RNA chaperones buffer deleterious mutations in E. coli
Figures
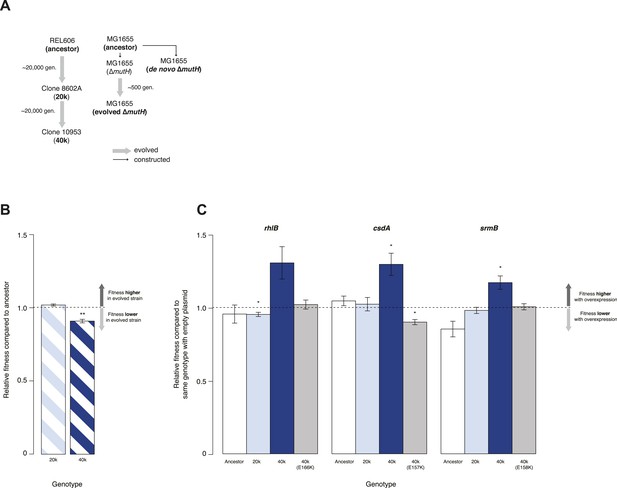
Relative fitness of Escherichia coli REL606-derived strains.
(A) Relationships between strains used in different competition assays. Short names of competed strains are given in bold; gen.: generations. (B) Relative fitness of the 20k and 40k genotypes, each competed against their REL606 ancestor. (C) Relative fitness of ancestral and evolved genotypes overexpressing one of three DEAD box RNA helicases (DBRHs) compared with identical strains carrying the empty control plasmid. E166K, E157K, and E158K: competitions in the 40k background where plasmids carried mutated versions of the respective DBRH. In each case, the central glutamic acid residue of the DEAD motif has been recoded to lysine, compromising the helicase activity. Bar heights indicate mean relative fitness across four biological replicates, with each mean derived by averaging over four technical replicates. Error bars represent standard errors of the mean. **p < 0.01, *p < 0.05 (one-sample t-test). Additional results for competitions terminated in mid-exponential phase (after 2 hr) are shown in Figure 1—figure supplement 1.
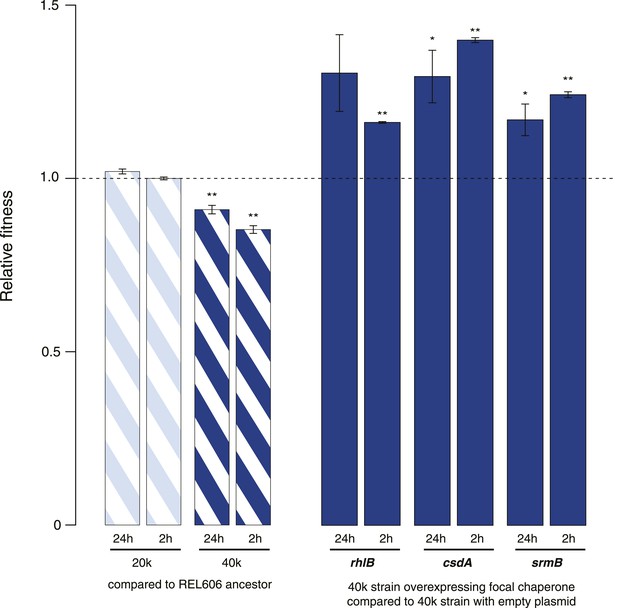
Relative fitness in competition experiments terminated in mid-exponential phase (REL606).
https://doi.org/10.7554/eLife.04745.004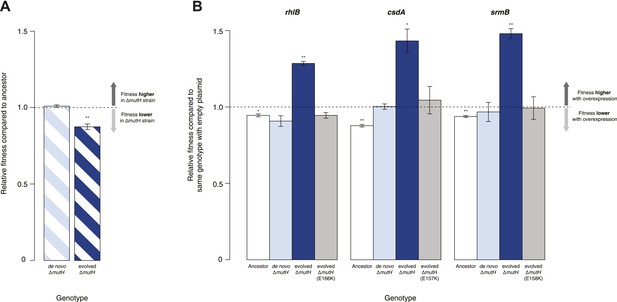
Relative fitness of Escherichia coli MG1655-derived strains.
