Identification of the transcription factor ZEB1 as a central component of the adipogenic gene regulatory network
Figures
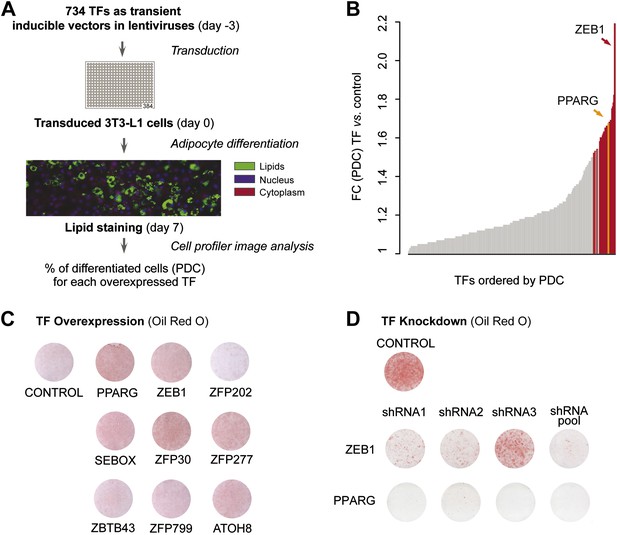
A large-scale TF overexpression screen identifies novel positive regulators of adipogenesis.
(A) Schematic overview of high-throughput screening illustrating how 3T3-L1 cells were transduced with 734 individual TFs in three replicates each, 3 days before induction of adipocyte differentiation (‘Materials and methods’). The effect of TF overexpression was quantified at differentiation day 7 by lipid, nucleus and cellular staining and summarized as a percentage of differentiated cells (PDC) per TF. (B) Overview of fold-changes (FC) compared to control for all TFs showing a differentiation FC > 1. TFs that significantly induced differentiation (FC ≥ 1.5, α = 0.05) are highlighted in red and PPARγ specifically in orange. (C) Effect of stably overexpressing eight putatively novel regulators of adipogenesis, PPARγ, or a control vector on 3T3-L1 differentiation as assessed by Oil Red O staining of lipid droplets at day 5 after induction. (D) Effect of knocking down ZEB1 or PPARγ (as a positive control), or the negative control (empty shRNA) on 3T3-L1 differentiation as assessed by Oil Red O staining at day 6 after induction. In the shRNA pool of ZEB1, shRNA2 was not used because the robustness of the cells after treatment was low. Examples of microscopic images illustrating the overexpression or knockdown (KD) effects on 3T3-L1 differentiation are shown in Figure 1—figure supplement 2 or Figure 1—figure supplement 3, respectively.
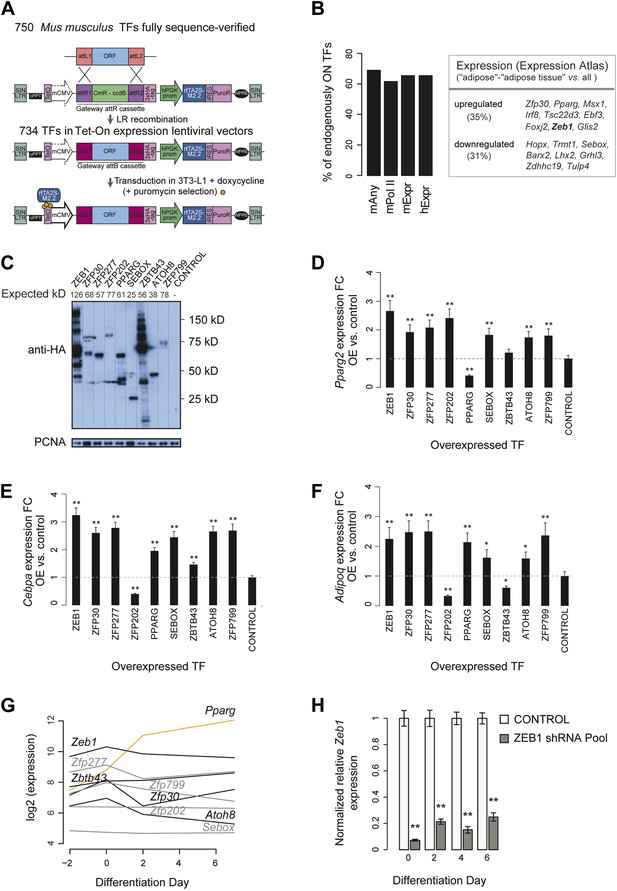
Large-scale TF overexpression screen identifies novel positive regulators of adipogenesis.
