Distinct mechanisms regulating mechanical force-induced Ca2+ signals at the plasma membrane and the ER in human MSCs
Figures
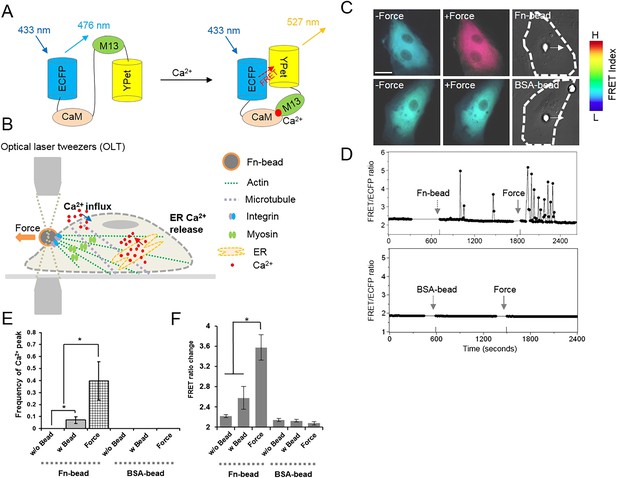
Intracellular Ca2+ oscillations in response to mechanical force in HMSCs with Ca2+-free medium.
(A) A schematic drawing of the activation mechanism of the Ca2+ FRET biosensor. (B) Beads coated with Fn or BSA were seeded onto the cell and mechanical force was applied by pulling a Fn-coated bead using optical laser tweezers. Both Ca2+ influx and ER Ca2+ release can contribute to force-induced Ca2+ signals. (C) Color images represent the YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor. The color scale bars represent the range of emission ratio, with cold and hot colors indicating low and high levels of Ca2+ concentration, respectively. (D) The time courses represent the YPet/ECFP emission ratio averaged over the cell body outside of nucleus upon seeding of Fn or BSA-coated beads and force application. (E–F) Bar graphs represent the frequency or ratio of the intracellular Ca2+ oscillations evoked by mechanical force. Error bars indicate standard error of mean; *p < 0.05, n = 14. (Scale bar: 10 µm).
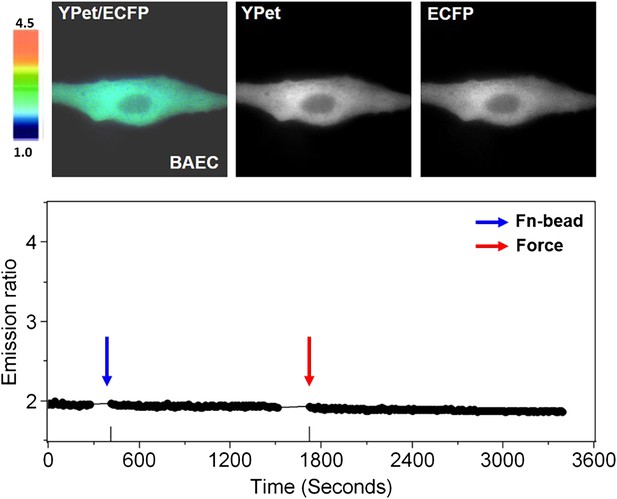
Laser-tweezer pulling of a Fn-coated bead on a BAEC in Ca2+-free medium.
Color images (upper panels) represent the YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor. The color scale bars represent the range of emission ratio, with cold and hot colors indicating low and high levels of Ca2+ concentration, respectively. The time courses of the YPet/ECFP emission ratio averaged over the cell body outside of nucleus is shown in the lower graph.
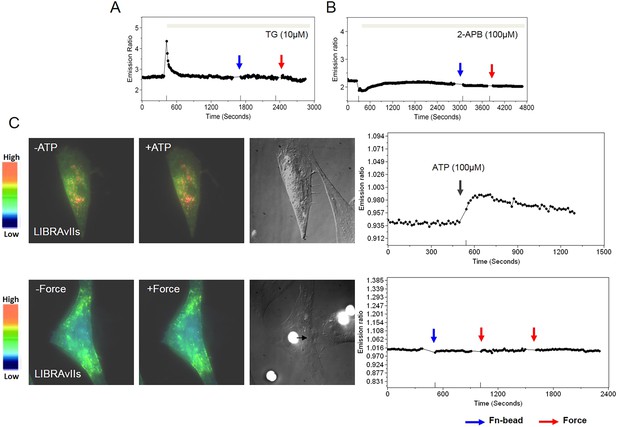
Mechanical force doesn’t induce any increase in IP3 level.