(A) Relative fitness of the evolved and de novo-constructed ΔmutH strains, each competed against their MG1655 ancestor. (B) Relative fitness of ancestral, evolved, and de novo ΔmutH genotypes overexpressing one of three DEAD box RNA helicases compared with identical strains carrying the empty control plasmid. E166K, E157K, and E158K, bar heights and error bars are as described in Figure 1. **p < 0.01, *p < 0.05 (one-sample t-test). Additional results for competitions terminated in the mid-exponential phase (after 2 hr) are shown in Figure 2—figure supplement 1.
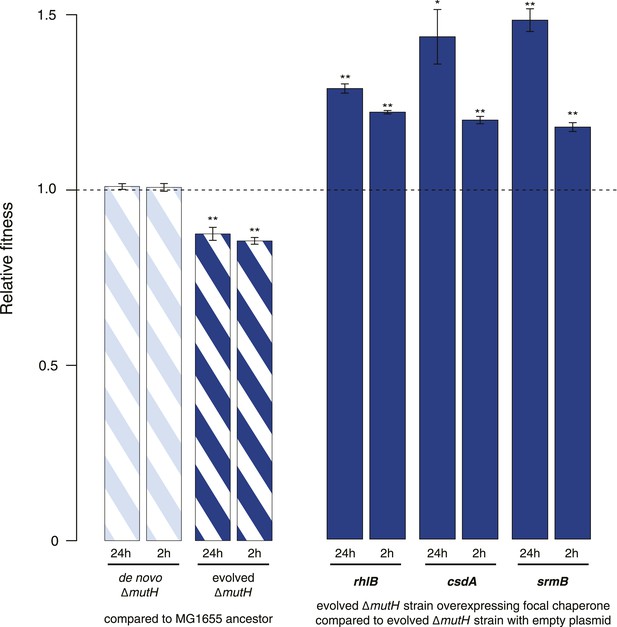
Relative fitness in competition experiments terminated in mid-exponential phase (MG1655).
https://doi.org/10.7554/eLife.04745.006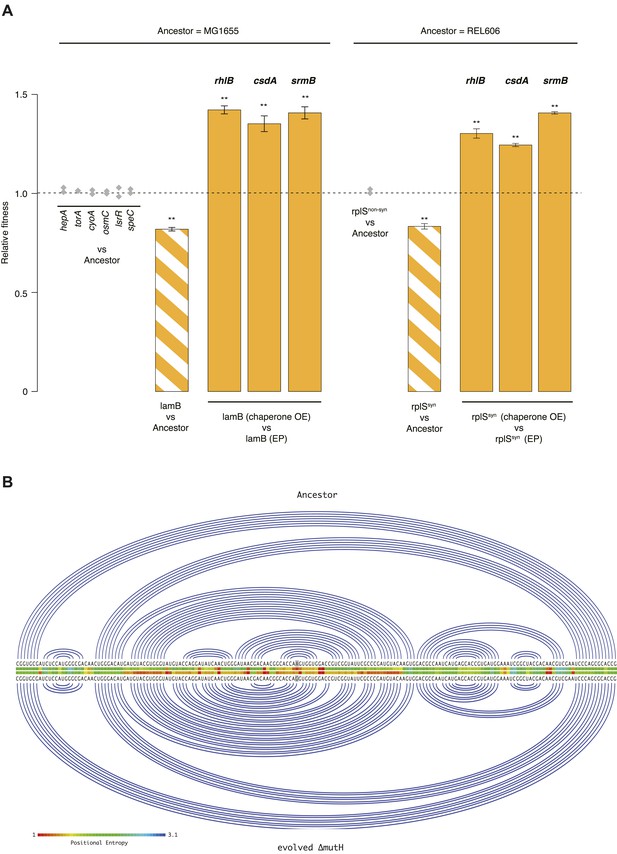
Fitness effects and buffering of individual mutations.
