Complete morphologies of basal forebrain cholinergic neurons in the mouse
Figures
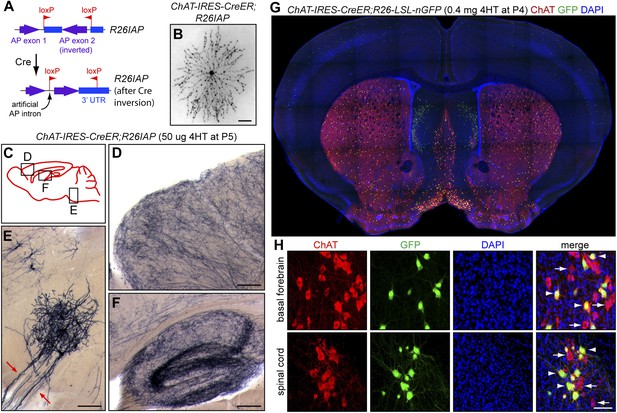
Cholinergic neuron specificity of Cre-mediated recombination.
(A) Structure of the R26IAP knock-in. In the absence of Cre-mediated recombination, the 3′ half of the AP coding region is inverted in the germline configuration. It assumes the correct orientation following Cre-mediated recombination between inverted loxP sites. (B) P30 retina from Chat-IRES-CreER;R26IAP mice treated with 4HT. AP histochemistry labels cholinergic (starburst) amacrine cells. Scale bar, 100 µm. (C–F) P30 brain from Chat-IRES-CreER;R26IAP mice treated with high dose 4HT at P5. AP histochemistry labels numerous axons throughout the cortex (D) and hippocampus (F), as well as cranial motor neurons (E), the axons of which are seen exiting the brain stem (red arrows). Scale bars in D–F, 200 µm. (G and H) Coronal sections of P30 forebrain from Chat-IRES-CreER;R26-LSL-nGFP mice treated with high dose 4HT at P4. Approximately 50% of cholinergic neurons in the basal forebrain, medial septal nucleus, striatum, and spinal cord (visualized with ChAT immunohistochemistry) are GFP+. Medial to the striatum, a distinctive group of GFP+ cell is ChAT−; these cells presumably expressed Chat (and, therefore, Cre) in the early postnatal period and then repress Chat expression in adulthood. In (H), arrows point to ChAT+;GFP− neurons and arrowheads point to ChAT+; GFP+ neurons. Scale bar, 50 µm.
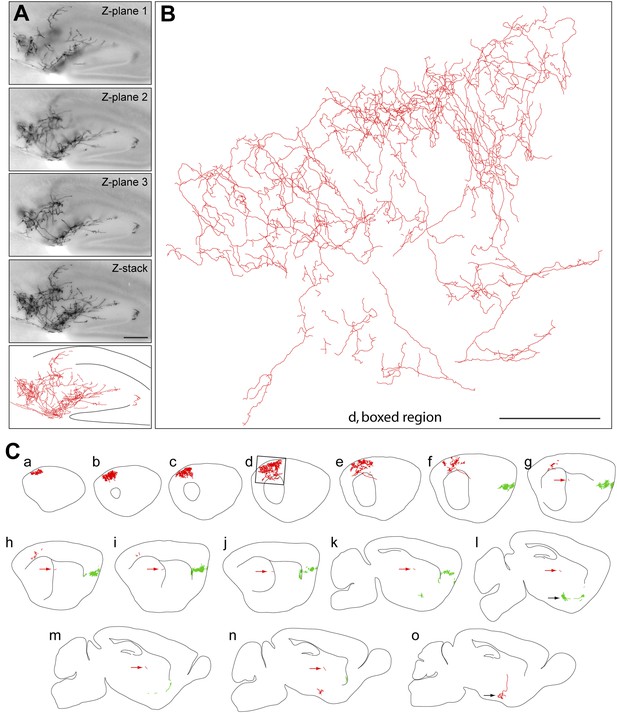
Axon arbors of forebrain cholinergic neurons from P30 Chat-IRES-CreER;R26IAP mice visualized with sparse Cre-mediated recombination.
