Evolutionary consequences of intra-patient phage predation on microbial populations
Figures
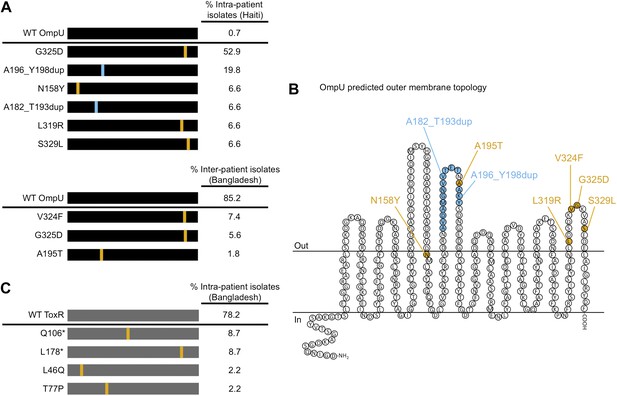
The presence of V. cholerae OmpU and ToxR mutants present within and between cholera patients.
(A) Graphical depiction and frequency of OmpU mutants found within a stool sample containing ICP2_2013_A_Haiti phage (108 PFU/ml) from a single Haitian patient (top) and from different patients in Bangladesh (n = 54) (bottom). (B) Predicted membrane topology of mature OmpU generated using Pred-TMBB (Bagos et al., 2004). Locations of amino acid substitutions or insertions carried by V. cholerae clinical isolates are indicated. (C) Graphical depiction and frequency of ToxR mutants found within a stool sample containing ICP2_2011_A (109 PFU/ml) from a single Bangladeshi patient. Amino acid substitutions or nonsense mutations (asterisks) are in orange and duplications are in blue.
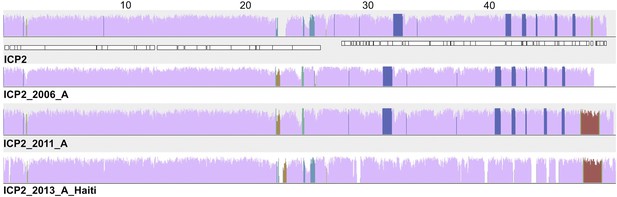
ICP2_2013_A_Haiti is closely related to ICP2 bacteriophages from Bangladesh.
Comparison of ICP2 genomes collected from cholera patients in Dhaka, Bangladesh in 2004 (ICP2), 2006 and 2011, and in Haiti in 2013 using progressiveMauve software. The degree of nucleotide similarity between aligned regions is indicated by the height of the similarity profile (colored blocks), where mauve represents the highly conserved backbone genome and other colors represent segments whose presence varies between isolates. Annotated genes in the ICP2 genome are shown as white boxes, with genes transcribed from the negative strand displaced downward. The numbers above the ICP2 genome show distance in kilobases.

Identification of OmpU mutants in samples collected at the International Centre for Diarrheal Disease Research, Bangladesh between 2001–2011.
(a) Single V. cholerae O1 El Tor isolates from different stool samples collected within a given year are indicated as closed circles. (b) The number of isolates with the noted mutation is indicated in a given year. If left blank, OmpU was wild-type.

OmpU expression of OmpU and ToxR mutants and their sensitivity to ICP2.
Outer membrane fractions were prepared from samples matched by equivalent OD600 units. Samples were separated by SDS-PAGE and subjected to Western blot analysis using rabbit polyclonal antisera against OmpU. The sensitivity of each strain to ICP2_2013_A_Haiti is represented as a histogram of the efficiency of plaquing, which is the plaque count ratio of a mutant V. cholerae strain to that of the wild-type strain. The limit of detection for plaque assays was 10−7.
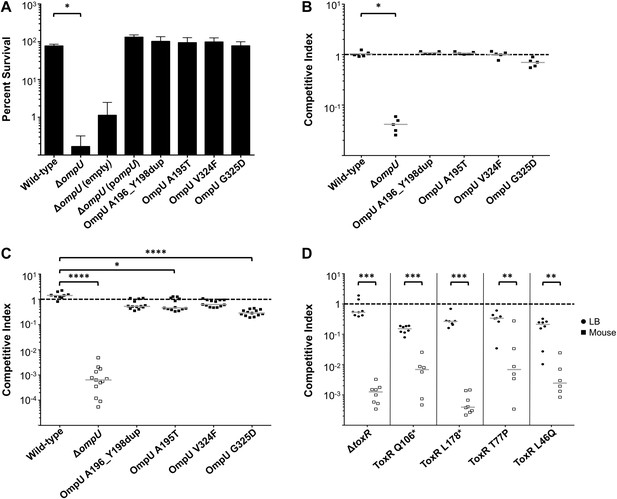
The fitness cost of clinically relevant OmpU and ToxR mutations.
