Tissue acidosis induces neuronal necroptosis via ASIC1a channel independent of its ionic conduction
Figures
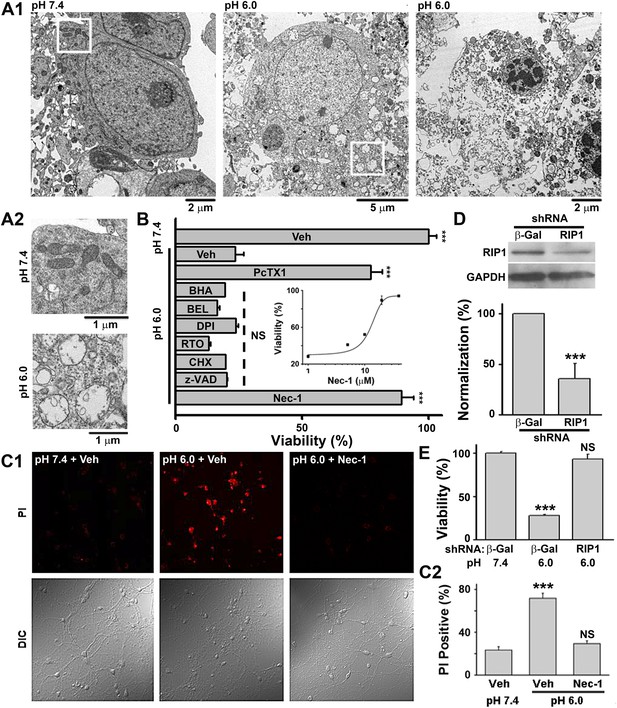
Acid (pH 6.0) induces RIP1-dependent necrotic cell death in cultured mouse cortical neurons.
(A1) Electron microscopy images of neurons treated with pH 7.4 (left) or pH 6.0 solution (middle and right). Of 57 cells counted in the pH 6.0-treated samples, 47 showed morphology similar to that shown in the middle and right panels. For pH 7.4-treated samples, the majority of cells had a similar morphology to that shown in the left panel; only 3 out of the 41 cells examined showed morphology that resembled that in the middle panel. (A2) Enlarged images from the white boxes in A1 showing swelling of organelles in pH 6.0- but not pH 7.4-treated neurons. (B) PcTX1 (10 nM) and Nec-1 (20 μM), but not BHA (100 μM), BEL (30 μM), DPI (15 μM), RTO (25 μM), CHX (100 μM), or z-VAD-fmk (10 μM), rescued cells from acid-induced neuronal death (indicated by the dashed line) (n=4–12, ***p<0.001; NS, no statistical significance, vs vehicle (Veh) at pH 6.0). Inset: dose-dependence of the rescue by Nec-1 (CTB assay, n=3–4). (C1) Rescue from acid-induced neuronal death by 20 μM Nec-1 (propidium iodide [PI] staining assay). (C2) Summary data for C1. At least 200 neurons were counted for each condition (***p<0.001; NS, no statistical significance, vs Veh at pH 7.4). (D) Knockdown efficiency of RIP1 shRNA as determined by Western blotting (***p<0.001, vs β-Gal). (E) Rescue of acid-induced neuronal death by RIP1 shRNA (CTB assay, n=3, ***p<0.001; NS, no statistical significance, vs β-Gal at pH 7.4).
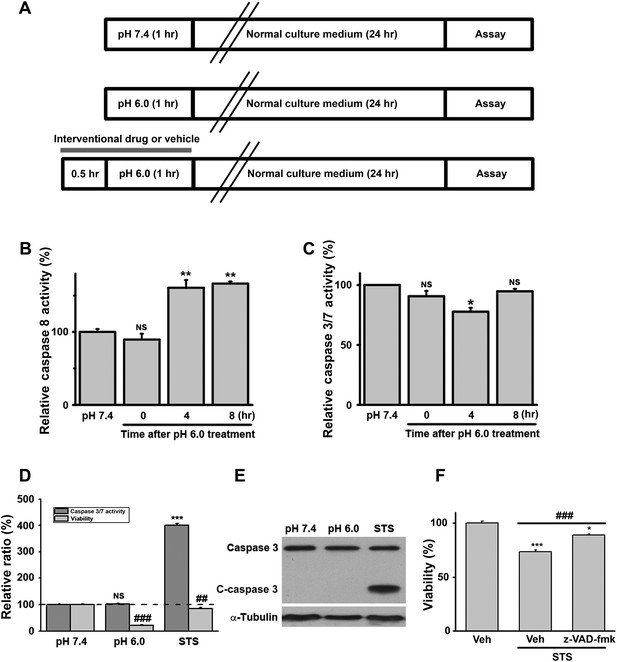
Acid (pH 6.0) treatment does not induce caspase 3/7 activation in cultured mouse cortical neurons.
