Head-to-tail interactions of the coiled-coil domains regulate ClpB activity and cooperation with Hsp70 in protein disaggregation
Figures
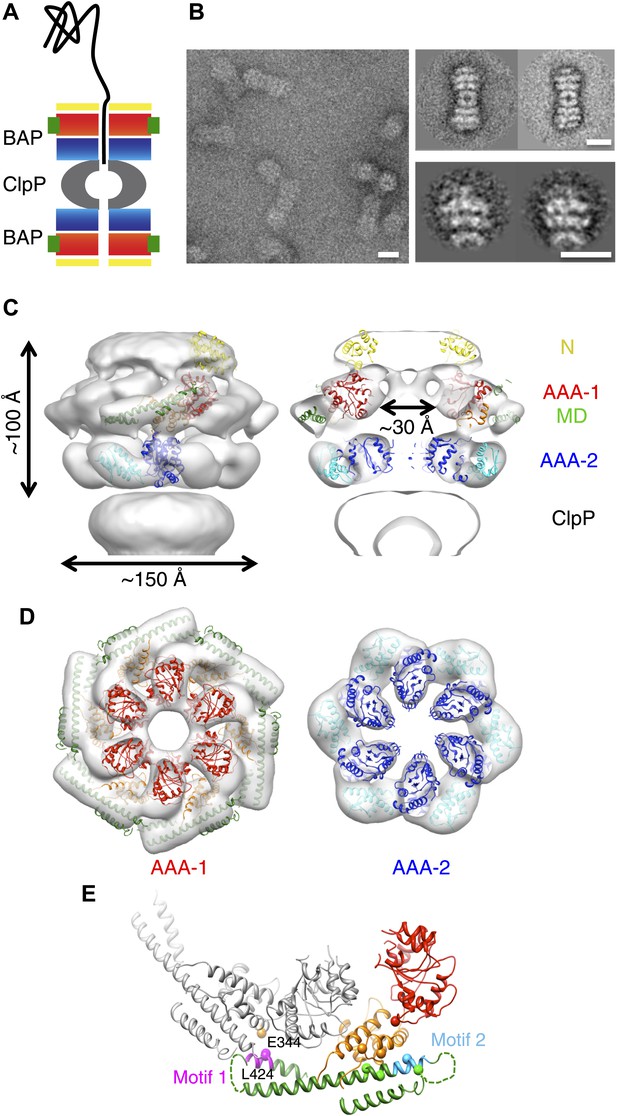
3D structure of the symmetrised BAP-ClpP complex.
(A) Schematic of the complex showing the threading of a polypeptide. ClpB domains are in yellow, N-terminus; red/orange, AAA-1 domain; green, MD and blue/cyan, AAA-2 domain. ClpP is in grey. (B) Negative stain EM image (left panel), class averages of 2:2 BAP-ClpP (upper right panel) and cut out 1:1 BAP-ClpP particles (bottom right panel). Scale bars are 150 Å. (C) BAP-ClpP structure with a fitted ClpB monomer (left) and central slice with fitted atomic coordinates (right). (D) AAA-1 and AAA-2 layers. (E) Two adjacent AAA-1 domains from the hexameric fit viewed in the plane of the ring. One monomer is in colour and the other in grey. Residues involved in engineered disulphide bonds are shown as spheres. Residues involved in intermolecular cross-links are labeled. The separation between cross-linking Cα pairs ranges from 8 to 14 Å.