The time courses represent the YPet/ECFP emission ratio of cytoplasmic Ca2+ in HMSCs pretreated with (A) Thapsigargin (TG, 10 µM), a SERCA pump blocker, or (B) 2-APB (100 µM), an IP3R blocker. (C) IP3 production is monitored by a FRET-based IP3 biosensor, LIBRAvIIs. ATP treatment induces IP3 increase, which can be clearly detected by an IP3 biosensor LIBRAvIIs (upper panels). However, laser-tweezer pulling of a Fn-coated bead to produce the mechanical force did not cause any increase in IP3 (lower panels).
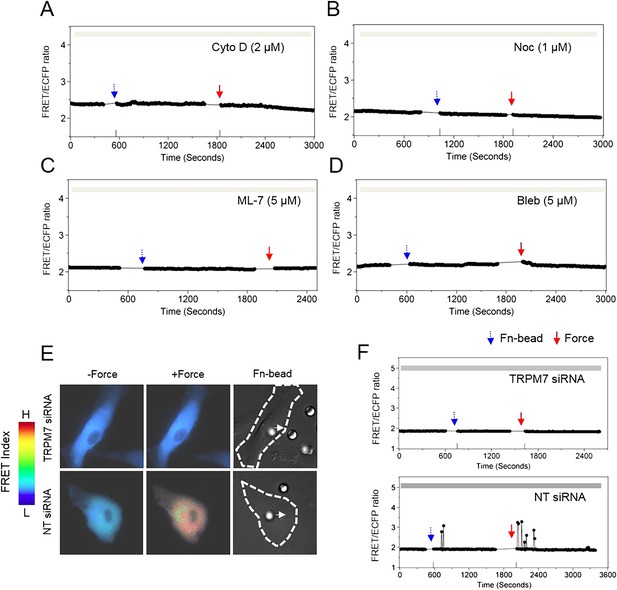
Cytoskeletal support, actomyosin contractility, and TRPM7 channels mediate the force-induced intracellular Ca2+ oscillations.
The time courses represent the YPet/ECFP emission ratio of cytoplasmic Ca2+ in HMSCs in the absence of extracellular Ca2+ when these cells were pretreated with (A) 2 μM Cyto D (n = 8), (B) 1 μM Noc (n = 8), (C) 5 μM ML-7 (n = 8), and (D) 5 μM Bleb (n = 8). (E) Color images represent the YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor in HMSCs transfected with NT or TRPM7 siRNA. The color scale bars represent the range of emission ratio, with cold and hot colors indicating low and high levels of Ca2+ concentration, respectively. (F) The time courses represent the YPet/ECFP emission ratio averaged over the cell bodies outside of nucleus treated with siRNA as indicated.
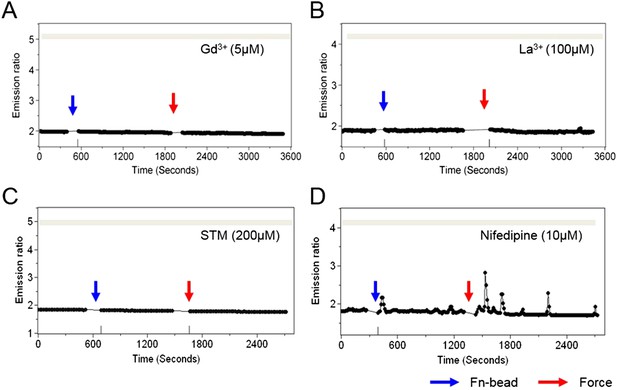
Stretch-activated or store-operated channels at the plasma membrane mediate force-induced Ca2+ release from ER.
