Expression site attenuation mechanistically links antigenic variation and development in Trypanosoma brucei
Figures
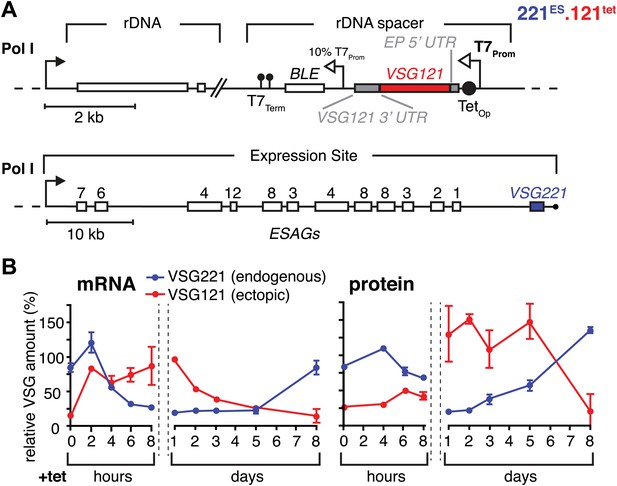
Inducible VSG121 expression leads to endogenous VSG silencing.
(A) Illustration of the ectopic VSG expression strategy. VSG121 integration into the transcriptionally silent rDNA spacer is achieved in the absence of tetracycline by using a constitutive 10% T7 promoter driving the BLE resistance cassette. High-level expression of ectopic VSG121 occurs upon tetracycline induction of a full T7 promoter. (B) Quantification of VSG mRNA and protein levels during the course of tetracycline-induced VSG121 expression. The values are percentages ± SD for two independent clones normalized to the parental 221ES or VSG121 wild-type cells. Total RNA samples were prepared at the time points indicated and analyzed by dot blotting. The blots were hybridized with VSG-specific fluorescent DNA probes and quantified by normalization to beta-tubulin mRNA using a Licor Odyssey near infrared scanner. Protein equivalents of 6 × 105 cells were dot blotted and incubated with anti-VSG121 or anti-VSG221 antibodies. Quantification was done by normalization to the paraflagellar rod (PFR) protein using the Licor Odyssey system.
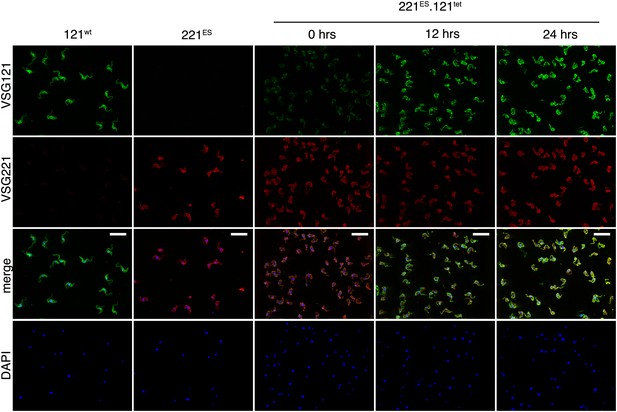
VSG immunofluorescence after tetracycline-induced VSG121 expression.
Double indirect immunofluorescence with mouse anti-VSG121 (green), rabbit anti-VSG221 (red) antibodies and DAPI staining (blue) was performed 0, 12, and 24 hr after tet induction. 121wt and parental 221ES cells served as controls for antibody specificity. Scale, 20 µm.
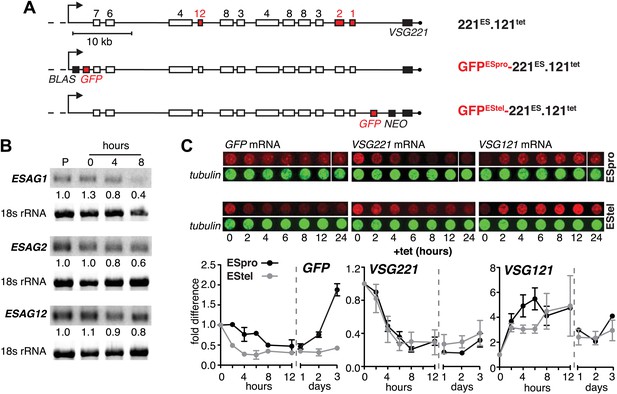
Ectopic VSG121 expression induces gradual ES attenuation.