(A) Part of the arbor of a forebrain cholinergic neuron in a P30 hippocampus visualized in a single 300 µm section at three Z-planes and in a Z-stacked image. Bottom, the traced arbor. Scale bar, 200 µm. (B and C) Fifteen consecutive 300 µm sagittal sections from a single P30 hemisphere (C) with two fully traced AP+ forebrain cholinergic neurons, colored red and green. Black arrows in panels l and o, the two cell bodies. Red arrows in g–n, the proximal axon segment for the red neuron trace. (B) An enlarged view of the boxed region of section d in (C). Scale bar in (B), 500 µm (corrected for tissue shrinkage in BBBA).
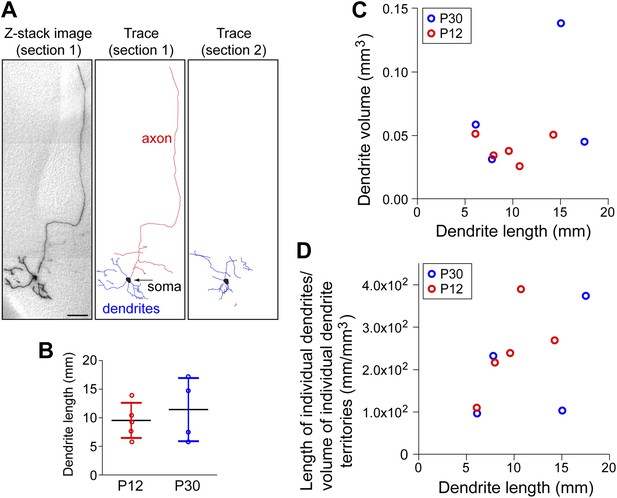
Dendrite structure among forebrain cholinergic neurons.
(A) Left, Z-stack image of a 300 µm sagittal brain section (‘section 1’) shown in panel ‘o’ of Figure 2C. Middle, trace of AP+ dendrites, part of the soma, and proximal axon arbor for the forebrain cholinergic neuron in section 1. Right, trace of the remainder of the dendritic arbor and the second part of the soma from the adjacent 300 µm sagittal brain section (‘section 2’) shown in panel ‘n’ of Figure 2C. Scale bar, 200 µm. (B) Total dendrite lengths for five P12 and four P30 forebrain cholinergic neurons. Bars show the mean +/− SD. (C) Dendrite arbor volume measured with the polygon method vs dendrite length for the same five P12 and four P30 forebrain cholinergic neurons plotted in panel B. (D) Individual dendrite arbor density (dendrite length divided by dendrite arbor volume) vs dendrite length for the same five P12 and four P30 forebrain cholinergic neurons plotted in panel B.
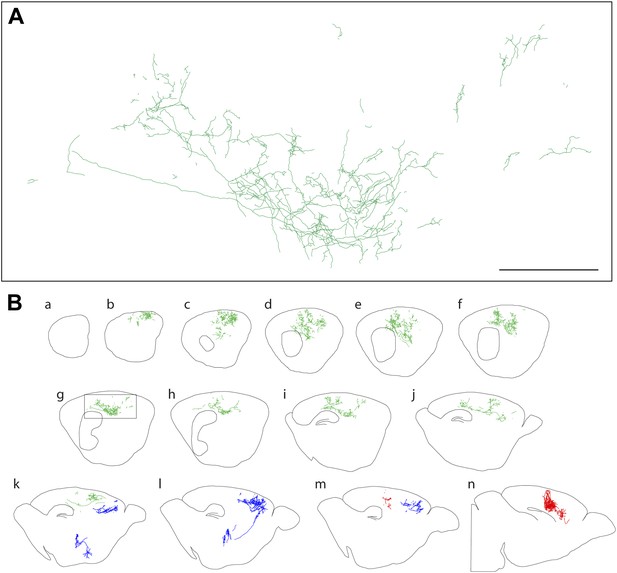
Axon arbors of forebrain cholinergic neurons from P12 Chat-IRES-CreER;R26IAP mice visualized with sparse Cre-mediated recombination.