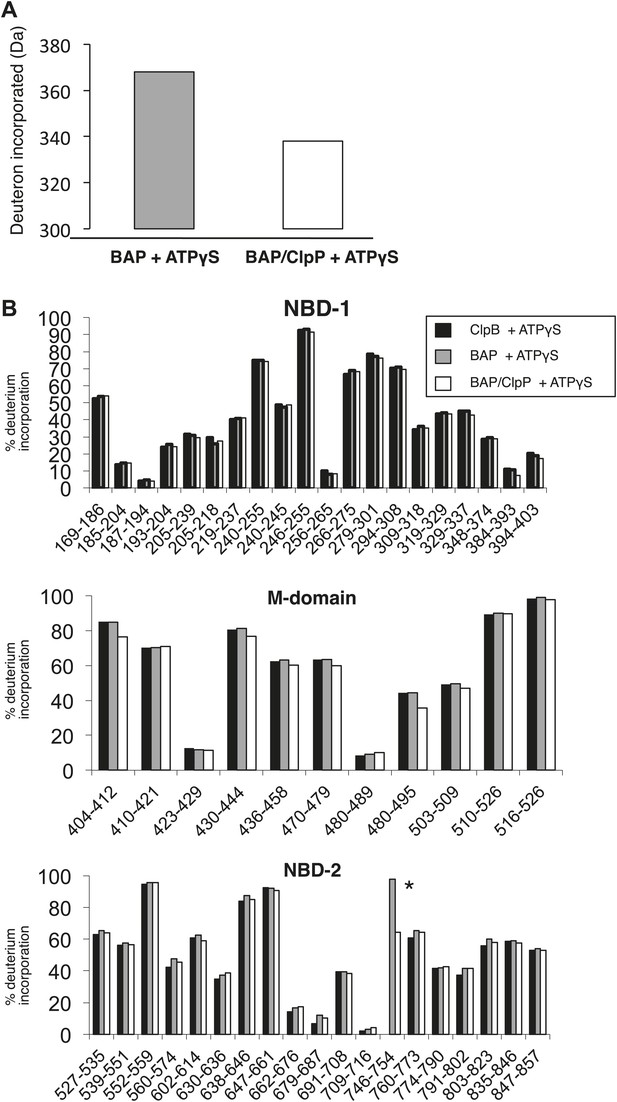
Complex formation between BAP and ClpP does not change MD protection pattern in HX experiments.
(A) BAP and ClpP form a stable complex during HX experiments causing stabilization of BAP. Protein samples preincubated with ATPγS (BAP, BAP-ClpP) were diluted 20-fold into D2O buffer and incubated for 60 s before quenching the proton-deuteron exchange reaction. The mass of full length BAP after deuteron incorporation was determined. Complex formation with ClpP protects 30 amide protons in BAP from exchange. (B) ClpB, BAP and BAP-ClpP were incubated with ATPγS and HX values of peptic peptides were determined after 60 s. Peptide 746–754 (*, KKIFRPEFI) includes the ClpP-binding loop of ClpA and is only present in BAP. Relative (%) deuterium incorporation levels are given for each peptide.

The BAP N-terminus is highly mobile.
(A) Class averages of BAP-ClpP. The arrows indicate the N-terminal (yellow), AAA-1 (red), AAA-2 (blue) and ClpP (black) tiers. (B) Eigen images showing that high variance is localized in the N-terminal ring. Scale bars are 50 Å.

Fourier Shell Correlation plots to estimate EM map resolution.
For each sample, FSC curves of C6 and C1 reconstructions are plotted together. The C1 curves show lower resolution in each pair. Example class averages are shown in the left column with corresponding re-projections on the right. Scale bar is 50 Å. FSC plots were deposited in the 3D-EM database EM (www.emdatabank.org).
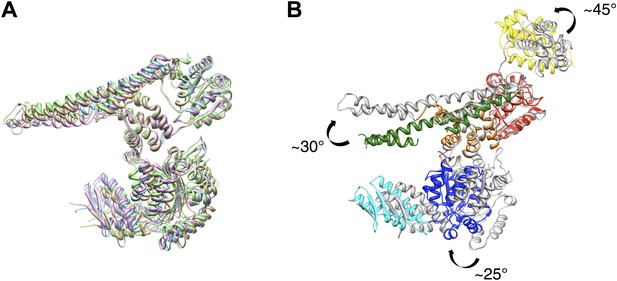
Crystal structure of E. coli ClpB and EM based model.
(A) Superimposed chains of E. coli ClpB and T. thermophilus crystal structures. For clarity, the N-terminus is not shown. (B) Comparison between the ClpB coordinates fitted into the EM maps (in colours) and the ClpB crystal structure (grey). In the EM model the MD is more tilted than in the crystal structures. AAA-2 is rotated by ∼25°, and the N-terminus is rotated by 45° in the plane of the ring.