The time courses represent the YPet/ECFP emission ratio of cytoplasmic Ca2+ in HMSCs pretreated with (A) GdCl3 (5 µM), a broad spectrum Ca2+ channel inhibitor, (B) LaCl3 (100 µM), a broad spectrum Ca2+ channel blocker, (C) streptomycin (200 µM), a mechanosensitive channel inhibitor, or (D) Nifedipine (10 µM), an L-type Ca2+ channel inhibitor, all in the absence of extracellular Ca2+.
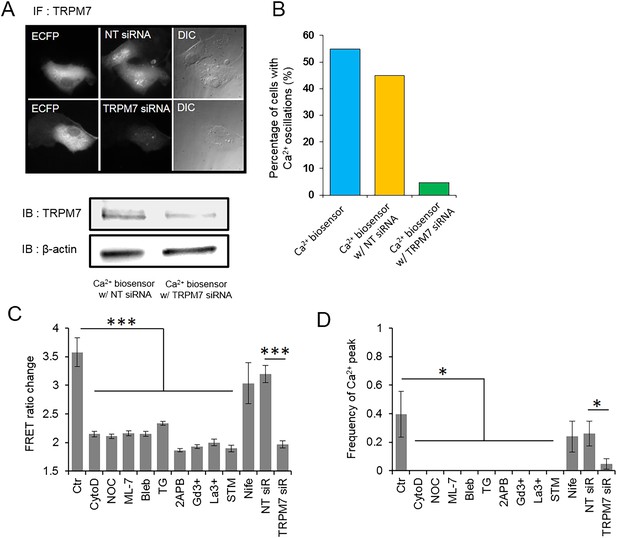
TRPM7 channels as well as cytoskeletal support and actomyosin contractility mediate the force-induced intracellular Ca2+ oscillations.
(A) HMSCs expressing Ca2+ biosensor and transfected with non-targeting (NT) or TRPM7 siRNA were immunostained (upper images) or immunoblotted (lower panels) with polyclonal TRPM7 antibody to assess the amount of TRPM7. TRPM7 specific siRNA induced knockdown of TRPM7. (B) Bar graphs represent the percentile of HMSCs showing intracellular Ca2+ oscillations. Three groups of cells (1: Ca2+ biosensor only, 2: biosensor and non-targeting (NT) siRNA, and 3: biosensor and TRPM7 siRNA) were measured and compared. The number of cells displaying Ca2+ oscillations in both control group (55%, 11 of 20 cells) and NT-siRNA group (45%, 9 of 20 cells) was approximately 9–11 fold higher than that of TRPM7 siRNA group (4.76%, 1 of 21 cells). (C–D) Bar graphs represent the frequency or ratio of the intracellular Ca2+ oscillations induced by mechanical force in the presence of different inhibitors. Error bars indicate standard error of mean; *p < 0.05.
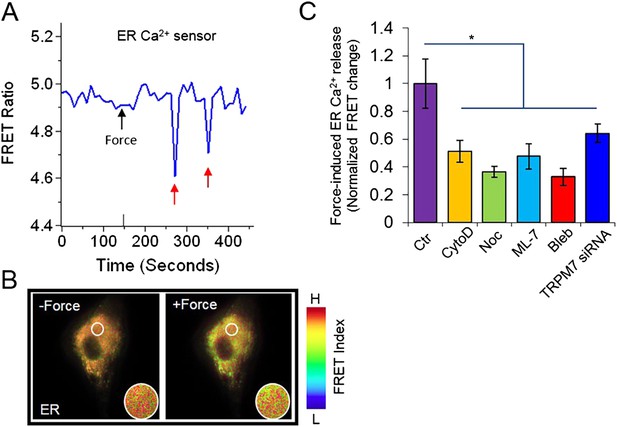
The visualization of force-induced Ca2+ release from ER using a FRET-based ER Ca2+ biosensor.
(A) The time course and (B) the color images of YPet/ECFP emission ratio in HMSCs expressing the D3ER biosensor before and after force application. The red arrows indicated episodes of Ca2+ release from ER. (C) The bar graphs represent the normalized changes of YPet/ECFP emission ratio of the D3ER in HMSCs upon force application without extracellular Ca2+ in the untreated cells as the control group (n = 3) or cells pretreated with CytoD (n = 5), Noc (n = 5), ML-7 (n = 6), Bleb (n = 5), or TRPM7 siRNA (n = 9) as indicated. * represents p < 0.05.
