Mitotic fidelity requires transgenerational action of a testis-restricted HP1
Figures
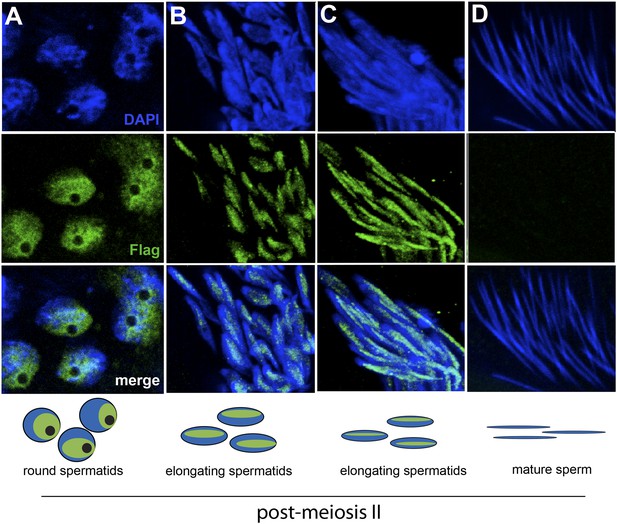
HP1E localization in D. melanogaster spermatogenesis.
We highlight four stages of spermatogenesis in D. melanogaster testes: (A) round spermatids, (B, C) elongated spermatids, and (D) mature sperm. HP1E localization was visualized with a Flag epitope-tagged HP1E transgene driven by a native promoter. We find that Flag-HP1E protein (green) localizes to the DNA (blue) of post-meiotic spermatids and persists until sperm maturation but is not present on mature sperm.
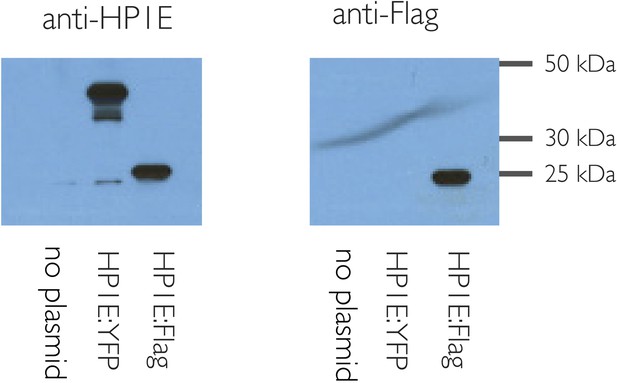
HP1E antibody is specific.
HP1E antibody recognizes heat-shock induced HP1E expression in cell culture (S2 cells). Predicted sizes: native HP1E 20.3 kD, Flag:HP1E, 24 kD, YFP:HP1E 46 kD.
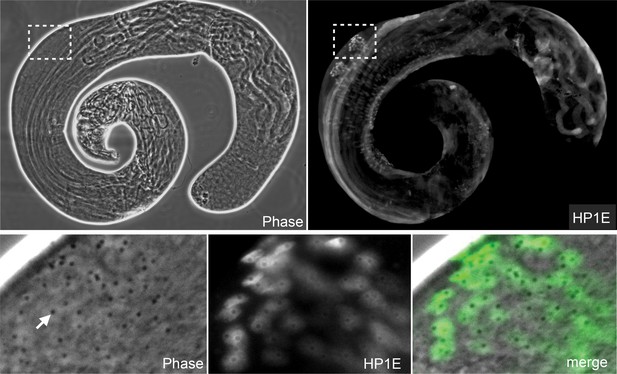
HP1E protein localizes to post-meiotic spermatids.
HP1E-stained whole-mount testis (upper panel). HP1E localizes to spermatids after meiosis II. HP1E first appears during spermatogenesis on round spermatids with a prominent ‘protein body’ or ‘pseudonucleus’ (arrow), which corresponds to the region of HP1E exclusion. A protein body is diagnostic of a post-meiotic stage.