(A) Relative fitness of strains carrying single point mutations introduced into the relevant ancestral background competed against the respective ancestor. The mutations correspond to those listed in Supplementary files 2, 3 for the respective genes. Initial screening for fitness defects involved two biological replicates (grey diamonds). For the two mutations where the initial screen suggested a measurable fitness deficit, lamB and rplSsyn, all competitions were carried out in quadruplicate. Bar heights and error bars are as described in Figure 1. **p < 0.01, *p < 0.05 (one-sample t-test). OE: overexpression; EP: empty plasmid. Additional results for competitions terminated in mid-exponential phase (after 2 hr) are shown in Figure 3—figure supplements 1, 2. (B) Linear Feynman graph of the lamB region that harbours the mutation in the evolved ΔmutH strain (highlighted in grey). We predicted RNA secondary structure for the entire malK-lamB-malM transcription unit (RegulonDB identifier: ECK120009315) and its mutated counterpart using RNAfold (Lorenz et al., 2011). The malK-lamB-malM operon contains a repetitive extragenic palindromic (REP) element downstream of lamB, which prevents premature degradation of the lamB cistron following cleavage from malM. Resolution of this REP element as part of regulated degradation was previously shown to require RhlB (Khemici and Carpousis, 2004). However, comparison of predicted minimum free energy structures between wild type and mutant malK-lamB-malM transcripts suggested structural changes that do not interfere with REP element formation but rather lead to decreased positional entropy at the local level, as highlighted here.
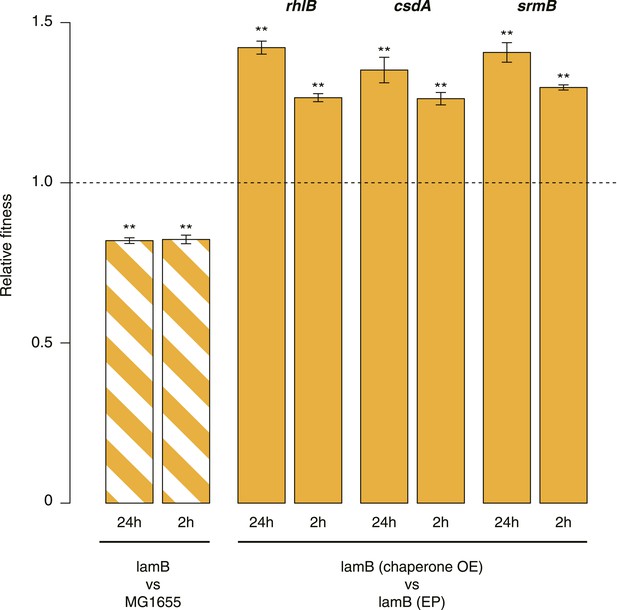
Relative fitness in competition experiments terminated in mid-exponential phase (lamB).
OE: overexpression; EP: empty plasmid.
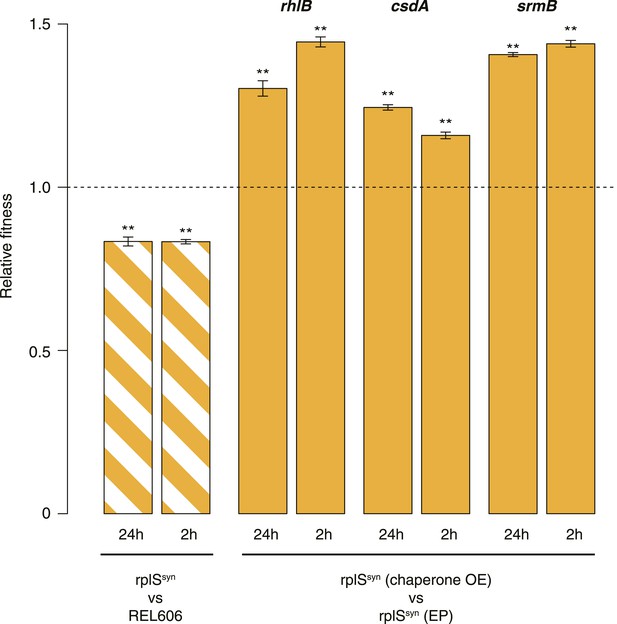
Relative fitness in competition experiments terminated in mid-exponential phase (rplSsyn).
OE: overexpression; EP: empty plasmid.
