Requirement for highly efficient pre-mRNA splicing during Drosophila early embryonic development
Figures
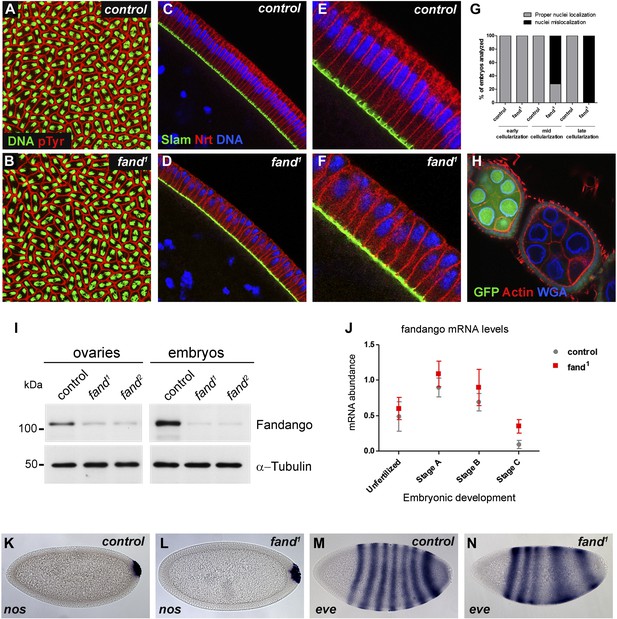
Drosophila Fandango/Xab2 is required for blastoderm cellularization.
(A and B) Panels show embryos with normal syncytial blastoderm nuclear divisions in control embryos (hs-FLP; FRT42B) (A) and fand1 germ-line clone embryos (hs-FLP; FRT42B fand1, maternal mutant) (B). Embryos were stained for DNA (green) and p-Tyrosine (red). (C–F) Panels show blastoderm cellularized embryos. Control embryos showed normal epithelial architecture with elongated nuclei and columnar cell shape (C). fand1 germ-line clone mutant embryos showed abnormal epithelial architecture, the cortical nuclei failed to elongate and became mislocalized (D). (E and F) Magnification of C and D, respectively. Embryos were stained for Slam (green), Neurotactin (red), and DNA (blue). (G) Quantification of fandango maternal mutant embryo phenotype during blastoderm cellularization. Early cellularization: control: 100% normal (n = 44), fand1: 100% normal (n = 49); mid cellularization: control: 100% normal (n = 25), fand1: 28% normal (n = 21); late cellularization: control: 100% normal (n = 42), fand1: 0% normal (n = 38). (H) Maternally controlled oogenesis was normal in fandango mutant clones. Absence of endogenous nGFP (green) indicated that the cells were homozygous for fand1 mutation. Ovaries were stained for F-actin (red) and WGA (blue). (I) Western blot of whole protein extracts from embryos and ovaries mutant for fand1 and fand2 alleles (germ-line clones) showed a clear reduction in Fandango protein levels compared to control tissues. It should be noticed that due to experimental constraints the total protein extracts from mutant ovaries included not only signal from mutant germ-line cells (homozygous for fand1), but also the tightly associated heterozygote somatic follicle cells. α-Tubulin was used as a loading control. (J) Real-time qPCR analysis showed no significant differences in fandango mRNA levels between control and fand1 embryos during development (Two-way ANOVA p>0.05 ns.). fandango mRNA levels were normalized with β-actin mRNA levels. (K–N) in situ hybridization for nanos RNA (maternal) and even-skipped RNA (early zygotic) in blastoderm cellularized embryos. Both control (K) and fand1 mutant (L) embryos showed normal nos localization pattern in the pole cells. fand1 embryos (N) showed A–P patterning defects of eve compared to control embryos (M).

fandango mutant alleles contain changes in highly conserved amino acids.
