Actin foci facilitate activation of the phospholipase C-γ in primary T lymphocytes via the WASP pathway
Figures
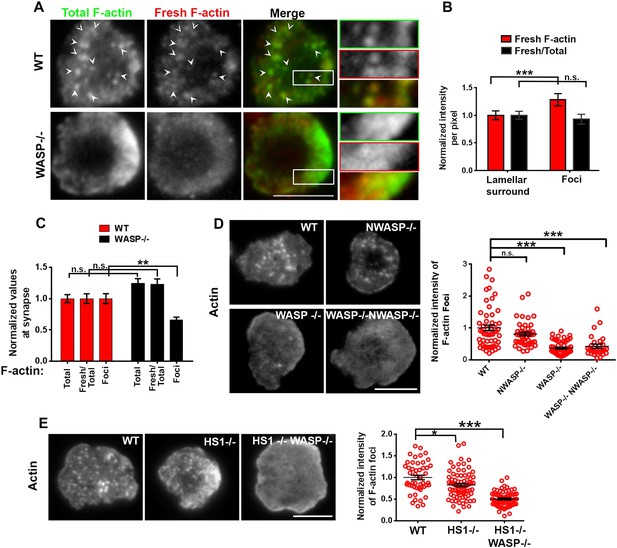
WASP dependent dynamic F-actin foci at the T cell synapse.
(A) Barbed-end decoration of freshly polymerized actin filaments reveals the dynamic behavior in WT (top panel) and WASP−/− (bottom panel) T cell synapses. CD4 T cells isolated from WT and Was−/− C57BL/6J mice were processed for barbed end labeling (fresh actin) to identify the actin incorporation sites within 1 min of polymerization, as well as total F-actin labeling, as described in ‘Materials and methods’. Arrowheads indicate the sites of F-actin foci in both ‘Total’ as well as ‘Fresh’ F-actin images. (B) The graph shows average incorporation of Rhodamine actin per pixel within the foci or the surrounding lamellar pixels (Fresh F-actin); and a ratio of Rhodamine and Alexa488 actin intensities in foci or surrounding pixels, in the WT cells. The foci areas in the total F-actin image were identified and outlined by intensity rank-based filtering as described in ‘Materials and methods’. The synaptic area outside the foci was defined as lamellar surround. These outlined areas were then analyzed in both ‘fresh F-actin’, and ‘total F-actin’ raw images and per pixel intensity is plotted in the graph. Note that while the rate of polymerization (Fresh/Total) ratio is not altered, there is higher incorporation of actin at foci sites (Fresh). n = 20 cells, p1 < 0.0001, p2 = 0.181 (Wilcoxon nonparametric pairwise comparison). (C) The graph shows total F-actin intensity, fresh F-actin intensity, or the ratio of the two at the synapse, each point represents value obtained from single cell, n in WT = 33, n in WASP−/− = 35; p1 = 0.06, p2 = 0.059, p3 = 0.0008. Scale bars, 5 μm. (D) WASP is critical for F-actin foci generation. Freshly purified CD4 T cells from WT 129 (top left), Was−/− 129 (bottom left), Wasl−/− (top right), or Was/− Wasl−/− 129 (bottom right) mice were activated on SLB containing ICAM1 and anti-CD3 antibody, fixed and stained with Alexa488-phalloidin. Note that only the lack of WASP, and not N-WASP, causes loss of actin foci. n1 = 54, n2 = 45, n3 = 53, n4 = 28. p1 = 0.30, p2 < 0.0001, p3 < 0.0001. (E) F-actin foci in CD4 T cells freshly purified from wild type (WT) mouse (left) or from Hcls1−/− mouse (center) or Hcls1−/− Was−/− (right) C57BL/6J mice were incubated with bilayer containing anti-CD3 antibody and ICAM1 for 2 min. Cells were then processed for F-actin staining (Alexa488-phalloidin) and visualized. Note that, while there is a minor alteration in F-actin foci in HS1−/− T cells, there is a gross deficit in HS1−/− WASP−/− double knockout T cells. The graph shows the quantification of total intensity of F-actin foci at the synapse in individual T cells derived from the indicated backgrounds. n1 = 48, n2 = 70, n3 = 64, p1 = 0.031, p2 = 0.0001.
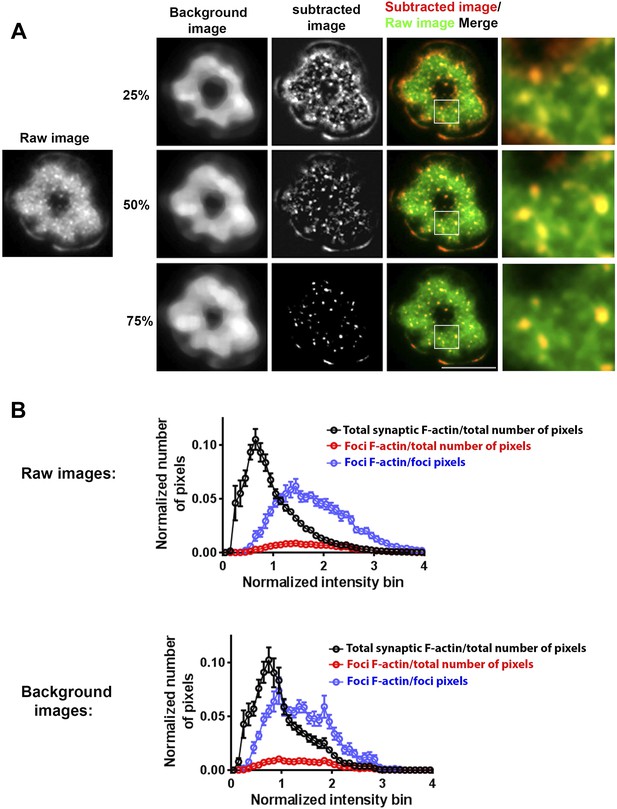
Method of analyzing F-actin foci from the raw F-actin TIRF image.
(A) The raw image (left) was processed for local background subtraction. Briefly, we first used a rank filter selecting for the 25th, 50th (median) or 75th percentile intensity value in a 1.6 × 1.6 µm moving window, to extract the ‘background image’. This ‘background image’ was then subtracted from the raw image to generate a high-pass filtered image where we could clearly visualize F-actin foci. The processed image was manually checked for co-localization of F-actin foci with the F-actin foci in the raw image. As shown in the ‘merge’ panel, the 50% rank filter was found to be most optimal for F-actin foci extraction, while the 25% filter overestimated and 75% filter underestimated the number of F-actin foci. The 50% rank filter corrected image was therefore used to quantify F-actin foci per cell, as indicated in the corresponding figure legends. In some experiments, this processing was also carried out for TCR and ICAM1 image, to estimate co-localization between F-actin foci and TCR MC. (B) Characterization of range of F-actin foci intensities to assess the efficacy of foci extraction method. Intensities of pixels corresponding to foci were quantified in raw images (upper graph) or in images generated using 50% intensity rank filtering (‘Background images’, lower graph). CD4 T cells were incubated with bilayer containing MHCp and ICAM1 for 2 min, fixed, and stained with Alexa488-phalloidin (F-actin). TIRF images of the T cell synapse were processed to extract F-actin foci as described above in (A). These foci were used to create a binary mask to outline the foci or total synaptic cell regions in either original raw images (upper graph) or background images (lower graph). Each graph shows the intensity distribution of individual pixels corresponding to foci or total synapse area, and the X-axis represents the binned pixel intensity, normalized to the mean synaptic F-actin intensity from each cell's raw image. The Y-axis represents the number of pixels in the foci or synapse area, normalized to either the total number of pixels in the synapse (‘/total number of pixels’ traces), or to only the total number of pixels in the foci area (‘/foci pixels’ trace). The data is represented as the mean number of pixels from 13 cells ±SEM. It is notable that the number of pixels encompassing foci regions is <10% of total number of pixels (top left graph). Also note that in both raw and background images, the pixels in foci do not exclude the high-intensity range, suggesting that our foci identification method does not preclude foci in high intensity neighborhoods, and identifies higher intensity patches even in high background neighborhood.
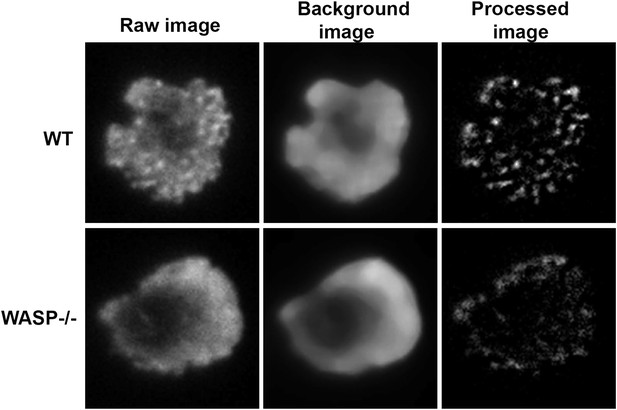
Test of rank-filter based processing method for foci detection.
T cells purified from WT or Was−/− mice were incubated with anti-CD3/ICAM1 reconstituted lipid bilayers for 2 min and then stained with Alexa488-phalloidin. WT and WASP−/− cells have comparable levels of total F-actin at synapse (raw images in left panels), as shown in Figure 1. However, WASP−/− cells visibly lack F-actin foci. When processed using local background correction method, the processed images indeed show quantifiably different numbers of foci.
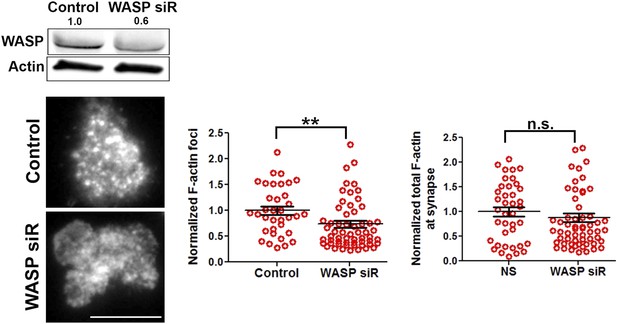
WASP silencing causes a reduction in F-actin foci.
Mouse CD4 T cell blasts were electroporated with WASP siRNAs, or with non-targeting sequences (control). The extent of depletion of protein levels was estimated using western blots. ∼40% of protein depletion was achieved using WASP siRNAs (relative densitometry value mentioned on top of gel lanes). Cells electroporated with WASP siRNA exhibited significantly fewer F-actin foci. AND T cell blasts electroporated with siRNA sequences, as described above, were incubated with bilayer containing anti-CD3 and ICAM1 for 2 min, followed with fixation and staining with Alexa488-phalloidin (left panels), and imaged using TIRF microscopy. The graphs show quantitation of total F-actin (right graph) or F-actin foci (left graph) per cell, in control or WASP siRNA-treated cells. n1 = 35, n2 = 60, p = 0.238 (right), p = 0.0025 (left graph).