(A) Scheme of acid treatment and assay protocols. Unless indicated otherwise, all interventional drugs were applied 30 min before and were present during the pH 6.0 treatment. (B) Moderate activation of caspase 8 after pH 6.0 treatment (n=3, **p<0.01; NS, no statistical significance, vs pH 7.4). (C) Caspase 3/7 were not activated after pH 6.0 treatment (n=3, *p<0.05; NS, no statistical significance, vs pH 7.4). (D) Caspase 3/7 activity and neuronal death were tested 8 hr after 1 hr of pH 6.0 treatment from the same batch of neurons. Staurosporine (STS, 100 nM) strongly increased caspase 3/7 activity. However, no increase in caspase 3/7 activity was observed even though severe neuronal death occurred (n=3, for caspase 3/7 activity, ***p<0.001; NS, no statistical significance, vs pH 7.4; for neuronal death, ##p<0.01; ###p<0.001, vs pH 7.4). The dashed line indicates caspase 3/7 activity and neuronal viability at pH 7.4. (E) No cleavage of caspase 3 (C-caspase 3) was detected after treatment with the pH 6.0 solution. Cleavage was seen with the STS treatment. (F) z-VAD-fmk significantly inhibited STS-induced apoptotic neuronal death (n=3, ***p<0.001; NS, no statistical significance, vs vehicle (Veh); ###p<0.001, vs STS+Veh), demonstrating that z-VAD-fmk was effective (control for Figure 1B).
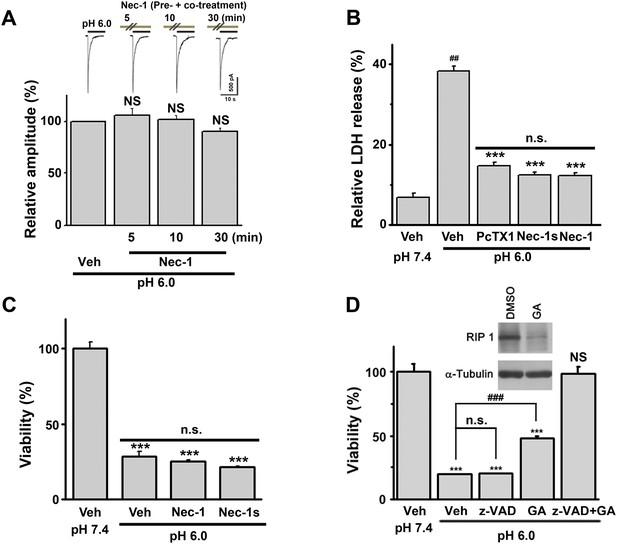
RIP1 mediates ASIC1a-dependent acid-induced neuronal death in cultured mouse cortical neurons.
(A) No inhibitory effect of Nec-1 (40 μM, pre+co-treatment) on I6.0 in cultured mouse cortical neurons (n=5) was observed. Neurons were stimulated with the pH 6.0 solution and then treated with Nec-1 for 5, 10, and 30 min at pH 7.4 before the pH was switched to 6.0 in the presence of Nec-1. (B) Pre+co-application of 7-Cl-O-Nec-1 (Nec-1s, 20 μM), Nec-1 (20 µM), or PcTX1 (10 nM) with the pH 6.0 solution suppressed acidic neuronal death (LDH assay, n=3–5, ##p<0.001, vs vehicle (Veh) at pH 7.4; ***p<0.001, vs Veh at pH 6.0; n.s., no statistical significance, vs PcTX1). (C) No neuroprotection by Nec-1 and Nec-1s when co-administered with the pH 6.0 solution was observed (CTB assay, n=3–5, ***p<0.001, vs Veh at pH 7.4; n.s., no statistical significance, vs Veh at pH 6.0). (D) Geldanamycin (GA) treatment (1 mg/ml, 18 hr) decreased the RIP1 expression level and acid-induced neuronal death. Note: GA also caused cell death by apoptosis, which was abolished by z-VAD-fmk (CTB assay, n=3–5, ***p<0.001; NS, no statistical significance, vs Veh at pH 7.4; ###p<0.001; n.s., no statistical significance, vs Veh at pH 6.0).
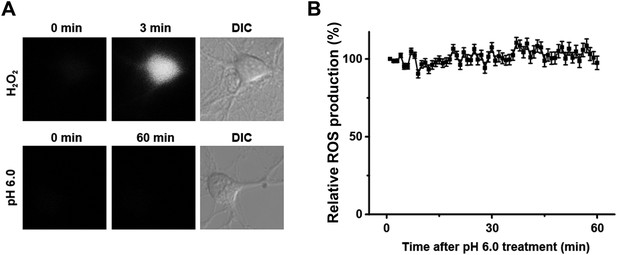
Acid (pH 6.0) treatment does not induce reactive oxygen species (ROS) production in cultured mouse cortical neurons.
(A) H2O2 but not the pH 6.0 solution caused a rapid increase in intracellular ROS (stained with CM-H2DCFDA). (B) No change in the ROS level was observed during 1 hr treatment with the pH 6.0 solution (n=25).