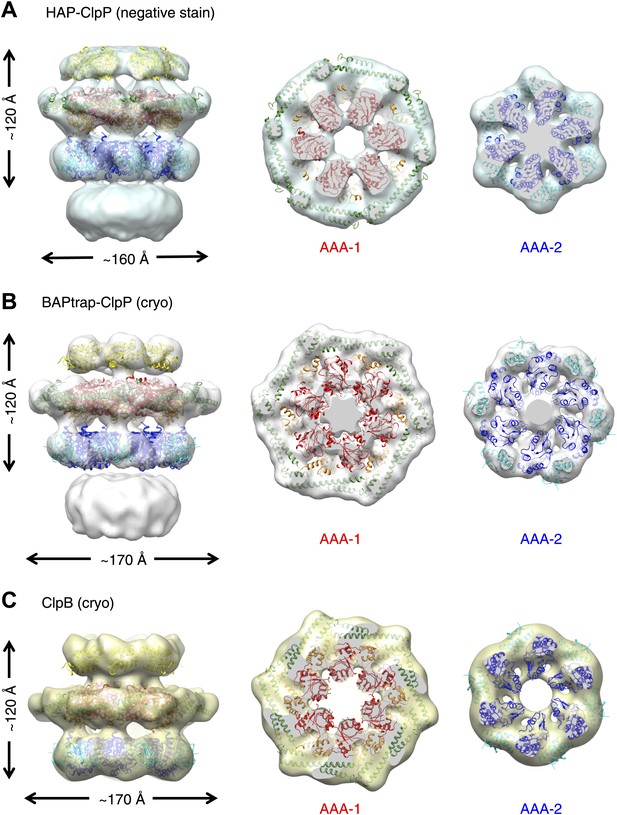
Independently determined maps and fitted hexameric models of HAP-ClpP, BAPtrap-ClpP and ClpB.
(A) Negative stain EM map of HAP-ClpP. From left to right: surface side-view, AAA-1 layer and AAA-2 layer. The central channel enclosed by the AAA-2 ring is filled with density. (B) Cryo EM map of BAP-ClpP formed with the BAP variant that traps the substrate inside. (C) Cryo EM map of wild type ClpB alone.
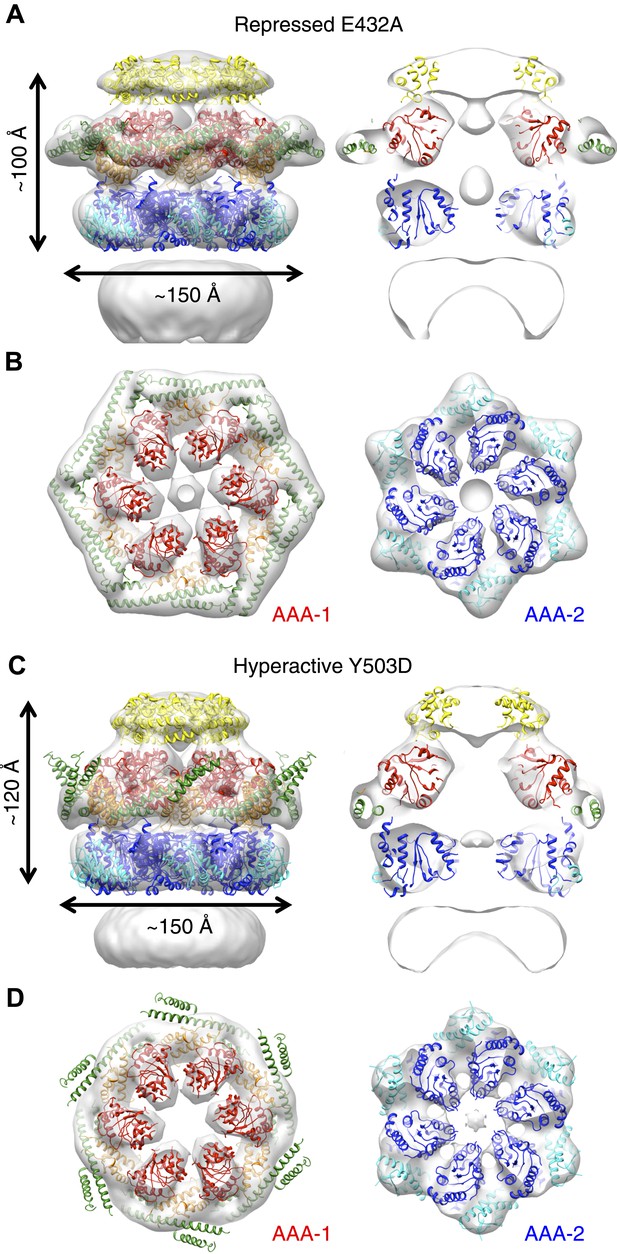
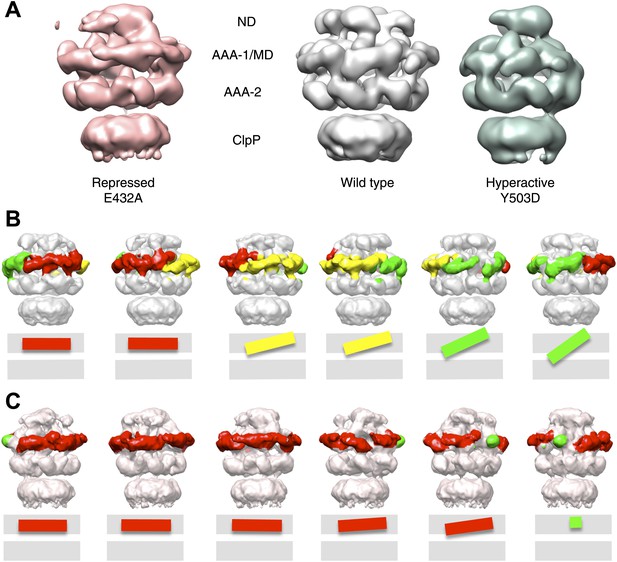
Structures of repressed and hyperactive BAP-ClpP mutants.