(A) Workflow for transferring mouse TF open reading frames (ORFs) into a lentiviral vector to overexpress HA-tagged TFs in 3T3-L1 cells. 750 fully sequence-verified entry TF clones were transferred using LR Gateway cloning into the Tet-On expression vector (derived from the original TRE_GOI_rtTA_hPGK vector [Barde et al., 2006], ‘Materials and methods’), during which the attL sites recombine with the attR sites. 734 ORF TFs were successfully transferred. (B) Barplots: percentage of expressed/transcribed TFs of all TFs that significantly enhance adipogenesis (positive candidates) in mouse 3T3-L1 cells based on microarray expression data in mouse 3T3-L1 (mExpr) and human hASC (hExpr) as well as POLII signal over genes (mPolII) and combined POLII signal and expression (mAny) in mouse 3T3-L1 cells (Nielsen et al., 2008; Mikkelsen et al., 2010); table: positive candidates that are significantly up- or down-regulated in mouse adipose tissue compared to other probed tissues based on ArrayExpress Expression Atlas (Kapushesky et al., 2012). (C) Protein levels of stably overexpressed HA-tagged TFs selected for follow-up in 3T3-L1 cells (follow-up TFs). The expected molecular mass for each protein is indicated above the image. Note that especially for ZEB1, several bands were detected which likely correspond to cryptic translation or specific protein degradation products given that they stem from the same open-reading frame construct and that they are all tagged by HA. (D–F) Relative (to control): Pparg2 (D), Cebpa (E), and Adipoq (F) mRNA fold-changes (FCs) in 3T3-L1 cells stably overexpressing each follow-up TF, as measured by qPCR. To measure Pparg2 mRNA levels, primers were used that target the 5′ UTR of the endogenous transcript, allowing us to differentiate between the overexpression and endogenous Pparg transcripts. (G) Microarray-based (Mikkelsen et al., 2010) expression analysis of follow-up TFs during 3T3-L1 adipogenesis. In contrast to Pparg (orange), most follow-up TFs are at their maximal expression level already prior to induction of differentiation (days −2 and day 0). (H) Relative (to control; i.e. transduced with the shEmpty vector) Zeb1 mRNA FCs in knockdown 3T3-L1 cells at four different time points during adipogenesis, as measured by qPCR. Error bars depict the standard error of the mean from three biological replicate experiments. **p ≤ 0.01 and 0.01 < *p ≤ 0.05.
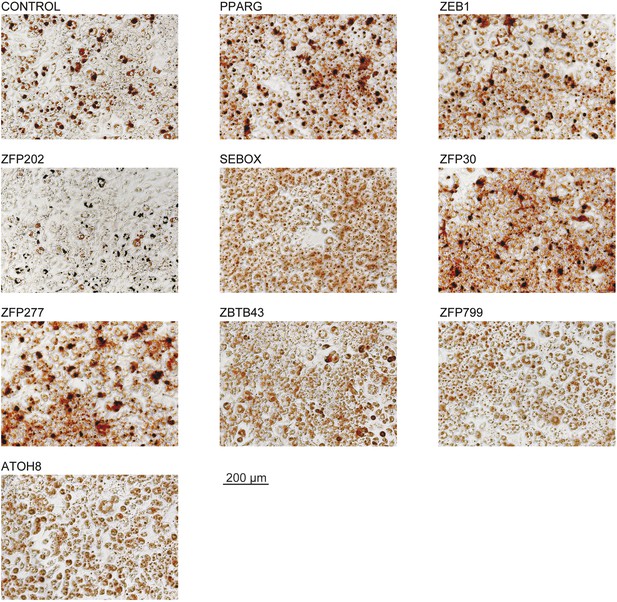
Microscopic images of Oil Red O stained 3T3-L1 adipocytes after overexpression of candidate TFs.
These images were acquired from the wells presented in Figure 1C.
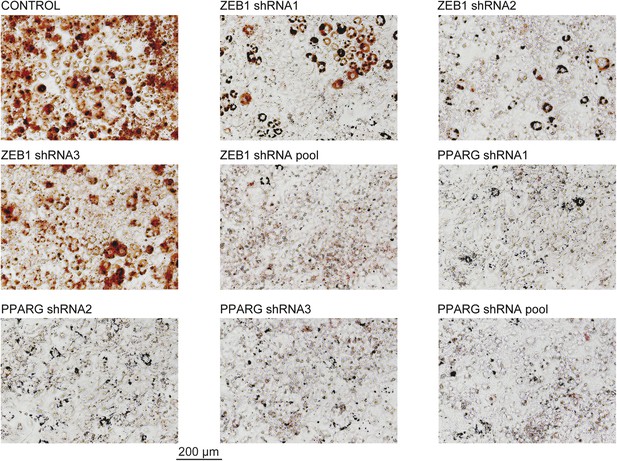
Microscopic images of Oil Red O stained 3T3-L1 adipocytes after ZEB1 and PPARG KD using distinct KD constructs.
These images were acquired from the wells presented in Figure 1D.
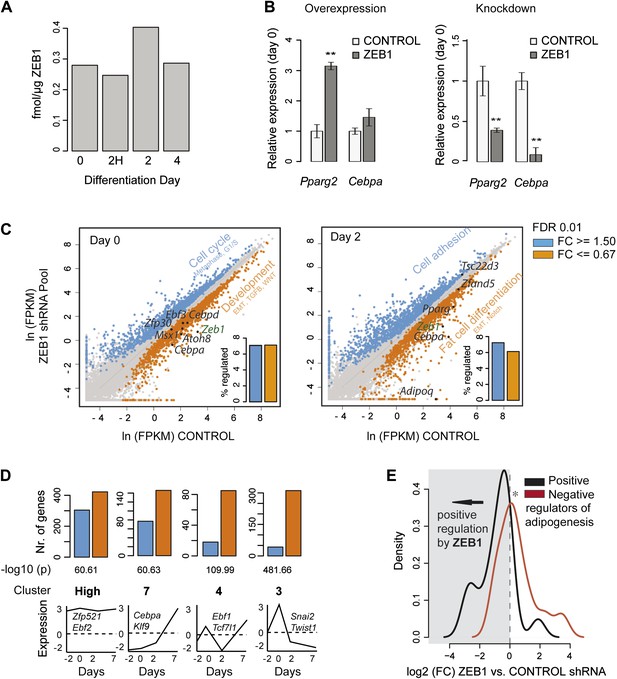
ZEB1 knockdown perturbs the expression of adipogenic regulators.