(A) Panels show the rescue of the transheterozygous zygotic lethality of fand1 and fand2 alleles. Only transheterozygous mutant females carrying the wild-type copy of fandango (wt-fandango; FRT42B, fand1/fand2) were viable (dark gray box). Transheterozygous mutant males without receiving the genomic fragment (FRT42B, fand1/fand2) died (light gray box). In the reciprocal cross, both females and males transheterozygous mutants carrying the wild-type copy of fandango, were viable (dark gray box). (B) Fandango has multiple copies of a tetratricopeptide repeat (TPR) motif, a protein–protein interaction module found in a number of functionally different proteins. A scheme displaying the distribution of conserved TPR protein domains in Fandango (top). Mutations of fand1 and fand2 alleles affect highly conserved amino acids of the TPR domains 7 and 6, respectively. fand1 contained a missense point mutation changing an alanine to a valine at aminoacid position 401 (A401V) and fand2 contained a microdeletion which resulted in loss of six conserved amino acids from position 355 to 360 (Δ355–360). Partial alignment of Fandango (Drosophila melanogaster CG6197, ref.NP_610891.1) with orthologous Xab2 (Homo sapiens, ref. NP_064581.2), Xab2 (Mus musculus, ref. NP_080432.1), Xab2 (Danio rerio, ref.NP_001038248.1), C50F2.3 (Caenorhabditis elegans, ref. NP_491250.1), cwf3 (Schizosaccharomyces pombe, ref. NP_596612.1), and SYF1 (Saccharomyces cerevisiae, ref. NP_010704.1) (bottom).
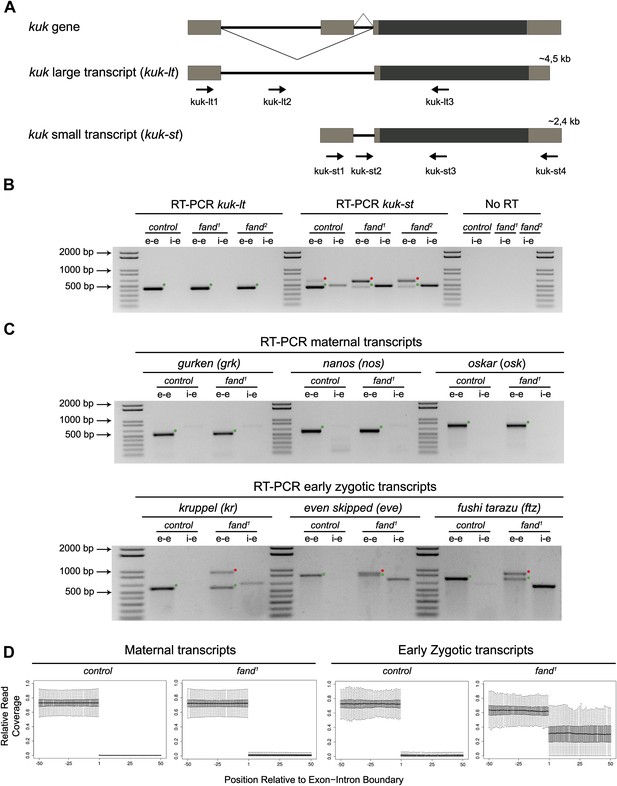
Splicing of early zygotic but not maternally encoded pre-mRNAs is affected in fandango mutants.