(A) Enlarged view of the boxed region from panel g in (B). Scale bar, 500 µm (corrected for tissue shrinkage in BBBA). (B) Fourteen consecutive 300 µm sagittal sections from a single P12 hemisphere (a–n) with three traced AP+ cholinergic neurons, colored red, green, and blue. The blue neuron is shown in its entirety, including the cell body and dendrites in the basal forebrain; for the green and red neurons, only the cortical axon arbors are shown.
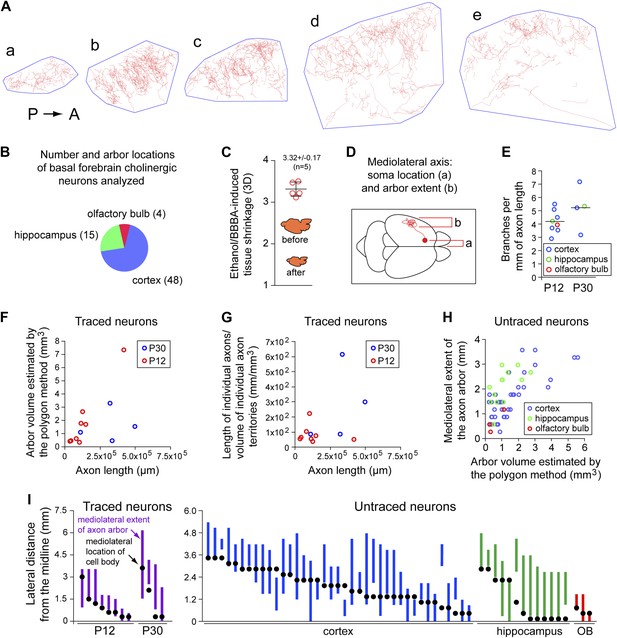
Quantitative analysis of morphologic parameters for cholinergic axon arbors.
(A) The polygon method for estimating the target area for a single cholinergic axon arbor. Traced axon arbor images are shown for each 300 µm sagittal section from the P30 brain in Figure 2C, panels a–e. A minimal convex polygon has been drawn around each trace, providing an upper estimate of the cortical territory that is directly influenced by the arbor. As seen in panels d and e, the polygon method somewhat overestimates the target area by including regions that are relatively far from the axon. P, posterior. A, anterior. (B) Axon arbor locations for the 67 basal forebrain cholinergic neurons analyzed. (C) Quantification of tissue volume shrinkage due to dehydration in ethanol and BBBA. (D) Schematic of a forebrain cholinergic neuron in a dorsal view of the mouse brain showing the mediolateral cell body and arbor locations, the parameters displayed in panels (H) and (I). (E) The number of branch points per mm of axon length for the 12 forebrain cholinergic neurons that were traced. These data were obtained from two 300 µm sections per arbor by measuring the total axon length and counting all branch points for the AP+ arbor within each section. (F) Scatter plot of arbor volume (estimated using the polygon method) vs axon length for the 12 forebrain cholinergic neurons that were traced. (G) Scatter plot of axon density (length divided by arbor volume) vs axon length for the 12 forebrain cholinergic neurons that were traced. (H) Scatter plot of the mediolateral extent (defined in panel D) vs arbor volume (estimated using the polygon method) for the 55 forebrain cholinergic arbors that were not traced. (I) Mediolateral cell body and arbor locations for the 12 traced neurons (left) and the 55 untraced neurons (right). Black dots represent cell body location and the vertical bar represents the mediolateral extent of the axon arbor. OB, olfactory bulb.