(A) Protein levels (fmol/μg nuclear extract) of ZEB1 during 3T3-L1 differentiation (one representative biological replicate). (B) Pparg2 and Cebpa mRNA levels after ZEB1 knockdown and overexpression in un-induced 3T3-L1 pre-adipocytes as measured by qPCR. (C) Expression levels [ln(FPKM), ‘Materials and methods’] of mouse genes in ZEB1 KD vs. control cells at day 0 and day 2 after differentiation induction as measured by RNA-seq. Significantly up-regulated genes (FC ≥ 1.5, padj ≤ 0.01) are highlighted in blue, down-regulated genes in orange (FC ≤ 0.67, padj ≤ 0.01), significantly de-regulated follow-up TFs as well as adipogenic TFs such as PPARγ and C/EBPs are indicated in black. Bar plots represent the percentage of genes that are significantly up- or down-regulated. Representative enriched GeneGO pathway categories for up- or down-regulated genes are highlighted (complete results in Supplementary file 1C [Gubelmann et al., 2014]). (D) Number of significantly up- or down-regulated genes belonging to previously defined expression clusters (High/7/4/3) (Mikkelsen et al., 2010). The typical expression pattern of genes in each cluster as well as of representative members that are significantly down-regulated upon ZEB1 KD is sketched. Clusters are sorted by increasing enrichment of down-regulated genes and corresponding p-values (chi-square test) are listed. (E) Distribution of gene expression FCs at day 0 after ZEB1 KD for genes annotated as positive or negative regulators of adipogenesis (Supplementary file 1B [Gubelmann et al., 2014]). Error bars depict the standard error of the mean. **p ≤ 0.01 and 0.01 < *p ≤ 0.05.
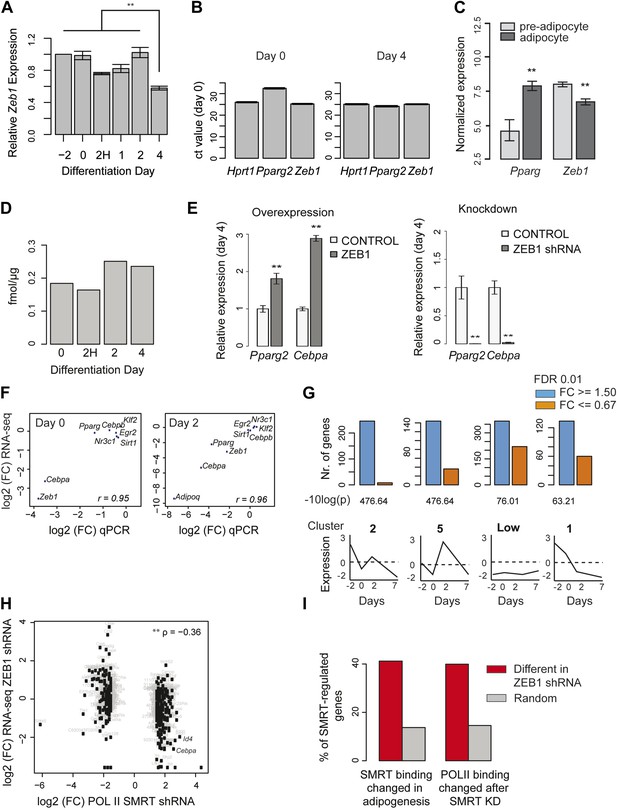
ZEB1 knockdown perturbs the expression of adipogenic regulators.
(A) Relative (to day −2) Zeb1 mRNA levels in wild-type 3T3-L1 cells during differentiation, as measured by qPCR. (B) Raw Ct values for Pparg, Zeb1 as well as the housekeeping gene HPRT1 at days 0 and 4 of 3T3-L1 differentiation as measured by qPCR. (C) Pparg and Zeb1 mRNA levels in pre-adipocytes and adipocytes derived from publicly available data through ArrayExpress (Rustici et al., 2013). (D) Protein levels (fmol/μg nuclear extract) of ZEB1 during 3T3-L1 differentiation (biological replicate of data shown in Figure 2A). (E) Pparg2 and Cebpa mRNA levels after ZEB1 knockdown and overexpression at day 4 after adipogenic induction as measured by qPCR. (F) Fold-changes of expression levels of selected adipogenic factors in response to ZEB1 KD as measured by qPCR and RNA-seq at day 0 and day 2 of 3T3-L1 differentiation. The Pearson's correlation coefficient (r) is indicated. (G) Number of significantly up- and down-regulated genes after ZEB1 knockdown belonging to previously defined expression clusters (2/5/Low/1) (Mikkelsen et al., 2010). The typical expression pattern of genes in each cluster is sketched. Clusters are sorted by decreasing enrichment of up-regulated genes and corresponding p-values (chi-square test) are listed. (H) Changes in mRNA levels and POLII binding over gene bodies after ZEB1 and SMRT knockdown, respectively (Raghav et al., 2012). The Spearman's ρ is indicated, showing a significantly negative correlation. (I) Percent of genes with dynamic SMRT binding during adipogenesis (red), of genes that lose/gain POLII upon SMRT KD (red), and of random genes (grey) that are significantly differentially expressed upon ZEB1 KD. Error bars depict the standard error of the mean. **p ≤ 0.01 and 0.01< *p ≤ 0.05.
