Mechanical stress contributes to the expression of the STM homeobox gene in Arabidopsis shoot meristems
Figures
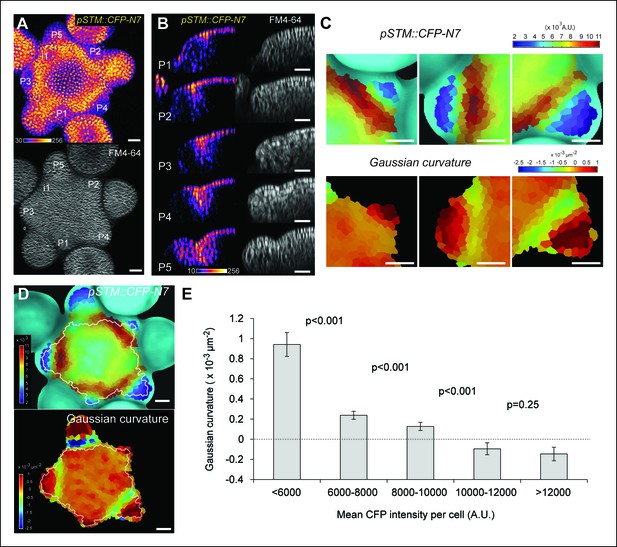
Correlation between pSTM::CFP-N7 expression level and tissue folding at the boundary.
(A) pSTM::CFP-N7 expression pattern in the SAM. Membranes are labeled with FM4-64 (white, lower panel) and pSTM::CFP-N7 expression is shown using the Fire lookup table in ImageJ (upper panel, n > 20). (B) Longitudinal optical sections (5 μm thick maximal projection of orthogonal views) through the middle of five successive boundaries of a representative meristem expressing pSTM::CFP-N7. Note the increase of pSTM::CFP-N7 signal intensity in the boundary as the crease between organ and meristem becomes deeper. (C) Close-ups showing a correlation between pSTM::CFP-N7 signal intensity (upper panels) and Gaussian curvature (lower panels, see Material and methods) in three successive boundaries of the meristem presented in A. (D and E) Quantification of the correlation between pSTM::CFP-N7 signal intensity (upper panel) and Gaussian curvature (lower panel) in the meristem presented in A (see Material and methods). (D) The white outline encloses the cells that are used for the graph presented in E. (E) pSTM::CFP-N7 signal intensity is plotted against Gaussian curvature. Values are compared using a bilateral Student test. The same correlation was observed in 5 independent meristems. Scale bars, 20 µm.

Time lapse imaging of a meristem recovering from NPA treatment and expressing pSTM::CFP-N7.
i1 and i2 marks the presence of new initia where the CFP-N7 signal decreases and the white arrows points toward a new developing boundary where the CFP-N7 signal increases. Note that t = h corresponds to 24 h after transfer to a NPA free medium. Scale bars: 20 µm.
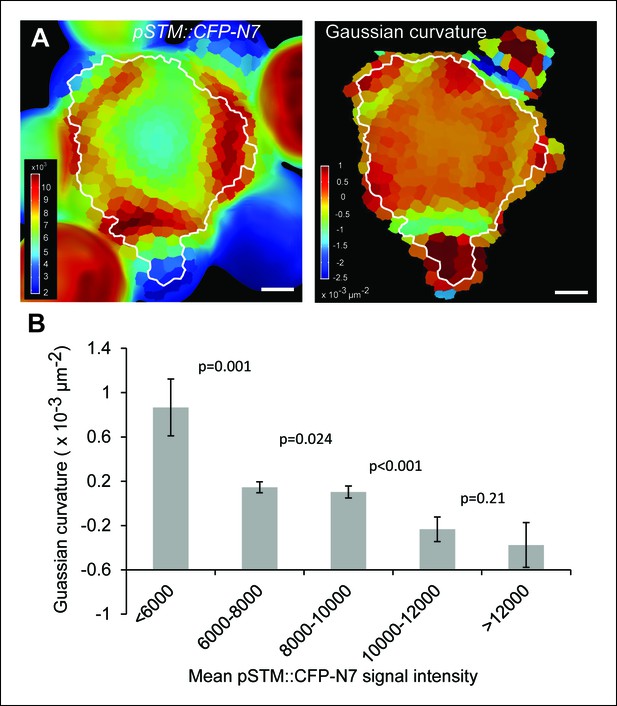
Correlation between pSTM::CFP-N7 expression level and tissue folding at the boundary.
(A) Correlation between pSTM::CFP-N7 signal intensity and Gaussian curvature in another meristem than the one presented in Figure 1. The white outline encloses the cells that are used for the graph presented in B (See Material and methods). (B) Quantitative measurement of the negative correlation between pSTM::CFP-N7 intensity and Gaussian curvature in the meristem presented in A (n = 231 cells). Successive values are displayed with a Student confidence interval (α = 0.05) and compared using a bilateral Student test. Scale bars: 20 µm.