(A) Clinically relevant OmpU mutants retain fitness in the presence of bile. *p < 0.05 significantly different means for the compared data sets (Mann–Whitney U Test). (B) OmpU mutants retain competitive fitness in pond water. *p < 0.05 significantly different from wild-type control (Kruskal–Wallis and post hoc Dunn's multiple comparison tests). (C) OmpU mutants have slight competitive fitness defects when serially passaged in Luria–Bertani broth (for ca. 58 generations). *p < 0.05 or ****p < 0.0001 significantly different from wild-type control (Kruskal–Wallis and post hoc Dunn's multiple comparison tests). (D) ToxR mutants are attenuated in vivo using the infant mouse colonization model. **p < 0.01 or ***p < 0.001 significantly different from the in vitro median (Mann–Whitney U Tests). The horizontal bars indicate the median of each data set. Open symbols represent data below the limit of detection.
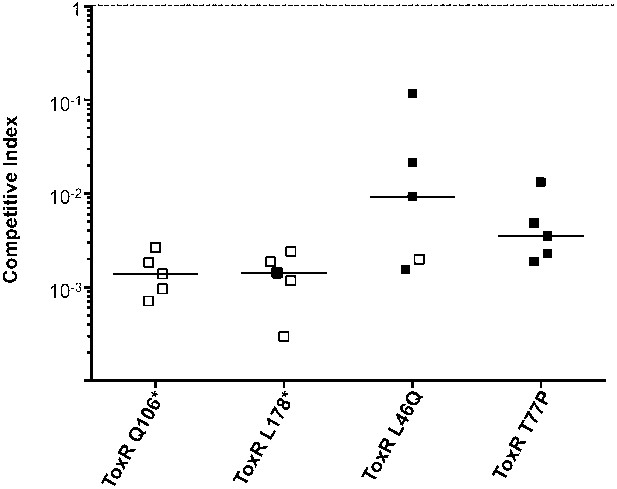
Clinical isolates harboring ToxR mutations are severely attenuated for infection.
Competitions indices (CI) in infant mice after 24 hr of infection were determined between each clinical ToxR mutant strain and its isogenic ToxR wild-type revertant strain carrying a lacZ deletion. The lacZ deletion allowed for differentiation of the competing strains upon plating on LB agar plates supplemented with X-gal. Each symbol represents the CI for an individual animal. The horizontal bars indicate the median of each data set. The open symbols represent data below the limit of detection for the ToxR mutant strain.
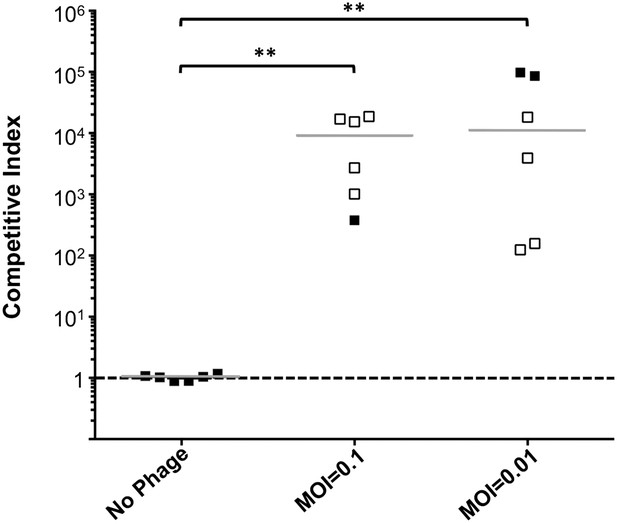
Phage predation leads to enrichment of OmpU mutant over wild-type in vivo.
Competitive indices (CI) were determined between wild-type ΔlacZ and OmpU G325D in the absence or presence of ICP2_2013_A_Haiti at the multiplicity of infection (MOI) indicated in infant rabbits 12 hr post-infection. Each symbol represents the CI for an individual rabbit and the horizontal lines indicate the median for each condition. The open symbols represent data below the limit of detection for the wild-type strain. **p < 0.01, significantly different from no phage control (Kruskal–Wallis and post hoc Dunn's multiple comparison tests).
Additional files
-
Supplementary file 1
V. cholerae isolates recovered from the ICP2-positive Haitian patient sample are isogenic except for mutations in ompU.
- https://doi.org/10.7554/eLife.03497.010
-
Supplementary file 2
V. cholerae isolates recovered from the ICP2-positive Bangladeshi patient sample are isogenic except for mutations in toxR.
- https://doi.org/10.7554/eLife.03497.011
-
Supplementary file 3
Strains used in this study.
- https://doi.org/10.7554/eLife.03497.012