FGF14 modulates resurgent sodium current in mouse cerebellar Purkinje neurons
Figures
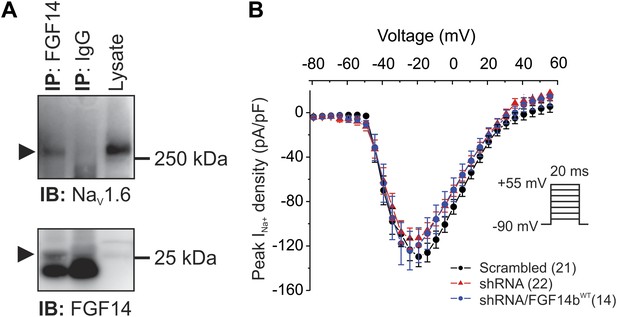
FGF14 is a component of the NaV1.6 macromolecular complex in mouse cerebellum, but shRNA knockdown does not affect NaV current density in cultured cerebellar Purkinje neurons.
(A) Co-immunoprecipitation (IP) of NaV1.6 by FGF14 from mouse cerebellum and immunoblot (IB) with the indicated antibodies. Mw markers are indicated on right. Arrowheads indicate NaV1.6 (top panel) and FGF14 (bottom panel). Intense signal in bottom panel below the FGF14 signal is immunoglobulin light chain. (B) Current–voltage relationships (normalized to cell capacitance) of transient NaV currents from cerebellar Purkinje neurons transfected with Scrambled control shRNA (Scrambled), FGF14 shRNA (shRNA), or FGF14 shRNA plus the shRNA-resistant FGF14bWT (shRNA/FGF14WT). The number of neurons tested, N, is in parentheses. Inset shows schematic of voltage protocol.
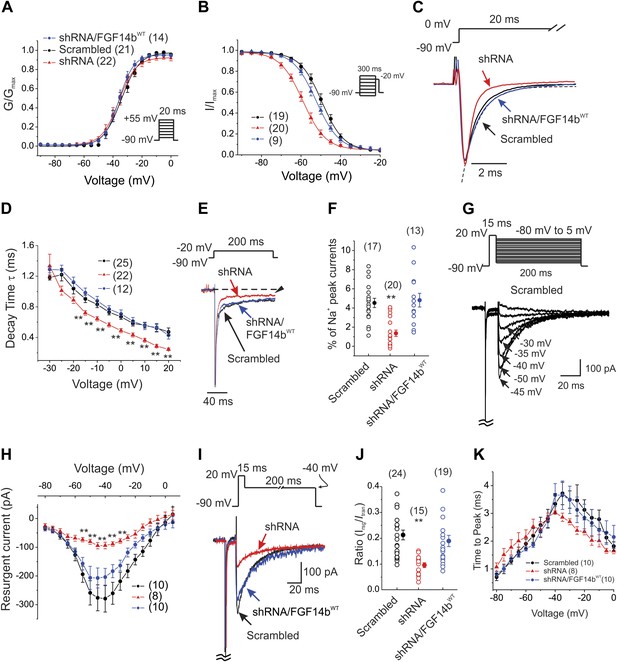
FGF14 knockdown in cerebellar Purkinje neurons affects multiple NaV channel biophysical properties.
(A and B) Voltage-dependence of NaV channel activation and steady-state inactivation in cerebellar Purkinje neurons transfected with Scrambled control shRNA (Scrambled), FGF14 shRNA (shRNA), or FGF14 shRNA plus the shRNA-resistant FGF14bWT (shRNA/FGF14WT). The number of neurons tested, N, is in parentheses. Inset shows schematic of voltage protocol. (C) Exemplar normalized NaV current traces elicited with a step depolarization to 0 mV from a holding potential of −90 mV and an exemplar single exponential fit (for Scrambled) for the time constant (τ) of inactivation (dotted line). (D) τ of inactivation at the indicated test voltages. The number of neurons tested, N, is in parentheses. **p < 0.01. (E) Exemplar normalized TTX-sensitive late NaV currents at −20 mV (measured at 150 ms, arrowhead) from a holding potential of −90 mV. (F) Amplitude of late NaV current as a % of peak (transient) NaV current. The number of neurons tested, N, is in parentheses. **p < 0.01. (G) Voltage-clamp protocol and exemplar TTX-sensitive resurgent NaV currents recorded from cerebellar Purkinje neurons transfected with Scrambled control shRNA. The transient current has been clipped. (H) Current–voltage relationship of NaV resurgent currents. The number of neurons tested, N, is in parentheses. **p < 0.01. (I) Overlay of NaV resurgent currents for the indicated conditions recorded with the indicated voltage protocol. The transient current has been clipped. (J) Ratio of peak NaV resurgent current (at +20 mV) to transient NaV current (at −10 mV). The number of neurons tested, N, is in parentheses. **p < 0.01. (K) Time to peak of NaV resurgent current over a broad range of voltages. The number of neurons tested, N, is in parentheses. **p < 0.01.