(A) The kugelkern (kuk) locus encodes two transcripts of different size, kuk-lt containing a large intron and kuk-st with a short intron. Orientation and position of primers used for splicing analysis is indicated (arrows). (B) RT-PCR analysis of kuk transcripts. Control embryos yielded PCR products in the size predicted for the properly spliced forms of both kuk transcripts using exon–exon (e–e) primers (green dots, kuk-lt: 431 bp and kuk-st: 437 bp). fandango maternal mutant embryos (fand1 and fand2 alleles) showed splicing defects only in the kuk-st transcript; PCR products were detected by e–e primers in the size expected for intron retention (red dots, kuk-st: 596 bp) and by intron–exon (i–e) primers (kuk-st: 474 bp). Splicing of the kuk-lt was not affected in fandango mutant background; PCR products were only detected with e–e primers in the predicted size for the correctly spliced pre-mRNA (green dots, kuk-lt: 431 bp). ‘No RT’ controls (only total RNA as template) yielded no amplification, meaning there was no contamination with genomic DNA in the samples tested. (C) RT-PCR analysis of maternal and early zygotic genes. Maternal transcripts were properly spliced, in both, control and fand1 mutant embryos; PCR products were only detected using e–e primers (green dots, grk: 527, nos: 581, osk: 762 bp). In contrast, early zygotic transcripts were correctly spliced only in control embryos (green dots, kr: 559, eve: 828, ftz: 753 bp). fand1 mutant embryos yielded PCR products in the size predicted for intron retention with e–e primers (red dots, kr: 932, eve: 899, ftz: 900 bp) and with i–e primers (kr: 629, eve: 720, ftz: 595 bp). All PCR bands showed in the panels were cloned and sequenced to confirm their identity. Green dots indicate correctly spliced transcripts, red dots indicate unspliced transcripts (intron retention). (D) RNA-Seq data confirmed that zygotic but not maternally encoded transcripts displayed a large fraction of splicing defects (intron retention) in fand1 mutant embryos. The panel shows box plot of the distribution of numbers of reads per bp relative to the total number of reads falling inside a 100 bp window centered around the 5′splice sites of zygotic (n = 408 splice sites from 270 genes) or maternal genes (n = 5876 splice sites from 2048 genes).
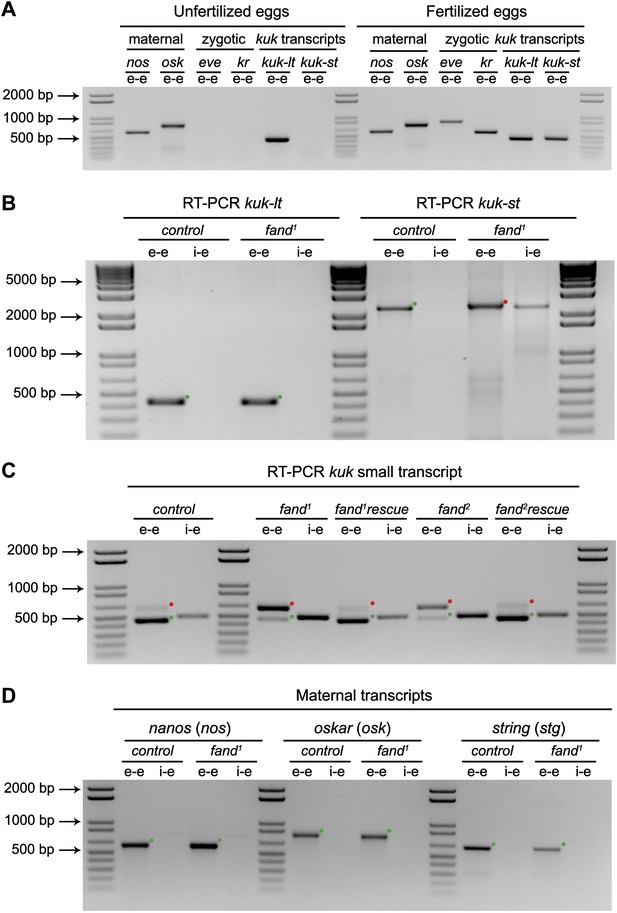
Splicing of early zygotic but not maternally encoded pre-mRNAs is affected in fandango mutants.