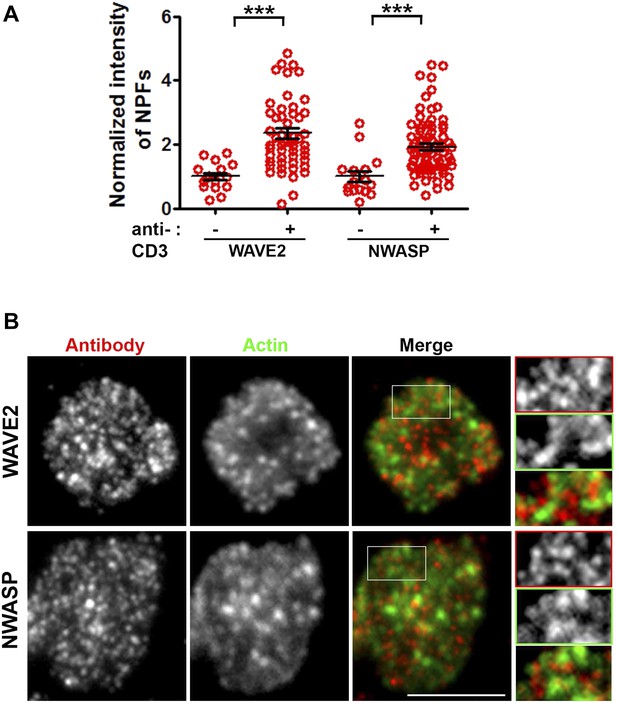
WASP family members NWASP and WAVE2 are not associated with F-actin foci.
(A) TCR-induced recruitment of NWASP and WAVE2 to IS. Mouse primary WT CD4 T cells were incubated with bilayer containing ICAM1 alone (−) or both ICAM1 and anti-CD3 (+) for 2 min at 37°C, fixed and immunostained for endogenous proteins. Stained cells were visualized using TIRF microscopy. The graph shows quantitation of antibody fluorescence at IS, where each point represents the value obtained from a single cell. n1 = 16, n2 = 54 (for WAVE2), n3 = 16, n4 = 78 (for NWASP); p1, p2 < 0.0001. Each point represents average levels of indicated protein at synapse in a single cell. (B) The images shown are TIRF plane distributions of the indicated proteins. As elaborated in the magnified areas marked with white boundary in original ‘merge’ image, there is a lack of co-localization between either of these proteins and TCR MCs. Scale bar, 5 μm. Insets in (B) have been intensity scaled differently from original ‘Merge’ panel to highlight protein distribution with more clarity.
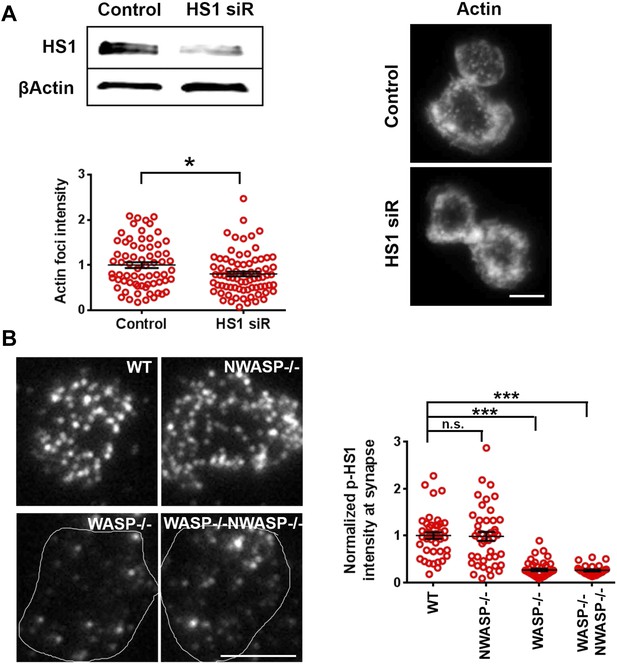
WASP dependent HS1 recruitment and F-actin foci.
(A) HS1 silencing and F-actin foci. AND CD4 T cell blasts were electroporated with non-specific (control siRNA oligos) or oligos targeting HS1. The depletion of protein was assessed using western blotting (top left, gel). Cells electroporated with control or HS1 siRNA oligos were incubated with bilayers for 2 min at 37°C, fixed, and stained with Alexa488-phalloidin, and then imaged using TIRF illumination. The images (right) show F-actin distribution in their respective backgrounds. The graph shows quantification of F-actin foci in control or HS1 depleted cells. n1 = 70, n2 = 100, p = 0.0268. (B) WASP determines phospho-HS1 levels at the synapse. CD4 T cells purified from WT-129 mouse, Was−/−, or Wasl−/− 129 mice were incubated with bilayer containing ICAM1 and anti-CD3 for 2 min, and were fixed and stained with anti-phospho-HS1 antibody. The TIRF images were quantified for phospho-HS1 levels. Each dot represents average phospho-HS1 intensity per cell. n1 = 42, n2 = 43, n3 = 46, n4 = 27. p1 = 0.637, p2, p3 < 0.0001.
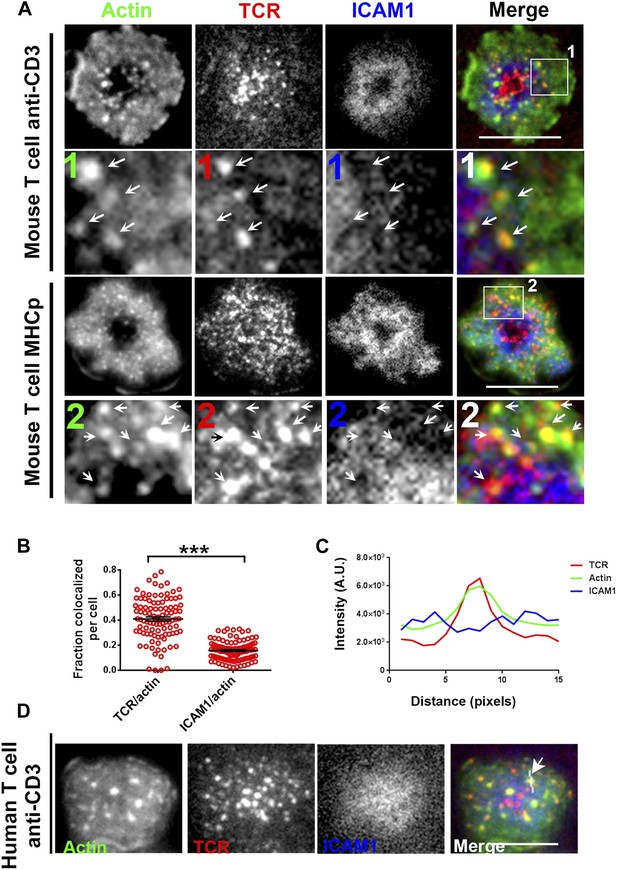
F-actin foci co-localize with TCR MC and not ICAM1.
(A) Freshly isolated mouse AND CD4 T cells were incubated with lipid bilayer reconstituted with Alexa568 tagged anti-CD3 (TCR, red) and Alexa647-ICAM1 (blue), for 2 min at 37°C. Post incubation, cells were fixed and stained for F-actin using Alexa488-phalloidin (green), and imaged using TIRF microscopy. The region marked 1 in the ‘merge’ panel is magnified to clearly show the co-localization of actin foci with TCR-containing MCs. Lower panels: AND mouse CD4 T cell blasts exhibit F-actin enrichment at TCR MCs sites. AND mouse CD4 T cell blasts were labeled with Alexa568-H57 Fab (TCR), and incubated with bilayer reconstituted with MHCp and Alexa405-ICAM1 for 2 min at 37°C, fixed and stained for F-actin. Region marked 2 in the ‘merge’ image is further magnified to show the overlap between TCR and F-actin (arrows). The insets 1 and 2 in both MHC-activated and anti-CD3-activated mouse T cells are contrasted differently from the original ‘merge’ image to highlight the TCR and actin distribution. (B) Quantitation of the fraction of TCR or ICAM1 localized with F-actin foci. AND CD4 T blasts were incubated with antigen containing bilayer for 2 min, as described above and the images acquired were processed for colocalization assessment as described in ‘Materials and methods’ section. Each point represents fraction of total synaptic TCR or ICAM1 associated with F-actin foci in a single cell. n1 = 99, n2 = 100. p < 0.0001. (C, D) Freshly isolated human peripheral blood CD4 T cells were incubated with bilayer containing Alexa568 tagged anti-CD3 (TCR), Cy5-ICAM1 at 37°C, fixed and stained with Alexa-488 phalloidin and subsequently imaged using TIRF microscopy. The image shows a freshly attached cell to the bilayer. The line marked by the arrow in the ‘merge’ panel shows the line-scan profile plotted in (C), where relative intensities of ICAM1 and actin with single TCR MC across the pixels marked in the ‘merge’ image are shown. Scale bar, 5 μm.
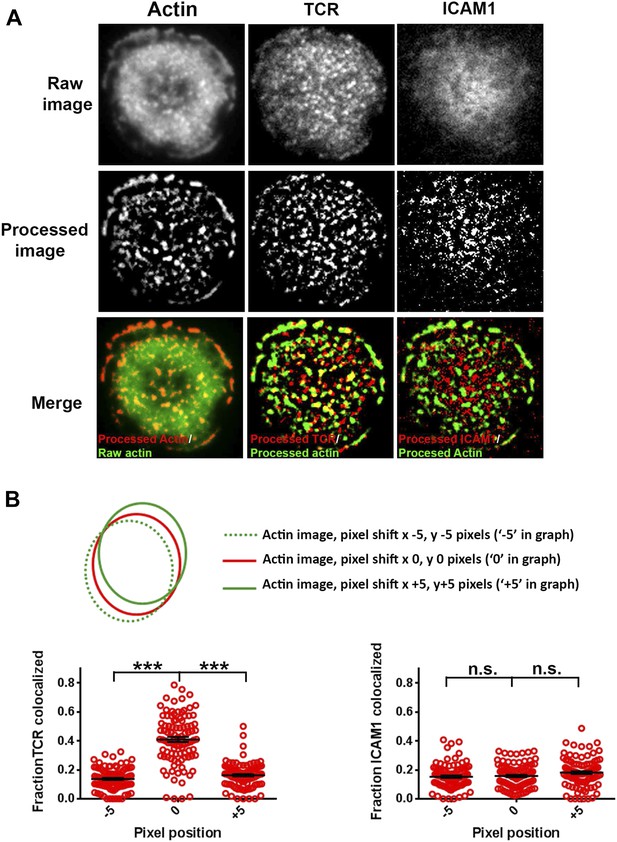
Test for co-localization of foci and MCs.
(A) An example of image analysis carried out to assess co-localization between F-actin foci and TCR/ICAM1, using 50% rank-filter based processing, as described in Figure 1–figure supplement 1A (B) TCR MC and F-actin foci co-localization is significantly higher than chance overlap. To measure chance level co-localization, the actin images from Figure 1 were shifted by 5 pixels in x and y dimensions (schematic on top), and analyzed for co-localization with respect to the unshifted TCR (left graph) or ICAM1 (right graph) images. Note that while pixel shift results in significant reduction TCR-F-actin foci co-localization (left graph, p < 0.0001), levels of ICAM1/F-actin foci association are unaffected (right graph, p = 0.53).