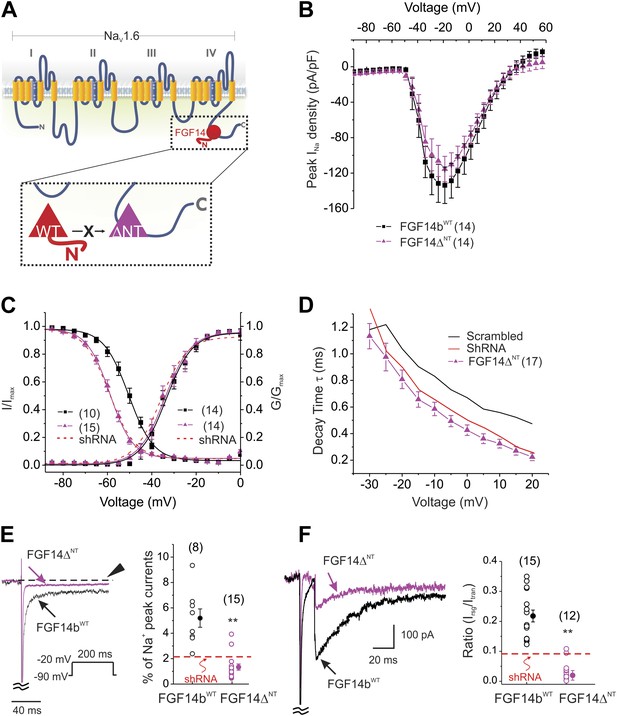
Expression of the dominant negative FGF14ΔNT affects multiple NaV channel biophysical properties indicating essential roles for the FGF14 N-terminus.
(A) Schematic of endogenous wild type (WT) FGF14 binding to the C-terminus of the NaV1.6 α subunit (top) and magnified schematic of the expressed FGF14ΔNT preventing binding of the WT FGF14 (in box). (B) Current–voltage relationships (normalized to cell capacitance) of transient NaV currents from cerebellar Purkinje neurons transfected with FGF14bWT or FGF14ΔNT. The number of neurons tested, N, is in parentheses. The current–voltage relationship for Scrambled control shRNA (from Figure 1) is shown for comparison. (C) Voltage dependence of NaV channel activation and steady-state inactivation in cerebellar Purkinje neurons transfected with FGF14bWT or FGF14ΔNT. The number of neurons tested, N, is in parentheses. The curves for Scrambled control shRNA (from Figure 1) are shown for comparison. (D) τ of inactivation at the indicated test voltages. The number of neurons tested, N, is in parentheses. (E) Exemplar normalized TTX-sensitive late NaV currents at −20 mV (measured at 150 ms, arrowhead) from a holding potential of −90 mV. The number of neurons tested, N, is in parentheses. **p < 0.01. (F) Overlay of NaV resurgent currents recorded from cerebellar Purkinje neurons transfected with FGF14ΔNT or FGF14bWT and ratio of peak NaV resurgent current (at +20 mV) to transient NaV current (at −10 mV). The number of neurons tested, N, is in parentheses. The average for FGF14 shRNA knockdown (Figure 1) is shown for comparison. **p < 0.01.
