Genomic variations of the mevalonate pathway in porokeratosis
Figures
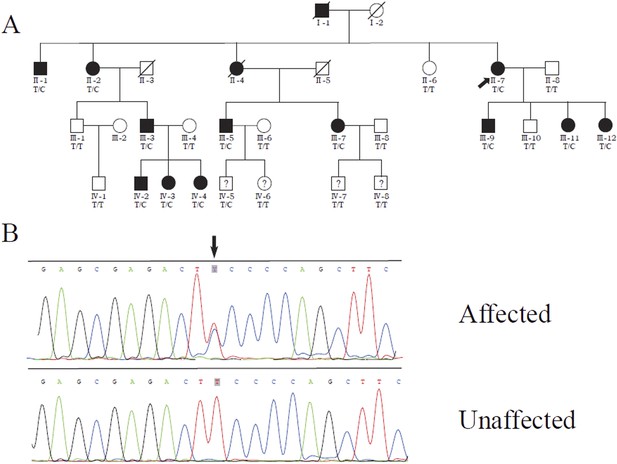
Identification of a MVD mutation in a porokeratosis (PK) family.
(A) c.746T>C (p.Phe249Ser) in MVD displayed 100% co-separation with PK phenotype in this family (Luan et al., 2011). (B) Sanger sequencing chromatograms of proband (II-7, affected) and normal control (II-8, unaffected) at the c.746T>C mutation site indicated by arrow.
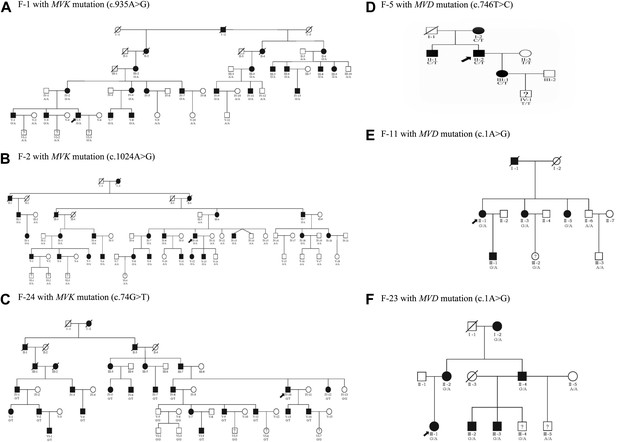
Examples of six pedigree charts showing that each mutation displayed 100% co-segregation with the porokeratosis (PK) phenotype in the family.
https://doi.org/10.7554/eLife.06322.004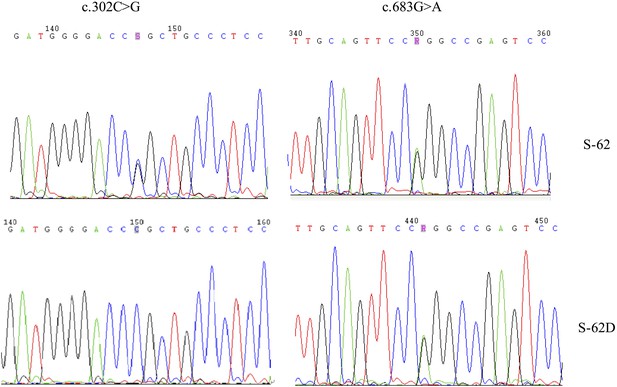
Two MVD mutations, c.302C>G (p.Pro101Arg) and c.683 G>A (p.Arg228Gln), for S-62 were located in the trans position because his daughter (S-62-D) carried only one of them.