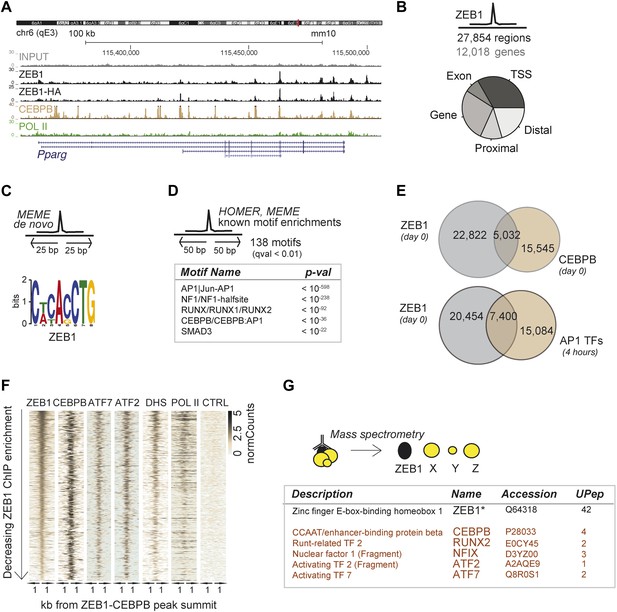
ZEB1 co-binds the genome with established adipogenic regulators such as C/EBPβ.
(A) ZEB1, C/EBPβ, POLII, and Control (CTRL) read density tracks at the Pparg locus. (B) Number of ZEB1-bound regions and of their proximal (≤ 10 kb) genes in 3T3-L1 cells. Distribution of ZEB1 binding with respect to genomic annotation (‘Materials and methods’). (C) De novo motif discovery using MEME and a 50 bp sequence centered on ZEB1 peak summits reveals the canonical ZEB1 motif (p = 10−8, ‘Materials and methods’) (D) Motif enrichment analysis in a 100 bp window around ZEB1 peak summits reveals 138 significantly enriched motifs (complete results in Supplementary file 1E). Highlighted here are motif names of the known early adipogenic regulators C/EBPβ, NFI, and AP1 factors as well as RUNX and SMAD3. (E) Peak overlap between ZEB1 and C/EBPβ (day 0) as well as AP1 factors (day 0, 4 hr) in 3T3-L1 cells. (F) Overview of ZEB1, C/EBPβ, AP1 proteins ATF2 and ATF7, POLII normalized ChIP-seq as well as DNase-seq (DHS) enrichments (‘Materials and methods’) in a 2 kb window around the summits of ZEB1 peaks that overlap C/EBPβ binding. Intervals are sorted based on decreasing ZEB1 enrichment. (G) Summarized results from mass spectrometry experiments of proteins that were identified when pulling down ZEB1 (complete results in Supplementary file 1F [Gubelmann et al., 2014]).
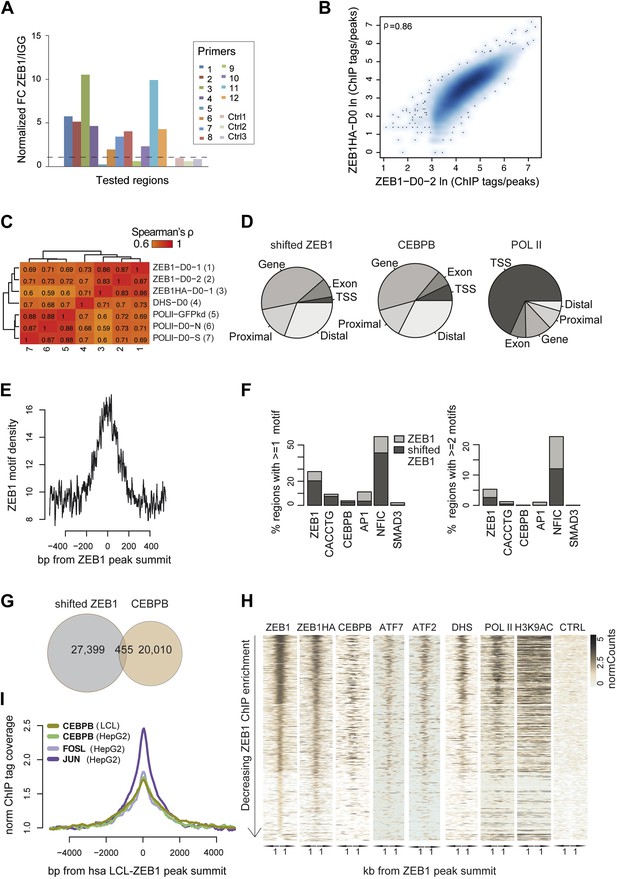
ZEB1 co-binds the genome with established adipogenic regulators such as C/EBPβ.
