinase mutants suppress Ptrn-1 overextension phenotypes.
(A) Single, double, and triple mutant combinations of ptrn-1(tm5597), dlk-1(km12) and rpm-1(ok364) were grown at 22°C, scored as L4 larvae for PLM neurite morphology defects using jsIs973(Pmec-7:mRFP), and for the presence of collateral branch presynaptic varicosities using jsIs821(Pmec-7::GFP-RAB-3). Since PVM forms small GFP puncta in the ventral nerve cord (see Figure 1A,B), PLM collateral varicosities which retained a small GFP-RAB-3 puncta in the ventral nerve cord cannot be unambiguously distinguished from PLM collaterals completely lacking presynaptic components. n = 200 PLM cells from 100 animals. (B) Midbody of L4 animals expressing mRFP in TRNs under Pmec-7 (jsIs973) showing the wild-type and mutant PLM morphology associated with ptrn-1 and dlk-1. Elongated varicosity in ptrn-1, rpm-1, dlk-1 triple mutant identified with double arrows. Scale bar = 10 µm.
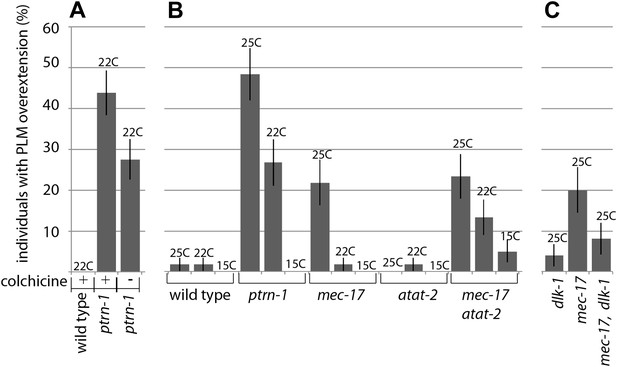
Manipulating the microtubule cytoskeleton leads to terminal neurite extension and ventral nerve cord targeting.
(A) Wild-type and ptrn-1(js1286) mutants were grown for two generations on plates containing 1 mM colchicine and scored for PLM neurite overextension (n = 80). Error bars represent SEP. (B) Wild-type, ptrn-1 mutants and mutants with lesions in microtubule acetylases (paralogs mec-17 and atat-2) were scored for PLM neurite overgrowth defects at various temperatures (n = 55-60). Error bars represent SEP. (C) dlk-1 suppresses the PLM neurite overextension phenotypes of mec-17 (n = 50). Error bars represent SEP.
Additional files
-
Supplementary file 1
Oligonucleotides used in this study.
- https://doi.org/10.7554/eLife.01637.015