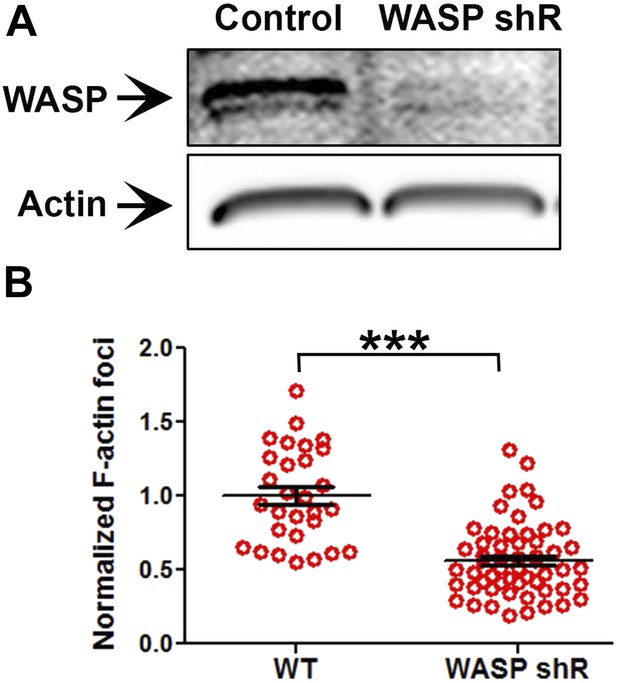
Loss of WASP in Human CD4 T cells, and its impact on foci induction.
(A) Human CD4 T cells were incubated with culture media containing lentiviral particles carrying WASP shRNA or non-specific (control) shRNA for 48 hr, and were lysed and assess for WASP depletion using western blotting (‘Materials and methods’). (B) Human CD4 T cells were incubated with lentiviral particles carrying WASP shRNA or mock shRNA (Control) for 60 hr, and were further activated using anti-CD3 and ICAM1 containing surface. Cells were then fixed, stained with Alexa488-phalloidin, imaged using TIRFM and analyzed for foci induction. Note that transduction with WASP shRNA leads to significant reduction in foci at synapse. The graph represents foci intensity per cell, normalized to mean intensity in Control T cells, n1 = 29, n2 = 56, p < 0.0001.
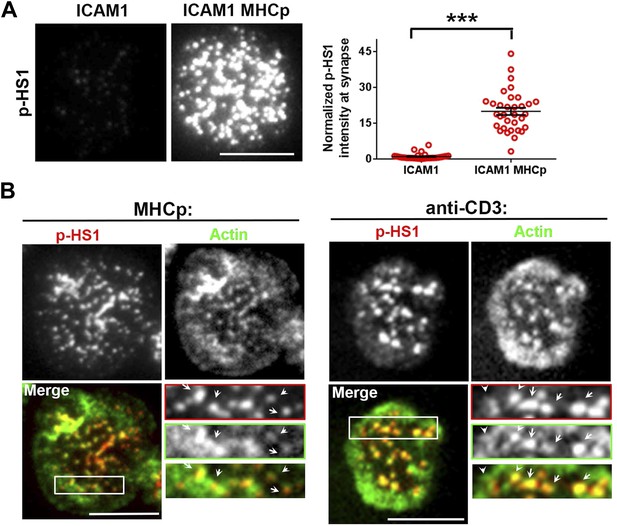
(A) Increased phosphorylation of HS1 in response to TCR activation.
AND CD4 T cell blasts were incubated with bilayers reconstituted with ICAM1 alone (left image) or both ICAM1 and MHCp (right image) for 2 min at 37°C, and then fixed and immunostained for phosphorylated HS1. The images show phospho-HS1 (p-HS1) signal in the cells acquired in the TIRF field. The graph (far right) shows quantitation of total p-HS1 intensities, where each point represents integrated intensity per cell in the TIRF field. n1 = 33, n2 = 33, p < 0.0001. (B) Phospho-HS1 localizes to F-actin foci and TCR MCs. Mouse AND CD4 T cell blasts (left panels) or primary CD4 T cells (panels on the right) were incubated with bilayer containing ICAM1 and MHCp (for blasts) or ICAM1 and anti-CD3 (right panels) for 2 min, fixed and stained with phospho-Y397 HS1 antibody (red) and Alexa488-phalloidin (green). The area marked in ‘Merge’ image has been further enlarged in insets; the arrows indicate F-actin foci sites that localize with phospho-HS1.
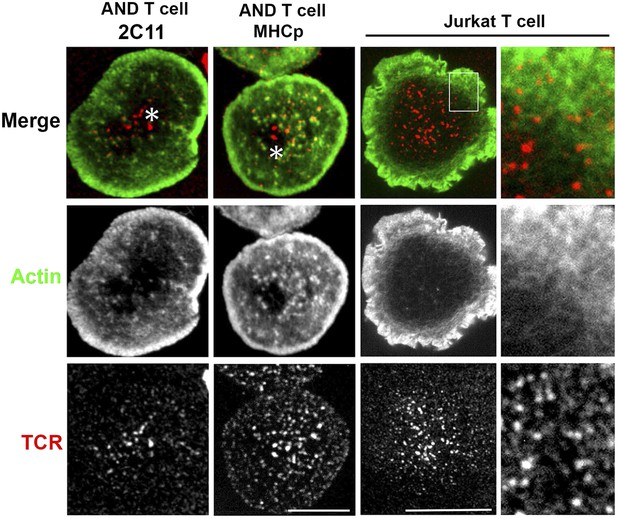
Lack of F-actin foci in the cSMAC of primary T cells and the Jurkat T cell line.
AND CD4 T cell blasts were either incubated with bilayers containing Alexa568 tagged anti-CD3 and ICAM1 (far left panels) or were labeled with Alexa568-H57 Fab and incubated with MHCp/ICAM1 bilayers (center left panels), for 2 min. Cells were fixed and stained for F-actin using Alexa488-phalloidin (middle row, green) and visualized using spinning disc confocal microscopy. Each image is a maximum intensity projection of the bottom three planes that show high intensity signals from the bilayer. Note that TCR MCs (bottom row, red) in the F-actin depleted central zone of the cell (asterisk) exhibit no significant co-localization with actin foci. This phenomenon where central microclusters lack F-actin foci was observed in >90% cells exhibiting well-defined cSMAC (number of experiments >3). F-actin foci were not detected in Jurkat T cells (center right panel, and far right insets). Jurkat T cells were activated on bilayer containing Aexa568 tagged anti-CD3 (TCR, red, bottom row) and ICAM1 for 2 min and were fixed and stained with Alexa488-phalloidin (Actin, green, middle row). As highlighted in insets, visible F-actin enrichment is missing from the TCR MCs in these cells. Scale bars, 5 µm (AND T cell) and 10 µm (Jurkat T cell).
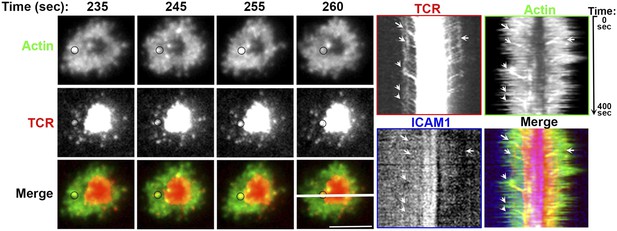
Association of TCR MC and F-actin foci in live T cell.
Live cell imaging of primary human CD4 T cell electroporated with Lifeact-GFP DNA expression construct (Actin, green, top panels) on lipid bilayer reconstituted with Alexa568 tagged anti-CD3 (TCR, pseudocolored in red, middle panels) and ICAM1. The images show snapshots of TIRF images for the indicated time points from the start of observation. The circle on the top left of the t = 235 s image tracks an example TCR MC that exhibits movement along with F-actin foci in subsequent frames, until it finally merges with cSMAC, concomitant with disappearance of actin foci. The images on the right show kymographs obtained from the central section of the cell (marked with white bar in 260 s ‘merge’ panel). The arrows point to the MCs that colocalize with LifeAct and co-migrate until the MC reaches the cSMAC. This association was readily observed in >60% cells monitored (n = 7).
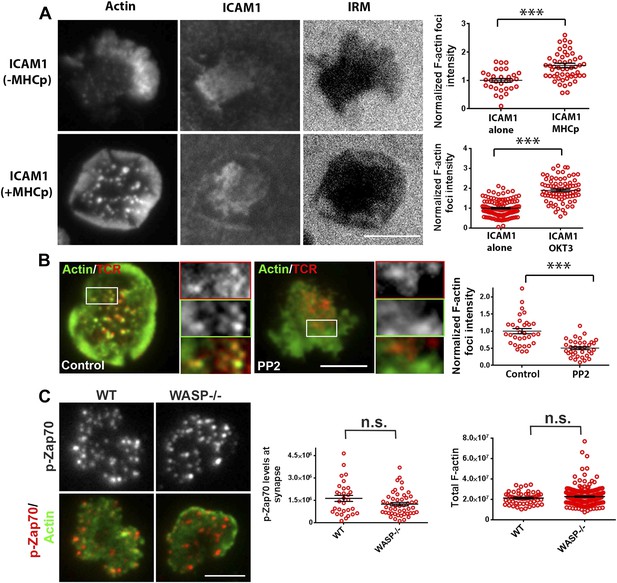
Relationship of F-actin foci to TCR engagement and TCR proximal signaling.
(A) AND mouse CD4 T cell blasts were incubated with lipid bilayer containing Alexa405-ICAM1 and MHCp, or bilayer containing Alexa405-ICAM1 alone (ICAM1) at 37°C for 2 min. The cells were fixed and stained with Alexa488-phalloidin (Actin) and imaged using TIRF microscopy. The images in the right panel are reflection (interference reflection microscopy – IRM) images in the two conditions, showing cell-bilayer contact area. Actin images were further high-pass filtered using a rank-filter based subtraction method (see ‘Materials and methods’) to reveal spatially localized actin features within each cell, under both incubation conditions. The graph (top right) shows average intensity of actin features (F-actin foci) per cell. n1 = 31, n2 = 53, p < 0.0001. The bottom graph shows foci induction in human CD4 T cells. Primary human CD4 T cells were incubated with bilayer containing Alexa405-ICAM1 alone, or both Alexa405-ICAM1 and Alexa568 tagged anti-CD3 at 37°C for 2 min, and were then fixed and stained for F-actin using Alexa488-phalloidin, for imaging using TIRF microscopy. The graph shows integrated intensity of actin spots per cell, each point on the graph represents a single cell. n1 = 117, n = 79, p < 0.0001 (B) Formation of TCR MC associated F-actin foci requires SFK signaling. AND T cell blasts were treated with PP2 for 10 min at 37°C and were labeled with Alexa568-H57 Fab. This was followed by further incubation with ICAM-1/MHCp containing bilayer at 37°C for 2 min, and fixation and staining for F-actin. The TIRF images show Actin (Alexa488-phaloidin, green) and TCR (red) distribution in DMSO treated (control, left) and PP2 treated (right) cells. Graph (far right) shows mean intensity of F-actin features in control and PP2 treated cells. n1 = 32, n2 = 35, p < 0.0001. (C) Phospho-Zap70 levels are normal in WASP deficient cells. Freshly isolated C57BL/6J WT or WASP−/− CD4 T cells were activated on bilayer containing anti-CD3 and ICAM1 for 2 min, fixed and stained with anti-phospho-Zap70 antibody (left graph). The top images show phospho-Zap70 intensity at the synapse, and the left graph shows integrated intensity of phospho-Zap70 per cell. n1 = 30, n2 = 49, p = 0.17. The WASP−/− cells display normal whole cell levels of TCR-induced F-actin, as marked by Alexa488-phalloidin staining (right graph). The cells were imaged under wide-field illumination settings – each point represents total F-actin intensity per cell. n1 = 53, n2 = 212, p = 0.583.