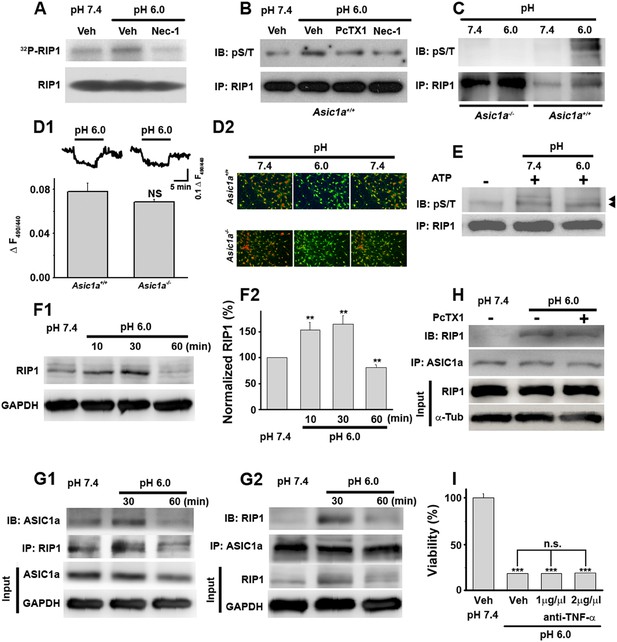
Acid (pH 6.0) induces RIP1 phosphorylation and physical association between RIP1 and ASIC1a.
(A) Acid-induced phosphorylation of RIP1 and its inhibition by 40 μM Nec-1, as measured by 32P incorporation. (B) Acid-induced phosphorylation of RIP1 and its inhibition by Nec-1 (40 μM) and PcTX1 (50 nM), detected using the anti-phospho S/T antibody. (C) Acid failed to induce RIP1 phosphorylation in neurons from Asic1a−/− mice. Five-fold more proteins were loaded for Asic1a−/− samples than for Asic1a+/+ samples. (D1, D2) Time courses (D1) and representative ratio images (D2) of intracellular acidification of cultured mouse cortical neurons from Asic1a+/+ and Asic1a−/− mice in response to extracellular pH decrease from 7.4 to 6.0, monitored using BCECF (n=30 for each genotype, peak changes summarized in D1). (E) In vitro RIP1 phosphorylation assay in pH 7.4 and pH 6.0 reaction solutions. Bands for phosphorylated RIP1 are indicated by the arrowheads. (F1, F2) Acid altered RIP1 expression levels with time. Shown are representative blots (F1) and summary data (F2) (n=5, **p<0.01 vs pH 7.4, by paired t test). (G1, G2) pH 6.0 treatment caused association of RIP1 with ASIC1a (G1, IP (immunoprecipitation) RIP1, IB (immunoblotting) ASIC1a; G2, IP ASIC1a, IB RIP1). (H) PcTX1 disrupted acid-induced ASIC1a–RIP1 association. (I) TNF-α neutralizing antibody (1 μg/μl and 2 μg/μl) failed to rescue pH 6.0 solution-induced neuronal death (CTB assay, n=3, ***p<0.001 vs vehicle (Veh) at pH 7.4; n.s., no statistical significance, vs Veh at pH 6.0).
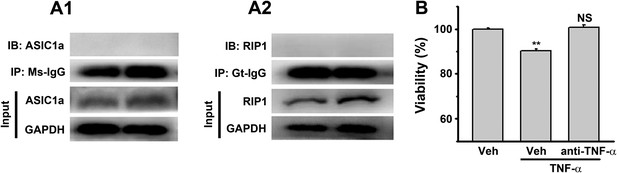
Controls for Figure 2G,H,I.
(A1, A2) No ASIC1a or RIP1 was immunoprecipitated by mouse IgG (Ms-IgG) or goat IgG (Gt-IgG). (B) TNF-α neutralizing antibody (1 μg/μl) rescued TNF-α (10 ng/ml, added in culture medium)-induced neuronal death (CTB assay, n=3, **p<0.01, vs vehicle (Veh) without anti-TNF-α).
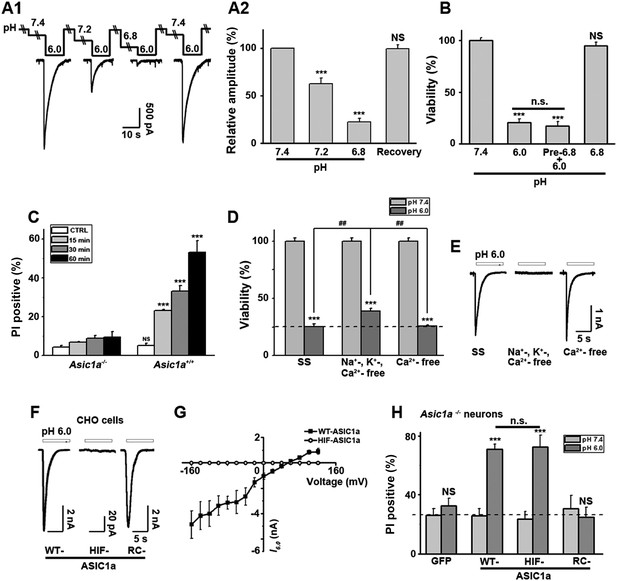
Ion-conducting function is not necessary for ASIC1a-mediated neuronal death.