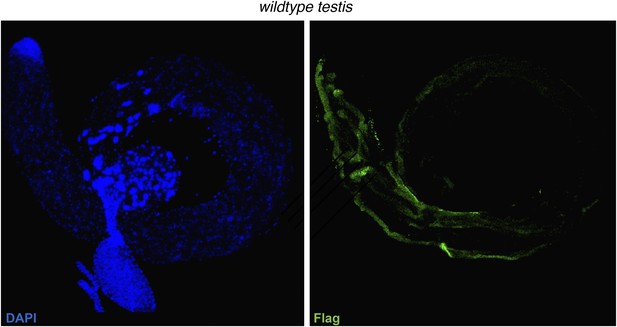
Anti-3xFlag (M2) exhibits no localization to spermatids in a non-Flag tagged HP1E genetic background.
https://doi.org/10.7554/eLife.07378.006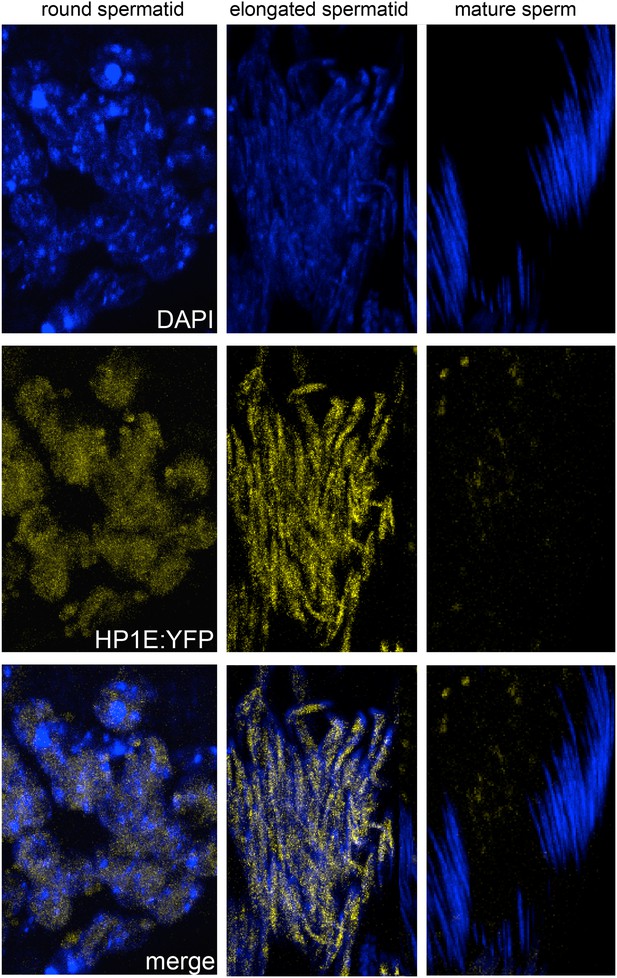
Fixed testis expressing HP1E-YFP driven by a native promoter recapitulates immunofluorescence results.
HP1E localizes to DNA of round and elongated spermatids. We observed no evidence of HP1E localization to DNA of mature sperm.
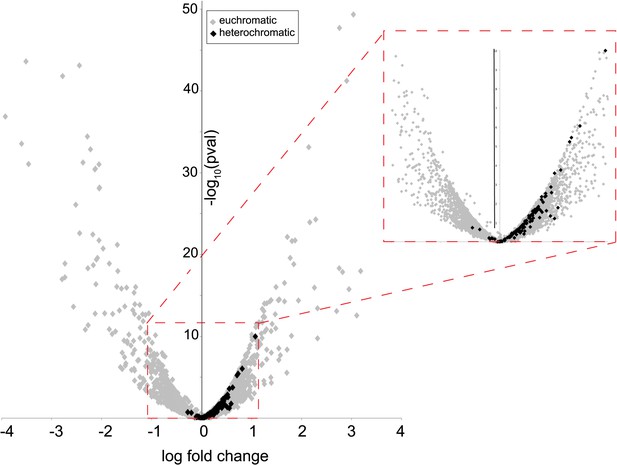
Heterochromatin-embedded genes are globally perturbed upon HP1E-depletion.