https://doi.org/10.7554/eLife.06322.005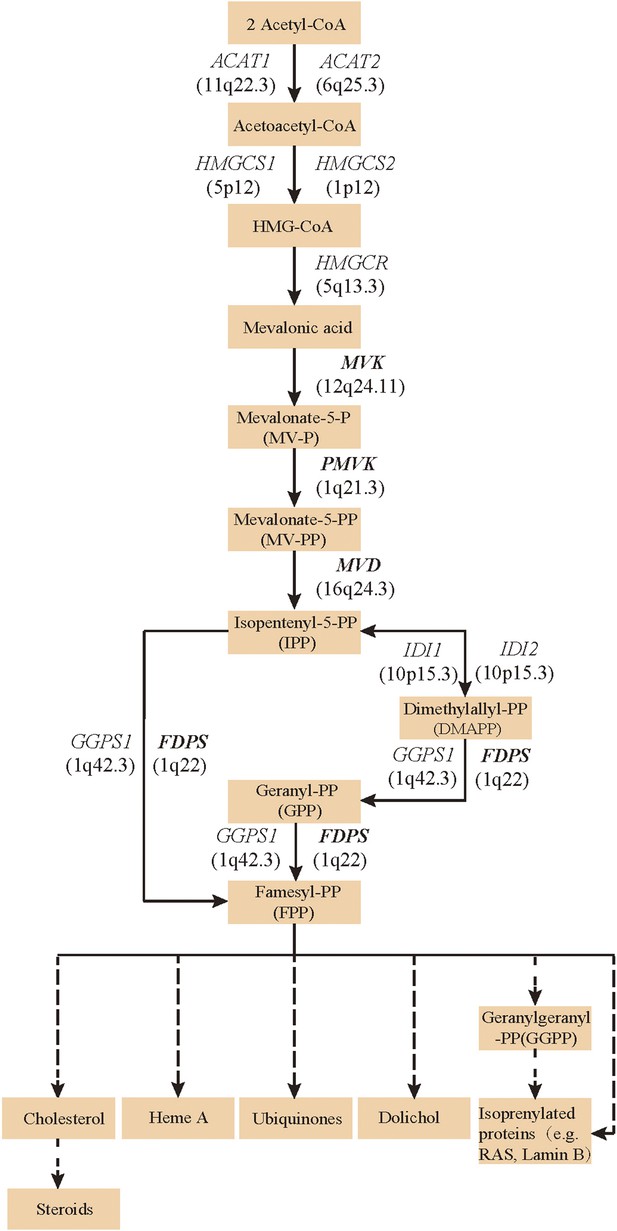
Isoprenoid biosynthesis via the mevalonate pathway.
12 member genes (ACAT1, ACAT2, HMGCS1, HMGCS2, HMGCR, MVK, PMVK, MVD, IDI1, IDI2, FDPS, GGPS1) were subject to mutation screening. The genomic loci of the 12 member genes are provided in parentheses. The illustration is adapted from the 00900 interactive map of the Kyoto Encyclopedia of genes and genomes (KEGG) (Kanehisa et al., 2012).
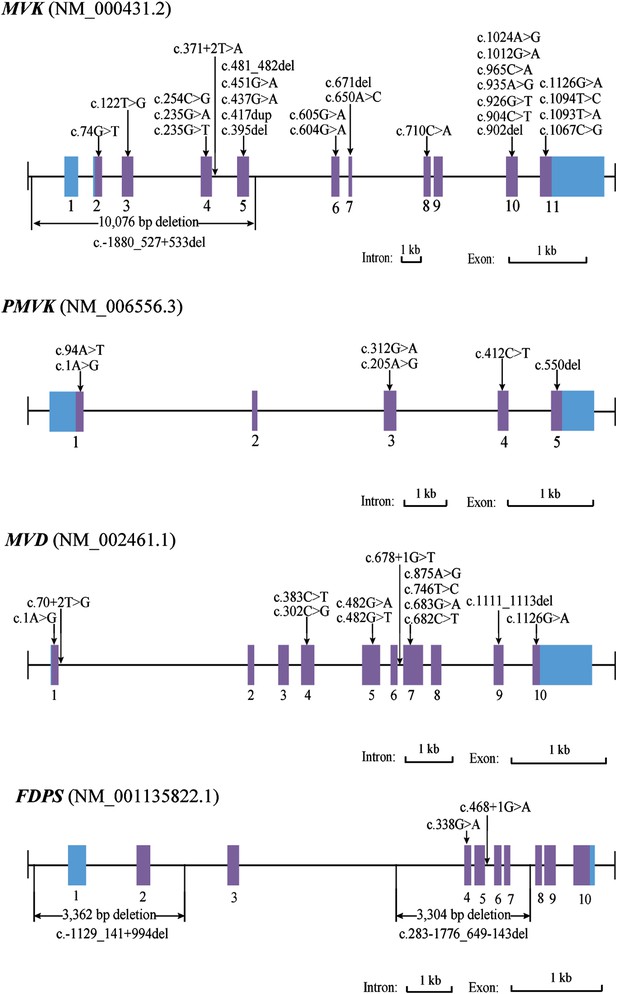
Mutational spectrum of MVK, PMVK, MVD and FDPS in 113 of the 134 porokeratosis (PK) patients.