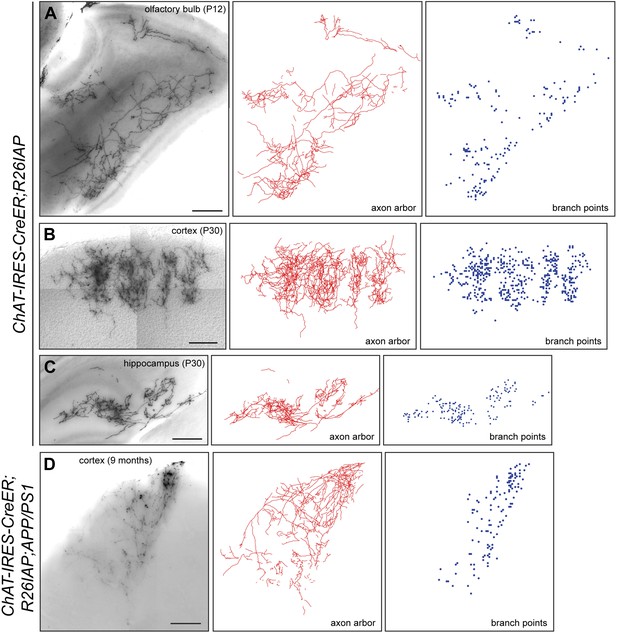
Cholinergic axon arbors: Z-stacked images, traces, and branch point locations.
(A–D) Axon arbors from single cholinergic neurons visualized in 300 µm sagittal sections from olfactory bulb (A), cortex (B and D), and hippocampus (C): Z-stacked images (left), traces (center), and branch points (right). Z-stacked images (A–C) are from Chat-IRES-CreER;R26IAP brains at P12 or P30 and image (D) is from a Chat-IRES-CreER;R26IAP;APP/PS1 brain at 9 months. Scale bars, 200 µm.
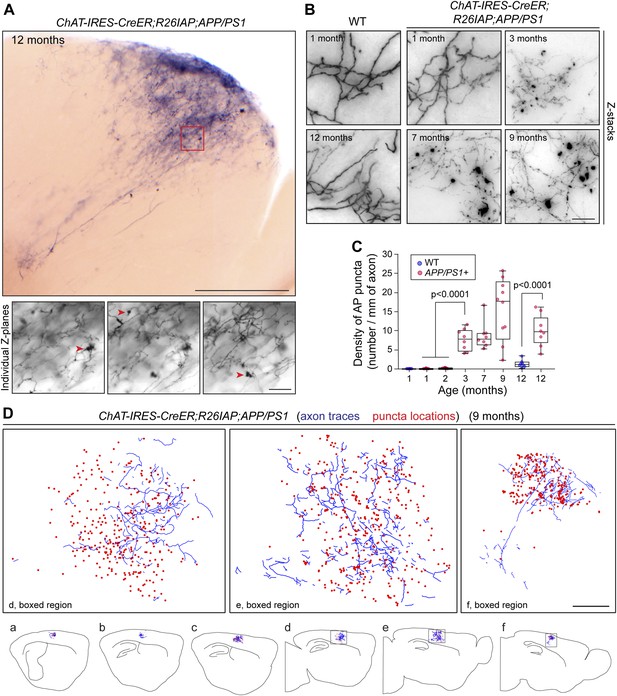
Disruption of cholinergic axon arbors in Chat-IRES-CreER;R26IAP;APP/PS1 mice.
(A) Upper panel, 300 µm sagittal section of a 12 month old Chat-IRES-CreER;R26IAP;APP/PS1 brain showing part of a single AP+ axon arbor. The olfactory bulb is visible at lower right. Lower panels, three Z-planes enlarged from the region enclosed in the red square in the upper panel. Red arrowheads point to clumps of AP+ material (puncta). Scale bars: upper panel, 500 µm; lower panels, 50 µm. (B) Comparison of representative regions from forebrain cholinergic axon arbors in the cortex of Chat-IRES-CreER;R26IAP brains (WT; left) and Chat-IRES-CreER;R26IAP;APP/PS1 brains (right), between one and 12 months of age. Structural heterogeneity, including the clumping of AP + material (puncta) and loss of AP staining intensity, increases with age in the APP/PS1 background. Scale bar, 50 µm. (C) Quantification of AP+ puncta in the cortex and hippocampus of Chat-IRES-CreER;R26IAP (i.e., WT) and Chat-IRES-CreER;R26IAP;APP/PS1 mice at different ages. Puncta appear at 3 months in Chat-IRES-CreER;R26IAP;APP/PS1 mice. The box plots indicate the extreme data points (top and bottom bars), the 25–75% interval (box), and the median (central line). p-values, student's t test. (D) Complete tracing of an AP+ cortical cholinergic arbor (blue) with the locations of AP + puncta (red dots) indicated. Panels a–f show six adjacent 300 µm sagittal sections within which this arbor resides. The three enlarged images above correspond to the boxed regions in panels d–f. Scale bar, 500 µm (corrected for tissue shrinkage in BBBA).