HP1E depletion in testis results directly or indirectly in mis-regulation of hundreds of genes. Volcano plot illustrates the fold up- and down-regulation of euchromatin-embedded genes (gray points) and heterochromatin-embedded genes (black points).
-
Figure 2—source data 1
Results of RNA-seq comparisons between testes of wild-type vs HP1E-depleted males, rank-ordered by the false discovery rate.
- https://doi.org/10.7554/eLife.07378.009
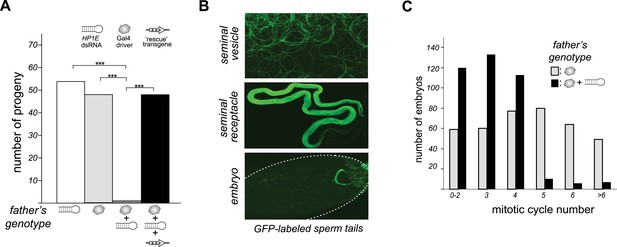
HP1E is a paternal effect lethal in D. melanogaster.
(A) HP1E knockdown via simultaneous presence of both a UAS-driven HP1E dsRNA gene and a Gal4 driver results in highly penetrant male sterility. Fertility can be fully restored by an HP1E transgene recoded at all synonymous sites, driven by a native HP1E promoter (‘rescue transgene’). Please refer to Figure 3—source data 1. (B) HP1E-depleted males produce abundant motile sperm (seminal vesicle), which are efficiently transferred to females (seminal receptacle), and fertilize the egg (embryo). We visualized sperm tails using the ‘don juan-GFP’ transgene (29). (C) Unlike wild-type embryos (gray), embryos fathered by HP1E-depleted males (black) arrest after 3–4 rounds of nuclear divisions (Mann–Whitney U: p < 0.0001). Embryos were collected in the 5–70 min window post-fertilization. Please refer to Figure 3—source data 2.
-
Figure 3—source data 1
Number of progeny fathered by males encoding both the UAS-HP1E hairpin and the Gal4 driver (24196/A5C) compared to fathers encoding the Gal4 transgene alone (w1118/A5C), hairpin alone (24196/CyO), or both plus the native promoter-driven, HP1E recoded transgene (24196/A5C + transgene).
- https://doi.org/10.7554/eLife.07378.011
-
Figure 3—source data 2
Mitotic cycle number (0, 1, 2 etc) of embryos fathered by wild-type males (24196/TM6) or PEL embryos fathered by HP1E-depleted males (24196/A5C) collected during the 75-min window post-oviposition.
- https://doi.org/10.7554/eLife.07378.012
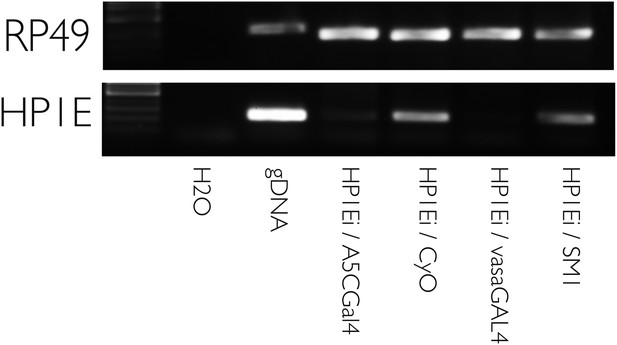
HP1E knockdown using multiple drivers is efficient.
RT-PCR on testis cDNA prepared from testis encoding a UAS-HP1E hairpin alone (‘HP1Ei’) or HP1E hairpin + Gal4 driver. Ubiquitous (‘A5CGal4’) and testis-restricted (‘vasaGal4’) drivers of the UAS_HP1E hairpin knockdown HP1E expression. Rp49 is the positive control. ‘CyO’ and ‘SM1’ refer to balancer chromosomes and represent wild-type HP1E expression levels.

DNA sequence of recoded HP1E transgene.
All third position sites were changed to either the preferred codon (if un-preferred was encoded) or the next preferred codon (if the preferred codon was encoded) following the Drosophila melanogaster preferred codon usage table.

Nucleotide and amino acid sequence of the HP1E mutant.