(A) RT-PCR analysis of kuk transcripts from unfertilized and fertilized eggs. kuk-lt is maternal and kuk-st only zygotically expressed. kuk-st (437 bp) transcripts are only detected in fertilized eggs, as are other well-known early zygotic transcripts (kr: 559, eve: 828 bp). kuk-lt (431 bp) is amplified from both fertilized and unfertilized eggs, as are other known maternal transcripts (nos: 581, osk: 762 bp). (B) RT-PCR analysis of kuk transcripts with a specific reverse primer for kuk-st (kuk-st4). Control embryos yielded PCR products with the size expected for properly spliced kuk transcripts using e–e primers (green dots, kuk-lt: 431 bp and kuk-st: 2257 bp). fand1 maternal mutant embryos showed splicing defects in kuk-st; PCR products were detected in the size expected for intron retention with e–e primers (red dot, kuk-st: 2413 bp) and by i–e primers (kuk-st: 2293 bp). Splicing of the kuk-lt was not affected in fandango mutant background; a PCR product was only detected with e–e primers in the expected size for correctly spliced pre-mRNAs (green dot, kuk-lt: 431 bp). (C) RT-PCR analysis of kuk-st showed the rescue of splicing defects observed in both fandango alleles by a genomic fragment construct derived from the third chromosome that contained a wild-type copy of fandango. Embryos analyzed were laid by GLC females FRT42B fand1/CyO; wt-fandango or FRT42B fand2/CyO; wt-fandango. GLC FRT42B and mutant GLC fand1 and fand2 embryos were used as controls. (D) RT-PCR analysis of maternally encoded transcripts from wild-type and fandango mutant ovaries (germ-line clones) failed to detect any splicing defects. The PCR products detected in both samples were of the size predicted for properly spliced pre-mRNAs (green dots, nos: 581, osk: 762, stg: 614 bp). All PCR products shown in these panels were cloned and sequenced to confirm their identity. Green dots indicate correctly spliced transcripts and red dots indicate unspliced transcripts (intron retention).
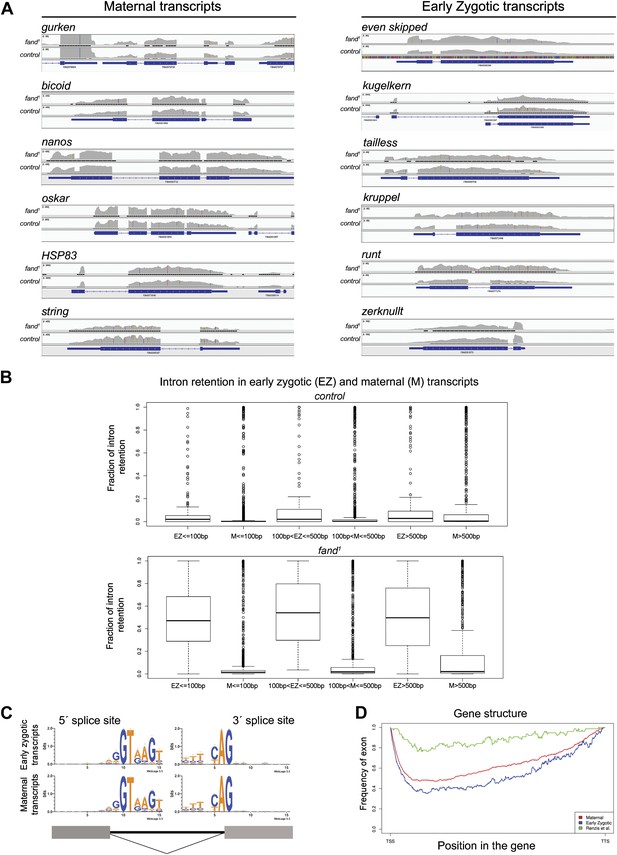
Early zygotic but not maternally encoded pre-mRNAs shows significant intron retention in fandango mutants.