(A) Side view (left) and 35 Å section (right) of the BAP-E432A map with the ClpB hexamer fit. (B) Fitting of the BAP-E432A AAA+ rings. (C) Side view (left) and 35 Å section of the fitted BAP-Y503D structure. MD is only partially in density and is more tilted than in the wild type and the BAP-E432A repressed mutant. The ND is smeared out as in all BAP forms due to disorder, but has a more vertical orientation in the hyperactive state. (D) Fitting of the BAP-Y503D AAA+ rings. The motif 2 region protrudes from the density.
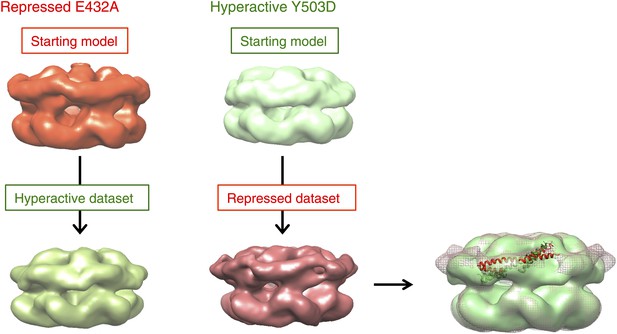
Test of EM map refinements after interchange of starting models.
https://doi.org/10.7554/eLife.02481.011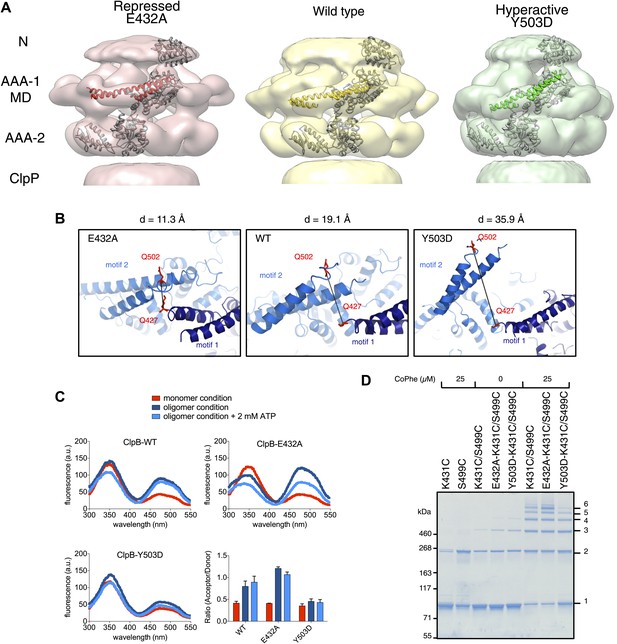
Analysis of MD movements.