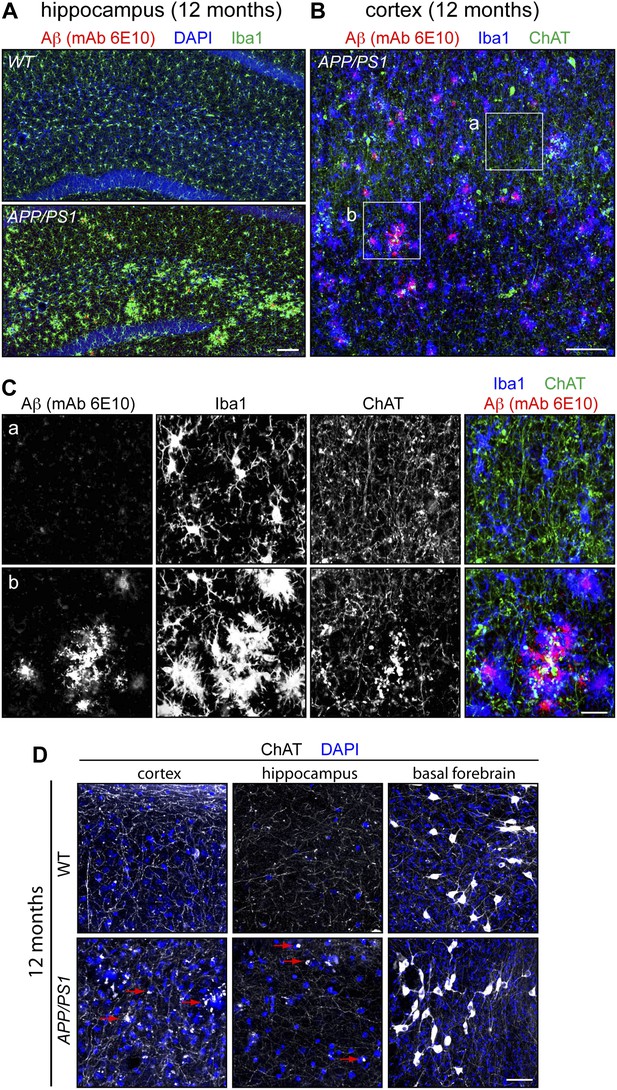
Aβ deposition, microglial reorganization, and disorganization of cholinergic fibers in the APP/PS1 brain.
(A) The hippocampus in WT and APP/PS1 mice at 12 months. Numerous Aβ deposits are seen in the APP/PS1 hippocampus, and are associated with migration of microglia, visualized by ionized calcium binding adaptor molecule 1 (Iba1). Scale bar, 100 µm. (B) The cortex of an APP/PS1 mouse at 12 months. Numerous Aβ deposits are present, along with an irregular distribution of microglia. Scale bar, 100 µm. (C) Enlarged views of the boxed regions in panel (B). Boxed region ‘a’ lacks large Aβ deposits. Boxed region ‘b’ has numerous large Aβ deposits surrounded by microglia. ChAT immunostaining shows numerous puncta in the region corresponding to Aβ deposits. Scale bar, 25 µm. (D) Cholinergic axons in the cortex and hippocampus and cholinergic cell bodies in the basal forebrain in WT and APP/PS1 brains at 12 months. Cholinergic cell bodies in the basal forebrain show little or no difference between the two samples. Cholinergic axons show numerous puncta in the APP/PS1 cortex and hippocampus, some of which are highlighted by red arrows.