(A1, A2) Two-minute pretreatment with mild acidification greatly suppressed I6.0 in cultured mouse cortical neurons. Shown are representative current traces at −60 mV (A1) and summary data for peak currents (A2) (n=4–6, ***p<0.001; NS, no statistical significance, vs pH 7.4). (B) Pretreatment with pH 6.8 (Pre-6.8) for 10 min failed to prevent acid-induced neuronal death (CTB assay, n=4–6, ***p<0.001, NS, no statistical significance, vs pH 7.4; n.s., no statistical significance, vs pH 6.0). Note: treatment with the pH 6.8 solution for 1 hr only did not alter neuronal viability. (C) Summary of propidium iodide (PI)-positive neurons for Asic1a+/+ and Asic1a−/− cultures treated with pH 7.4 (CTRL) or pH 6.0 solutions for 15, 30, and 60 min. Representative images are shown in Figure 3—figure supplement 1B. At least 400 neurons were counted for each condition (***p<0.001; NS, no statistical significance, vs corresponding Asic1a−/− cultures). (D) Neuronal death induced by 1 hr treatment with the pH 6.0 solution in normal (SS, standard external solution), Na+-, K+-, Ca2+-free (NMDG replacement), and Ca2+-free conditions (CTB assay, n=3, ##p<0.01 vs Na+-, K+-, Ca2+-free in pH 6.0; ***p<0.001 vs pH 7.4 under the same cation conditions). Dashed line, pH 6.0 solution-induced neuronal death under normal SS condition. (E) Representative traces of I6.0 for cortical neurons in normal (SS), Na+-, K+-, Ca2+-free and Ca2+-free conditions. (F) Representative traces of I6.0 for wild type (WT) ASIC1a and its HIF and RC mutants expressed in CHO cells. (G) Current–voltage relationship of WT-ASIC1a (filled squares) and HIF-ASIC1a (open circles) in response to the pH 6.0 solution. Note: no current was induced by pH 6.0 in CHO cells that expressed HIF-ASIC1a at a broad range of holding voltages. (H) Summary data of cell death induced by 1 hr pH 6.0 solution treatment in Asic1a−/− neurons expressing GFP vector, WT-ASIC1a, and its HIF and RC mutants, based on PI staining of GFP-labeled neurons (see Figure 3—figure supplement 6 for representative images, n=100 for each condition, ***p<0.001; NS, no statistical significance, vs pH 7.4 of the corresponding transfection; n.s., no statistical significance, vs WT-ASIC1a).
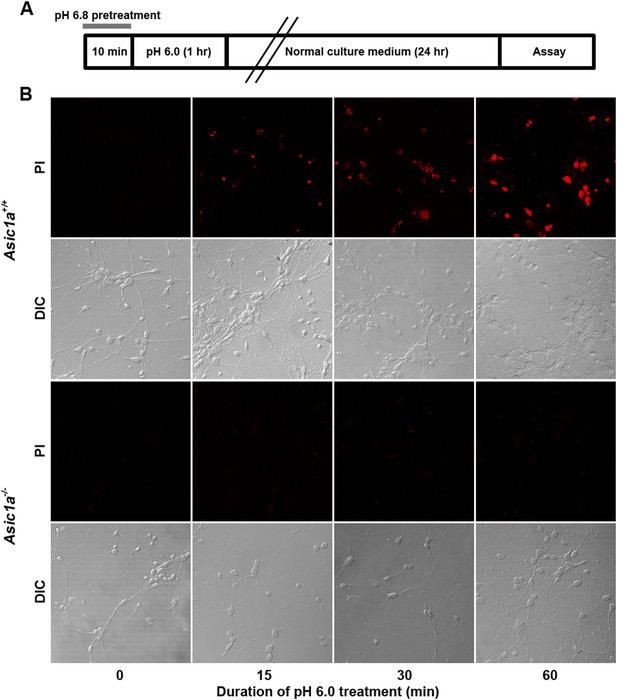
Time-dependence of acid-induced neuronal death.
(A) Experimental scheme for Figure 3B, where mild acid pretreatment was used to induce ASIC1a channel steady-state desensitization before pH 6.0 treatment and subsequent death assay. (B) Dependence of neuronal death (propidium iodide [PI] staining assay) on the duration of exposure to pH 6.0 solution. Neurons were identified by DIC.
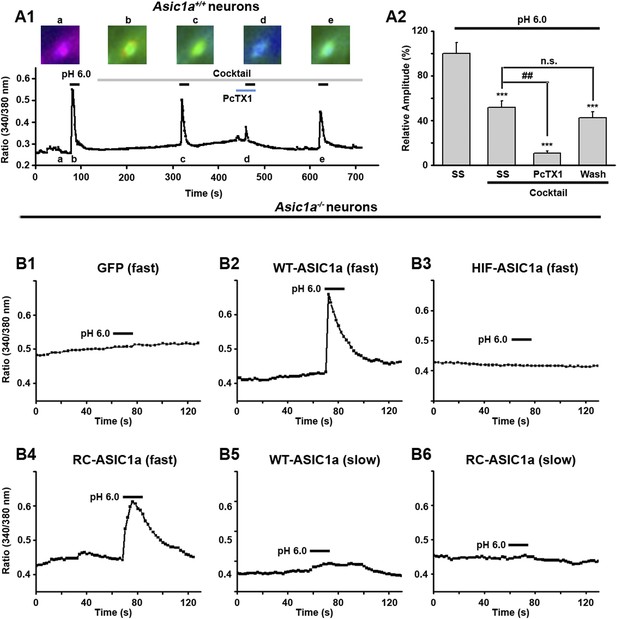
Fast perfusion is necessary for acid to induce [Ca2+]i elevation in ASIC1a-expressing neurons.