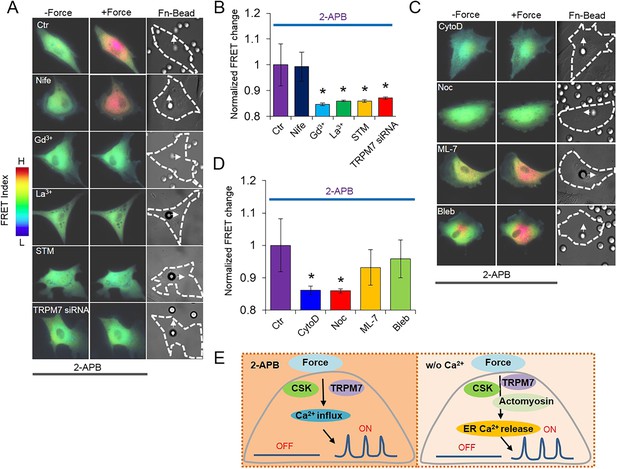
Ca2+ influx in response to mechanical force in Ca2+-containing medium. Ca2+ release from ER in all the HMSCs was blocked by pretreatment with 2-APB.
(A, C) Color images represent the YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor in control cells treated by 2-APB only (n = 5) or those co-treated by Nifedipine (n = 5), Gd3+ (n = 3), La3+ (n = 6), STM (n = 8), or TRPM7 siRNA (n = 9), CytoD (n = 8), Noc (n = 4), ML-7 (n = 9) or blebbistatin (n = 6). Arrows in DIC images point to the direction of applied force. (B, D) Bar graphs represent the normalized change of YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor under different conditions as indicated in (A, C). Error bars indicate standard errors of mean; * represents p < 0.05. (E) The models depicting the mediators of mechanical force-induced Ca2+ influx or ER Ca2+ release.
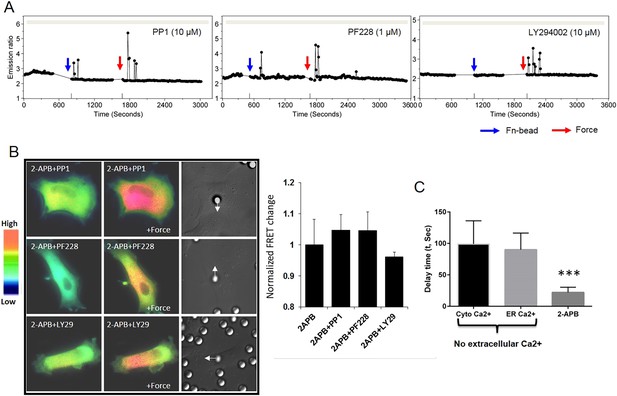
Src, FAK or PI3K has no effect on the mechanical force-induced Ca2+ signals.
(A) The inhibition of neither Src by PP1 (n = 4), FAK by PF228 (n = 4), nor PI3K by LY294002 (n = 3) abolished the force-induced cytosolic Ca2+ oscillations in HMSCs without extracellular Ca2+. (B) The Ca2+ release from ER in all the HMSCs was blocked by pretreatment with 2-APB. Color images represent the YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor in cells pretreated by 2-APB together with PP1 (n = 5), PF228 (n = 4), or LY294002 (n = 6). Arrows in DIC images point to the direction of applied force. Bar graphs represent the normalized change of YPet/ECFP emission ratio of the cytoplasmic Ca2+ biosensor under different conditions as indicated in (A). (C) The average delay time between force application and the first Ca2+ signals in three groups. First two groups were ER Ca2+ release monitored by either cytoplasmic or ER Ca2+ biosensor. The last group was Ca2+ influx in the presence of 2-APB monitored by cytoplasmic Ca2+ biosensor. (***, p < 0.001) Error bars indicate standard errors of mean.