An HP1E-targeted TALEN deleted 7 bp starting at coding sequence base pair 58 (above). Translation of coding sequence encoding the lesion. ‘*’ = stop codon (below).
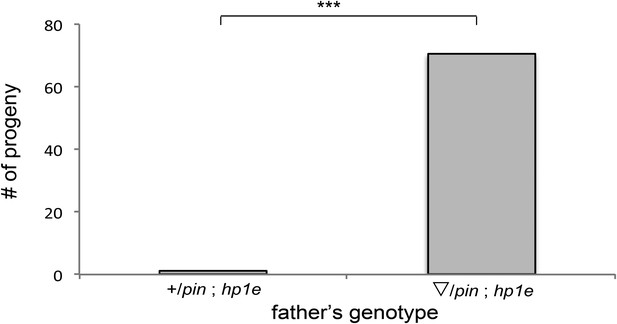
HP1E mutant recapitulates male fertility defect.
‘pin’ refers to a marked second chromosome, triangle refers to the native promoter driven HP1E transgene inserted on the second chromosome. ‘***’ refers to a p-value < 0.0001 in a Mann–Whitney U test.
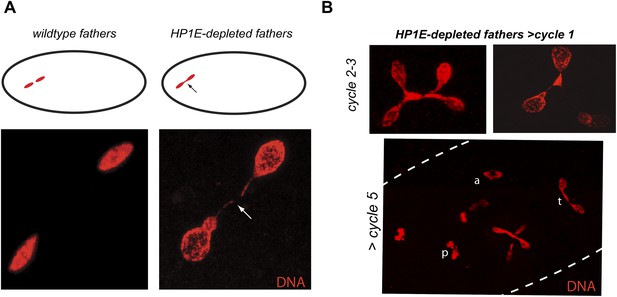
HP1E depletion in testis results in failed first embryonic mitosis and later mitotic catastrophe.
(A) We observed a chromatin bridge (arrow) in the first zygotic telophase in PEL embryos fathered by HP1E-depleted, but not wildtype, males. (B) Embryos aged beyond first mitosis exhibit increasingly aberrant nuclear morphology and asynchrony across nuclei (p = prophase, a = anaphase, t = telophase).
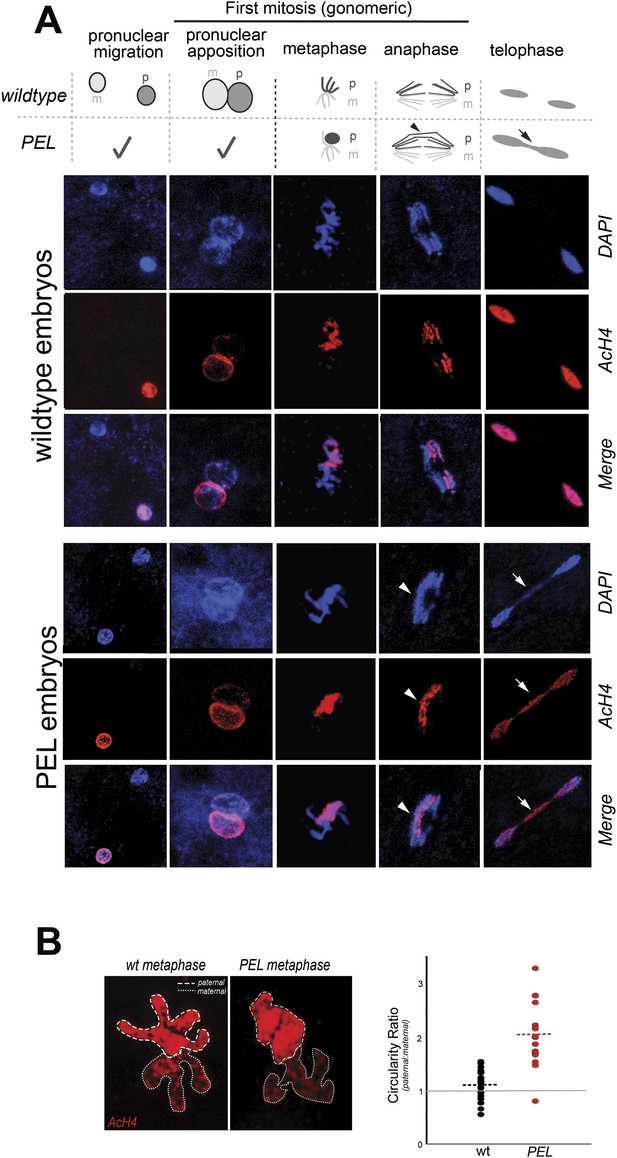
HP1E depletion in fathers results in mitotic arrest due to a paternal chromatin defect.