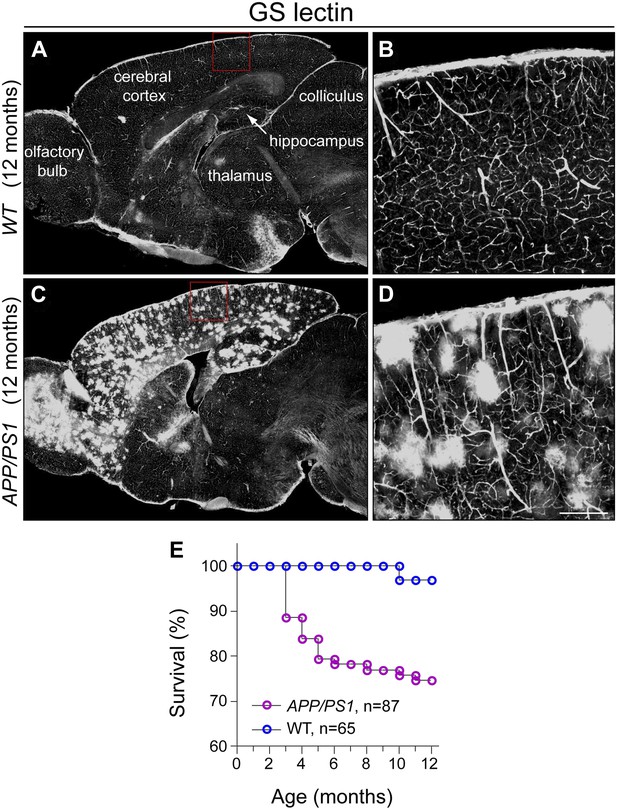
Characterization of APP/PS1 mice: survival and microglial activation.
(A and B) Sagittal section of a 12 month old WT brain stained with GS-lectin, which visualizes vascular endothelial cells and activated microglia. In the cortex and hippocampus, binding is limited to the vasculature. The boxed region in (A) is enlarged in (B). (C and D) Sagittal section of a 12 month old APP/PS1 brain stained with GS-lectin as for panels (A and B). In the cortex, hippocampus, and olfactory bulb, GS-lectin binding is observed in the vasculature and in numerous clusters of activated microglia. Activated microglia are not present in other brain regions. The boxed region in (C) is enlarged in (D). (E) Kaplan–Meier survival curves for Chat-IRES-CreER;R26IAP (i.e., WT) and Chat-IRES-CreER;R26IAP;APP/PS1 mice.
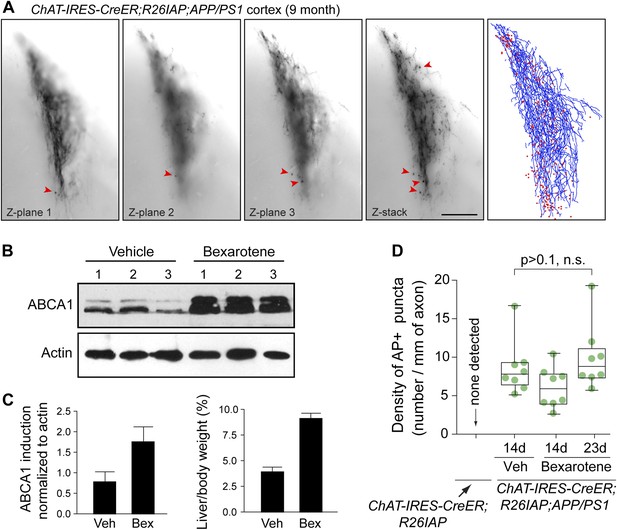
Tracing AP+ axons, and locating and quantifying AP+ puncta with and without bexarotene treatment.