(A) Zygotic genes display intron retention in the fand1 mutant, while maternal genes do not. This panel shows Integrative Genomics Viewer (IGV) screenshots of RNA-Seq read coverage from tophat alignments of a selected set of known early zygotic and maternally expressing genes. Within each gene, the image scale is identical for both fand1 and control. (B) Zygotic genes in fand1 mutant present clear evidence of intron retention, independent of intron size. The panel displays box plot distributions of percentage intron retention of exon–intron boundaries of early zygotic and maternal genes in fand1 and control. Exon–intron boundaries were divided in bins, by intron size (less than 100 bp; from 100 to 500 bp; and greater than 500 bp). Sizes were selected empirically to have comparable dataset sizes in each bin (150–200 boundaries for zygotic genes, 1000–3000 for maternal genes). The frequency of intron retention was determined by comparing the number of unsplit reads overlapping the splice site with the number of reads that showed an exon–exon split (see ‘Materials and methods’ for more details). (C) Splice sites in zygotic and maternal genes presented the same characteristic sequence pattern (5′ GT; 3′ AG). (D) Zygotic genes and maternal genes (see ‘Materials and methods’ for more details) did not reveal any distinguishing features in terms of exon–intron structure. The exon frequency is close to that expected from a random distribution, roughly 50% (with the obvious exception of gene endings—TSS and TTS). For comparison, the 59 early zygotic genes described by (De Renzis et al., 2007), 70% of which are intronless, display a very distinct, non-random, pattern.
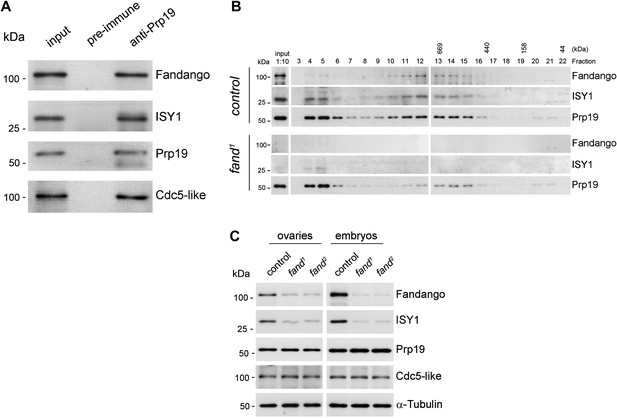
Fandango physically interacts with a similar group of splicing proteins during oogenesis and embryogenesis.
(A) Pull down assay from nuclear-enriched protein extracts using a polyclonal antibody of Prp19. Endogenous Prp19 interacts physically with Fandango and other subunits of the NTC/Prp19 complexes (ISY1 and CDC5L). Pre-immune serum was used in the control. (B) Size-exclusion chromatography of control and fand1 mutant protein extracts from 0–3 hr embryo collections using a Superose 6 10/300 column. After separation, each fraction was analyzed by Western blot. NTC/Prp19 complexes subunits (Prp19, Fandango, and ISY1) were part of a ∼600–800 kDa complex and also co-purified in a significantly larger complex (fraction 4 and 5). fand1 mutant protein extracts showed a significant reduction in levels of Fandango and ISY1 subunits and a size reduction of the Prp19-positive ∼600–800 kDa complex. (C) Western-blot analysis of total protein extracts from ovaries (left) and 0–3 hr embryos (right) from control and both fandango alleles, showed a reduction of Fandango and ISY1 protein levels in both tissues. Protein levels of Prp19 and CDC5L were not affected. α-Tubulin was used as loading control. Fandango Western blot is the same as shown in Figure 1I.
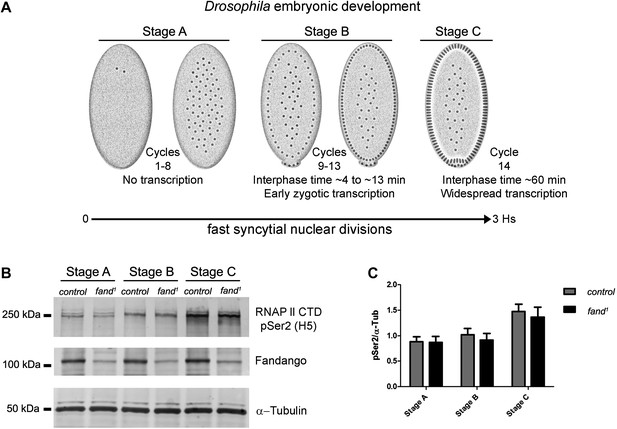
Early zygotic transcription is not affected during mid/late-syncytial blastoderm in fandango mutants.