https://doi.org/10.7554/eLife.06322.007-
Figure 3—source data 1
Sanger sequencing chromatograms of normal control and porokeratosis (PK) patients at 48 mutation sites in MVK, PMVK, MVD and FDPS.
- https://doi.org/10.7554/eLife.06322.008
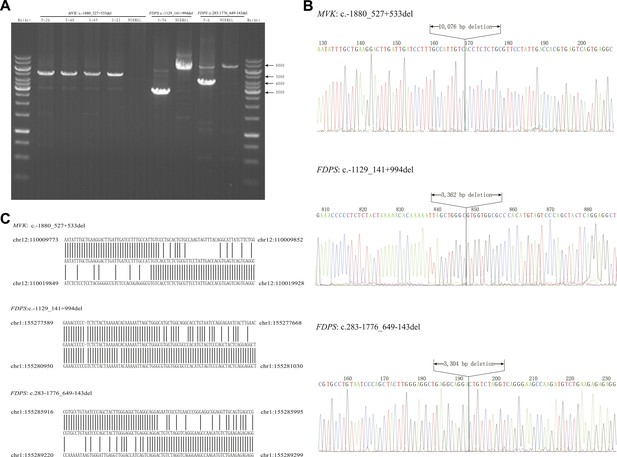
Breakpoint analysis for three large deletion mutations in MVK and FDPS genes.
(A) Agarose electrophoresis analysis of long PCR products from patients with deletion mutations and a normal control. (B) Sequencing chromatograms of long PCR products from deletion mutations. (C) Alignment of the sequences from three deletion mutations with human genome reference GRch38 primary assembly indicates a deletion of 10,076 bp, 3362 bp and 3304 bp, respectively.
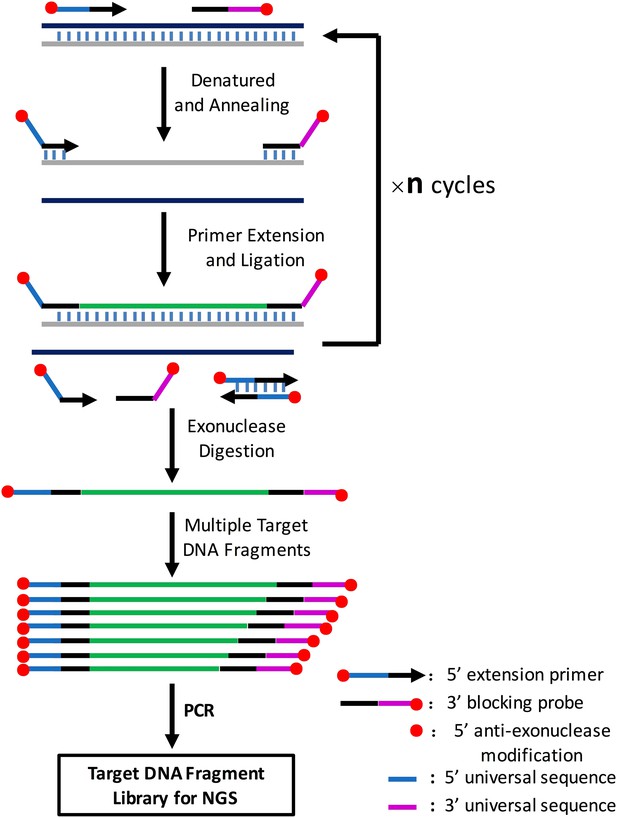
Illustration of amplification of multiple target DNA fragments mediated by cycled primer extension and ligation.