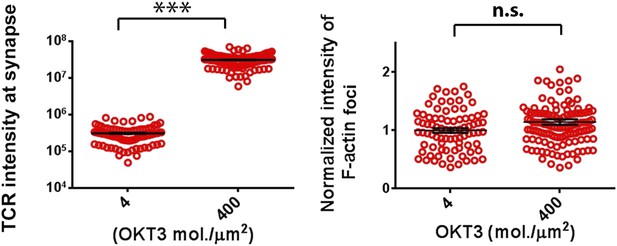
Effect of TCR-ligand concentration on F-actin foci formation.
CD4 human T cells were incubated with bilayer containing indicated concentration of Alexa568 tagged anti-CD3 and ICAM1 for 5 min, fixed and stained with Alexa488-phalloidin, and then imaged using TIRF microscopy. The graph on the left shows total intensities of anti-CD3 gathered by cells at synapse, at indicated anti-CD3 reconstitution densities. n1 = 84 n2 = 112, p < 0.0001. The graph on the right shows total intensity of actin features in the synapse, obtained using the same images used for analysis in the left graph. p = 0.06.
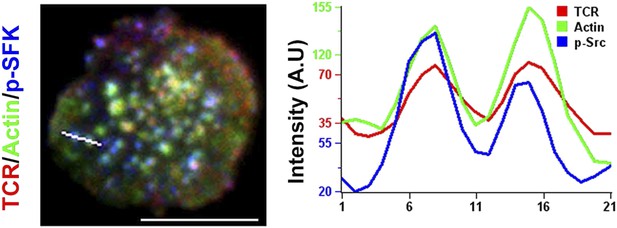
Localization of phosphorylated form of Src family kinases (SFK) at TCR MC/F-actin foci.
AND mouse CD4 T cell blasts were labeled with Alexa568-H57 Fab, incubated with MHCp/ICAM1 containing bilayer for 2 min, fixed and then immunostained for phospho-SFK (p-SFK). An example line scan profile for the line (white) indicated on the left image is displayed (right graph) to show the relative distribution of TCR, actin and phospho-SFK across the row of pixels. Scale bar, 6 μm.
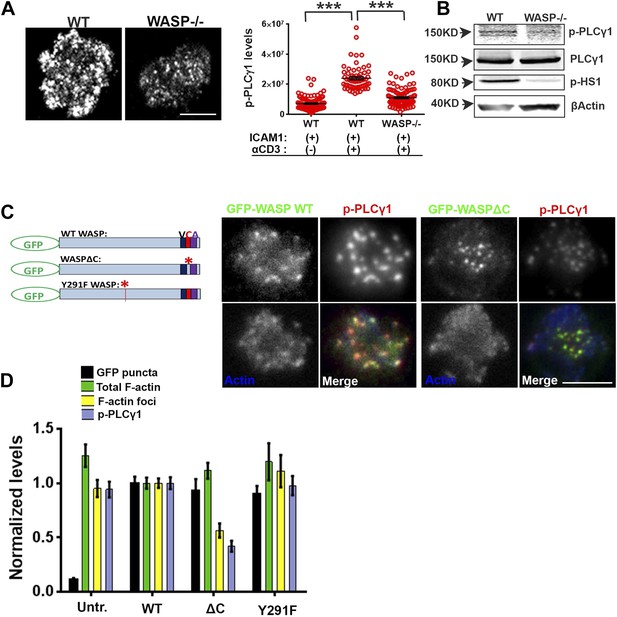
WASP regulates calcium ion signaling via generation of F-actin foci.
(A) WASP deficient T cells exhibit impaired TCR-induced PLCγ1-Y783 phosphorylation. CD4 T cells freshly isolated from C57BL/6J WT or Was−/− mice were activated with ICAM1 alone, or both ICAM1 and anti-CD3 for 2 min. Cells were fixed and immunostained for phospho-PLCγ1 (image not shown), and visualized using confocal microscopy. The images show phospho-PLCγ1 staining in the bottom section (synapse plane) of WT (left) or WASP−/− (right) cells. The graph on the left shows phospho-PLCγ1 levels in the synapse planes in the cells in WASP deficiency background. n1 = 104, n2 = 60, n3 = 94, p < 0.0001. (B) Assessment of total cellular PLCγ1 phosphorylation in WASP−/− T cells using western blot. Freshly isolated WT or WASP−/− CD4 T cells were incubated with anti-CD3/CD28 beads for 5 min, lysed, and the lysates were analyzed using western blotting. Note that, in WASP−/− T cells TCR-induced PLCγ1 phosphorylation is defective, while total PLCγ1 is comparable to the WT cells. HS1 phosphorylation was included as a control that exhibits diminished phosphorylation in WASP−/− T cells. These experiments were repeated twice with similar results. (C) Arp2/3 activation by WASP is essential for F-actin foci generation and optimal phospho-PLCγ1 at the synapse. Human CD4 T cells were transfected with GFP-WASP (WT), GFP-WASPΔC, or GFP-WASP291F (shown in the schematic on the left) for 16 hr and were then incubated with anti-CD3/ICAM1-reconstituted bilayers for 2 min, fixed and stained with Alexa568-phalloidin and anti-phospho-PLCγ1 antibody. The images show the GFP (green), F-actin (blue) and phospho-PLCγ1 (red) distribution at the synapse for WT (left) and WASPΔC (right) T cells. (D) The graph shows levels of GFP-tagged constructs at the synapse, analyzed and obtained via 50% rank filtering of images shown in (D), as described in ‘Materials and methods’, as well as the quantitation of total synaptic F-actin, foci and phospho-PLCγ1 in the same cells, normalized to mean values obtained for WT cells. n1 = 23, n2 = 27, n3 = 33, n4 = 27 p values, p < 0.0001 between WT and WASPΔC cells for foci and phospho-PLCγ1 levels, and between untransfected and cells expressing GFP tagged constructs for ‘GFP puncta’. For all other comparisons, p > 0.05.
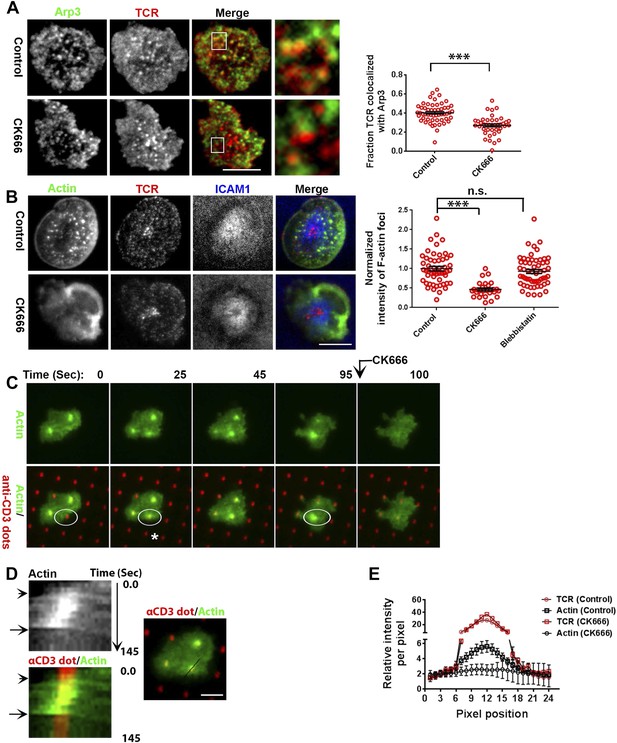
F-actin foci require activity of the Arp2/3 complex.
(A) Arp2/3 complex is localized at TCR MCs. DMSO (Control, top) or CK666 treated-(bottom) AND mouse T cell blasts were labeled with Alexa568-H57 Fab and were incubated with bilayer containing ICAM1 and MHCp for 2 min at 37°C, and then fixed and immunostained with anti-Arp3 antibody. The TIRF images show relative distribution of Arp3 (left, green) and TCR (middle, red) in the TIRF plane. Note that while Arp3 distribution overlaps with MCs in control cells, MCs are significantly devoid of Arp3 in CK666-treated cells (areas from ‘merge’ panels further magnified in insets). The graph (far right) shows the fraction co-localization of total synaptic TCR with Arp3. n1 = 54, n2 = 41, p < 0.0001. In this experiment, the mean intensity of Arp3 in synapse was 3.46 (±0.022) × 106 for control cells, and 2.38 (±0.015) × 106 for CK666 treated cells (data not shown), and it was repeated twice with similar results. (B) TCR MCs associated F-actin enrichment is generated by Arp2/3 complex activation. AND T cell blasts were treated with DMSO alone (control), or 100 μM CK666, or 100 μM blebbistatin for 10 min at 37°C, were labeled with Alexa568-H57 Fab (TCR, red), and incubated with Alexa405-ICAM1 (ICAM1, blue) and MHCp-containing bilayer in the presence of the specific inhibitors at 37°C for 2 min. Cells were subsequently fixed, stained with Alexa488-phalloidin and imaged using TIRF microscopy. Note that there is a substantial reduction in number of actin foci in the cells treated with CK666. The graph (far right) shows the quantitation of F-actin foci intensity in the cells in inhibitor treatment backgrounds. n1 = 52, n2 = 28, n3 = 56. p1 < 0.0001, p2 = 0.36. (C) Immobilized anti-CD3 microdots induce localized actin polymerization that is dependent on Arp2/3 complex. Human CD4 T cells were transfected with LifeAct-GFP plasmid (green), incubated with glass substrate coated with ICAM1 and printed with Alexa647 tagged anti-CD3 microdots (red), and subsequently imaged live. The images represent snapshots taken from a time-lapse sequence, at the indicated time points, ‘0 s’ represents onset of the video sequence acquisition. Note that immediately upon contacting the microdot, cells display localized actin polymerization (marked circle and *). CK666 treatment (arrow after 95 s panel) leads to immediate loss of F-actin from the foci. (D) Kymograph of F-actin events (upper left image) with anti-CD3 dot (bottom left image) during contact formation with the microdot prior to and during CK666 treatment. Arrowhead indicates the time of contact of the cell with anti-CD3 dot, and arrow indicates start of CK666 treatment. The line marked on the color combined image (right) shows the region of the cell used to create kymograph for the indicated time duration. (E) Enrichment of LifeAct (Actin, black) at the anti-CD3 microdot (TCR, red) in control and CK666-treated fixed cells. The graph shows the mean intensity per pixel obtained using the line-scan profile of LifeAct (actin) and anti-CD3 (TCR) across 31 microdots from 15 cells. Intensity profiles across identical lines for each microdot were obtained for both anti-CD3 and LifeAct; these values were normalized to the lowest pixel intensity for each line-profile, and their mean ± SEM values are plotted in the graph.