(A) ZEB1 ChIP-qPCR validation of ChIP-seq data at 12 selected ZEB1 target sites and three negative control (CTRL) regions during 3T3-L1 adipogenesis. (B) Scatterplot and Spearman's ρ of ZEB1 ChIP-seq and ZEB1-HA ChIP-seq read counts inside genomic intervals defined by ZEB1 binding in pre-adipocytes. (C) Spearman correlations between read counts for replicate ZEB1 ChIP-seq (including ZEB1-HA) as well as publicly available POLII and DNase-seq data (Siersbæk et al., 2011; Raghav et al., 2012) inside genomic intervals defined by ZEB1 binding in pre-adipocytes. (D) Distribution of randomly shifted ZEB1, C/EBPβ, and POLII peaks with respect to genomic annotation (‘Materials and methods’). (E) ZEB1 motif density at 800 bp centered on ZEB1 peak summits. (F) Fraction of ZEB1 and randomly shifted ZEB1 peaks (to show background values) that contain at least one or two, respectively, ZEB1, CACCTG (E-box), C/EBPβ, AP1, NFIC and SMAD3 motif hits (‘Materials and methods’). (G) Peak overlap between randomly shifted ZEB1 and C/EBPβ bound regions in 3T3-L1 pre-adipocytes. (H) Overview of ZEB1, ZEB1-HA, C/EBPβ, AP1 factors ATF2 and ATF7, POLII and H3K9AC normalized ChIP-seq as well as DNase-seq and control (CTRL) enrichments (‘Materials and methods’) in a 2 kb window around the summits of ZEB1 peaks. Intervals are sorted based on decreasing ZEB1 enrichment. (I) Mean C/EBPβ and AP1 complex proteins JUN and FOSL normalized (to total read number) ChIP-seq enrichments in human HepG2 and lymphoblastoid cell lines (LCLs) in a 8 kb window around the summits of ZEB1 peaks detected in LCLs.
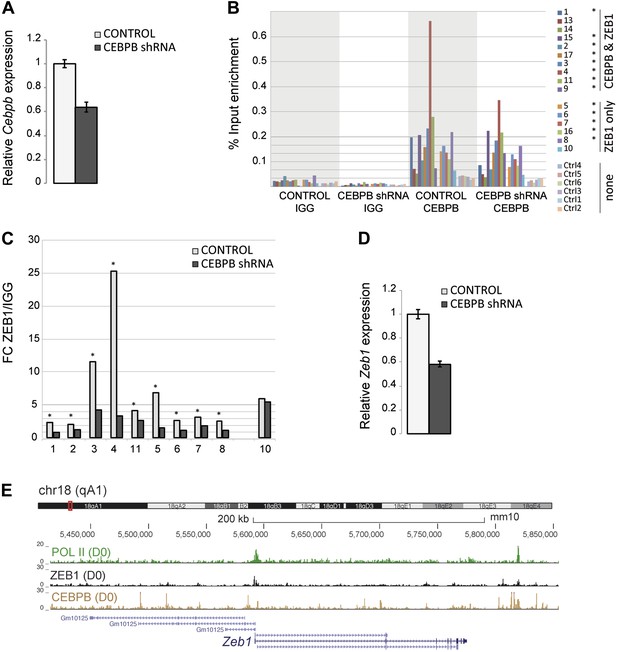
ZEB1 binding and expression is affected by C/EBPβ KD.
(A) C/EBPβ expression (mRNA level) in stable C/EBPβ KD and control 3T3-L1 pre-adipocytes as measured by qPCR. (B) C/EBPβ ChIP-qPCR at 10 C/EBPβ-ZEB1, 6 ZEB1-only and six negative control regions according to our ZEB1 D0 ChIP-seq data and publicly available C/EBPβ ChIP-seq data (Siersbæk et al., 2011). 5 out of 6 ZEB1-only regions also show C/EBPβ ChIP enrichment. *C/EBPβ-enriched regions (C) ZEB1 ChIP-qPCR at 9 C/EBPβ-ZEB1 regions as well as one ZEB1-only region (10) in C/EBPβ KD and control 3T3-L1 cells. * regions showing changes in ZEB1 enrichment after C/EBPβ KD (D) Zeb1 expression (mRNA level) in stable C/EBPβ KD and control 3T3-L1 cells as measured by qPCR. (E) POLII, ZEB1, and C/EBPβ read density tracks at the Zeb1 locus in 3T3-L1 pre-adipocytes.
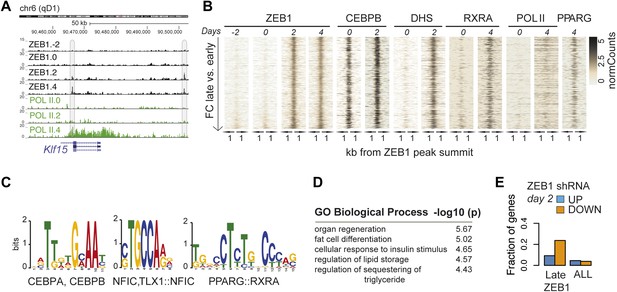
ZEB1 binding increases at adipogenic genes during 3T3-L1 differentiation.