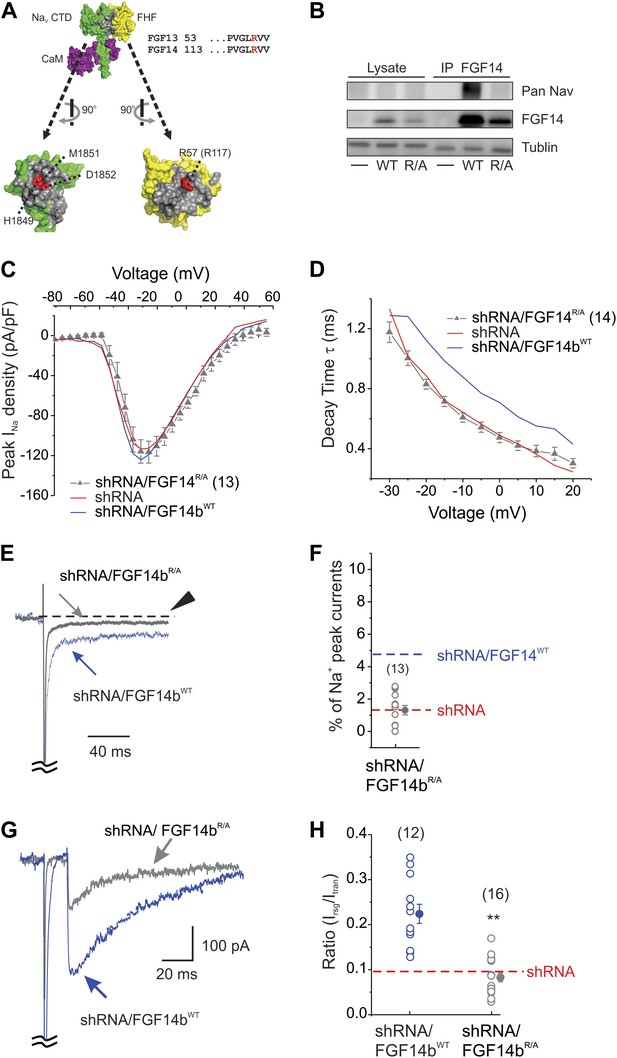
The FGF14bR/A mutant that prevents interaction with the NaV C-terminus cannot rescue NaV kinetic effects of FGF14 knockdown.
(A) Surface representation of the crystal structure of a ternary complex containing the NaV1.5 C-terminus domain (CTD, green), FGF13 (yellow), and calmodulin (purple); the interaction surfaces are colored gray (PDB ID: 4DCK). The critical R57 in FGF13 (equivalent to R117 in FGF14) is indicated in red, as is its binding pocket on the NaV1.5 CTD. (B) Co-immunoprecipitation (IP) of NaV1.6 and FGF14bWT or FGF14bR/A expressed in HEK293 cells, showing that the FGF14bR/A is unable to interact with the intact NaV1.6. Immunoblots (IB) were performed with the indicated antibodies. (C) Current–voltage relationship (normalized to cell capacitance) of transient NaV currents from cerebellar Purkinje neurons transfected with FGF14bR/A. The number of neurons tested, N, is in parentheses. The current–voltage relationship for FGF14 knockdown (shRNA) and knockdown rescued with shRNA-insensitive FGF14bWT (shRNA/FGF14bWT) from Figure 1 are shown for comparison. (D) τ of inactivation at the indicated test voltages. The number of neurons tested, N, is in parentheses. (E) Exemplar normalized TTX-sensitive late NaV currents at −20 mV (measured at 150 ms, arrowhead) from a holding potential of −90 mV and (F) amplitude of late NaV current as a % of peak (transient) NaV current. The number of neurons tested, N, is in parentheses. Averages for shRNA and for shRNA/FGF14bWT (see Figure 1) are shown for comparison. (G) Overlay of NaV resurgent currents recorded from cerebellar Purkinje neurons transfected with FGF14bR/A or FGF14bWT and (H) ratio of peak NaV resurgent current (at +20 mV) to transient NaV current (at −10 mV). The number of neurons tested, N, is in parentheses. The average for FGF14 shRNA knockdown (Figure 1) is shown for comparison. **p < 0.01.
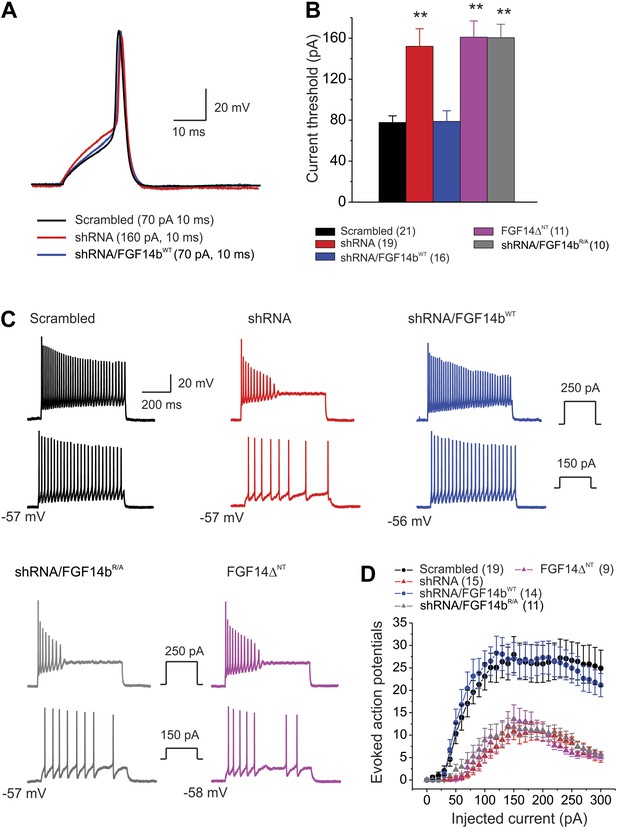
Action potential dynamics and repetitive spiking are dependent upon FGF14 and its N-terminus.