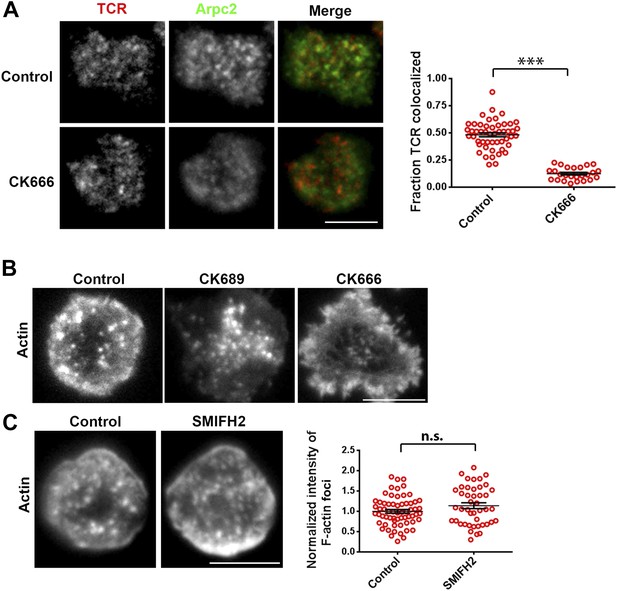
Arp2/3 and formin’s role in F-actin foci.
(A) Arp2/3 complex activation at the TCR MC. AND T cell blasts were labeled with Alexa568-H57 Fab fragments, and were then incubated with bilayer containing MHCp and ICAM1 for 2 min. Cells were then fixed and processed for p34-Arc (Arpc2) immunofluorescence. The images show relative distribution of TCR MCs (red, left) and Arpc2 (green, center), at the T cell interface. The graph (far right) shows the fraction of TCR localized with Arpc2/cell, in control and CK666-treated cells. n1 = 50, n2 = 25, p < 0.0001. This experiment was repeated twice with matching results. (B) Effect of CK666 and its inactive control isomer, CK689, on F-actin foci. AND T cell blasts were treated with DMSO alone (control, left image) or with 100 μM CK689 (center image) or CK666 (right image) for 10 min at 37°C, followed by incubation with ICAM1/MHCp-containing bilayer for 2 min. Cells were fixed and stained for F-actin with Alexa488-phalloidin. (C) Formin inhibition does not eliminate F-actin foci. C57BL/6J CD4 T cells were treated with DMSO (control, left image) or with 10 μM SMIFH2 (right image) for 30 min at 37°C, and were incubated with ICAM1/anti-CD3 bilayer in the presence of the inhibitor for 2 min. Cells were then fixed and stained with Alexa488-phalloidin. F-actin features were quantified in the images (graph on the right). n1 = 63, n2 = 44, p = 0.18. Each dot in the graph on the right represents total F-actin foci intensity per cell. Scale bar is 5 µm for all of the above images.
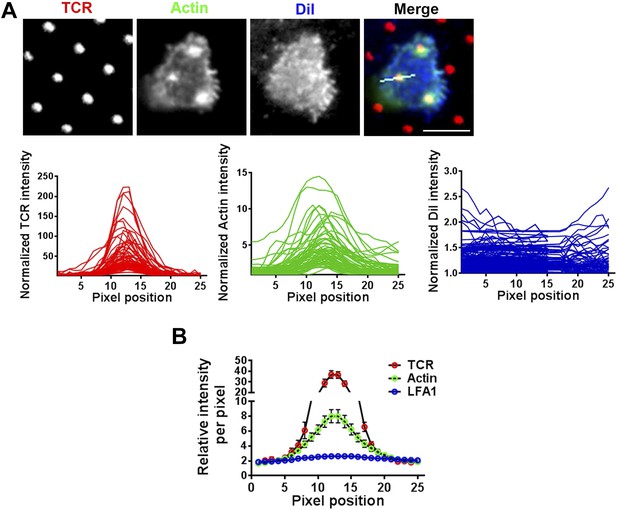
F-actin foci on microdots are not enriched in phospholipid membranes.
(A) F-actin enrichment at the microdot is not a consequence of cell morphology around the microdot. Human CD4 T cells were labeled with CM-DiI according to manufacturer's protocol and were then incubated with glass coverslip coated with patterned Alexa647 tagged anti-CD3 and ICAM1 for 5 min, fixed and stained with Alexa488-phalloidin. Cells were then imaged using TIRF microscopy. The graphs on the bottom panels show line-scan profiles of anti-CD3 (TCR, red, left), actin (Green, center) and DiI (blue, right) acquired from 70 different microdots. The TCR, actin and DiI intensities were measured from identical pixel positions for a given microdot, were then normalized by the lowest pixel intensity per microdot for a given fluorescent channel, and plotted. Note that while actin shows enrichment at the microdot site, DiI is not enriched at the same position. (B) The same procedure as described above, except with the use of Cy3-anti- LFA1 Fab fragments instead of DiI, was carried out in T cells, and the normalized line-scan intensities were plotted as average value per pixel position ±SEM. The graph represents mean of normalized intensities across 25 pixels, acquired using 74 different microdots. In both these experiments (A, B), n > 20.
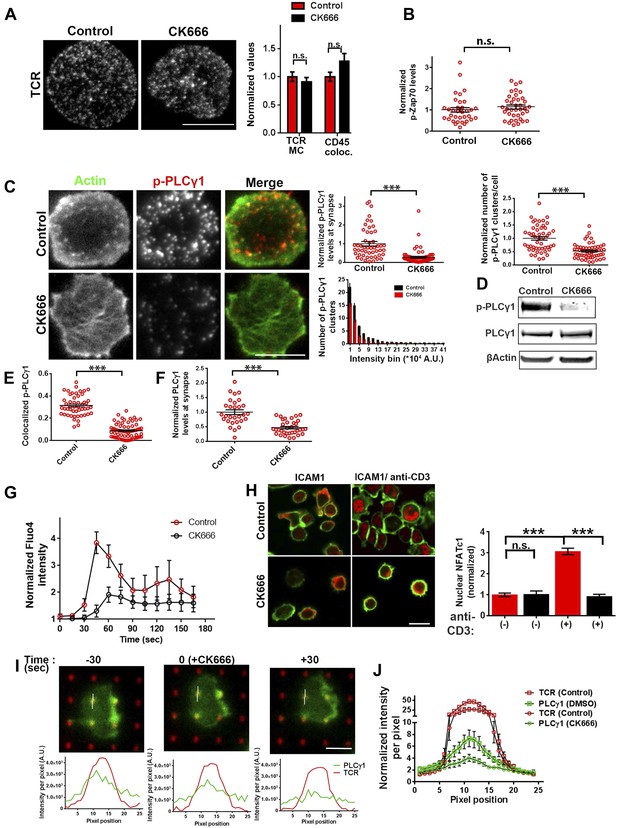
Arp2/3 inhibition leads to defective TCR-distal signaling.
(A) Formation of TCR MCs or CD45 exclusion does not require F-actin foci. AND CD4 T cell blasts labeled with Alexa568-H57 Fab (to assess TCR clustering), or Alexa488-CD45 Fab (for CD45 exclusion) at 4⁰C, were then incubated with bilayers containing ICAM1 and MHCp for 2 min, fixed (to assess TCR clustering) or visualized live (for CD45 exclusion) using TIRF microscopy. The images were processed to assess TCR clustering, or CD45 and TCR colocalization using rank filter based filtering, as described in ‘Materials and methods’ section. For the bars showing TCR cluster intensities per cell, n1 = 45, n2 = 28, p = 0.6; for CD45 co-localization, n1 = 26, n2 = 13, p = 0.09. (B) Phosphorylation of TCR-proximal molecule Zap70 is not reduced in the cells treated with CK666. T cells were treated with DMSO or CK666 for 10 min, then incubated with surface containing ICAM1/anti-CD3 for 3 min, and processed for Y319-phospho-Zap70 and imaged using TIRF. The images were quantified to obtain the synaptic levels phospho-Zap70, and plotted as normalized to mean value of the ‘control’ cells. In the graph shown here, n1 = 34, n2 = 38, p = 0.16. (C) Synaptic phospho-PLCγ1 levels are reduced in cells lacking F-actin foci. DMSO (control, top panel) or CK666 (bottom panel) treated AND CD4 T cell blasts were incubated with bilayer containing anti-CD3 and ICAM1 for 2 min, fixed and stained with Alexa488-phalloidin (green) and phospho-Y783-PLCγ1 (red), and visualized using TIRF microscopy. In these images, total synaptic levels of phospho-PLCγ1 were assessed (top left, graph). These images were also analyzed using integrated morphometry and total number of phospho-PLCγ1 events per cell (c, top right, graph) or intensity distribution of the events across the population of T cells (bottom right, histogram) were measured. Note that CK666 treatment leads to a uniform reduction in phospho-PLCγ1 events across different intensity ranges (c, bottom histogram). In all graphs in c, n1 = 50, n2 = 67, p < 0.0001 (D) Total cellular levels of phospho-PLCγ1 and PLCγ1 with or without treatment of cells with CK666. CD4 T cell blasts were treated with 100 μM CK666 or DMSO (control), and then incubated with anti-CD3/CD28 beads for 5 min in the presence of the inhibitor, lysed and analyzed using western blotting. (E) In CK666-treated cells, there is a substantial reduction in synaptic phospho-PLCγ1 co-localized with F-actin foci. Colocalization analysis was performed on images from (C), to estimate phospho-PLCγ1 and F-actin colocalization, as described in ‘Materials and methods’. (F) Pan- PLCγ1 levels at the synapse are reduced in CK666-treated cells. T cells were processed for PLCγ1 immunofluorescence and imaged as described above. In the graph, each dot represents PLCγ1 intensity per cell in the TIRF images. n1 = 29, n2 = 34, p < 0.0001. (G) Defective calcium ion mobilization in CK666 treated cells. AND CD4 T cell blasts were loaded with Fluo4, and treated with DMSO or 100 µM CK666 for 1 min, and then imaged live on anti-CD3 and ICAM1, to monitor calcium ion flux (‘Materials and methods’). Each point on the graph (far right) represents mean value of the baseline corrected fluorescence ±SEM (n = 30 cells). (H) Nuclear translocation of NFAT1 in cells treated with CK666. DMSO (control, top panels) or CK666-treated (bottom panels) AND CD4 T cell blasts were incubated with glass coverslips coated with ICAM1 alone (left), or ICAM1 and anti-CD3 (right) for 10 min at 37°C, fixed and immunostained for NFAT1 (red), and stained with Alexa488-phalloidin (green). Cells were subsequently imaged using confocal microscopy. The middle sections from the z-stack of the images of the cells were used to quantitate nuclear levels of NFAT1, by outlining phalloidin-free central area (nucleus) of the cell. The graph (right) shows nuclear NFAT intensity in the nuclear area, in a single section per cell. n1 = 98, n2 = 40, n3 = 156, n4 = 31. p-values, p1 = 0.92, p2, p3 < 0.0001. (I) PLCγ1 dynamics at TCR microdots. Human CD4 T cells transiently expressing PLCγ1-YFP (PLC, green) were incubated with glass coverslips patterned with Alexa647 tagged anti-CD3 microdots (TCR, red) and coated with ICAM1 and imaged live using TIRF microscopy. The graphs on the bottom show linescan intensity profiles of PLCγ1 and anti-CD3 (TCR) across the pixels marked in the corresponding image in the top panels. The middle panel shows the intensity profiles during the addition of CK666, and the right panel shows the profiles 30 s after the addition of CK666. The graph in (J) shows anti-CD3 (TCR) and PLCγ1-YFP profiles obtained from at least 30 different microdots from >12 cells in each control and CK666 treatment background. PLCγ1-YFP expressing human T cells were incubated with patterned substrate for (1 + 4) min in the presence of DMSO (Control) or CK666, fixed and imaged using TIRF microscope. The CK666 was added after 1 min of incubation of cells with the substrate. The graph shows mean value of pixel intensities calculated from the linescan profiles. The pixel intensities within a linescan were normalized to lowest pixel intensity within that linescan prior to calculating mean value across different linescans. For a given treatment background and linescan profile, TCR and PLCγ1 intensities were obtained from the identical pixel positions. Scale bar, 5 µm.