(A) Embryos were divided into three different groups according to developmental stage (‘Materials and methods’), stage A: early/mid-syncytial blastoderm embryos, stage B: mid/late-syncytial blastoderm embryos, and stage C: blastoderm cellularization embryos. (B) Western blot for RNApol II CTD Ser2 phosphorylation levels. Control and fand1 embryos showed a similar increase in the global levels of RNApol II CTD Ser2 phosphorylation over the course of early embryonic development. α-Tubulin was used as a loading control. (C) Quantification of the CTD Ser2 phosphorylation from five independent western blot assays showed no significant difference at any of the embryonic developmental stages analyzed (Two-way ANOVA p>0.05 ns.).
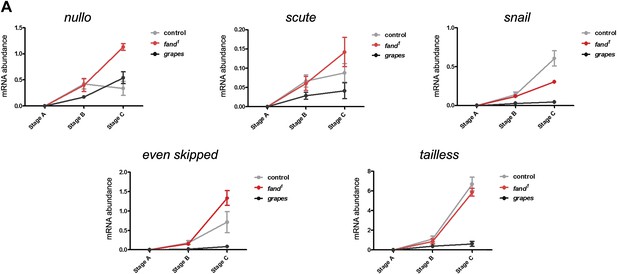
Early zygotic transcription is not affected during mid/late-syncytial blastoderm in fandango mutants.
(A) Real-time qPCR analysis to measure levels of early zygotic transcripts from intronless (nullo, snail, and scute) and containing introns genes (even-skipped and tailless) during embryonic development. mRNA levels from early zygotic genes were normalized with β-actin mRNA levels. At stage B, when early zygotic genes are transcribed, there was no significant differences in mRNA abundance for any of the genes analyzed in either control or fandango samples (Two-way ANOVA p>0.05 ns.).
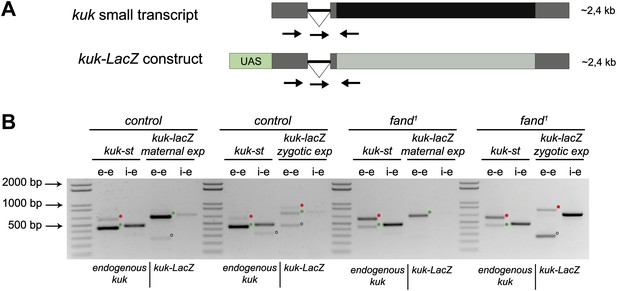
Ectopic maternal expression of an early zygotic transcript in the mutant background is sufficient to suppress its splicing defects.
(A) The kuk-LacZ construct was built using the 5′UTR, the intron and the 3′UTR of the kuk small transcript (dark gray), and replacing the kuk ORF (black) by the LacZ coding sequence (light gray). To induce the expression of this construct it was put under the control of the UAS promoter (green) to drive the tissue specific expression with GAL4 drivers. Orientation and position of primers used for splicing analysis is indicated (arrows). (B) RT-PCR analysis of the kuk-LacZ construct. When it was zygotically expressed, it was correctly spliced in control but not in fand1 embryos (similarly to the endogenous small kuk transcript). Intron retention with e–e primers (red dots, kuk-st: 596 bp and kuk-LacZ: 869 bp) and a PCR product with i–e primers (751 bp) were observed in the mutant. When it was maternally expressed, kuk-LacZ construct was correctly spliced both in control and fand1 embryos, being detected just the spliced form of the construct (green dots, kuk-st: 437 bp and kuk-LacZ: 713 bp). In contrast, the endogenous zygotically expressed small kuk transcript (kuk-st) is still poorly spliced in fand1 embryos carrying the kuk-LacZ construct. Open circles indicate unspecific PCR products (confirmed by sequencing). Green dots indicate correctly spliced transcripts, whereas red dots indicate unspliced transcripts (intron retention).