(A) Scheme illustrating the anatomy of the VSG221 expression site in a set of reporter lines. Cell line 221ES.121tet was used to quantify three different ESAG transcripts. In GFPESpro-221ES.121tet, a GFP reporter gene was inserted just downstream of the ES promoter and in GFPEStel-221ES.121tet just upstream of the endogenous VSG221. NEO, neomycin resistance; BLAS, blasticidin resistance; numbered boxes, ESAGs; arrow, ES promoter. (B) ESAG1 and 2 transcripts decrease faster than ESAG12 mRNA. After 0, 4, and 8 hr of ectopic VSG121 induction, mRNA levels of three ESAGs were quantified using 32P-labeled probes on Northern blots and normalized to 18S rRNA using a fluorescent probe. Values are relative to the parental 221ES cells (P). (C) Expression site attenuation starts at the telomere and is released in the reverse direction. The GFP reporter lines were induced for ectopic VSG121 expression and mRNA levels of GFP, VSG221 and VSG121 were quantified using fluorescently labeled probes, normalized to beta-tubulin mRNA (upper panel). Values are relative to the non-induced levels ± SD for two independent clones (lower panel).
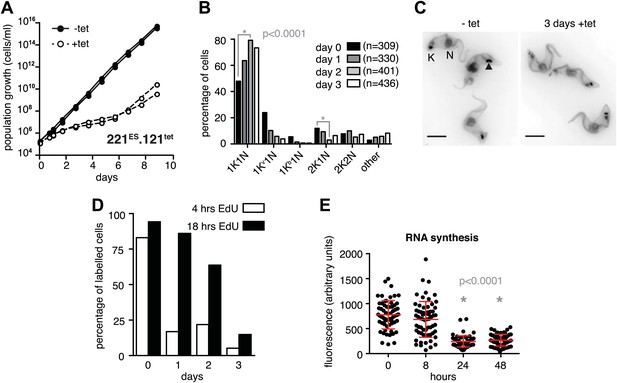
Expression site attenuation causes G1 retardation and transcriptional shut-down.
(A) Cumulative growth curve after induction of ectopic VSG121 expression. Two independent clones were analyzed for 9 days in the absence (−tet) or presence (+tet) of 1 µg/ml tetracycline. (B) Cell cycle analysis. 221ES.121tet cells were induced for VSG121 expression and fixed at the time points indicated. The configurations of the mitochondrial genome (kinetoplast, K) and the nucleus (N) were visualized by light microscopy following staining with DAPI. The configuration of K and N was subdivided into 1K1N for cells having single copies of each organelle, 1Kv1N for those with an early dividing kinetoplast and 1Kb1N with a late dividing kinetoplast. Cells in G2/M phase reveal 2K1N configuration and post-mitotic trypanosomes are marked by 2K2N. Abnormal configurations are scored as ‘other’. For each time point, n >300 cells were analyzed. Significance was determined using Fisher's exact test. (C) Cell cycle retardation has no effect on cell morphology. Trypanosomes before (−tet) and 3 days after VSG121 induction (+tet) were stained with DAPI and the cell surface labeled using AMCA-sulfo-NHS. The arrowhead points to a 1Kv1N cell. Scale bar: 5 µm. (D) Expression site attenuation leads to reduced growth, but not to cell cycle arrest. Incorporation of EdU into 221ES.121tet cells induced for VSG121 expression. The trypanosomes were induced for ectopic VSG121 expression and at 0, 1, 2, and 3 days, they were incubated with 50 µM EdU for both 4 and 18 hr. After chemical fixation the cells were co-stained with DAPI and analyzed by light microscopy (n >100 each). (E) Quantification of RNA synthesis rate after ectopic VSG121 expression. After 0, 8, 24, and 48 hr of tetracycline induction, cells were incubated in the presence of 0.5 mM BrUTP for 15 min at 33°C, subsequently chemically fixed and incubated with a monoclonal mouse anti-BrdU and an Alexa488-coupled goat anti-mouse antibody. The intensities of 3D images (100 images; z-step 100 nm) were measured in summed slice projections using the ImageJ software and are expressed as arbitrary units. To exclude variations in signal intensities due to different positions within the cell cycle, only G1 cells were analyzed. After 8 hr of tet induction, no altered fluorescence intensity was observed. After both 24 and 48 hr, fluorescence intensity was significantly decreased to 30% (p<0.0001; unpaired t test). For each time point more than 60 cells were analyzed. Red lines and error bars show mean ± SD.