In brief, for each target region, an extension primer and a block probe are designed. The extension primer has a 5′ exonuclease-resistance modification and the block probe has a 5′ phosphorate and 3′exonuclease-resistance modification. These two oligos are mixed with genomic DNA, heat-denatured, and then annealed to the same strand of the target DNA fragment. The primer extends and stops until it meets the block probe, and the extension product is then ligated with the block probe by a thermal stable ligase. The above procedure can be repeated n times on the PCR machine using a two-step PCR cycling program. The extension and ligation product is then purified by an exonuclase mixture digestion to remove any DNA fragment with no exonuclease-resistance modification at both ends such as remained primers or probes, primer dimers and genomic DNA, and then amplified using universal NGS PCR primer pairs. A CPELA reaction can include hundreds of extension primer-blocking probe sets and simultaneously amplify hundreds of target DNA fragments for subsequent massively parallel sequencing.
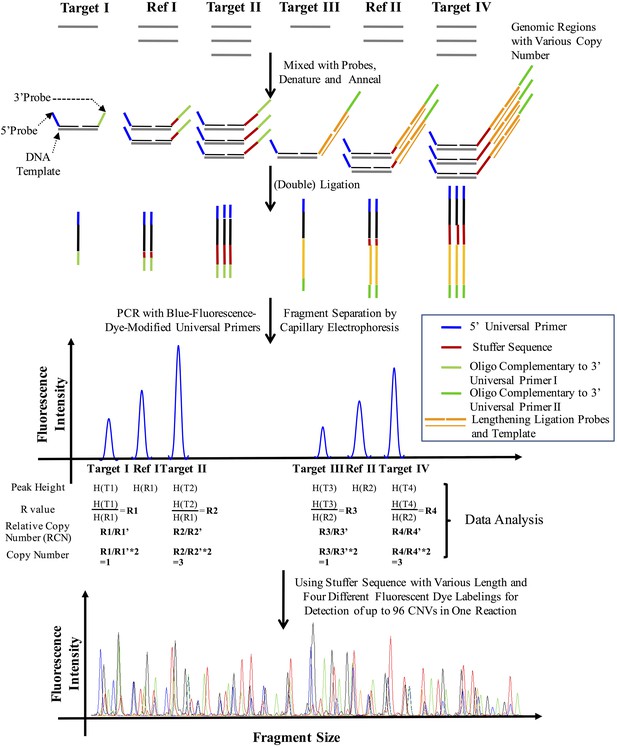
The principle of CNVplex technology.
https://doi.org/10.7554/eLife.06322.011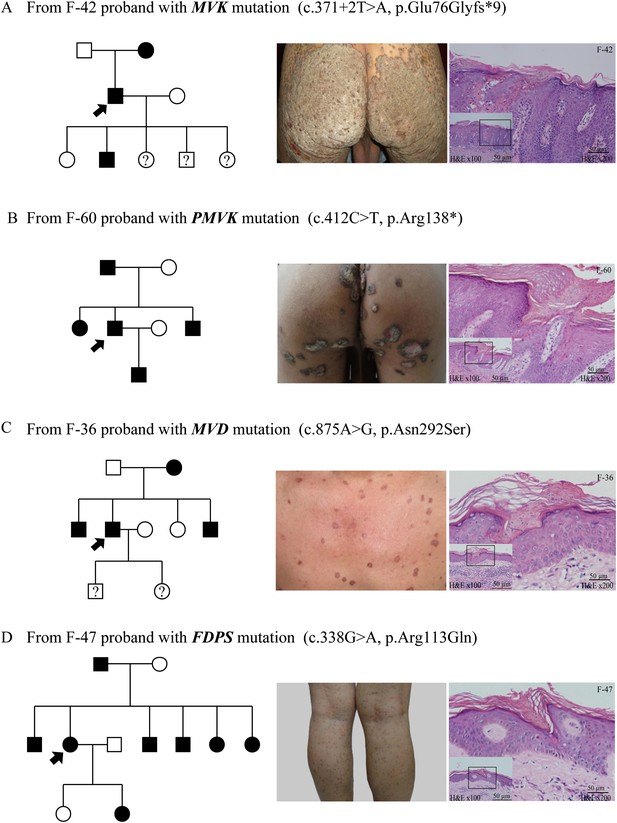
Representative clinical phenotypes and histopathology associated with the four genotypes.