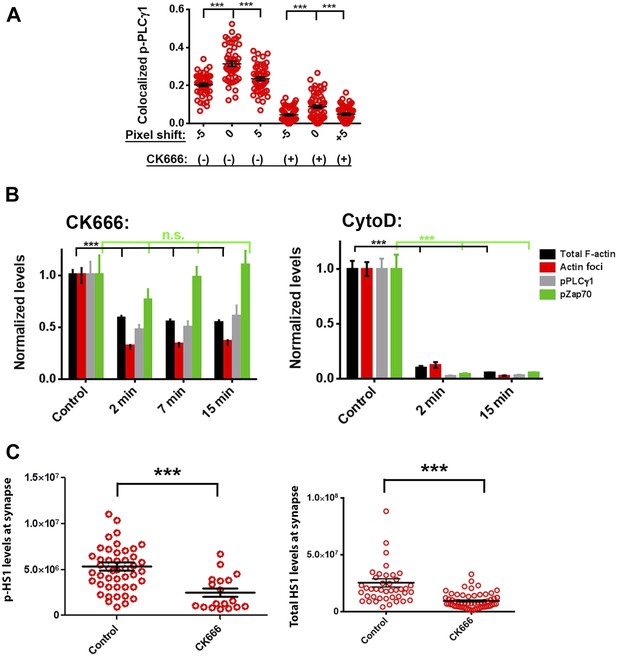
CK666 treatment leads to loss of foci-associated PLCγ1 and phospho-HS1 from the synapse.
(A) Images from Figure 4E were analyzed for phospho-PLCγ1 and F-actin foci chance level co-localization, after pixel shifts of phospho-PLCγ1 images by 5 pixels, as performed earlier, in Figure 3—figure supplement 1. Note that both in DMSO (control) as well as CK666 treated cells, a shift of 5 pixels (x and y) in phospho-PLCγ1 image results in significant reduction in co-localization, indicating that loss of overlap in CK666 treatment is not just due to reduction in total phospho-PLCγ1 levels. (B) Characterization of the effect of CK666 vs Cytochalasin D (CytoD) on total synaptic F-actin, F-actin foci, phospho-Zap70 and phospho-PLCγ1 levels. Mouse CD4 T cells were treated with 100 µM CK666 or 5 µM CytoD for indicated time duration, were activated on anti-CD3 and ICAM1 containing surface for 2 min in the presence of inhibitors, fixed and processed for immunofluorescence of the indicated molecules. Note that, while both CK666 as well as CytoD treatments lead to loss of F-actin foci, CytoD treatment causes a greater reduction in total F-actin and phospho-Zap70 levels. For the left graph, for total actin and F-actin foci, n1 = 123, n2 = 118, n3 = 100, n4 = 87; for phospho-Zap70, n1 = 49, n2 = 47, n3 = 53, n4 = 42; for phospho-PLCγ1, n1 = 78, n2 = 67, n3 = 48, n4 = 42. For total phospho-Zap70, p > 0.90, and for the rest of the datasets, p < 0.0001. In the CytoD graph, for total actin and F-actin foci, n1 = 128, 67, 88; for phospho-Zap70, n1 = 80, n2 = 27, n3 = 57; for phospho-PLCγ1 n1 = 52, n2 = 54, n3 = 31. For all of the data across treatment durations, p < 0.0001. (C) CK666-treated or untreated AND T cells were incubated with anti-CD3/ICAM1 containing bilayer, and stained with phospho-HS1 antibody (left graph) or pan-HS1 antibody (right graph). Cells were then imaged, and images were quantified for total synaptic phospho-HS1 (left graph) or total synaptic HS1 (right graph) intensities per cell. Each point on the graph represents integrated intensity of the indicated protein per cell. In the left graph, n1 = 47, n2 = 18, p < 0.0001; for the graph on the right, n1 = 42, n2 = 59, p < 0.0001.
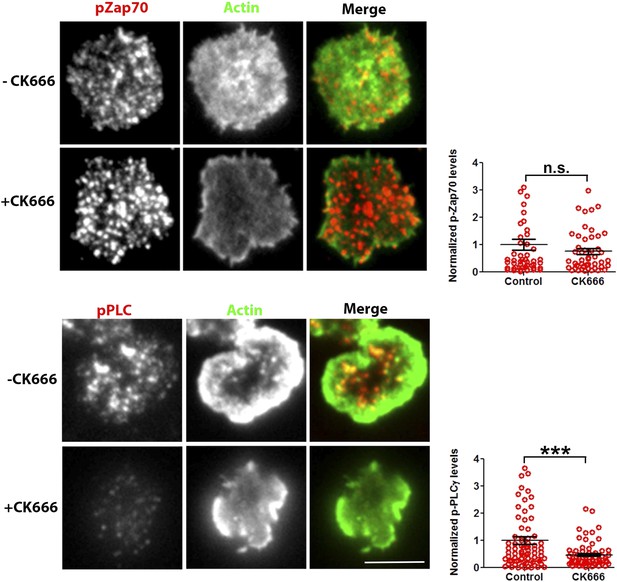
Effect of CK666 on TCR-induced Zap70 and PLCγ phosphorylation in human T cells.
Freshly purified human CD4 T cells were treated with DMSO (control) or CK666, and incubated with bilayer reconstituted with anti-CD3 and ICAM1 for 2 min, in presence of the inhibitor. Cells were then fixed and stained with Alexa488 phalloidin (Actin, green) and either of anti-phospho Zap70 (upper panels) or anti-phospho-PLCγ1 (Lower panels) antibodies, and subsequently imaged using TIRFM. The graph represents phospho-Zap70 (upper graph) or phospho-PLCγ1 levels at synapse, normalized to the mean value in control cells. For upper graph, n1 = 49 n2 = 47, p = 0.976; for the lower graph, n1 = 78, n2 = 67, p = 0.004.
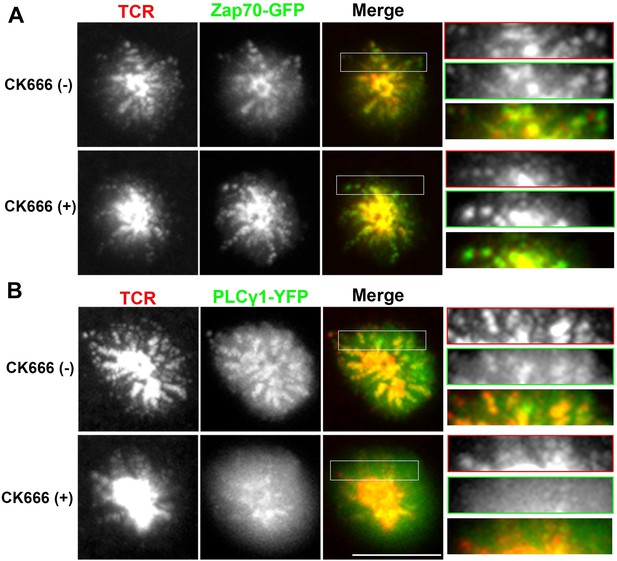
Arp2/3 complex inhibition and synaptic dynamics of Zap70 and PLCγ1 in live cells.
Human CD4 T cells were transfected with Zap70-GFP (A) or PLCγ1-GFP (B) for 16 hr, were then incubated with bilayers containing ICAM1 and Alexa568-OKT3 and imaged live at a rate of 1 frame per 5 s. The images represent maximum intensity projection of five frames (25 s of imaging) immediately before (-CK666) or after (+CK666) CK666 treatment onstage. The projections have been scaled to an identical extent before (upper panels) and after (lower panels) CK666 treatment. An area in the ‘Merge’ panel has been further magnified in the insets (rightmost panels). The insets have been scaled differently from the original ‘Merge’ panels (but similarly between + CK666 and–CK666 cases), to visualize local protein distribution in better details. Note that while Zap70 localization to mobile TCR MC is maintained after CK666 treatment, PLCγ1 localization is severely reduced.
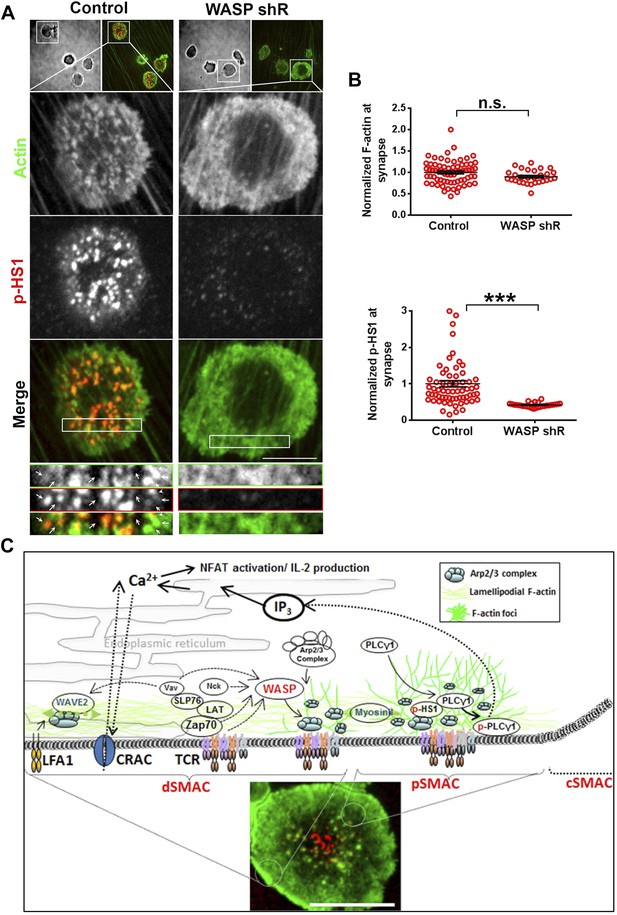
ILP signaling in T-EC immunological synapse is WASP dependent.