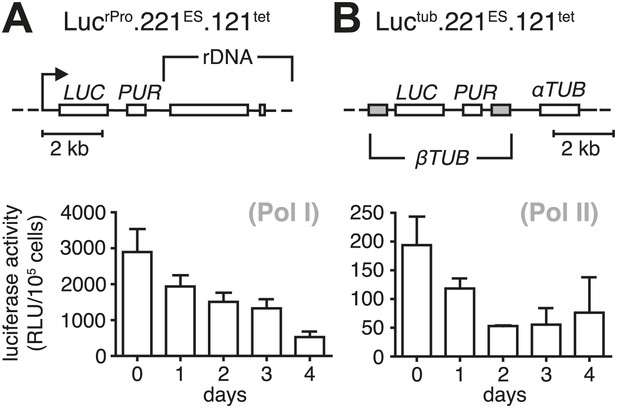
Growth retardation is accompanied by a reduced transcriptional rate of Pol I and Pol II.
Ectopic expression of VSG121 causes reduction in gene expression. (A) Luciferase reporters were inserted downstream of an rDNA polymerase I promoter and (B) into the polymerase II-transcribed tubulin locus. Values are given as mean RLU ± SD of 1 × 105 cells for three (Luctub.221ES.121tet) and two (LucrPro.221ES.121tet) independent clones.
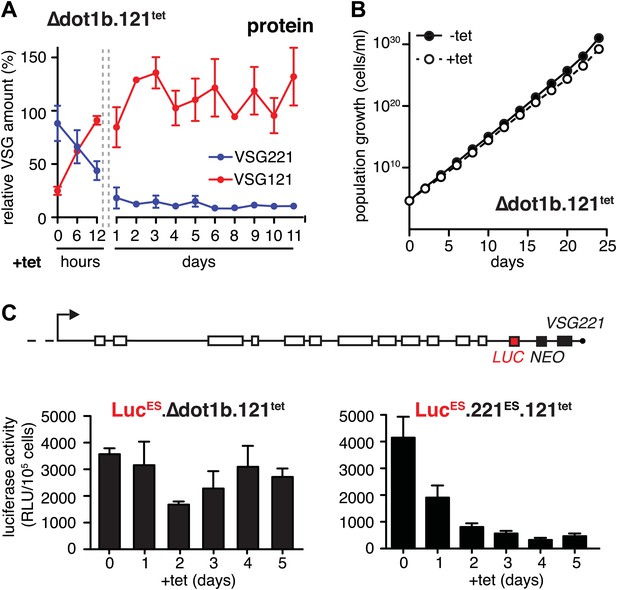
Expression site attenuation requires the histone methyltransferase DOT1B.
(A) Quantification of VSG protein levels during the course of tetracycline-induced VSG121 expression in DOT1B-depleted cells. Quantification was done by normalization to the paraflagellar rod protein (PFR), using the Licor Odyssey system. The values are percentages ± SD for two independent clones normalized to the parental Δdot1b or VSG121 wild-type cells. (B) Growth curve after induction of ectopic VSG121 expression in a Δdot1b cell line. Two independent clones were analyzed for 25 days in the absence (−tet) or presence (+tet) of 1 µg/ml tetracycline. (C) A luciferase reporter gene (LUC) was inserted into the active VSG221 ES (upper panel) of Δdot1b.121tet and 221ES.121tet cells. VSG121 expression was induced in the resulting LucESΔdot1b.121tet and LucES.221ES.121tet cells and luciferase activity of three (LucESΔdot1b.121tet) or two (LucES.221ES.121tet) independent clones was measured at the time points indicated and expressed as relative light units (RLU) ± SD.
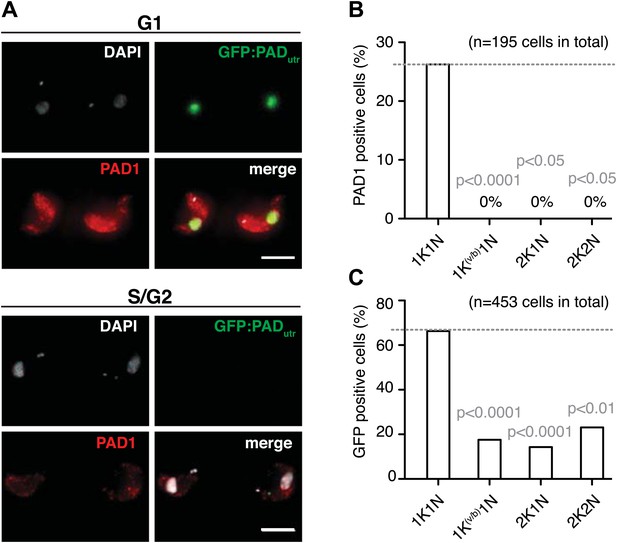
Expression site attenuation leads to PAD1 surface expression.