From left to right, pedigree charts, clinical phenotypes and the corresponding histopathology photos are shown correspondingly. (A) Family (F)-42 proband with MVK mutation showed giant hyperkeratotic plaque-type porokeratosis ptychotropica. (B) F-60 proband with PMVK mutation showed tumor-like porokeratoma in the genitogluteal region. (C) F-36 proband with MVD mutation showed discrete, red-brown annular keratotic papules or maculopapules on the chest. (D) F-47 proband with FDPS mutation showed multiple, small, superficial, annular papules with thread-like ridges on the legs. All histopathology showed cornoid lamella, a histological hallmark of porokeratosis with vertical columns of parakeratosis overlying an area of hypogranulosis with dyskeratotic cells.
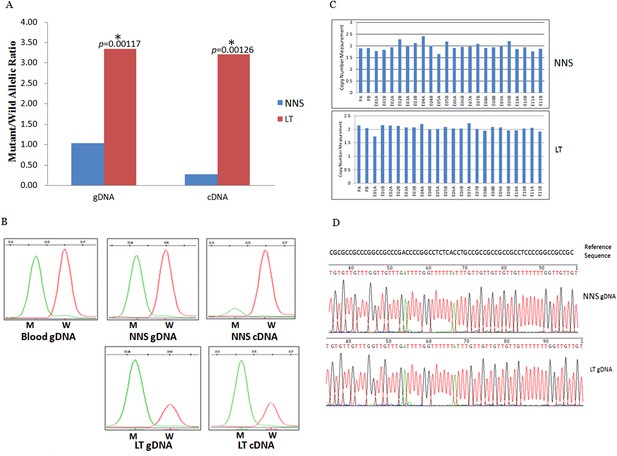
Gene conversion of the wild to mutant allele was identified in a buttock lesion from F-31 carrying a MVK mutation of c.395delT.
(A) The mutant/wild allelic ratios in genomic DNA (gDNA) and complementary DNA (cDNA) of lesional tissue (LT) and neighboring normal-appearing skin (NNS). The quantity of the mutant allele was about threefold and 10-fold more than the quantity of the wild allele in gDNA and cDNA, respectively. (B) The chromatograms of single nucleotide extension targeting the c.395delT mutation using the SNaPshot kit for five DNA samples (blood gDNA, NNS gDNA, LT gDNA, NNS cDNA and LT cDNA). The mutant peak was overpresented in both LT gDNA and LT cDNA. (C) No copy number change in genomic DNA of NNS and LT. PA and PB are probes in the promoter region, E01 to E11 designates exon 1 to exon 11, and A or B indicate two different probes in the same exon. (D) The bisulfite sequencing of NNS gDNA and LT gDNA. No methylation was observed for the targeted CpG sites in the promoter region of MVK.
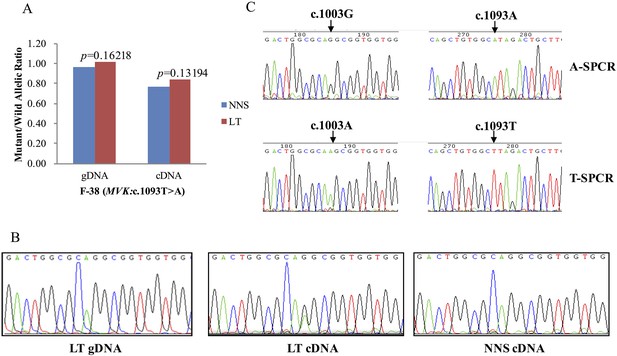
G-to-A RNA editing at position 1003 of the wild allele (T) of c.1093T>A in MVK was detected in a left forearm lesion from F-38.
(A) No allelic expression imbalance was observed in lesional tissue (LT) from F-38. (B) A mutation of c.1003G>A was identified in LT cDNA, but not in neighboring normal-appearing skin (NNS) cDNA or LT gDNA. (C) Sequencing the c.1093A and c.1093T allele-specific PCR products indicated the mutant allele(A) of c.1003G>A was in the cis position with the wild allele(T) of c.1093T>A. A-SPCR, c.1093A–specific PCR; T-SPCR, c.1093T-specific PCR.