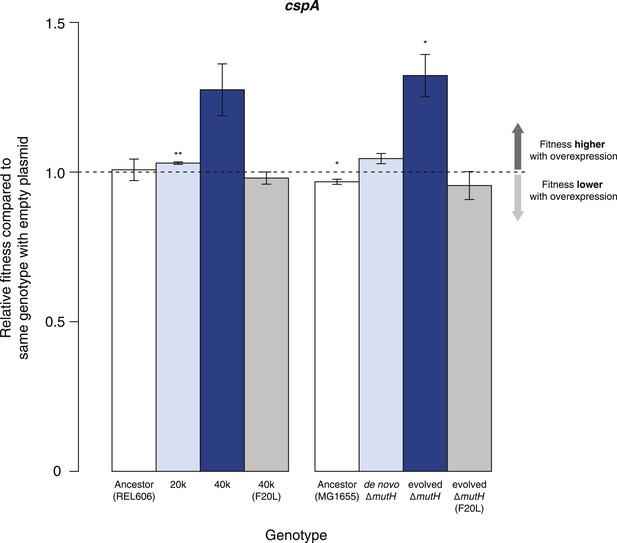
Effects of CspA overexpression on relative fitness.
Relative fitness of REL606- and MG1655-derived strains overexpressing CspA compared with strains of the same genotype carrying the empty control plasmid. F20L: competitions in the 40k and evolved ΔmutH backgrounds, respectively, where plasmids carried a mutated version of the cspA gene yielding a protein with compromised nucleic acid binding ability (Hilier et al., 1998). Bar heights and error bars are as described in Figure 1. **p < 0.01, *p < 0.05 (one-sample t-test). Additional results for competitions terminated in mid-exponential phase (after 2 hr) and competitions involving the lamB mutant in the evolved ΔmutH strain and the rplSsyn mutant in the 40k strain are shown in Figure 4—figure supplement 1.
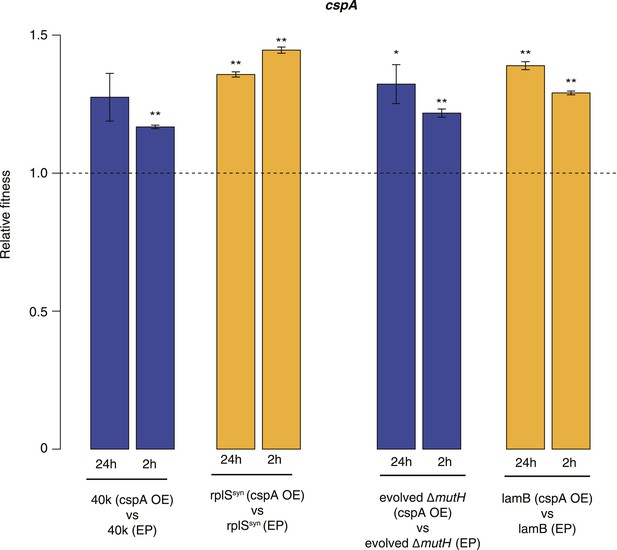
Relative fitness in competition experiments terminated in mid-exponential phase (cspA).
OE: overexpression; EP: empty plasmid.
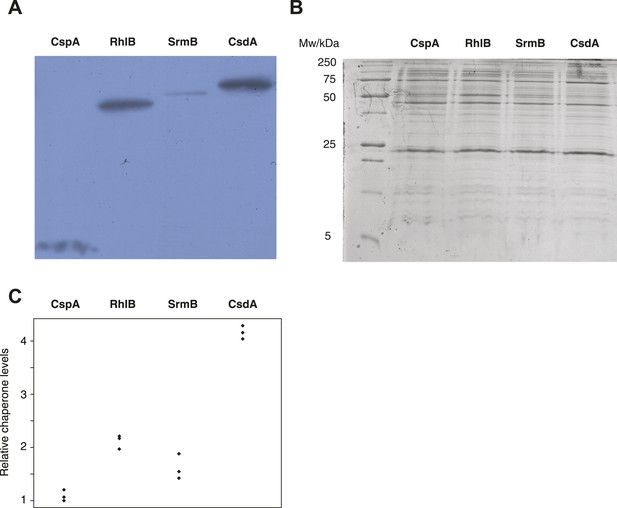
Relative chaperone abundances.