(A) GFP:PADutr and endogenous PAD1 are both expressed following ES attenuation. Maximum intensity projections of three-channel, 3D images (100 images, z-step: 200 nm) of GFP:PADutr.221ES.121tet cells induced for VSG121. After 48 hr, the cells were chemically fixed and incubated with an anti-PAD1 antibody (red) and counterstained with DAPI (grey). The GFP:PADutr reporter is expressed in the nucleus (green). The upper panel shows an example of G1 cells co-expressing the native PAD1 (red) and the reporter GFP:PADutr (green). S- and G2-phase cells did not show PAD1 expression (lower panel). Scale bar: 10 µm. (B and C) Cell cycle distribution of trypanosomes expressing the native PAD1 protein (B) and the GFP:PADutr reporter (C). Both the GFP-reporter and the PAD1 protein are expressed specifically in G1 cells. PAD1 expression is limited to G1. GFP:PAD1utr is visible in more G1 cells and also in other cell cycle stages. Values show percentages of positive cells of the indicated cell cycle stages and p-values were calculated using the Fisher's exact test.
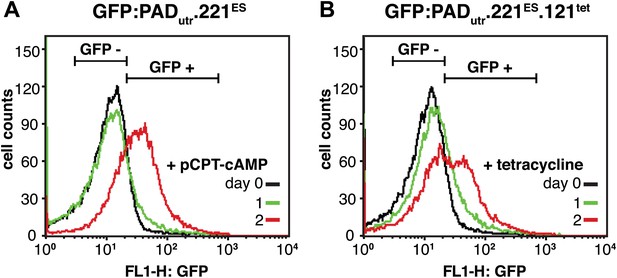
Flow cytometry analysis of GFP:PADutr expression in live cells.
(A) GFP expression of the parental 221ES cell line with the GFP:PADutr reporter inserted into the tubulin locus. Cells were analyzed for GFP expression before (day 0) and 1 and 2 days after the addition of 100 µM pCPT-cAMP, which is a membrane permeable analogue of cAMP and induces stumpy gene expression. At day 2, 42.3(±3.7)% of the cells were GFP positive (mean ± SD for two clones). (B) Flow cytometry analysis of 221ES.121tet cells with the GFP:PADutr reporter. Cells were induced for VSG121 expression and analyzed for GFP expression at the indicated times. At day 2, 31.1(±0.1)% of the cells showed GFP expression (mean ± SD for two clones).
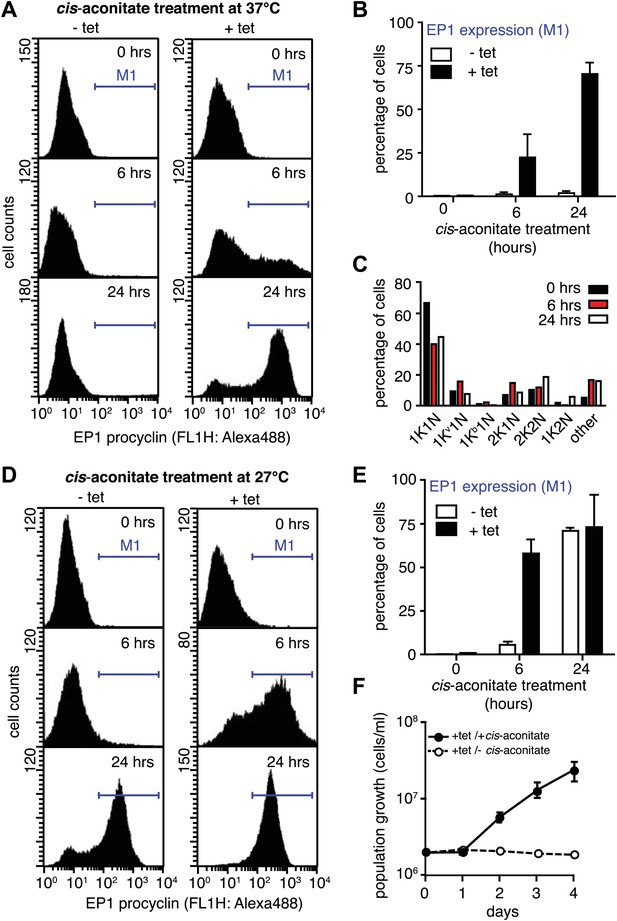
G1-retardation renders trypanosomes sensitive to the differentiation trigger cis-aconitate and provides full developmental competence at 37°C.