(A1) Acid (pH 6.0)-induced changes in Fura-2 ratio in cultured wild type (WT) mouse cortical neurons in the absence and presence of a cocktail of inhibitors (20 μM AP5, 20 μM CNQX, 1 μM TTX, and 5 μM nimodipine) without or with 50 nM PcTX1. Representative ratio images at indicated time points are shown above the trace. Fast perfusion (∼50 µl/min) was used. (A2) Summary data of normalized ratio changes after baseline subtraction for A1 (n>10, ***p<0.001, vs SS without cocktail; ##p<0.001; n.s., no statistical significance, vs SS with cocktail). (B1–B4) pH 6.0-induced Fura-2 ratio changes in cultured cortical neurons from Asic1a−/− mice transfected with control vector (GFP, B1) and GFP-tagged WT-ASIC1a (B2), HIF-ASIC1a (B3), or RC-ASIC1a (B4) via fast perfusion (∼50 µl/min). Note: there was no change in control (B1) and HIF-ASIC1a (B3)-transfected neurons. (B5, B6) pH 6.0-induced Fura-2 ratio changes in cultured cortical neurons from Asic1a−/− mice transfected with GFP-tagged WT-ASIC1a (B5) and RC-ASIC1a (B6) via slow perfusion (∼15 µl/min). Similar results were obtained for at least 10 neurons each for B1–B6.
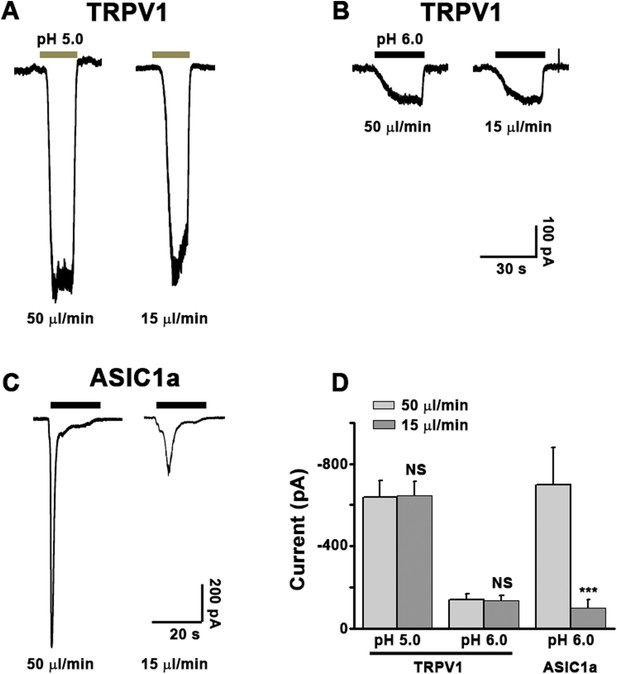
Validation of the Y-tube apparatus at fast and slow perfusion rates.
CHO cells were transiently transfected with the cDNA for rat TRPV1 (A, B, EGFPC1-TrpV1) or mouse ASIC1a (C, pEGFPC3-ASIC1a) and used for whole-cell patch clamp recordings after 24–30 hr. The cell was held at −60 mV while an acidic solution (pH 5.0 for A, pH 6.0 for B, C) was applied either at a fast (50 μl/min) or slow (15 μl/min) rate as indicated. The same Y-tube apparatus was used for all experiments and in the same manner as used for the Ca2+ imaging experiments shown in Figure 3—figure supplement 2. As summarized in D, the perfusion rate does not alter proton activation of the non-desensitizing TRPV1 currents, suggesting that the acidic solution effectively reached the cell under both conditions. Note: because the pH 6.0-evoked TRPV1 currents were small, we also used pH 5.0 to elicit larger and more reliable TRPV1 currents. On the other hand, the development of fast desensitizing ASIC1a currents was strongly affected by the perfusion rate of the pH 6.0 solution, giving slower and smaller currents at 15 μl/min than at 50 μl/min.
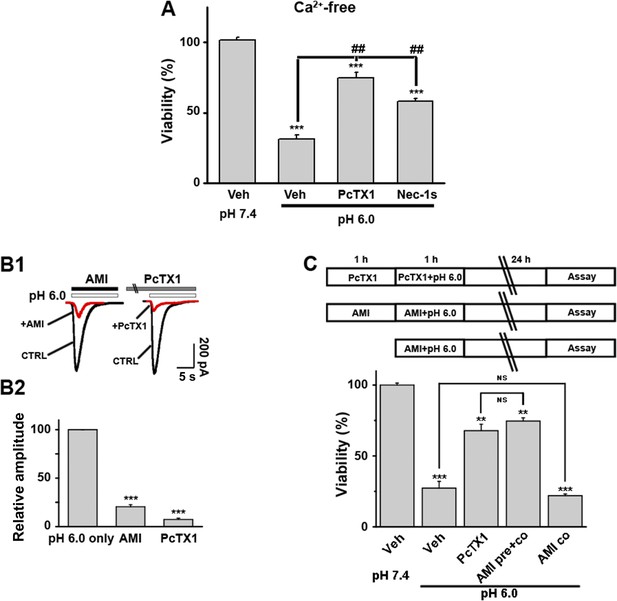
Acid-induced neuronal death is independent of Ca2+-flux and ionic currents via ASIC1a.