(A) ZEB1 and POLII read density tracks at the Klf15 locus during 3T3-L1 differentiation (days −2, 0, 2, and 4). Late-only bound regions are highlighted. (B) ZEB1, C/EBPβ, RXRα, PPARγ, POLII normalized ChIP (‘Materials and methods’) as well as DHS enrichments in a 2 kb window around the summits of late-only (days 2 and 4 but not days −2 and 0; padj ≤ 0.1, FC ≥ 2) ZEB1-bound regions during 3T3-L1 differentiation. (C) Differential motif discovery using MEME and a 50 bp sequence centered on summits of late-only vs. static ZEB1 peaks reveals adipogenic motifs: C/EBPα|C/EBPβ, NFIC and PPARG::RXR (p < 10−3, ‘Materials and methods’). (D) GREAT-based (McLean et al., 2010) Gene Ontology enrichment analysis of genes associated with late-only vs. static ZEB1 binding reveals terms associated with fat cell differentiation and function (complete results in Supplementary file 1G [Gubelmann et al., 2014]). (E) Fraction of genes associated with late-only ZEB1 binding and fraction of all genes significantly up (blue) and down (orange)-regulated after ZEB1 KD as measured at differentiation day 2 (complete results in Figure 4—figure supplement 1G).
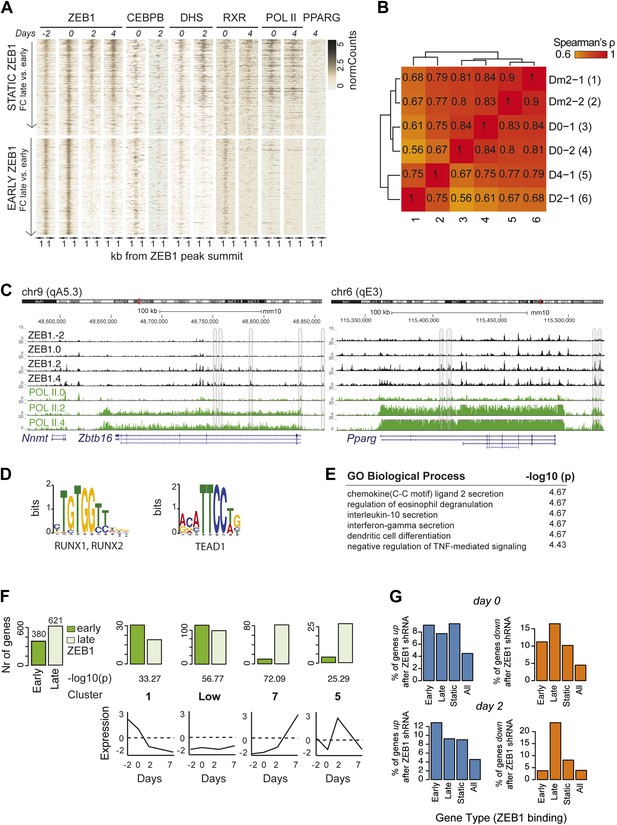
ZEB1 binding increases at adipogenic genes during differentiation.
(A) Overview of ZEB1, C/EBPβ, RXRα, PPARγ, POLII normalized ChIP-seq as well as DNase-seq enrichments (‘Materials and methods’) in a 2 kb window around the summits of static (padj ≥ 0.1 or FC < 2) and early-only (days-2 and 0 but not days 2 and 4; padj ≤ 0.1, FC ≥ 2) ZEB1-bound regions during 3T3-L1 differentiation. (B) Spearman correlations between read counts for ZEB1 ChIP-seq data at distinct adipogenic time points (days −2, 0, 2, and 4) inside genomic intervals defined by ZEB1 binding at any of these time points. (C) ZEB1 and POLII read density tracks at the Zbtb16 and Pparg loci during 3T3-L1 differentiation (days-2, 0, 2, and 4). Summarized genome-wide results are included in Supplementary file 1G. (D) Differential motif discovery using MEME and a 50 bp sequence centered on summits of early-only vs static ZEB1 peaks reveals non-adipogenic motifs: RUNX1/2 and TEAD1 (p < 10−5, ‘Materials and methods’). (E) GREAT-based (McLean et al., 2010) Gene Ontology enrichment analysis of genes associated with early-only vs static ZEB1 binding reveals terms associated with chemokine secretion and non-adipogenic functions. Full results are displayed in Supplementary file 1G. (F) Number of significantly up- or down-regulated genes associated (≤10 kb) with at least one early-only or late-only ZEB1 bound region, respectively, belonging to previously defined expression clusters (1/Low/7/5) (Mikkelsen et al., 2010). The typical expression pattern of genes in each cluster is sketched. Clusters are sorted by increasing enrichment of late-only ZEB1-bound genes and corresponding p-values (chi-square test) are listed. Only clusters showing a highly significant p-value (p < 10−10) are shown. (G) Fraction of genes associated with early-only, late-only, and static ZEB1 binding as well as the fraction of all genes significantly up (blue) and down (orange)-regulated after ZEB1 KD as measured at differentiation days 0 and 2.
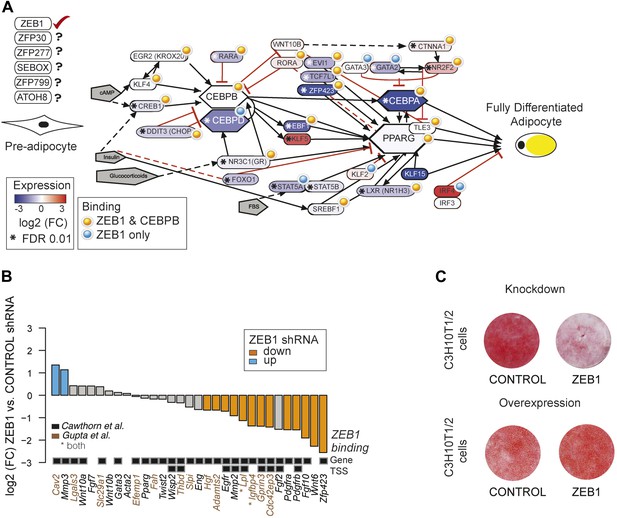
ZEB1: a central component of the adipogenic regulatory network.