The response of trypanosomes to cis-aconitate was determined 48 hr after induction of ectopic VSG121 expression. (A) Flow cytometry of EP1 surface expression at 37°C. The induced cell line 221ES.121tet was treated with cis-aconitate at 37°C in bloodstream form medium for 0, 6, and 24 hr (+tet). Non-induced cells (−tet) were used as a control. (B) Percentage of cells expressing EP1 at the surface in (A). Values represent mean ± SD of two independent clones. (C) Cell cycle analysis after treatment with cis-aconitate for various lengths of time. At least 100 cells were analyzed for each time point. For details see legend to Figure 2. (D) Flow cytometry analysis of EP1 surface expression at 27°C. The experiments were conducted as described in (A) except that cells were grown in differentiation medium DTM at 27°C. (E) Percentage of cells expressing EP1 at the surface in (D). Values represent mean ± SD of two independent clones. As expected for differentiation at 27°C, after 24 hr of cis-aconitate treatment, both populations were expressing EP1. This is due to cold-shock induction of EP1 in the presence of the dominant differentiation trigger. Values are mean ± SD of two independent clones. (F) Cis-aconitate treatment releases growth attenuation. After induction for ectopic VSG121 expression for 48 hr, 221ES.121tet cells were transferred to differentiation medium at 27°C in the presence or absence of cis-aconitate and kept at a density of 2 × 106 cells/ml. Cumulative cell numbers per ml are shown as mean ± SD of two independent clones.
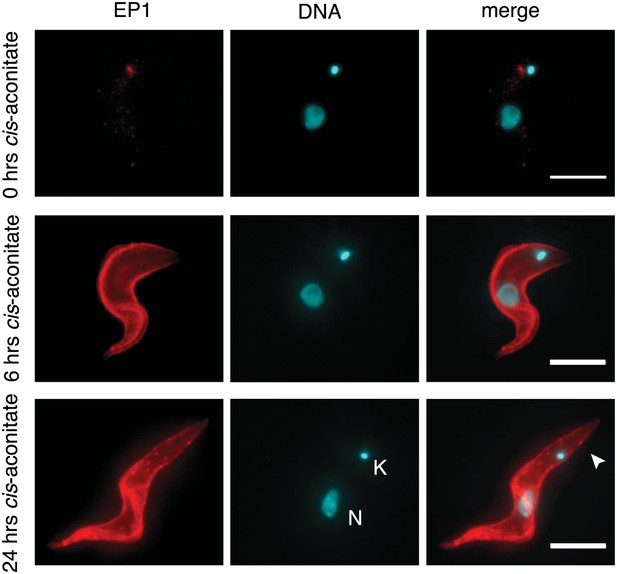
Immunofluorescence detection of the insect stage surface protein EP1.
The trypanosomes were incubated at 37°C with 6 mM cis-aconitate in bloodstream form culture medium HMI-9 for 0, 6, and 24 hr. Red indicates EP1 fluorescence and blue is DAPI staining of the nucleus (N) and kinetoplast (K). Note the characteristic posterior elongation of the cell marked by an arrowhead. Scale bar, 5 µm.
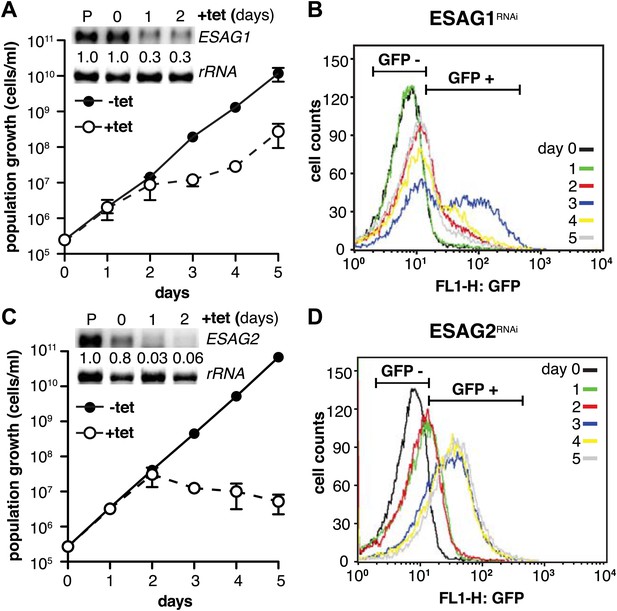
Depletion of ESAG 1 or 2 by RNAi causes distinct growth defects and induction of PAD1 expression.