Human CD4 T cells were incubated with culture media containing lentiviral particles carrying WASP shRNA or non-specific (control) shRNA for 48 hr (A) T cells transduced with WASP shRNA or control shRNA carrying lentiviral particles were incubated with endothelial monolayer for 10 min, fixed and processed for Alexa594-phalloidin (pseudo-colored green) and phospho-HS1 (pseudo-colored red) immuno-staining. The conjugates were then imaged using an EMCCD-coupled spinning disc confocal microscope. Each image represents a single confocal plane of T cell synapse, where the planar endothelial interface is in focus. The area outlined in ‘merge’ panels was further scaled and magnified to show the details with more clarity (bottom panels). The top panels show the image of the field of view in DIC (left image) or fluorescence settings. (B) A reduction in WASP levels results in defective phospho-HS1 accumulation at T cell-endothelial cell synapse. The upper graph shows quantitation of phalloidin intensity in the synaptic plane, while the lower graph shows phospho-HS1 levels in the same plane. For both the upper and lower graphs, n1 = 68, n2 = 29, p1 = 0.071, p2 < 0.0001. This experiment was repeated twice with similar results. (C) Model of temporal sequence of events leading to F-actin foci formation and PLCγ signaling at the immunological synapse. Multiple pathways can result in actin polymerization and remodeling at the synaptic interface, contributing to F-actin organization in different SMAC zones. One such pathway involves WAVE2 recruitment by activated LFA1, followed by WAVE2 dependent Arp2/3 complex activation resulting in thick lamellipodial (dSMAC) and lamellar (pSMAC) F-actin meshworks. WAVE2-dependent F-actin pool is required for calcium-dependent calcium entry via the CRAC channel. Additional pathways including MyosinII-mediated actin remodeling is required for maintaining lamellar actin flow and directional persistence of microclusters (MCs) towards the cSMAC, and formin-mediated nucleation of F-actin promotes MTOC docking and stability of synapse. Another pool of F-actin or ‘F-actin foci’ is generated by the activity of WASP protein in the p- and dSMAC zones. Following TCR triggering, WASP is recruited at TCR signalosome via several possible mechanisms – such as via Vav, via NCK, via Zap70 and CrkL mediated WIP release and other effector mechanisms, and, through Fyn or PIP2 or PTP-PEST-binding at the plasma membrane (PM). Once activated, WASP recruits Arp2/3 complex to the MC, which then leads to actin branch nucleation and polymerization at the MC, over and above the local background actin. This process continues even during MC movement in the lamellar region, with a high F-actin turnover at the foci until its delivery to the cSMAC. In the foci, HS1 is recruited via binding both the Arp2/3 complex as well as F-actin, and is subsequently phosphorylated. As a consequence of early TCR signaling, PLCγ1 is also recruited to the MC signalosome, where it is stabilized via interactions with both F-actin, and foci residing HS1. F-actin foci dynamics in the proximity of the plasma membrane further support PLCγ1 phosphorylation, potentially by facilitating its interaction with PM-bound, upstream activators such as Itk. Phosphorylation of PLCγ1 by Itk then triggers phosphoinositide signaling, which in turn initiates calcium ion flux and NFAT1 activation. WASP deficiency or failure to activate Arp2/3 complex by WASPΔC mutant leads to selective loss of nucleation of foci at the MC. As a result, early signaling is not affected, however, both HS1 and PLCγ1 levels are severely reduced at the microcluster sites. The remaining PLCγ1 at synapse allows cell spreading and synapse formation, however, it is not sufficient to achieve calcium flux comparable to the control cells. Direct pharmacological inhibition of Arp2/3 complex using CK666 yields similar results; early TCR signaling is preserved while PLCγ1 phosphorylation and late signaling are severely perturbed. As actin polymerizing processes other than WASP also utilize Arp2/3 Complex, CK666-treated cells show a general reduction in lamellipodial and lamellar actin as well. However, the remaining F-actin levels are sufficient to support early TCR signaling. In contrast, total F-actin depolymerization at the synapse using CytochalasinD results in defects in early as well as late signaling, as has been reported in earlier studies. The image on the bottom shows a maximum intensity projection of synaptic contact interface of a human primary CD4 T cell, acquired using spinning disc confocal microscope. This cell was activated on a bilayer reconstituted with Alexa568 tagged anti-CD3 (red) and ICAM1 (unlabeled), for 2 min, fixed and stained for F-actin (green), and imaged.
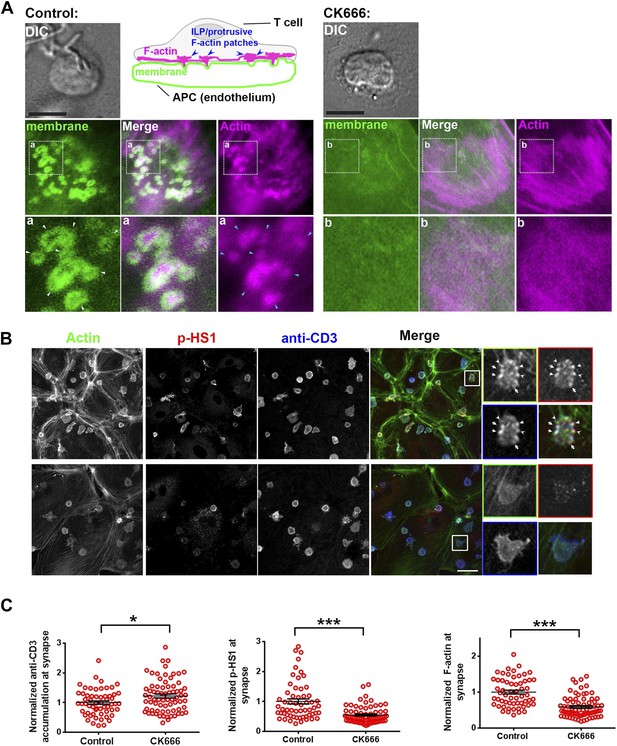
ILP F-actin and signaling in T-EC immunological synapse is dependent on Arp2/3 activity.
(A) Visualization of loss of F-actin in ILPs following CK666 treatment. T cells were incubated with membrane-YFP (green) expressing endothelial cells in the presence of DMSO (control, left) or CK666 (right) for 5 min, then fixed, permeabilized and stained with Alexa594-phalloidin (‘Actin’, pink). Note that F-actin rich protrusions are missing at the CK666-treated T cell interface. Scale bar, 5 µm. (B) Loss of ILPs correlated with reduced phospho-HS1 at the T cell-endothelial cell synaptic interface. T cells were incubated with endothelial cell layer, as described in ‘Materials and methods’ for 5 min and were then fixed and stained with Alexa594-phalloidin (‘Actin’, green), phospho-HS1 (red) and anti-CD3 (blue). Cells were then imaged using confocal microscopy. The insets show single T cells marked in the ‘Merge’ image. Scale bar, 10 µm. (C) The intensity of the indicated proteins was then analyzed in the synaptic plane without and with CK666 treatment. n1 = 58, n2 = 71, p1 = 0.031, p2, p3 < 0.0001.
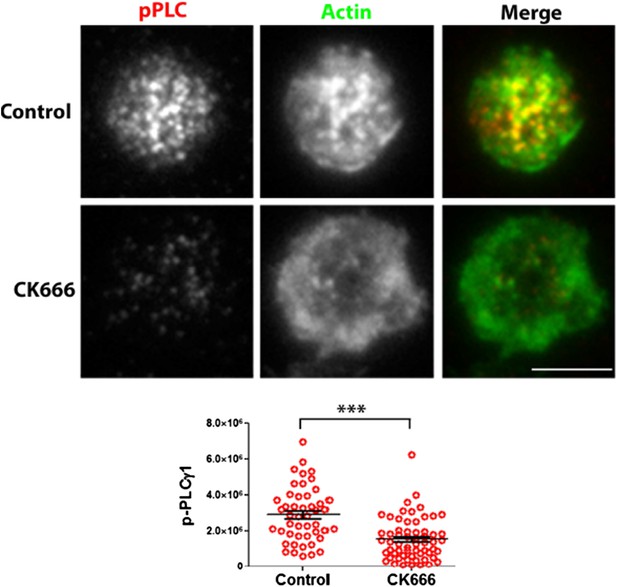
CK666 treatment results in reduced phosphorylation of PLCã1 at the T cell synapse activated on MHCp. AND CD4 T cell blasts treated with DMSO (Control, upper panels) or CK666 (lower panels) were incubated with bilayers containing ICAM1 and MHCp, for 2 min at 37⁰C. Cells were then fixed, immunostained with anti-phospho-PLCã antibody and Alexa488-phalloidin, and visualized using TIRF microscopy. The graph shows phosphoPLCã levels at synapse, in control and CK666 treated cells. n1=47, n2=60, p <0.0001.
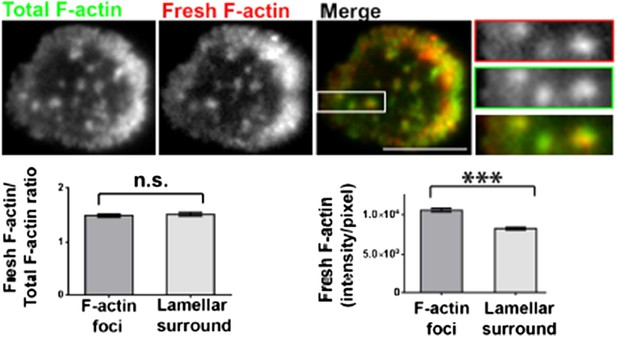
Barbed-end labeling of polymerizing actin filaments in Human CD4 T cells. T cells were incubated with coverslips coated with anti-CD3 and ICAM1 for 5 min, and then processed for barbed end labeling and visualized using TIRF microscopy as described in Methods and Figure 1A. The images show total F-actin (Alexa488-phalloidin, left image) and barbed ends (Rhodamine-phalloidin or ‘fresh F-actin’, middle image) labeled within 1 min of actin polymerization at the synapse. The foci areas in the total F-actin image were identified and outlined by intensity rank-based filtering. These outlined areas were then analyzed in both ‘fresh F-actin’ (barbed end labeled), and ‘total F-actin’ images. The average intensity per pixel within foci regions (‘F-actin foci’), and outside the foci area (lamellipodial surround), was measured and plotted (bottom right graph). The bottom left graph shows the normalization of fresh F-actin (graph on the right) with total F-actin in the same area. n=17 cells, for right graph, p<0.0001, for the graph on the left, p=0.9.