(A) Example of a cortical arbor from a Chat-IRES-CreER;R26IAP;APP/PS1 brain at 9 months of age visualized in a single 300 µm sagittal section at three Z-planes (left three panels) and in a Z-stacked image (fourth panel). Arrowheads point to some of the larger puncta. Fifth panel, the traced arbor (blue) and AP+ puncta (red). Scale bar, 200 µm. (B) Induction of ABCA1 in cerebral cortex by bexarotene treatment, assessed by anti-ABCA1 immunoblotting. 7-month old mice received vehicle (DMSO; n = 3) or 100 mg/kg bexarotene in DMSO (n = 3) once daily for 7 days by oral gavage. The six mice were sacrificed on day 8. (C) Quantification of ABCA1 immunoblot signal intensity (panel B) and the ratio of liver weight to body weight in mice that received vehicle (DMSO; ‘veh’) or 100 mg/kg bexarotene in DMSO (‘bex’) once daily for 7 days by oral gavage. Bexarotene treatment induces a rapid hepatomegaly. (D) Quantification of AP+ puncta in the cortex and hippocampus of untreated Chat-IRES-CreER;R26IAP (i.e., WT) mice and Chat-IRES-CreER;R26IAP;APP/PS1 mice treated with daily 100 µl gavages of DMSO for 14 days or bexarotene in DMSO for 14 or 23 days. The box plots indicate the extreme data points (top and bottom bars), the 25–75% interval (box), and the median (central line). There is no statistically significant effect of bexarotene treatment on the density of AP+ puncta. p-value, student's t test.

Quantifying ChAT+ axon density in P30 mouse cortex.
(A) Coronal section of P30 mouse motor cortex following ChAT immunostaining. The cortical surface is at the top; the base of the cortex is at the bottom. Confocal images of the fluorescently immunostained tissue (converted to grey scale and inverted) were captured at Z-plane separations of 2 µm. Z-stacks encompassing planes 3–6 and 7–10 are shown. The traced axons for both sets of Z-stacks are color-coded with axons in stacks 3–6 in green, axons in stacks 7–10 in red, and regions of overlap in blue. Scale bar, 25 µm. (B) Axon tracings corresponding to the region in the right panel in (A) that is demarcated by the vertical red line. Scale bar, 25 µm. (C) ChAT immunostaining corresponding to the region adjacent to the left panel in (A) that is demarcated by the vertical black line. Scale bar, 25 µm. (D) The red rectangle shows the region of motor cortex analyzed in (A), at approximately Bregma −1.06.
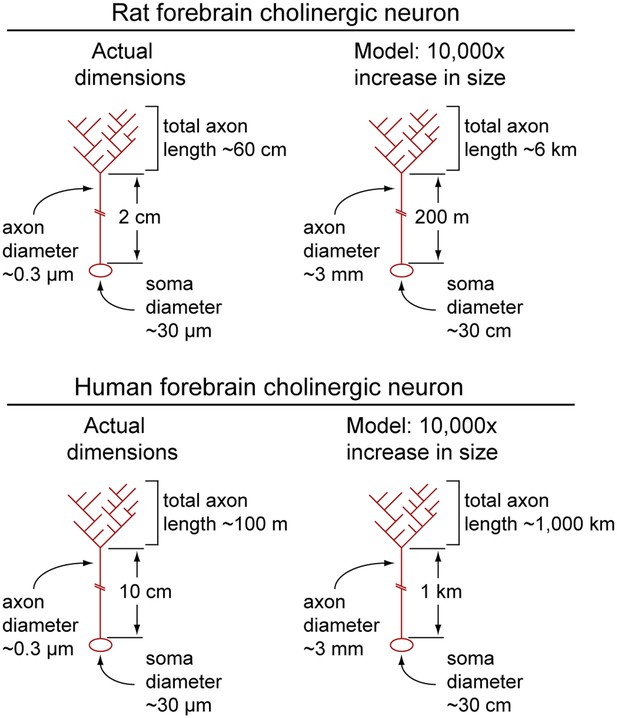
Calculated dimensions of rat and human forebrain cholinergic neurons.
