Ventromedial prefrontal cortex stimulation enhances memory and hippocampal neurogenesis in the middle-aged rats
Figures
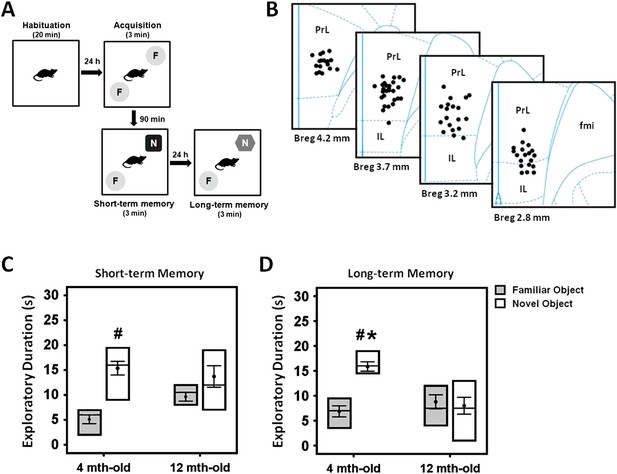
Experimental protocol of the novel-object recognition test (A), and representative illustration of the stimulating electrode localization in the vmPFC (B).
The box plots show the comparisons between young (4 month old) and middle-aged (12 month old) animals on the short- and long-term memory retention interval in the novel-object recognition task (C, D). Note: there was a decrease of time spent in the novel object exploration in the middle-aged animals as compared to the young rats, suggesting a possible manifestation of memory deficit in this animal model. Indication: *, significant difference from the middle-aged rats; #, significant difference from the familiar object of respective age animals, (p < 0.05).
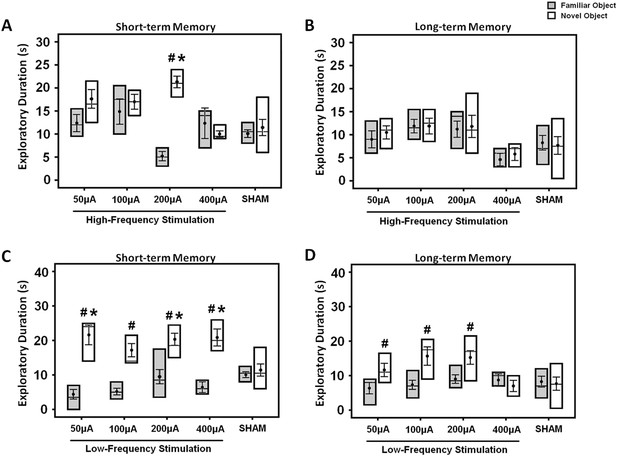
The box plots show the effects of either high- (A, B) or low-frequency (C, D) stimulation at amplitudes varying across 50, 100, 200, and 400 μA in the middle-aged animals.
Both the short- and long-term memory functions were tested using the novel-object recognition test. Note: HFS (100 Hz) at 200 μA and LFS (10 Hz) at 50, 200, 400 μA significantly increased the novel object exploration as compared to the sham animals, respectively. Indication: *, significant difference from the sham rats; #, significant difference from the familiar object of respective stimulation amplitude, (p < 0.05).
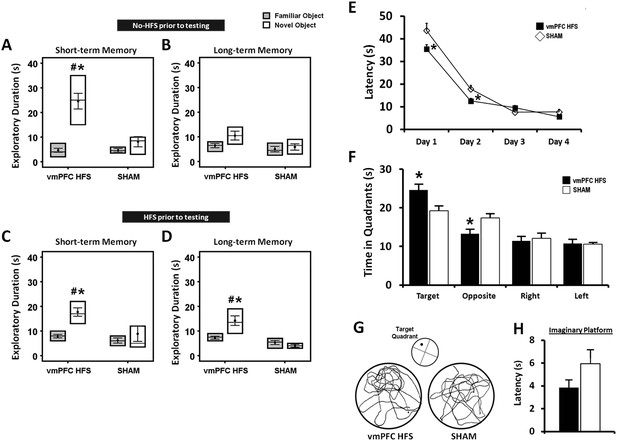
Effects of chronic stimulation on the short- and long-term memory retention interval in the novel-object recognition test and Morris water-maze task.