(A) Paternal chromatin (marked by anti-AcH4 (red)) morphology mirrors maternal chromatin in wild-type embryos but differs in PEL embryos. In both wild-type and PEL embryos, the female and male pronuclei ‘migrate’ toward each other, ‘appose’, and then enter mitosis. However, in PEL embryos, metaphase appears asynchronous between maternal and paternal chromatin, an AcH4-enriched chromatin bridge appears in anaphase (arrowhead) and persists at telophase (arrow). (B) We calculated a ‘circularity ratio’ (1 = perfect circle, 0 = starfish) for the first metaphase in wild-type and PEL embryos. We found that the paternal chromatin was significantly more circular i.e., less condensed than maternal chromatin in PEL (red dots) but not wild-type embryos (black dots), (Mann Whitney-U test, p < 0.0001). Dotted lines refer to sample means. A circularity ratio of 1 (gray solid line) refers to paternal and maternal chromatin with equivalent circularity.
-
Figure 5—source data 1
Independent measurements of ‘circularity’ of maternal to paternal nuclei at first metaphase in embryos fathered by either wild-type (‘wt’) or HP1E mutant males (‘HP1E’).
- https://doi.org/10.7554/eLife.07378.019
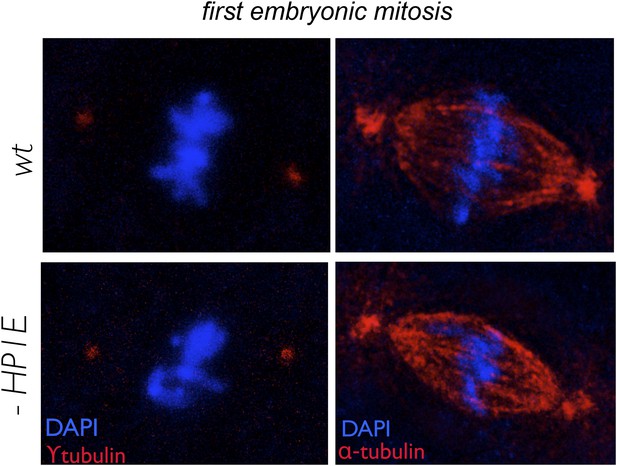
In embryos fathered by both wild-type and HP1E knockdown males, metaphase centrosomes (red, left panel) and spindle (red, right panel) are indistinguishable.
Genotype of father appears on the left.
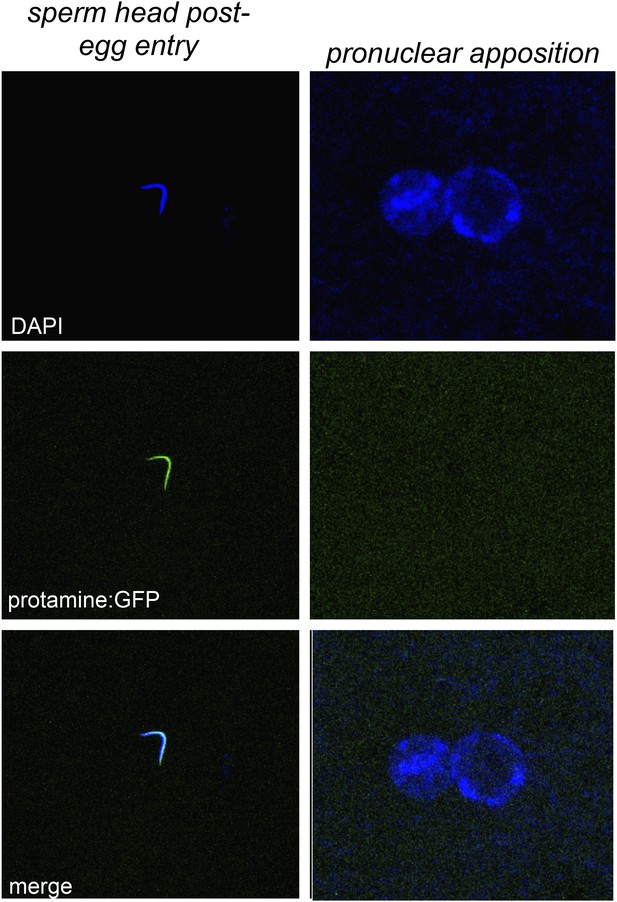
Embryos fathered by HP1E knockout males eject protamines.