(A) Effect of ZEB1 knockdown on the adipogenic gene regulatory network. The network was assembled on the ‘Adipogenesis’ Pathway scaffold in WikiPathways as well as reviews and most recent publications of novel adipogenic regulators (Rosen and MacDougald, 2006; Kelder et al., 2012; Siersbæk et al., 2012). ZEB1 and C/EBPβ-bound regions that are proximal (within 500 bp) to TSSs or genes in pre-adipocytes are highlighted. *Significant (padj ≤ 0.01) expression changes after ZEB1 KD at day 0 of 3T3-L1 differentiation. Other candidate adipogenic regulators identified by our high throughput screen are listed. (B) Expression changes of adipogenic commitment genes after ZEB1 KD in 3T3-L1 pre-adipocytes as measured by RNA-seq. Displayed genes are either part of the pre-adipocyte expression signature derived by Gupta et al. (2010) or of the list of pre-adipocyte commitment factors compiled by Cawthorn et al. (2012). Lpl and Igfbp4 occur in both lists. Significant differences in expression (padj ≤ 0.01) are marked in orange (FC ≤ 0.67) and blue (FC ≥ 1.5). Black-grey squares depict ZEB1 binding to TSSs or gene bodies. (C) Effect of ZEB1 knockdown and overexpression on C3H10T1/2 adipogenesis as assessed by Oil Red O staining at day 7 and day 8, respectively after induction.
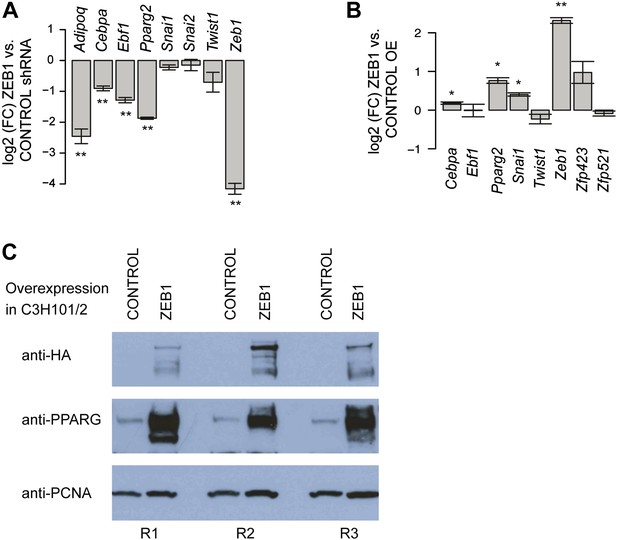
ZEB1 regulates adipogenic commitment factors.
(A) Effect of ZEB1 knockdown on the expression [log2 (FC) mRNA] of adipogenic (Adipoq, Cebpa, Ebf1, Pparg2) and EMT (Snai1, Snai2, Twist1) factors as measured by qPCR at day 8 after induction. (B) Effect of ZEB1 overexpression on the expression [log2 (FC) mRNA] of adipogenic (Cebpa, Ebf1, Pparg2), pre-adipogenic (Zfp423, Zfp521), and EMT (Snai1, Twist1) factors as measured by qPCR at day 0 after induction of differentiation. (C) Western Blot showing PPARγ induction upon ZEB1 overexpression (visualized using anti-HA antibody) in C3H10T1/2 cells using PCNA as a normalization control. R1-3 indicates biological replicates. Error bars depict the standard error of the mean. **p ≤ 0.01 and 0.01 < *p ≤ 0.05.
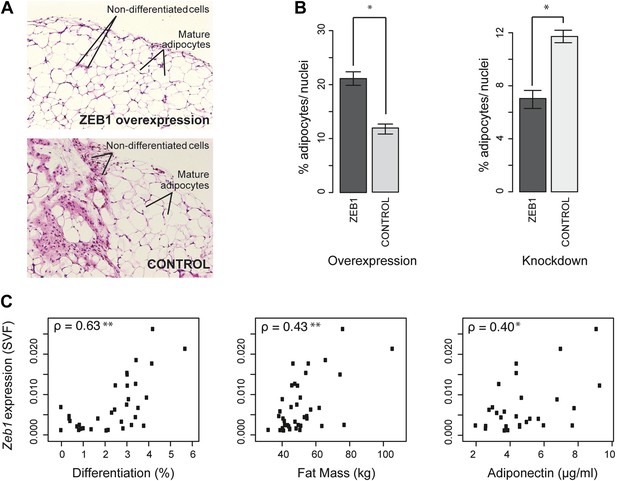
ZEB1 is required for adipogenesis in vivo in mouse and its expression levels correlate with adipogenic indicators in humans.