(A) Overlay of single action potentials evoked with a 10 ms current injection (current amplitude shown in parentheses). (B) Current threshold to induce action potentials for the indicated conditions. **p < 0.01. The number of neurons tested, N, is in parentheses. Additional summary data are presented in Table 2. (C) Example evoked action potentials for the indicated treatments at two separate current injection amplitudes (shown in insets). The resting membrane potential is indicated. (D) The number of evoked action potentials for the indicated amplitude of current injection. The number of neurons tested, N, is in parentheses.
Tables
NaV current activation and inactivation parameters in cerebellar Purkinje neurons
| Activation | Inactivation | |||||
|---|---|---|---|---|---|---|
| V1/2 (mV) | K | n | V1/2 (mV) | K | n | |
| Scrambled | −34.4 ± 1.5 | 4.0 ± 0.3 | 21 | −49.7 ± 1.2 | 4.2 ± 0.1 | 19 |
| shRNA | −35.0 ± 1.9 | 4.3 ± 0.5 | 22 | −59.0 ± 1.2** | 4.7 ± 0.2 | 20 |
| shRNA/FGF14bWT | −35.5 ± 1.5 | 3.9 ± 0.3 | 14 | −52.0 ± 0.9 | 5.2 ± 0.3 | 9 |
| shRNA/FGF14RA | −33.2 ± 1.5 | 3.5 ± 0.2 | 12 | −50.9 ± 1.4 | 4.4 ± 0.3 | 11 |
| FGF14bWT | −33.1 ± 1.4 | 4.5 ± 0.5 | 14 | −50.6 ± 1.3 | 4.9 ± 0.3 | 10 |
| FGF14∆NT | −33.9 ± 2.0 | 3.9 ± 0.5 | 14 | −59.1 ± 0.8** | 4.7 ± 0.3 | 15 |
-
Mean ± s.e.m. (n), **p < 0.01 compared to Scrambled control.
Intrinsic membrane properties and single action potential (AP) characteristics measured in cerebellar Purkinje neurons
| Scrambled | shRNA | shRNA/FGF14bWT | shRNA/FGF14bRA | FGF14∆NT | |
|---|---|---|---|---|---|
| Input resistance (MΩ) | 221.8 ± 15.8 (7) | 238.0 ± 49.5 (11) | 266.6 ± 24.5 (11) | 225.9 ± 10.8 (6) | 269.5 ± 30.2 (6) |
| Resting membrane potential (mV) | −54.5 ± 1.3 (21) | −55.8 ± 1.2 (19) | −54.4 ± 1.0 (16) | −55.4 ± 0.7 (10) | −54.9 ± 1.4 (11) |
| Current threshold (pA) | 77.8 ± 6.4 (21) | 142.1 ± 18.5 (19)** | 78.8 ± 10.4 (16) | 170.5 ± 16.7 (10)** | 191.0 ± 22.1 (11)** |
| AP threshold (mV) | −35.6 ± 1.0 (21) | −32.8 ± 1.2 (19) | −34.5 ± 0.8 (16) | −32.8 ± 1.4 (10) | −32.7 ± 0.8 (11) |
| AP amplitude (mV) | 78.8 ± 3.2 (21) | 77.5 ± 2.8 (19) | 78.9 ± 4.5 (16) | 79.1 ± 4.3 (10) | 73.1 ± 4.7 (11) |
| AP duration (ms) | 2.3 ± 0.2 (21) | 2.0 ± 0.1 (19) | 2.2 ± 0.1 (16) | 1.8 ± 0.1 (10) | 2.1 ± 0.3 (11) |
-
Mean ± s.e.m. (n), **p < 0.01 compared to Scrambled control.