Actual dimensions of human and rat forebrain cholinergic neurons, calculated from the data in Table 1 (left image in each pair). A macroscopic model in which the linear dimensions of each cholinergic neuron have been multiplied 10,000-fold (right image in each pair). Soma and axons are shown; dendrites have been omitted.
Tables
Axon arbor lengths for diffuse projection neurons
| Basal forebrain cholinergic neurons (nucleus basalis of Meynert to cortex) | |||||
|---|---|---|---|---|---|
| Species | Number of neurons | Axon density in cortex | Axon length in cortex | Cortical volume | Mean axon length/neuron |
| Mouse | 6632 (a) | 1300 m (b) | 109 mm3 (c) | 20 cm | |
| 4500 (b) | 1300 m (b) | 109 mm3 (c) | 29 cm | ||
| Mouse | 6632 (a) | 0.020–0.044 µm/µm3 (d; this study) | 109 mm3 (c) | 33–72 cm | |
| 4500 (b) | 0.020–0.044 µm/µm3 (d; this study) | 109 mm3 (c) | 48-107 cm | ||
| Mouse* | 4 traced neurons following CreER/loxP labeling (this study) | 31 cm | |||
| Rat | 7,312 (d) | 0.0113 µm/µm3 (e) | 400 mm3 (f) | 62 cm | |
| Chimp | 315,000 (g) | 0.066 µm/µm3 (h) | 147 cm3 (i) | 31 m | |
| Human | 435,000 (g) | 0.080 µm/µm3 (h) | 583 cm3 (i) | 107 m | |
| Nigro-striatal dopaminergic neurons | ||||
|---|---|---|---|---|
| Species | Number of neurons | Varicosities per axon length | Number of TH + varicosities in the striatum | Mean axon length/neuron |
| Rat | 7000 (j) | 5–7 varicosities/7 µm (j) | 3.4 × 109 per side (j) | 55–77 cm |
| Rat* | 8 traced neurons following sparse GFP virus infection (k) | 47 cm | ||
| Serotonergic neurons (dorsal raphe nucleus to cortex) | ||||
|---|---|---|---|---|
| Species | Number of neurons | Axon density in cortex | Cortical volume | mean axon length/neuron |
| Rat | 11,500 (l) | 0.023 µm/µm3 (m) | 400 mm3 (f) | 80 cm |
| 15,191 (n) | 0.023 µm/µm3 (m) | 400 mm3 (f) | 61 cm | |
| Human | 80,386 (o) | 0.048 µm/µm3 (r) | 583 cm3 (i) | 348 m |
| 86,565 (p) | 0.048 µm/µm3 (r) | 583 cm3 (i) | 323 m | |
| 165,000 (q) | 0.048 µm/µm3 (r) | 583 cm3 (i) | 170 m | |
| Hippocampal CA3 pyramidal neurons | ||
|---|---|---|
| Rat* | 1 traced neuron following neurobiotin injection (s) | 48 cm |
| Cutaneous sensory neurons with free endings in back skin | ||
|---|---|---|
| Mouse* | 7 traced neurons following CreER/loxP labeling (t) | 71 cm |
-
References: (a) Perez et al., 2007; (b) Boncristiano et al., 2002; (c) Kovacević et al., 2005; (d) Miettinen et al., 2002; (e) Mechawar et al., 2000; (f) Mengler et al., 2013; (g) Raghanti et al., 2011; (h) Raghanti et al., 2008a; (i) Rilling and Insel, 1999; (j) Anden et al., 1966; (k) Matsuda et al., 2009; (l) Descarries et al., 1982; (m) Cunningham et al., 2005; (n) Vertes and Crane, 1997; (o) Underwood et al., 2007; (p) Underwood et al., 1999; (q) Baker et al., 1991;(r) Raghanti et al., 2008b; (s) Wittner et al., 2007; (t) Wu et al., 2012. The asterisk (*) marks experiments in which individual axon arbors were traced.