The protamine-GFP packaged sperm head is the only phase post-sperm entry when GFP signal was observed.
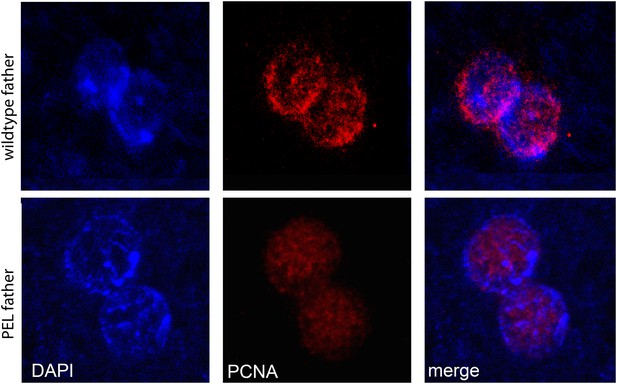
PCNA (replication factor) is recruited to both maternal and paternal pronuclei at apposition in wild-type and PEL embryos.
https://doi.org/10.7554/eLife.07378.022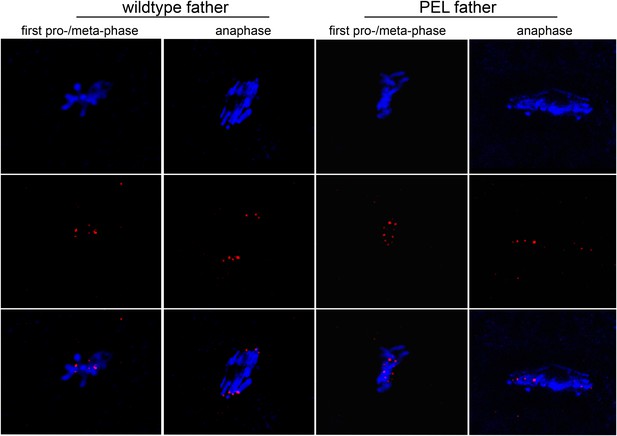
HP1E PEL embryos initiate kinetochore assembly.
Kinetochores of embryos fathered by HP1E knockout males visualized with anti-Cenp-C. In the left panel, the paternal DNA appears in the lower half of the chromosome conformation.
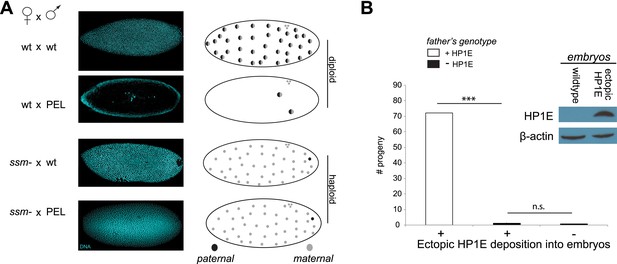
Embryonic mitosis can be rescued by excluding paternal chromatin but not by ectopic embryonic deposition of the HP1E protein itself.