(A and B) Adipocyte differentiation in stromal vascular fraction (SVF) transplants from different donor mice (as indicated) fed a high-fat diet for 6 weeks (Meissburger et al., 2011). (A) Fat sections from representative samples of ZEB1-overexpressing and control SVF transplants stained with Hematoxylin (blue) and Eosin (pink). (B) Fat cell content of the transplanted SVF cells containing ZEB1 and control overexpression or knockdown constructs. Error bars depict the standard error of the mean. *p = 0.05, one-sided Wilcoxon-rank sum test. (C) Zeb1 mRNA expression normalized to 36B4 in human subcutaneous SVF of obese subjects plotted against percent ex vivo differentiated adipocytes of human subcutaneous SVF, subject fat mass, and adiponectin levels. Spearman's ρ is indicated, **p ≤ 0.01 and 0.01 < *p ≤ 0.05.
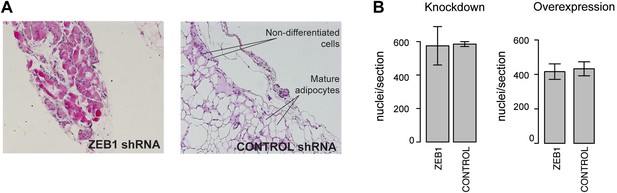
Analysis of the functional involvement of ZEB1 in mouse in vivo adipogenesis.
(A) Hematoxylin (blue) and Eosin (pink)-stained fat sections from representative samples of mouse ZEB1 knockdown transplants as well as the corresponding control (scrambled siRNA). (B) Number of nuclei per section (average over three sections) from ZEB1 overexpression, KD, or control SVF transplants.
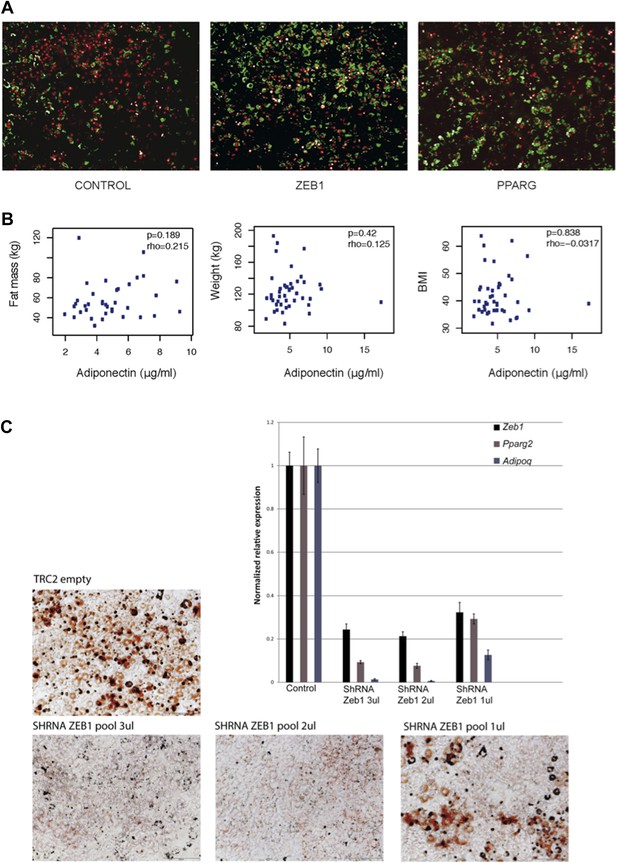
(A) Microscopic images of BODIPY (lipids, in green) and Hoechst (nuclei, in red)-stained 3T3-L1 cells transduced with control, ZEB1 and PPARG overexpression constructs. (B) Adiponectin levels plotted against fat mass, weight and BMI of obese subjects. Spearman’s ρ and p-values are indicated. (C) Microscopic images of Oil Red O-stained 3T3-L1 adipocytes after control and ZEB1 KD using three different quantities (measured in terms of µl) of pooled shRNAs at day 6. The relative expression of Zeb1, Pparg and Adipoq at day 2 are shown in the associated bargraphs.
Additional files
-
Supplementary file 1
Gubelmann et al. (2014). (A) Transcription factor screen. Percentage differentiated cells in response to TF overexpression. Raw values, fold-changes with respect to control, p-values and associated annotations are included for all TFs showing positive effects on adipogenesis. TFs significantly enhancing differentiation are highlighted in green (FC ≥ 1.5, Bonferroni α = 0.05). (B) mRNA levels upon ZEB1 knockdown. Estimated expression levels in all RNA-seq replicates (shZEB1 and shEmpty at days 0 and 2), fold-changes and p-values as well as information ZEB1 and C/EBPβ binding at gene TSS, gene bodies or in gene proximity. (C) Pathway Enrichments. Pathways enriched in genes significantly de-regulated upon ZEB1 knockdown at days 0 and 2. (D) Gene Ontology Enrichments Genomic features enriched at ZEB1-bound locations. (E) Motifs in ZEB1 bound regions. Motifs enriched in 100 bp centered on ZEB1 peak summits. (F) Mass Spectrometry results. List of proteins detected by at least one peptide in any of the two performed ZEB1 immunoprecipitation experiments. (G) GREAT Gene Ontology enrichments. GO Terms enriched in genes proximal to late-only and early-only ZEB1-bound regions. (H) Clinical data: Correlation of Zeb1 and Rorg mRNA levels in human obese subjects with a range of adipogenesis-relevant measures. Raw values are included for the significant correlations discussed in the manuscript. (I) qPCR primers. List of primers used for gene expression analyses and ChIP-qPCR.
- https://doi.org/10.7554/eLife.03346.018