Animals were tested in two different conditions with either no-HFS (A, B) or HFS (C, D) prior to the task. Effects of chronic stimulation on the memory performance in the Morris water-maze test (E, F). Note: VmPFC HFS significantly enhanced the short- and long-term memory performances in the novel-object recognition test (C, D). In the Morris water-maze experiment, there was an improvement on learning and memory after vmPFC HFS in both the training (E) and probe test (F) phases. Representative swimming paths (G) in the probe test, demonstrating vmPFC HFS increased duration within the virtual zone around the platform's location. Latency to reach the imaginary platform showed no difference between the vmPFC HFS and sham animals during the probe test (H). Indication: *, significant difference from the sham rats, (p < 0.05); #, significant from the familiar object of respective stimulation amplitude, (p < 0.05).
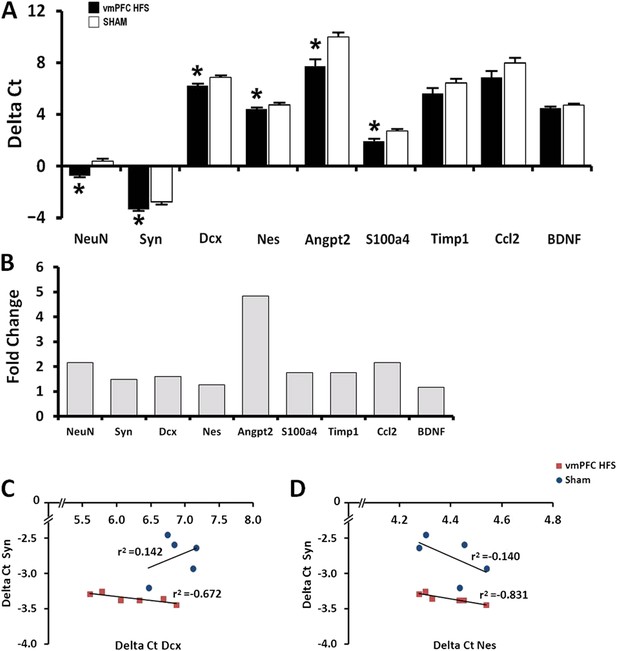
Effects of chronic vmPFC HFS on the mRNA gene expression related to neuroplasticity in the hippocampus (A).
Note: vmPFC HFS upregulated genes involved in proliferation and neurogenesis-related functions including the NeuN, Syn, Dcx, Nes, Angpt2, and S100a4 relative to sham. No changes were found for Timp1, Ccl2, and BDNF. Calculation for the fold-change values indicating that vmPFC HFS induced approximately 4.8-fold (Angpt2), 2-fold (NeuN), and >1-fold (Syn, Dcx, Nes, S100a4) increase of gene expression relative to the sham (B). Interestingly, scatter plots show significant correlation between the Syn and the Nes/Dcx (C, D), indicating that these genes are strongly related to each other for neuroplasticity in the hippocampus after chronic vmPFC HFS. Gene expression was expressed as the change in Ct of the gene of interest compared to the sham (Delta Ct); and relative expression was calculated using the comparative CT method with fold change 2-Delta (Delta Ct). Indication: *, significant difference from the sham rats, (p < 0.05).
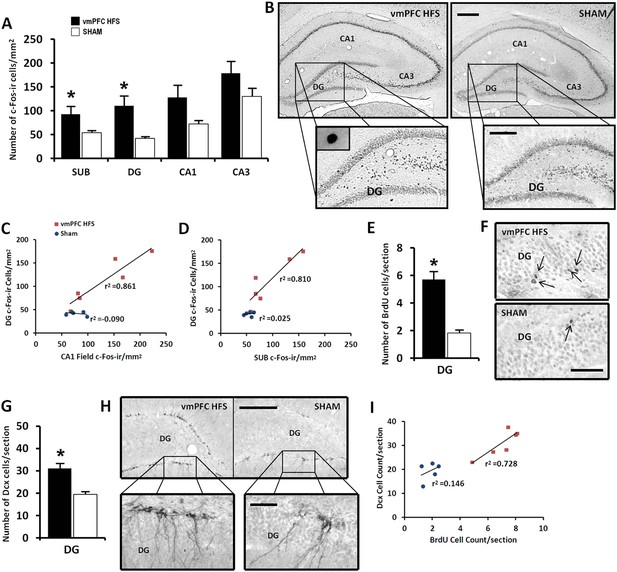
Effects of chronic vmPFC HFS on the hippocampal neuronal activity by c-Fos-ir (A–B) and the morphological changes related to neurogenesis functions (E–I).