(A) HP1E-knockdown males crossed to wild-type mothers father embryos that undergo early arrest (also see Figure 3C). However, both HP1E-depleted and wild-type males crossed to sesame (ssm) mothers father maternal haploid embryos that surpass mitotic cycle 12. Black and gray circles refer to the paternal and maternal DNA contributions, respectively, to the zygotic nuclei. Embryos were imaged after fixation and DAPI staining. Please refer to Figure 6—source data 1. (B) We observe no evidence of rescue when HP1E is deposited ectopically into the egg (‘+’) prior to fertilization (‘***’ refers to a p-value < 0.0001 in a Mann–Whitney U test, ‘n.s.’ = not signficant). Western blot probed with the HP1E antibody shows an absence of native HP1E in embryos of wild-type mothers and HP1E deposition into early embryos of the experimental females. Like wild-type females (‘−’ on the x-axis), these experimental females (‘+’ on the x-axis) fail to mother viable progeny when crossed to HP1E-depleted (black bar) males. Please refer to Figure 6—source data 2.
-
Figure 6—source data 1
Number of embryos generated by ssm- females that arrested earlier than cycle 3 (‘ARREST’) or after cycle 7 (‘NOarrest’) fathered by wild-type males (24196/TM6) or HP1E-depleted males (24196/A5C).
- https://doi.org/10.7554/eLife.07378.025
-
Figure 6—source data 2
Number of progeny generated from crosses between mothers encoding a Gal4 driver alone (MTD/CyO) or Gal4 driver plus UAS-HP1E construct and males heterozygous (‘HP1E/TM6’) or homozygous (HP1E-) for the HP1E mutant chromosome.
- https://doi.org/10.7554/eLife.07378.026
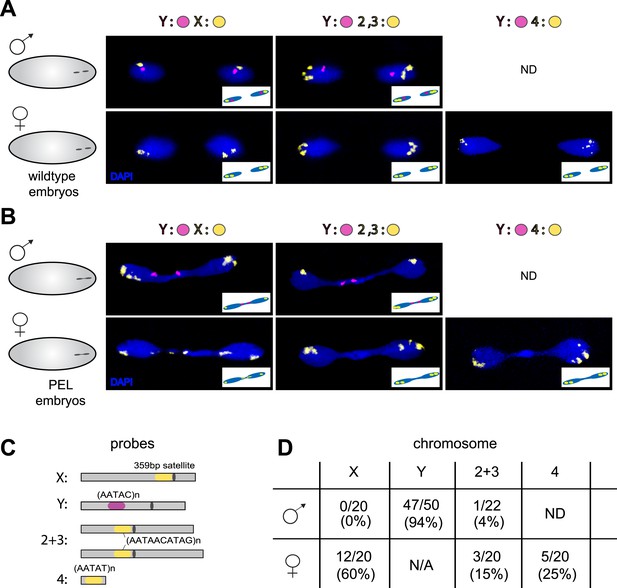
Sex chromosomes are especially vulnerable to HP1E depletion in D. melanogaster.
Representative images of fluorescent in situ hybridization (FISH) analyses of first zygotic telophase in (A) wild-type and (B) PEL embryos using chromosome-specific satellite probes (C), which recognize chromosome-specific repetitive elements (Dernburg, 2011). FISH probes against the Y chromosome were tested together with probes against either the chr. X probe (left) or chr. 2 + 3 probe (middle) or chr. 4 probe (right). (D) Using at least 20 images per probe pair per sex, we find that sex chromosomes are statistically enriched in the telophase bridge of PEL embryos. Quantification of chromosomal element appearance in the first telophase bridge in male and female embryos (PEL embryos) fathered by HP1E-depleted males. Hybridization of the fourth chromosome probe to the Y chromosome precluded data collection for this probe in male embryos. Data are reported as ‘obs/total/(%)’ where ‘obs’ = number of embryos observed with the probe appearing in the telophase bridge, ‘total’ = total number of embryos sampled per probe, and ‘%’ = obs × 100/total.