(A) Repressed (red), wild-type (yellow) and hyperactive (green) forms of BAP-ClpP with a fitted subunit. (B) Distances (D) are shown between Cα atoms of Q427 (motif 1) and Q502 (motif 2) of neighbouring subunits based on the symmetrised EM reconstructions of wild-type ClpB and MD mutants. (C) Fluorescence energy transfer between motif 1 and motif 2 of adjacent ClpB subunits. Emission spectra of ClpB-Q427W-Q502C-IAEDANS in the monomeric (high salt), oligomeric (low salt) and ATP state (2 mM ATP) are shown. Effects of MD mutations on FRET efficiency were determined. The ratio of acceptor to donor fluorescence (derived from the areas under the curves) was calculated as readout for FRET efficiency. (D) Intermolecular disulfide crosslinking between MD motif 1 and motif 2 of adjacent subunits. Reduced and oxidized ClpB-K431C-S499C and repressed (E432A) and hyperactive (Y503D) variants were analyzed by non-reducing SDS-PAGE. Positions of monomers (1) and crosslinked dimers (2), trimers (3), tetramers (4), pentamers (5) and hexamers (6) are indicated. A protein standard is given. CoPhe at 25 µM was used as oxidizing agent.
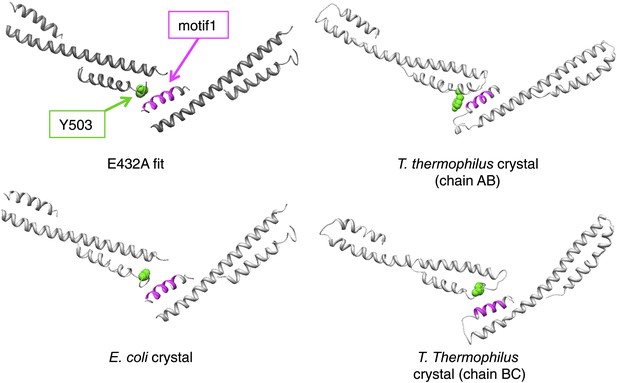
Contacts between adjacent MDs in the EM model compared to those found in the ClpB crystal structures.
https://doi.org/10.7554/eLife.02481.013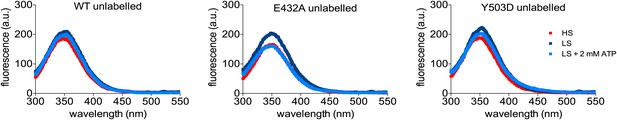
Tryptophan emission spectra of unlabeled ClpB-Q427W-Q502C and of corresponding MD mutants in the monomeric (HS, high salt), oligomeric (LS, low salt) and ATP-loaded state (LS + 2 mM ATP) are given.
High salt (400 mM KCl) prevents ClpB oligomerisation.
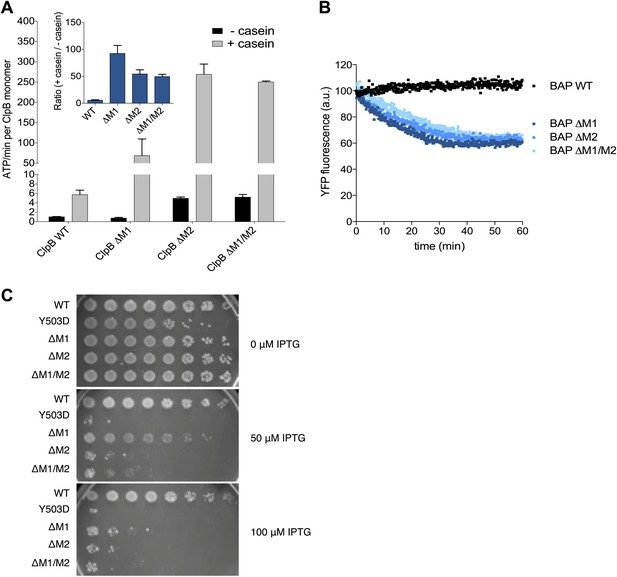
Deletion of MD motif 1 causes ClpB activation.