(A) PcTX1 and Nec-1s rescued acid-induced neuronal death in the Ca2+-free solution (CTB assay, n=3–5, ***p<0.001, vs vehicle (Veh) at pH 7.4; ##p<0.001, vs Veh at pH 6.0). (B1, B2) Inhibition of I6.0 by AMI (100 µM) and PcTX1 (10 nM) in cultured mouse cortical neurons. AMI was applied together with acid. PcTX1 was applied 60 min before acid. Both greatly suppressed I6.0. Shown are representative current traces at −60 mV (B1) and summary data for peak currents (B2) (n=4–6, ***p<0.001 vs pH 6.0 only). (C) Upper, scheme of AMI, PcTX1, and acid (pH 6.0) treatment of cortical neurons. Lower, summary data for neuronal death under the conditions indicated (CTB assay, n=4–6, **p<0.01, ***p<0.001 vs pH 7.4). AMI pre+co: AMI pretreatment plus AMI co-application with acid (pH 6.0); AMI co: AMI co-application with acid (pH 6.0).
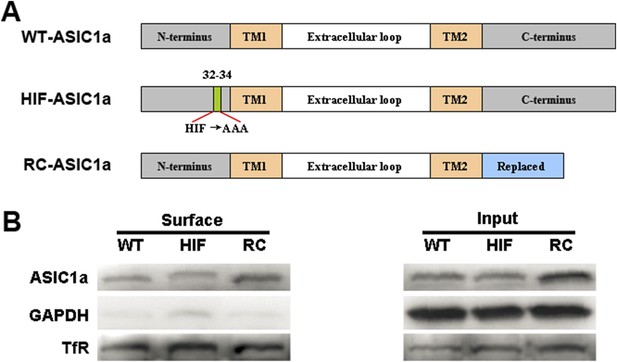
HIF and RC mutants of ASIC1a are normally expressed on the plasma membrane.
(A) Diagrams of the wild-type (WT) and mutant ASIC1a constructs. (B) Membrane expression of WT-ASIC1a and its HIF and RC mutants as determined by surface biotinylation experiments.
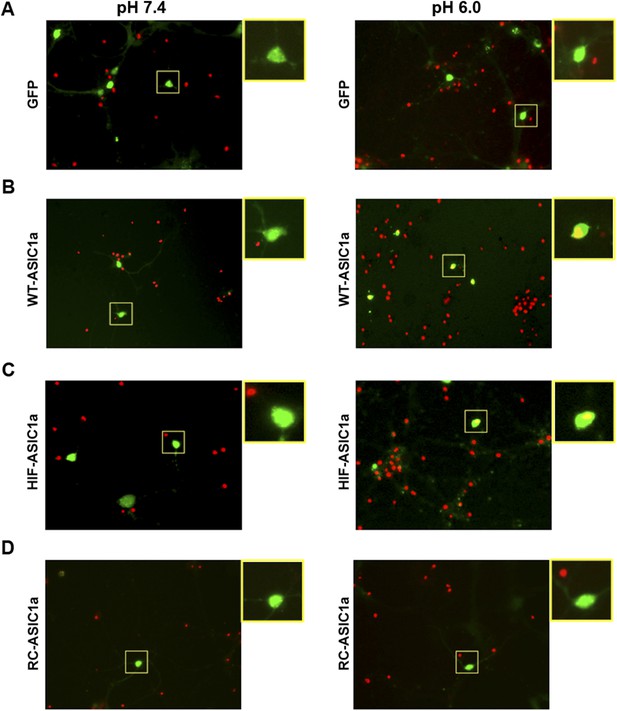
Expression of WT-ASIC1a and HIF-ASIC1a, but not RC-ASIC1a, in Asic1a−/− neurons resulted in acid-induced death.
Representative images for data shown in Figure 3H. Asic1a−/− neurons were transfected with GFP vector (A), GFP-tagged WT-ASIC1a (B), and its HIF (C) and RC (D) mutants. Transfected cells were identified by the green fluorescence signal. Cell death induced by 1 hr treatment with the pH 6.0 solution was assessed by propidium iodide (PI) staining (red). Note the nuclear PI labeling of green cells (yellow areas) in enlarged images from white boxes in B and C of pH 6.0-treated samples. These cells were counted as PI-positive.
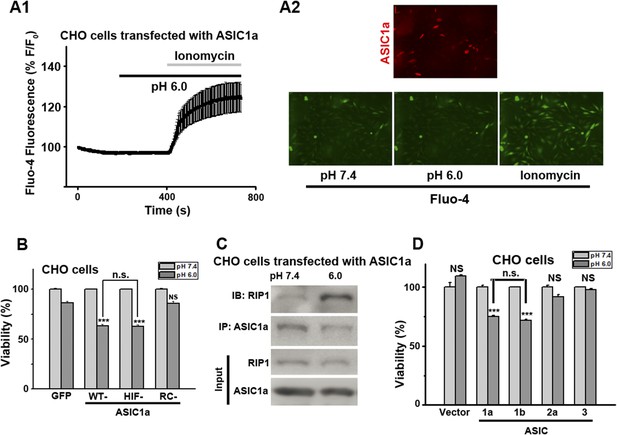
CHO cell death induced by acid (pH 6.0) is dependent on ASIC1 and involves recruitment of RIP1.