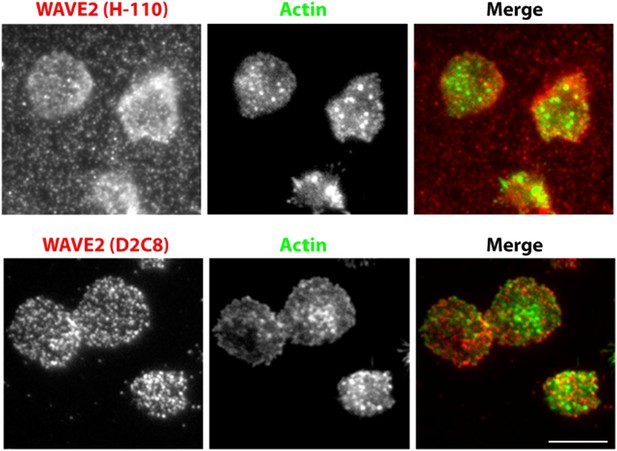
A comparison of endogenous WAVE2 staining, obtained using two different rabbit anti-mouse WAVE2 antibodies, and identical immunostaining procedure as described in Methods. Primary mouse CD4 T cells were activated on anti-CD3 and ICAM1 coated surface for 5min, fixed and processed for indirect immunofluorescence using H-110 clone (upper panels) or D2C8 clone (lower panels) and Alexa647 tagged secondary donkey antirabbit antibody. Cells were also stained with Alexa488-phalloidin. Note that H110 staining results in high fluorescence background in the areas surrounding cells (arrows). This pattern of staining was observed in two independent experiments.
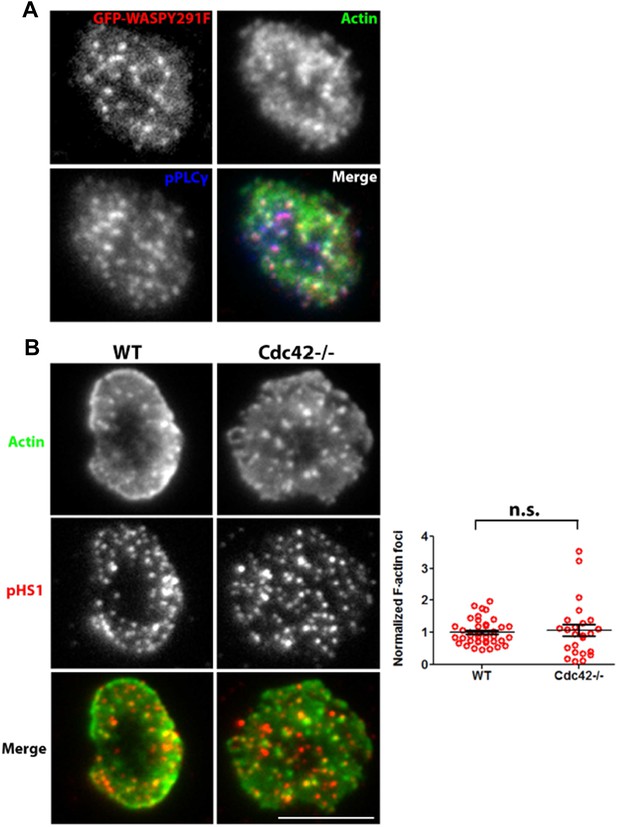
(A) Distribution of overexpressed GFP-WASPY291F in synaptic plane. A representative cell from the results presented in Figure 4d is shown here. Human CD4 T cells were transfected with GFP-WASP291F and utilized to assess the effect of their overexpression on foci formation, as described in Figure 4d. Note that GFP fluorescence (top left panel) is seen in a punctate form that colocalizes with actin Foci/phospho-PLCγ1 distribution. When assessed for overall intensity of GFP puncta at the synapse, Y291F shows comparable extent of recruitment as GFP-WT WASP (GFP-WT, 1.00±0.06, n=27; GFP-WASPY291F, 0.898±0.075, p=0.332). (B) Loss of Cdc42 does not reduce F-actin foci formation. Freshly isolated primary CD4 T cells from wild type (WT, left panels) or Cdc42 -/- mice (right panels) were incubated with 2C11 and ICAM1 containing bilayer for 2 min, fixed and stained with anti-phospho HS1 antibody and Alexa488-phalloidin. Cells were imaged using TIRFM, and images were quantified for foci formation (Graph on right). Note that lack of Cdc42 does not impair foci formation. This experiment was repeated twice with similar results. In the graph, n1=40, n2=24, p=0.632.
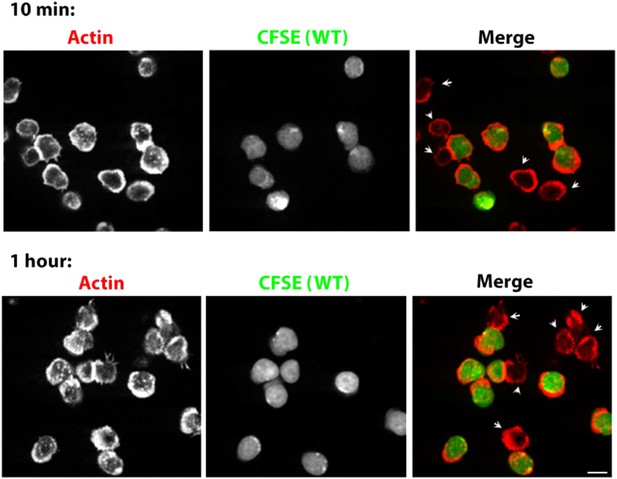
Prolonged Lack of foci in WASP-/- T cells. CD4 T cells from WT mice were isolated and labeled with CFSE (carboxyfluorescein diacetate, succinimidyl ester, 5μM, pseudocolored green). CFSE labeled WT cells were mixed with WASP-/- T cells, incubated with surface coated with 2C11 and ICAM1 for 10 min (upper panels) or 1 hour (bottom panels), fixed and stained with Alexa568-phalloidin (pseudocolored red). The synaptic contacts cells were then imaged using spinning disc confocal microscope. Note that while WT cells (CFSE positive, green) maintain foci at 1 hour post activation, WASP-/- T cells (arrowheads) persistently lack foci. This lack of foci is clearly visible in more than 85% of WASP-/- T cells, both at 10 min and 1 hour (n>48).
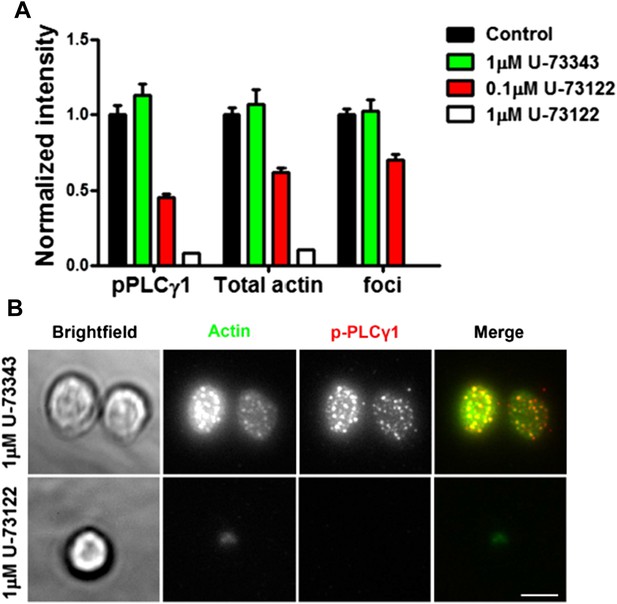
(A) Effect of PLC inhibition on synapse formation and foci induction. Mouse primary CD4 T cells were treated with indicated concentrations of PLC inhibitor, U-73122, or inactive control U-73343, or DMSO alone (Control) for 5 min at room temperature, then with 2C11 and ICAM1 coated surface for additional 5 min in the presence of above-mentioned inhibitors. Cells were then fixed, stained with Alexa-488 phalloidin and anti-phospho PLCγ1 antibody, and imaged using TIRF microscopy. The graph shows synaptic levels of phospho-PLCγ1, total F-actin or foci in different treatment backgrounds, normalized to the values obtained in the case of DMSO control (n in control= 98, in U-73343= 91, in 0.1μM U-73122=113, and in 1μM U-73122= 17). The foci could not be analyzed in cells treated with 1μM U-73122, since synapse spreading was severely diminished along with suppressed F-actin polymerization and reduced synaptic attachment (Author response image 6B).
In all of the above figures, scale bar is 5μm.
Videos
AND CD4 T cells were incubated with bilayer containing ICAM1 and Alexa568 tagged anti-CD3 (red) for 2 min, fixed and stained with Alexa488-phalloidin (green).
Cells were then visualized using spinning disc confocal microscopy. Video shows 3D reconstruction of images acquired.
AND CD4 T cells were labeled with Alexa568-H57 Fab (red), were activated on bilayer containing MHCp and ICAM1 for 2 min, then fixed and stained with Alexa488-phalloidin (green), and eventually imaged using spinning disc confocal microscopy.
Video shows 3D reconstruction of images acquired.
Human CD4 T cells were transfected with Lifeact-GFP (pseudocolored green) and were incubated with bilayer containing ICAM1 and Alexa568 tagged anti-CD3 (pseudocolored red).
The video was acquired with 5 s intervals between frames. The play rate is 80 times faster than the acquisition rate. Scale bar, 5 μm.
Jurkat T cells were transfected with Lifeact-GFP (pseudocolored green) and incubated with bilayer containing ICAM1 and Alexa568 tagged anti-CD3 (pseudocolored red).
The video was acquired with 15 s interval between frames. The play rate is 33 times faster than the acquisition rate. Scale bar, 10 μm.
Human CD4 T cells were transfected with LifeAct-GFP plasmid (green) and incubated with ICAM1-coated glass substrate with 1 µm anti-CD3 printed dots (red) on stage at 37°C and imaged live.
The play rate is 25 times faster than the acquisition rate. In all of the above images and videos, scale bar is 5 µm, unless otherwise noted.
Actin foci associate with TCR MC as soon as synapse forms. Human CD4 T cells were transfected with LifeAct-GFP (green) and incubated with SLB reconstituted with Alexa568-OKT3 (anti-CD3, red), and ICAM1. Cells were imaged live using TIRFM at the rate of 1 time frame per 5 sec. The movie shows sequence of events during the first 200 sec of synapse formation. Left panel shows F-actin dynamics using the raw images, while the panel on the right shows a time lapse from spatial frequency filtered F-actin images (Methods, Figure 1–figure supplement 1). The play rate of the movie is 33 times faster than the original image acquisition frame rate.