(A) Basal and substrate-stimulated ATPase activities of ClpB wild type and indicated MD deletions were determined in the absence and presence of casein. Relative ATPase activations by casein were calculated (inset). (B) Unfolding of Casein-YFP by BAP wild type and indicated MD deletions in the presence of ClpP was monitored by YFP fluorescence. Initial YFP fluorescence was set to 100%. ΔM1: ΔE410-Y455, ΔM2: ΔS456-E520, ΔM1/M2: ΔE410-E520. (C) E. coli ΔclpB cells expressing the indicated plasmid-encoded clpB alleles under control of an IPTG-regulatable promoter were grown overnight at 30°C. Various dilutions (10−1–10−7) were spotted on LB plates containing the indicated IPTG concentration and incubated at 37°C for 24 hr.
Variations in MD orientations around the ring.
(A) Asymmetric reconstructions of repressed BAP-E432A, wild type BAP-ClpP and hyperactive BAP-Y503. (B) Side views of the BAP-ClpP wild type structure rotated successively 60°. Red MDs are oriented horizontally and make motif 1 to motif 2 contact as in the repressed state. Yellow MDs have slightly tilted orientations similar to the wild type. Green MDs are tilted as in the hyperactive state. One of the MDs contacts AAA-2 of the same subunit (last right panel). (C) Equivalent views of the BAP E432A-ClpP repressed mutant structure. Color code for the MD orientations is the same as above.
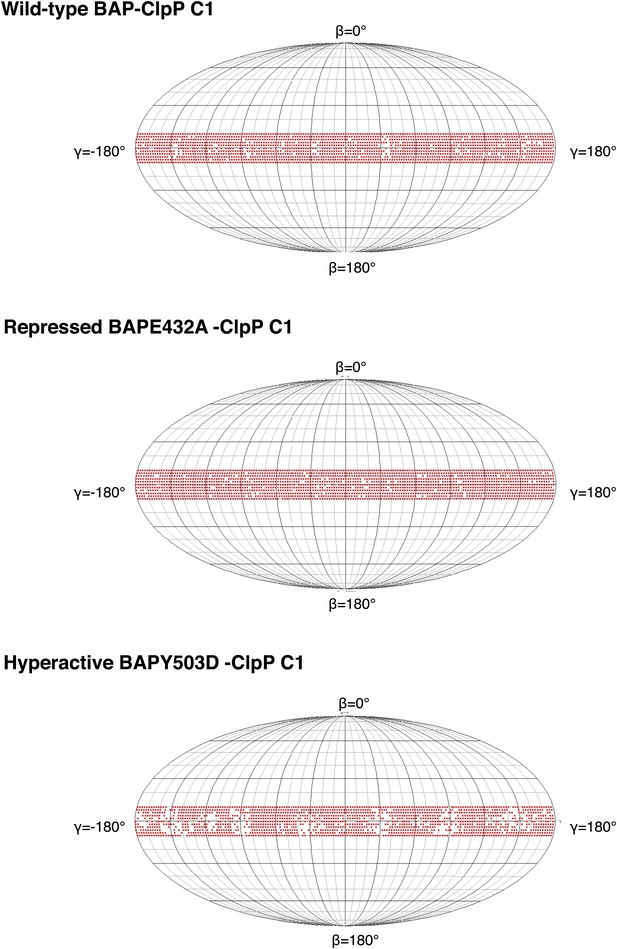
Plots showing the angular distribution of single particles (red dots) around the Euler sphere for each asymmetric reconstruction.
Angle search was done with 2° sampling. The equatorial band reflects the Euler angle distribution around the BAP-ClpP central axis.