(A1, A2) Acid (pH 6.0) induced no detectable [Ca2+]i elevation (by Fluo-4) in CHO cells transfected with mCherry-ASIC1a. Transfected CHO cells were identified by mCherry (A2, red image). Ionomycin (2 μM) was added at the end of the experiment to show that cells were properly loaded and viable. (B) Cell death induced by 1 hr treatment with the pH 6.0 solution in CHO cells stably expressing GFP vector, WT-ASIC1a, and its HIF and RC mutants (CTB assay, n=3, ***p<0.001; NS, no statistical significance, vs pH 7.4; n.s., no statistical significance, vs WT-ASIC1a). (C) Acid caused ASIC1a–RIP1 association in CHO cells that expressed WT-ASIC1a. Cells were treated or not with the pH 6.0 solution for 90 min. Cell lysates were subjected to immunoprecipitation by anti-ASIC1a and then Western blotting by anti-RIP1. (D) Cell death induced by 1 hr treatment with the pH 6.0 solution in CHO cells transfected with pEGFP vector, ASIC1a, ASIC1b, ASIC2a, and ASIC3 (CTB assay, n=3, ***p<0.001; NS, no statistical significance, vs pH 7.4; n.s., no statistical significance, vs ASIC1a). Both ASIC1a and ASIC1b, but not ASIC2a and ASIC3, mediated acid-induced cell death.
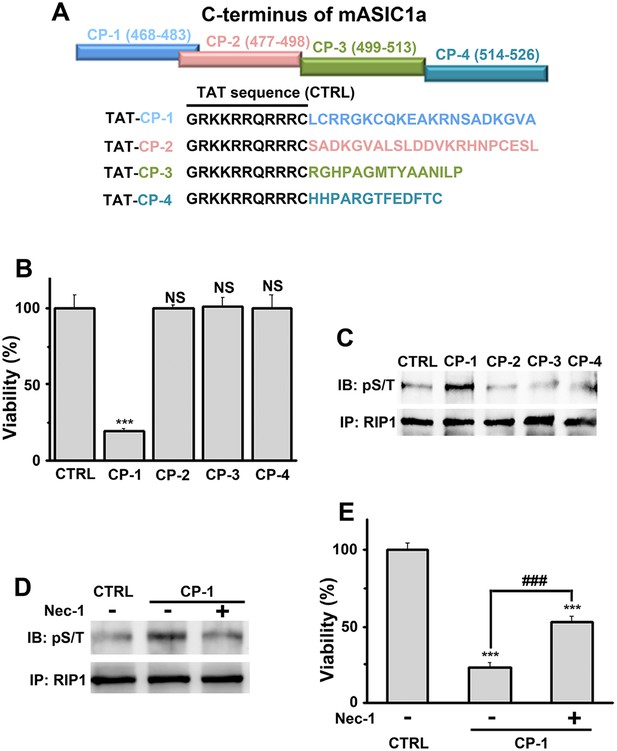
A peptide representing the proximal C-terminal region of ASIC1a induces RIP1 phosphorylation and neuronal death.
(A) Relative positions and amino acid sequences of four peptides representing different regions of the mouse ASIC1a (mASIC1a) C-terminus. (B) TAT-tagged CP-1, but not CP-2, CP-3, or CP-4, peptide (10 μM, 24 hr) induced neuronal death at pH 7.4 (CTB assay, n=3, ***p<0.001; NS, no statistical significance, vs control [CTRL, the TAT peptide alone]). (C, D) CP-1, but not CP-2, CP-3, or CP-4, enhanced RIP1 phosphorylation (C) and the effect was blocked by Nec-1 (20 µM) (D). (E) Nec-1 (20 μM) partially rescued CP-1-induced neuronal death (CTB assay, n=3, ***p<0.001 vs CTRL; ###p<0.001 vs CP-1 alone).
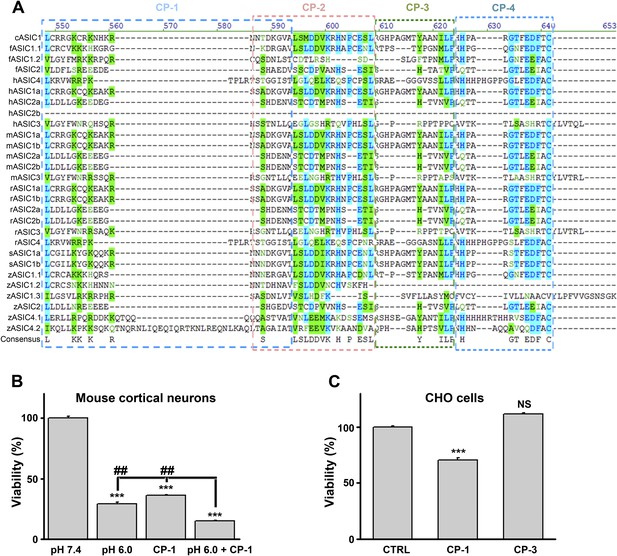
Design and characterization of ASIC1a-derived peptides.