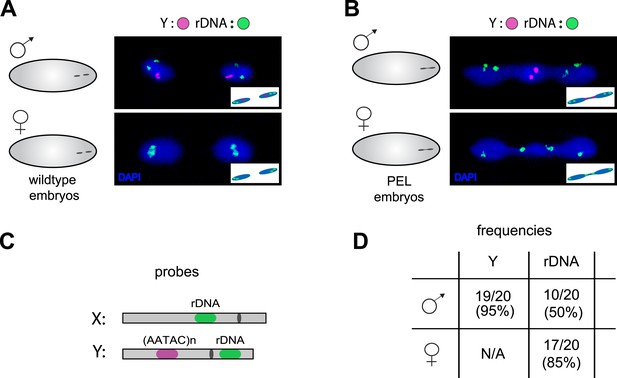
Localization of the paternal X- and Y-linked rDNA locus to the telophase bridge in female and male embryos fathered by the HP1E mutant (PEL embryos).
Representative images of FISH analyses of first zygotic telophase in (A) wild-type and (B) PEL embryos using (C) probes that recognize the Y-specific satellite AATAC (to determine sex of embryos) and rDNA (the intergenic spacer ‘IGS’ sequence). (D) Quantification of FISH signal in the first telophase bridge in male and female PEL embryos. Data are reported as ‘obs/total/(%)’ where ‘obs’ = number of embryos observed with the probe appearing in the telophase bridge, ‘total’ = total number of embryos sampled per probe, and ‘(%)’ = obs × 100/total.
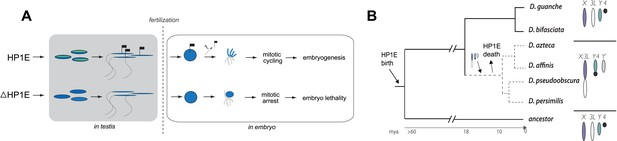
Proposed model for HP1E ‘hit and run’ priming of the paternal genome for timely entry into embryonic mitosis.
(A) HP1E localization to post-meiotic paternal chromatin directly or indirectly results in an epigenetic mark transferred to the embryo on sperm chromatin. This mark ensures synchronous paternal and maternal entry into the first embryonic mitosis. The absence of HP1E during postmeiotic sperm maturation leads directly or indirectly to the loss of an epigenetic mark (designated by the absence of the flag). Paternal chromatids fail to resolve and mitotic catastrophe ensues. (B) The loss of HP1E in the obscura group of Drosophila dates to the same 7 million-year long branch as a major karyotype innovation involving the sex chromosomes, including the birth of a neo-Y chromosome (Carvalho and Clark, 2005; Larracuente et al., 2010; Levine et al., 2012). For clarity, only the dynamic subset of the chromosomal elements is presented.
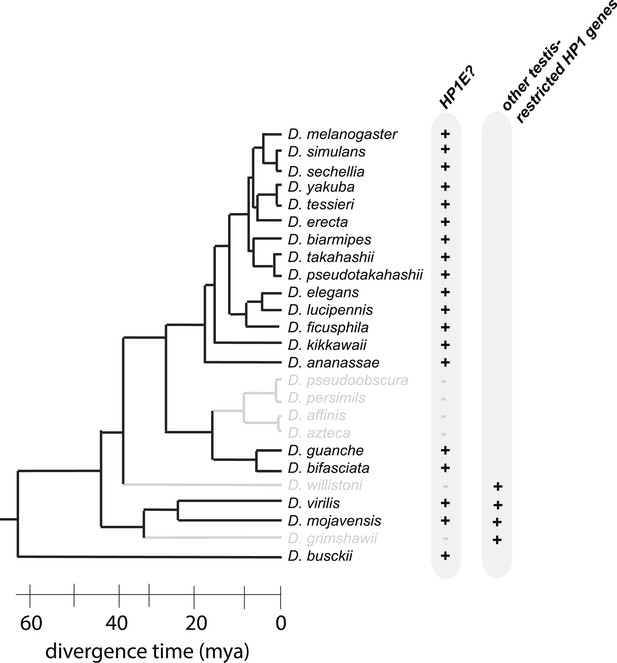
HP1E is present for more than 60 million years of Drosophila evolution but was lost at least three times over the Drosophila phylogeny.
D. willistoni and D. grimshawii each encode other testis-restrcited HP1 genes whereas D. pseuodobscura and its close relatives do not.