(A and C) Growth of GFP:PADutr cells induced for RNAi against either (A) ESAG1 or (C) ESAG2 is presented as the cumulative mean cell number ± SD for two (ESAG1) and three (ESAG2) independent clones. Upper panels in (A) and (C) show Northern blots specific for ESAG1 and 2, respectively, normalized to 18S rRNA. (B and D) Flow cytometric analysis of GFP:PADutr expression in a time course of (B) ESAG1-RNAi and (D) ESAG2-RNAi.
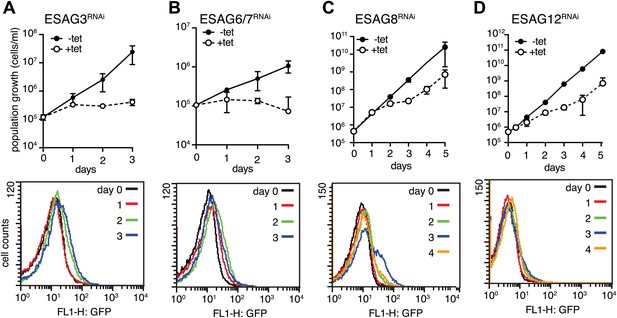
Down-regulation of a subset of three ESAGs induces GFP:PADutr.
Growth curves (upper panels) and flow cytometry analysis (lower panels) of GFP:PADutr cells induced for RNAi against several ESAGs. Depletion of ESAGs 3 (A) and 6/7 (B) causes severe growth phenotypes without induction of GFP:PADutr expression. In contrast, RNAi against ESAGs 8 (C) and 12 (D) leads to transient growth retardation after day 2, but only ESAG8 depletion leads to the appearance of a fraction of GFP-positive cells at day 3. All growth curves show the cumulative cell numbers/ml of uninduced (−tet) and induced (+tet) cells as mean ± SD for 2 (ESAGs 3, 8 and 12) and 3 (ESAG 6/7) independent clones.
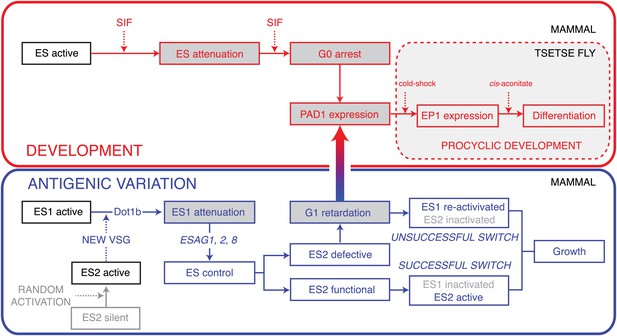
Model of the dual roles of expression site attenuation for antigenic variation and development of T. brucei.
Activation of a new ES leads to attenuation of the previously active one in a DOT1B-dependent manner. The attenuation and subsequent growth retardation allows ES quality control via ESAG1, 2 and 8. If the newly activated ES is functional, the old one is rapidly silenced, resulting in a successful VSG switch. If, however, a non-functional or non-compatible ES is activated, the cells react with a specific prolongation of the G1-cell cycle phase. This dormancy can last for up to 5 days and is fully reversible. Within this period the trypanosomes become competent for developmental stage transition, which involves expression of the stumpy marker PAD1. The stumpy stage, however, is irreversibly arrested in G0 and therefore committed to a life in the tsetse fly. In contrast, ES attenuation causes a reversible G1 dormancy, which allows either re-activation of the attenuated ES (unsuccessful switch) or survival by development in the insect vector. Thus, ES attenuation operates at the crossroads of antigenic variation and development.
Additional files
-
Supplementary file 1
Cell lines and targeting constructs.
- https://doi.org/10.7554/eLife.02324.016