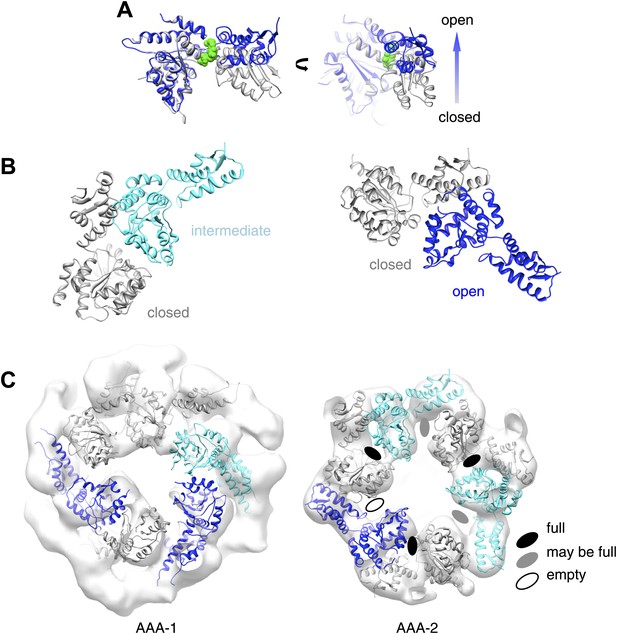
Open and closed conformations of AAA+ domains can be fitted into the asymmetric ClpB map.
(A) Superimposition of an open (blue) and a closed (grey) nucleotide pocket (ClpX 3HWS). ADP is depicted in green spheres. (B) EcoClpB AAA-2 dimers built with open, intermediate and closed conformations based on ClpX crystallographic dimers. (C) AAA-1 and AAA-2 layers of asymmetric wild-type ClpB reconstruction plus indication of nucleotide occupancy.
Asymmetric nucleotide binding to ClpB.
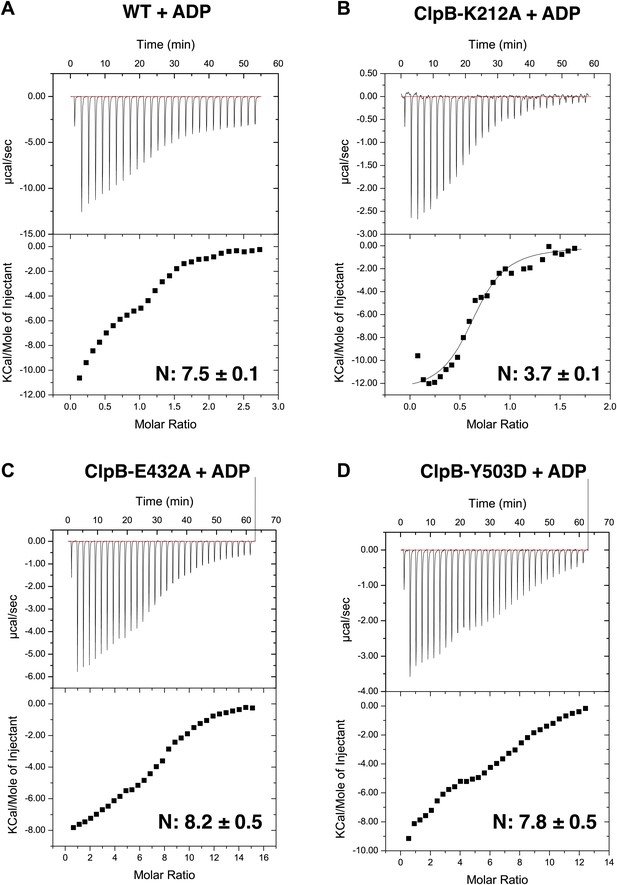
(A–D) Instrumental responses of successive injections of ADP (8.39 mM, 1.92 mM, 5.82 mM and 4.05 mM ADP for ClpB-WT, ClpB-K212A, ClpB-E432A, ClpB-Y503D, respectively) into a solution of ClpB-WT (678 μM), ClpB-K212A (239 μM), ClpB-E432A (467 μM) and ClpB-Y503D (396 μM). The molar ratio of ADP to ClpB hexamer is indicated. Raw isothermal titration calorimetry data are shown. Integrated data after base-line-correction and fitting to the respective binding isotherms to a single-site binding model are given (lower panel). The number of ADP binding sites was calculated.