Note: VmPFC HFS increased the number of c-Fos-ir positive cells in the subiculum, DG, and a marginal difference (p = 0.059) in the CA1 field of the hippocampus as compared to the sham (A–B; scale bars: 500 μm, low-power magnification; 250 μm, high-power magnification). In neurogenesis-related morphology, after chronic vmPFC HFS, there was an increase of surviving BrdU-positive cells (E–F, scale bar: 500 μm), and neural progenitors—Dcx-positive cells (G–H; scale bars: 300 μm, low-power magnification; 50 μm, high-power magnification). For correlational analysis, there was a strong relationship between the BrdU and Dcx cell-count (I). Importantly, the neurogenic zone of the DG was also highly correlated with the SUB and CA1 field of the hippocampus (C–D), indicating that these regions were functionally associated with memory functions after chronic vmPFC HFS. Abbreviations: SUB, subiculum; DG, dentate gyrus; CA1, CA1 field of the hippocampus; CA3, CA3 field of the hippocampus; vmPFC HFS, high-frequency stimulation of the ventromedial prefrontal cortex; BrdU, 5-bromo-2′-deoxyuridine; and Dcx, doublecourtin. Indication: *, significant difference from the sham rats, (p < 0.05).
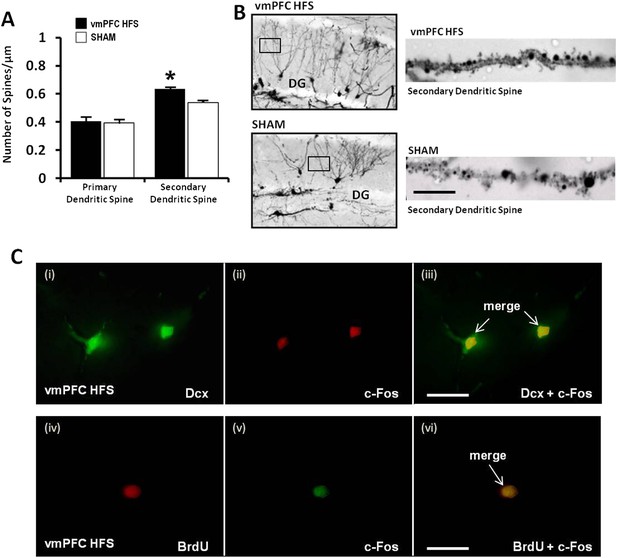
Effects of chronic vmPFC HFS on the Golgi measurement of dendritic spine density and immunofluorescence labeling of neurogenesis-related cell function in the hippocampal DG area.
Note: there was an increase in the secondary, but not the primary dendritic spine density of the Golgi-impregnated cells in the DG area of the hippocampus (A–B; scale bar: 10 μm). Representative confocal images (C) are demonstrated for the localization of Dcx (green, C-i), c-Fos (red, C-ii; or green, C-v), and BrdU-labeled (red, C-iv) immunofluorescence positive cells. Merged images showed the co-localization of Dcx and c-Fos (C-iii), as well as BrdU and c-Fos (C-vi) in the hippocampal DG region (scale bar: 40 μm). Indication: *, significant difference from the sham rats, (p < 0.05).