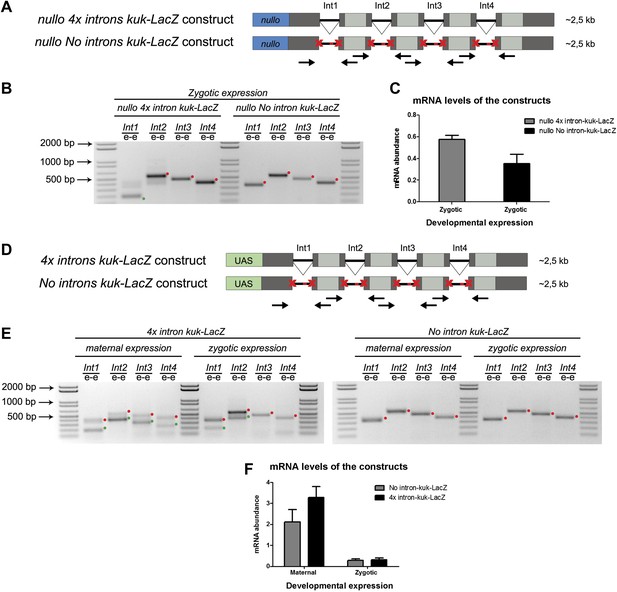
A small early zygotic transcript containing four introns is poorly spliced in wild-type embryos.
(A and D) The 4x intron kuk-LacZ construct was a variant of the kuk-LacZ that contains four copies of kuk small transcript intron (dark gray). Each intron is separated by 201 nucleotides of an in frame Lac-Z sequence (light gray). The no intron kuk-LacZ construct has all splice sites present in the 4x intron kuk-LacZ construct mutated to thymidines. The constructs were fused to a nullo minimal promoter (blue) (A), or fused to an inducible UAS promoter (green) (D). Orientation and position of primers used for splicing analysis is indicated (arrows). (B) RT-PCR analysis showed significant splicing defects (intron retention) of the 4x intron kuk-LacZ construct when expressed under the control of an endogenous early zygotic promoter (nullo promoter). The first intron was correctly spliced, being detected mainly the PCR product corresponding to the spliced form (green dot). The remaining introns (second, third, and fourth) were completely unspliced (red dots, intron retention). In the intronless (no intron kuk-LacZ) construct, under the control of the same nullo promoter were only observed PCR bands whose sizes correspond to unspliced forms (red dots, intron retention). (C) Real-time qPCR analysis showed that the 4x intron kuk-LacZ and no intron kuk-LacZ constructs were expressed to the same extent when under the control of the nullo minimal promoter (t test p>0.05 ns.). (E) RT-PCR analysis of the 4x intron kuk-LacZ construct showed significant splicing defects (intron retention) when zygotically expressed in wild-type embryos under the control of an inducible UAS promoter. Although the most 5′-localized introns (first and second) were still partially spliced, being observed two PCR bands corresponding to the spliced (green dots, int1: 191 and int2: 385 bp), and unspliced forms (red dots, int1: 347 and int2: 541 bp). The furthest 3′-localized introns (third and fourth) were completely unspliced, being only observed one PCR band with the size corresponding to intron retention (red dots, int3: 463 and int4: 385 bp). Maternal expression of the 4x intron kuk-LacZ construct was sufficient to significantly suppress splicing defects in the four introns analyzed (green dots, spliced forms: int1: 191, int2: 385, int3: 307, int4: 229 bp; red dots, unspliced forms: int1: 347, int2: 541, int3: 463, int4: 385 bp). Zygotic and maternal expression of the no intron kuk-LacZ construct only showed PCR bands with sizes corresponding to unspliced forms (red dots, intron retention). (F) Real-time qPCR analysis showed that the 4x intron kuk-LacZ and no intron kuk-LacZ constructs were expressed to the same extent both maternally (Two-way ANOVA p>0.05 ns.) and zygotically (p>0.05 ns.) in wild-type embryos. All PCR bands shown in these panels were cloned and sequenced to confirm their identity. Green dots indicate correctly spliced transcripts, red dots indicate unspliced transcripts (intron retention).