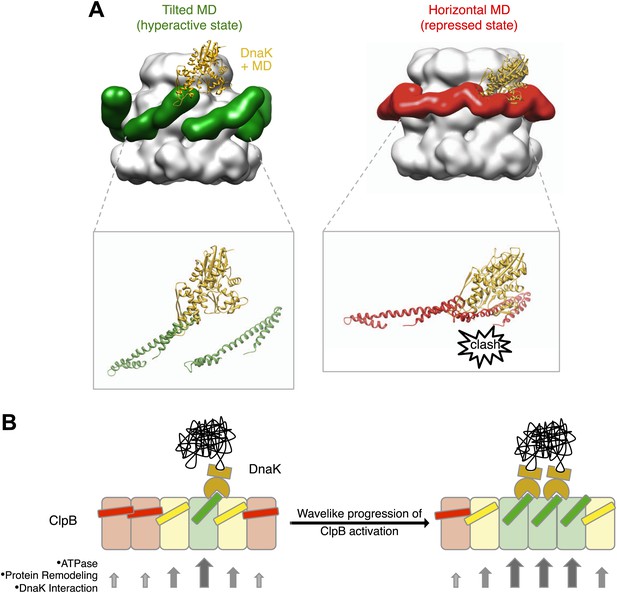
Interaction between the ClpB hexamer and DnaK.
(A) Binding of DnaK is only possible when the MD is tilted (green). DnaK cannot bind when two adjacent MDs occupy a more horizontal position (red) as it would clash with motif 1 of the neighbouring ClpB subunit. (B) Cartoon of an opened-out ClpB ring with two DnaK molecules bound. The MDs involved in DnaK binding are shown in green, and those that cannot bind to DnaK are shown in red. Release of the ends of the MD favours activation of adjacent subunits, and the active cluster can move around the ring in a wave-like manner. The model of DnaK binding to the MD is based on the NMR study in Seyffer et al. (2012).
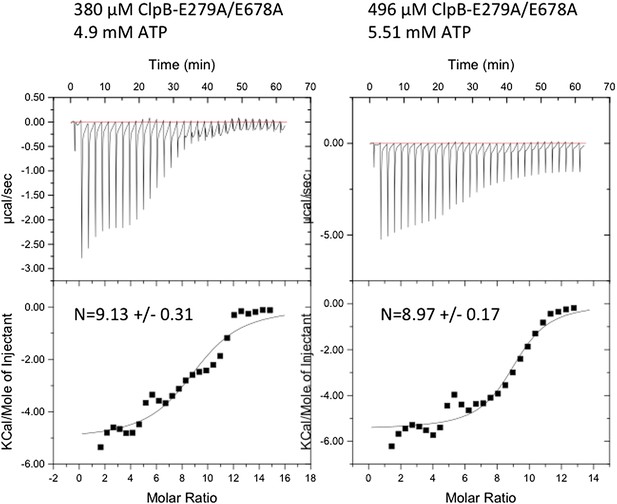
Instrumental responses of successive injections of ATP (4.9 mM or 5.51 mM) into a solution of 380 μM or 496 μM ClpB-E279A/E678A. The molar ratio of ATP vs. ClpB-E279A/E678A hexamer is indicated. Raw isothermal titration calorimetry data are shown. Integrated data after base-line correction and fitting to the respective binding isotherms to a single binding model are given (lower panel). The number of ATP binding sites was calculated.
Tables
X-ray data collection and refinement statistics
| Protein | ClpB E279A/E432A/E678A (SeMet) + ATP |
| Wavelength (Å) | 0.9794 |
| Space group | P65 |
| Unit cell (Å,°) | 127.34, 127.34, 119.86, 90, 90, 120 |
| Molecules | 1 |
| Resolution (Å) | 81.15–3.50 (3.69–3.50) |
| Reflections measured | 202965 |
| Unique reflections | 14024 |
| Rmerge | 8.8 (53.2) |
| Rpim | 3.0 (14.9) |
| I/σI | 16.8 (5.4) |
| Completeness (%) | 99.9 (100) |
| Redundancy | 14.5 (14.8) |
| Rwork/Rfree (%) | 22.5/24.5 |
| Protein residues | 657 (5257 atoms) |
| ADP | 2 |
| Rmsd bond lengths (Å) | 0.004 |
| Rmsd bond angles (°) | 0.94 |
| Average B-Factor Protein | 99.6 |
| Average B-Factor ADP | 87.7 |
| Ramachandran plot statistics | |
| Favored (%) | 95.5 |
| Allowed (%) | 4.1 |
| Generous (%) | 0.4 |
| Disallowed (%) | 0 |
| PDB entry code | 4CIU |