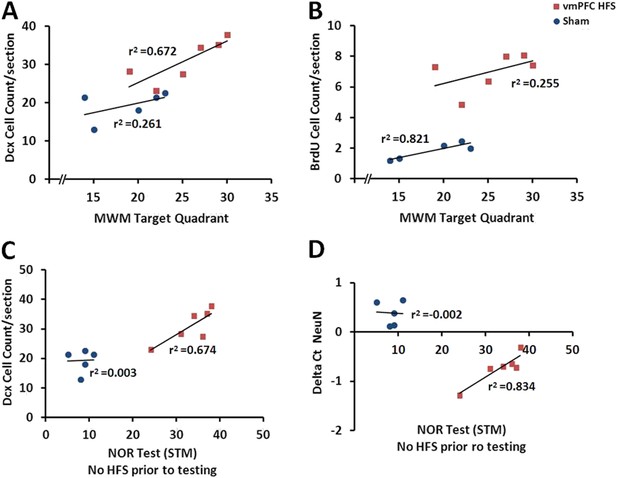
Scatter plots display the correlations between the variables related with the hippocampal neuroplasticity and the hippocampal-dependent memory behavioral tests.
Note: In the vmPFC HFS animals, the Morris water-maze target quadrant is positively correlated with the Dcx cell count (A), while the novel-object recognition task with no-HFS prior to testing shows positive correlation with the Dcx cell count (C) as well as the NeuN/Rbfox3 gene expression (D). No correlational association was found between the Morris water-maze and BrdU cell count after chronic vmPFC high-frequency stimulation (B).
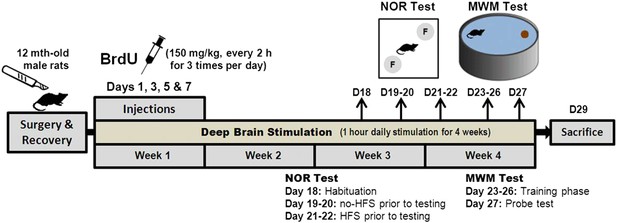
Schematic representation of the experimental design for chronic stimulation and behavioral testing of memory functions in the middle-aged animals.
https://doi.org/10.7554/eLife.04803.010Tables
The primers sequences used for real-time quantitative-PCR analysis
| Gene symbol | 5′–3′ primer sequence |
|---|---|
| NeuN (Rbfox3) | Fwd 5′–GGCTGGAAGCTAAACCCTGT–3′; Rev 5′–TCCGATGCTGTAGGTTGCTG–3′ |
| Syn | Fwd 5′–GTGCCAACAAGACGGAGAGT–3′; Rev 5′–TTGGTAGTGCCCCCTTTGAC–3′ |
| Dcx | Fwd 5′–ACGACCAAGACGCAAATGGA–3′; Rev 5′–AGGCCAAGGATCTGACTTG –3′ |
| Nes | Fwd 5′–TAAGTTCCAGCTGGCTGTGG–3′; Rev 5′–ATAGGTGGGATGGGAGTGCT–3′ |
| Angpt2 | Fwd 5′–GGACCCTGCAGCTACACATT–3′; Rev 5′–TGTCACAGTAGGCCTTGACC–3′ |
| S100a4 | Fwd 5′–CTTGGTCTGGTCTCAACGGT–3′; Rev 5′–GCAGCTTCGTCTGTCCTTCT–3′ |
| Timp1 | Fwd 5′–ACGCTAGAGCAGATACCACG–3′; Rev 5′– GATCGCTCTGGTAGCCCTTC–3′ |
| Ccl2 | Fwd 5′–AGCCAACTCTCACTGAAGCC–3′; Rev 5′–TGGGGCATTAACTGCATCTGG–3′ |
| BDNF | Fwd 5′–AGGACAGCAAAGCCACAATGTTC–3′; Rev 5′–TTGCCTTGTCCGTGGACGTTTG–3′ |
| HPRT | Fwd 5′–AGGCCAGACTTTGTTGGATT–3′; Rev 5′–GCTTTTCCACTTTCGCTGAT–3′ |
Additional files
-
Supplementary file 1
The tables show the total exploratory duration during the acquisition phase, short- and long-term memory retention intervals in the novel-object recognition test for animal experiments of comparisons between the young and middle-aged rats (A), acute stimulation (B, C), and chronic stimulation (D, E) studies. Indication: *, significant difference from the sham rats.
- https://doi.org/10.7554/eLife.04803.012
-
Supplementary file 2
The tables show the total exploratory duration for both identical object 1 and 2 during the acquisition phase for animal experiments of comparisons between the young and middle-aged rats (A), acute stimulation (B), and chronic stimulation (C) studies.
- https://doi.org/10.7554/eLife.04803.013