Tables
LC-MS analysis of co-immunoprecipitation assays from ovaries and embryos
| Drosophila | Human/yeast | Fandango-myc | Prp19-myc | |||||
|---|---|---|---|---|---|---|---|---|
| CG | gene | ovaries | embryos | embryos | ||||
| rep1 | rep2 | rep1 | rep2 | rep1 | rep2 | |||
| prp19 complex | ||||||||
| CG5519 | prp19 | PRP19/Prp19 | + | + | + | + | +++ | ++ |
| CG6905 | cdc5-like | CDC5L/Cef1 | + | + | + | + | +++ | ++ |
| CG1796 | Tango4 | PLRG1/Prp46 | + | + | + | + | + | + |
| CG4980 | - | BCAS2/Snt309 | – | + | - | - | + | + |
| CG12135 | c12.1 | CWC15/cwc15 | + | – | – | - | – | – |
| Prp19 related | ||||||||
| CG6197 | Fandango | Xab2/Syf1 | +++ | +++ | +++ | +++ | + | + |
| CG31368 | – | AQR/– | +++ | +++ | +++ | +++ | + | + |
| CG4886 | cyp33 | PPIE/– | ++ | ++ | + | ++ | + | + |
| CG9667 | – | ISY1/ISY1 | + | + | + | + | + | + |
| CG8264 | Bx42 | SNW1/Prp45 | + | + | + | + | + | + |
| CG14641 | – | RBM22/Cwc2 | – | + | + | + | + | – |
| CG3193 | Crn | CRNKL1/Clf1 | – | + | – | – | + | – |
| CG13892 | cypl | PPIL1/- | – | – | – | + | – | – |
| CG1639 | l(1)10Bb | BUD31/Bud31 | – | – | – | – | + | – |
-
Co-immunoprecipitations were performed using total protein extracts from the different tissues expressing Myc-tagged Fandango or Myc-tagged-Prp19. Human and yeast homologues and the different sub-complexes are shown as described in (Herold et al., 2009). (−), (+), (++), (+++) correspond to 0, 1–9, 10–19, and >20 non-repeated peptides respectively. None of the proteins shown were detected in the negative controls (for detailed LC-MS analysis see Supplementary file 1).
Additional files
-
Supplementary file 1
Complete list of proteins specifically co-immunoprecipitating with Myc-tagged Fandango and Myc-tagged Prp19.
List of proteins co-immunoprecipitating with Myc-tagged Fandango in ovaries (germ-line) and embryos, and with Myc-tagged Prp19 in embryos. Protein extracts from embryos and ovaries not expressing the Myc-tagged proteins were used as negative controls (−). Two replica experiments were performed for each condition. Proteins were identified by LC-MS-MS and data were blasted against the Flybase database using Mascot Search (see ‘Materials and methods’ for detail). The presence of a protein was measured from the total number of peptides (Matches), the number of non-repeated sequence peptides (Sequences) and the respective Score according to Mascot Search. Predictions of Human protein orthologous from Drosophila proteins were made using DIOPT (see ‘Materials and methods’ for detail). Proteins were grouped in NTC/Prp19-related complex (light gray), NTC/Prp19 complex (dark gray), general spliceosomal related proteins, miscellaneous, and ribosomal proteins. Non-reproducible (only 1 peptide in one replica) and non-specific proteins (more than 1 peptide in negative controls [−]) are listed separately. (*) Spliceosomal proteins in non-reproducible table, (**) NTC/Prp19 complex or NTC/Prp19-related complex subunits present in non-reproducible and non-specific tables.
- https://doi.org/10.7554/eLife.02181.014
-
Supplementary file 2
Complete list of primers.
Sequences of all the primers used in the RT-PCR and real-time qPCR assays presented in this manuscript (‘Materials and methods’).
- https://doi.org/10.7554/eLife.02181.015