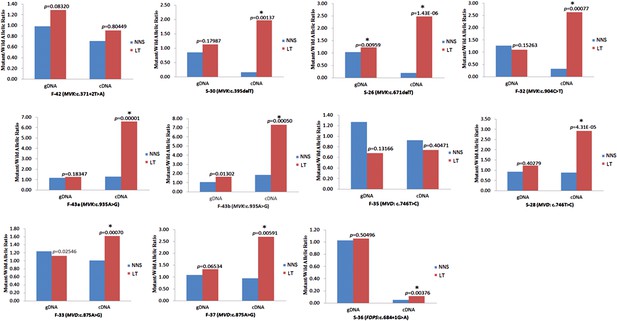
Significantly reduced expression of the wild allele in other nine lesion tissues.
The Student t-test was performed to measure the difference in mutant/wild allelic ratios in genomic DNA (gDNA) or complementary DNA (cDNA) from lesion tissue (LT) and neighboring normal-appearing skin (NNS) for each mutation. The test score (p value) is presented above the LT bars. The asterisk (*) designates a significance level of 1%. F-43a and F-43b indicates the tissue sets of the left forearm and left thigh, respectively, from the same F-43 patient.
Tables
Clinical characteristics and genetic causes of 134 index patients with PK
| Genetic causes of 134 index patients with PK | |||||
|---|---|---|---|---|---|
| MVK (39*) | PMVK (9) | MVD (62*) | FDPS (4) | Unknown (21) | |
| Sex | |||||
| Male | 27 | 9 | 38 | 1 | 17 |
| Female | 12 | 0 | 24 | 3 | 4 |
| Number of lesions | |||||
| 0–10 | 4 | 4 | 0 | 0 | 6 |
| 10–100 | 20 | 5 | 13 | 0 | 5 |
| 100–500 | 5 | 0 | 39 | 0 | 5 |
| >500† | 10 | 0 | 10 | 4 | 5 |
| Diameter of lesions | |||||
| Minimum | 2 mm | 5 mm | 1 mm | 1 mm | 1 mm |
| Maximum | 20 cm | 5 cm | 2 cm | 1 cm | 2 cm |
| Age of onset | |||||
| At birth | 0 | 0 | 0 | 0 | 3 |
| 0–20 | 23 | 4 | 20 | 1 | 0 |
| 20–40 | 12 | 3 | 28 | 1 | 9 |
| 40–60 | 3 | 1 | 12 | 2 | 5 |
| >60 | 1 | 1 | 2 | 0 | 4 |
| Variants of PK | |||||
| DSAP/DSP | 26 | 0 | 56 | 4 | 10 |
| SFP | 0 | 0 | 6 | 0 | 0 |
| PM | 23 | 9 | 0 | 0 | 6 |
| HPM | 13 | 3 | 1 | 0 | 0 |
| Giant plaque of PPt | 19 | 0 | 0 | 0 | 0 |
| Genital PK (localized) | 0 | 4 | 0 | 0 | 0 |
| Porokeratoma | 0 | 5 | 0 | 0 | 0 |
| LP | 1 | 1 | 3 | 0 | 5 |
| Comorbidity | |||||
| Psoriasis vulgaris | 4 | 0 | 2 | 0 | 0 |
-
*
One PK patient (proband of family-28, female), who has both the mutation c. 235G>A (MVK) and the mutation c. 746T>C (MVD), was included in both MVK and MVD groups.
-
†
The number of lesions is more than 500.
-
DSAP, disseminated superficial actinic porokeratosis; DSP, disseminated superficial porokeratosis; HPM, hyperkeratotic porokeratosis, LP, linear porokeratosis, PK, porokeratosis; PM, porokeratosis of Mibelli, PPt, porokeratosis ptychotropica; SFP, solar facial porokeratosis.
Additional files
-
Supplementary file 1
12 non-pathogenic rare missense or nonsense single nucleotide variants (SNVs) and major reasons for exclusion.
- https://doi.org/10.7554/eLife.06322.017
-
Supplementary file 2
Characterization of 51 mutations identified in 113 of the 134 porokeratosis (PK) patients.
- https://doi.org/10.7554/eLife.06322.018
-
Supplementary file 3
Mutant to wild allelic ratio measurements in both genomic DNA and complementary DNA of 13 pairwise tissue sets from 12 patients.
- https://doi.org/10.7554/eLife.06322.019