(A) Alignment of ASIC C-termini of different isoforms from various species. Starting positions are indicated in parentheses. Locations of the four mouse ASIC1a C-terminal peptides are shown by the boxes. c, chicken; f, toad fish; h, human; m, mouse; r, rat; s, shark; z, zebrafish. (B) CP-1 failed to inhibit acid-induced neuronal death (CTB assay, n=3, ***p<0.001 vs pH 7.4; ##p<0.001 vs pH 6.0+CP-1). (C) CP-1, but not CP-3, caused the death of CHO cells in the absence of ASIC1a (CTB assay, n=3, ***p<0.001; NS, no statistical significance, vs CTRL, the TAT peptide alone).
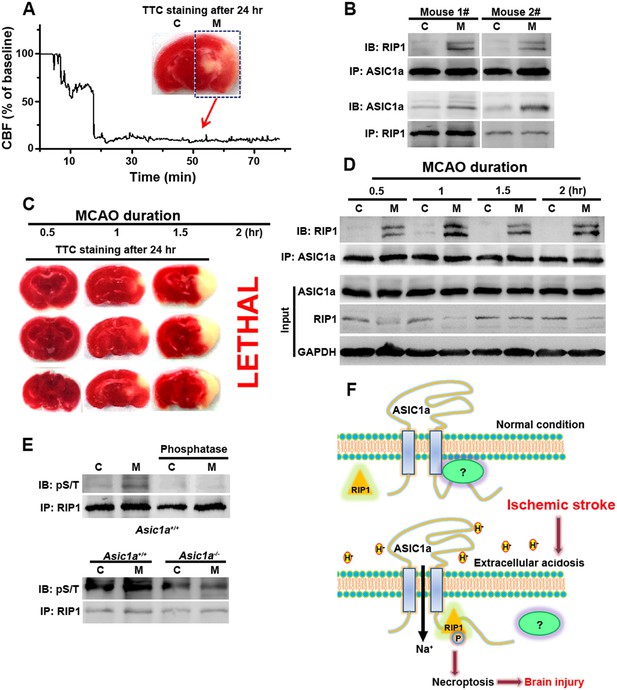
RIP1 is recruited to ASIC1a in ischemic brain.
(A) Cerebral blood flow (CBF) of ischemic brain hemisphere before and during middle cerebral artery occlusion (MCAO, arrow) was monitored by transcranial laser Doppler. C, control hemisphere; M, MCAO hemisphere. (B) One-hour MCAO treatment caused association of RIP1 with ASIC1a (upper, IP: ASIC1a, IB: RIP1; lower, IP: RIP1, IB: ASIC1a). Both bands in IB: RIP1 represent RIP1; the upper band may represent phospho-RIP1 or ubiquitinated-RIP1 as shown in previous studies (Cho et al., 2009; He et al., 2009). (C) TTC-staining of ischemic brain slices after 0.5, 1, and 1.5 hr of MCAO treatment followed by 24 hr reperfusion. The 2 hr MCAO was lethal. (D) RIP1-ASIC1a association in control (C) and MCAO hemisphere (M) following MCAO with durations as indicated (IP: ASIC1a; IB: RIP1). The levels of ASIC1a and RIP1 were assessed by IB. Note: total levels of RIP1 were reduced in the MCAO hemisphere compared to control, despite the increased association with ASIC1a. (E) Ischemia-induced RIP1 phosphorylation was abolished in Asic1a−/− brain. Upper, phosphorylation of RIP1 in ischemic brain of wild type (WT) mice, which was largely removed by 1 hr phosphatase treatment. Lower, Asic1a gene deletion prevented the ischemia-induced increase in RIP1 phosphorylation. (F) Schematic of a possible mechanism of acid-induced necroptosis. Upper, under normal physiological pH, the C-terminus of ASIC1a is protected by being buried inside or bound by an unknown protein; lower, acid stimulation exposes the CP-1 region of the ASIC1a C-terminus, allowing for association with and activation of RIP1, which in turn leads to necroptosis.
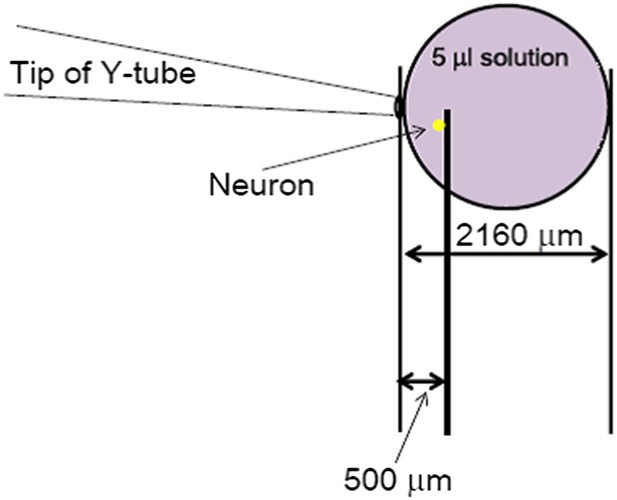
The most conserved estimate of space occupied by the 20s-administration of a perfusion solution with Y-tube at the flow rate of 15 µl/min. The solution coming out from the Y-tube was assumed to form a perfect sphere, which represents a large underestimate of the area it would cover at the bottom of the dish.