(A) Representative Western blot for evolved ΔmutH strains overexpressing one of the focal RNA chaperones. Molecular weights (from nucleotide sequence): CspA, 7.403 kD; RhlB, 47.126 kD; SrmB, 49.914 kD; CsdA, 70.546 kD. (B) Representative Coomassie-stained SDS-PAGE gel. (C) Relative chaperone levels are defined as the ratio of Western blot intensity to Coomassie intensity (see ‘Materials and methods’). The lowest ratio detected across triplicate experiments in all strains was set to one. Comparing these ratios between strains overexpressing different RNA chaperones gives a semi-quantitative indication of relative chaperone abundances. For example, CsdA levels in CsdA-overexpressing cells are ∼fourfold higher than CspA levels in CspA-overexpressing cells. Note that this metric does not allow conclusions about the absolute fraction of total protein that is occupied by each chaperone in the different strains.
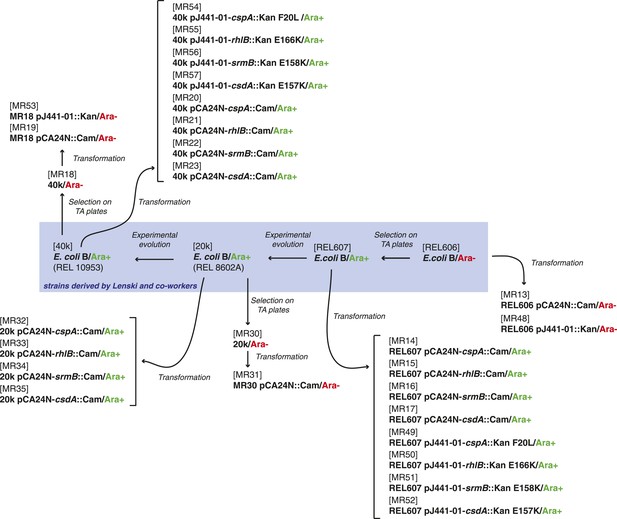
Relationship between REL606-derived strains.
https://doi.org/10.7554/eLife.04745.014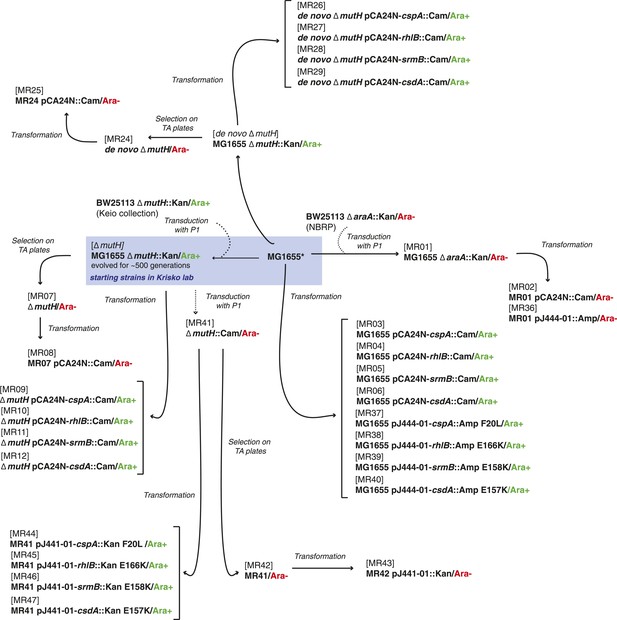
Relationship between MG1655-derived strains.
*Resequencing of the MG1655 laboratory strain revealed a single difference to the NC000913 reference genome, an intergenic dinucleotide insertion at position 4296380 (AC → ACGC).
Tables
Number of mutations in evolved mutator strains compared with their respective ancestors
Additional files
-
Supplementary file 1
Mutations in the 20k strain.
- https://doi.org/10.7554/eLife.04745.016
-
Supplementary file 2
Mutations in the 40k strain.
- https://doi.org/10.7554/eLife.04745.017
-
Supplementary file 3
Mutation in the evolved ΔmutH strain.
- https://doi.org/10.7554/eLife.04745.018
-
Supplementary file 4
List of Escherichia coli strains used in this study.
- https://doi.org/10.7554/eLife.04745.019
-
Supplementary file 5
Primers used for recombineering and sequencing.
- https://doi.org/10.7554/eLife.04745.020