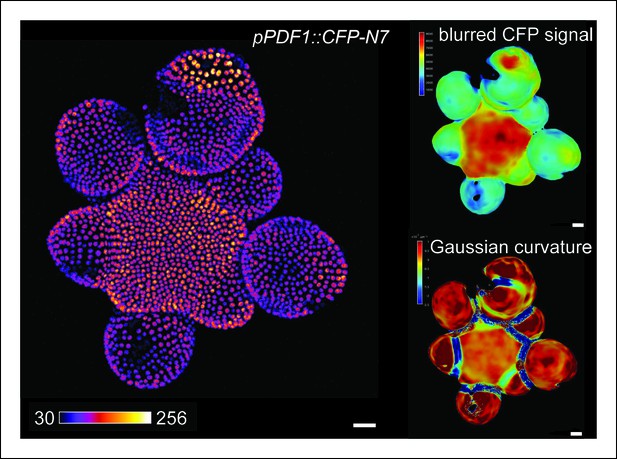
pPDF1::CFP-N7 expression pattern in the SAM.
Z-projection of a meristem expressing the L1 transcriptional reporter pPDF1::CFP-N7 (left panel). CFP-N7 Signal intensity map of the signal and Gaussian curvature extracted using the level set method and MorphoGraphX (right panels). Scale bars: 20 µm.
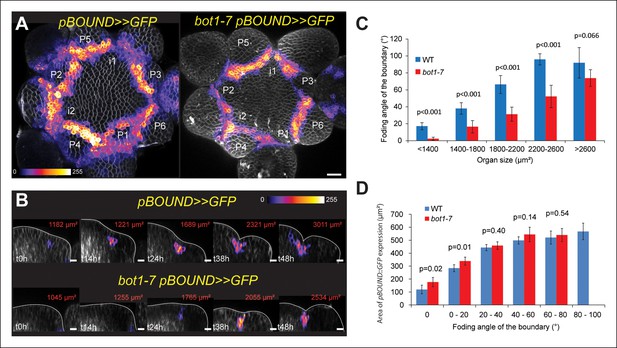
Correlation between pBOUND>>GFP expression level and tissue folding at the boundary in WT and bot1-7.
(A) pBOUND>>GFP expression pattern in WT (ecotype WS-4) and bot1-7 meristems. Membranes are labeled with FM4-64 (white) and pBOUND>>GFP expression is shown using the Fire lookup table in ImageJ. (B) Longitudinal sections through the middle of successive boundaries of the meristems presented in A (2 μm thick maximal projection of orthogonal views). Organ size (surface area as viewed from the top) is written in red for each stage. Note the delay in tissue folding and GFP signal expression in bot1-7 when compared to the WT. The white line marks the outer surface of the SAM. (C) Quantification of the delay in curvature at the boundary in bot1-7: Folding angle is measured on orthogonal views and organ size is estimated from the measurement of surface area on top views. (D) The correlation between the folding angle of the boundary and the area of pBOUND>>GFP expression is maintained in bot1-7 (both parameters are measured on orthogonal sections; WT: n = 130 from 5 SAM followed during a time lapse of 5 time points during 48 h, bot1-7: n = 79 from 3 SAM followed during a time lapse of 5 time points during 48 h). Values are displayed with a Student confidence interval (α = 0.05) and compared using a bilateral Student test. Scale bars: 20 µm.
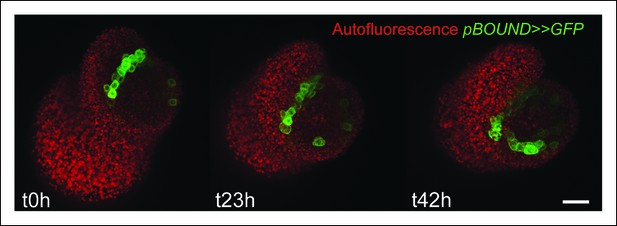
Time lapse of a pBOUND>>GFP meristem recovering from NPA treatment.
Note the signal induction in old and new boundaries. Scale bar, 20 µm.
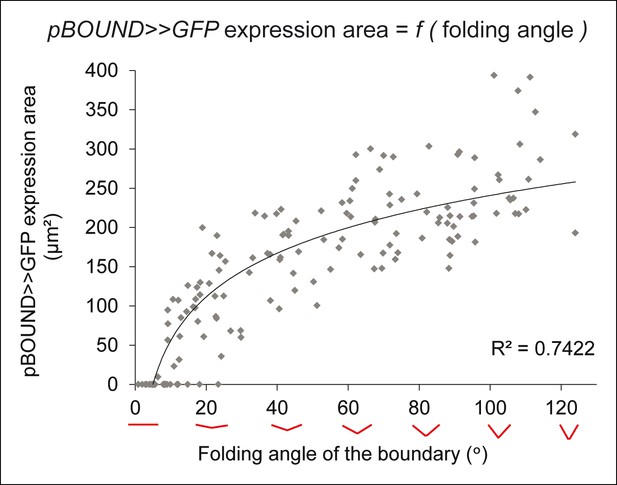
Correlation between pBOUND>>GFP expression area and tissue folding at the boundary.
Quantitative correlation between the folding angle of the boundary and the area of pBOUND>>GFP expression on longitudinal sections from another independent time course as the one presented in Figure 2 (n = 154, 5 meristems imaged 5 times over a time course of 48 hr).
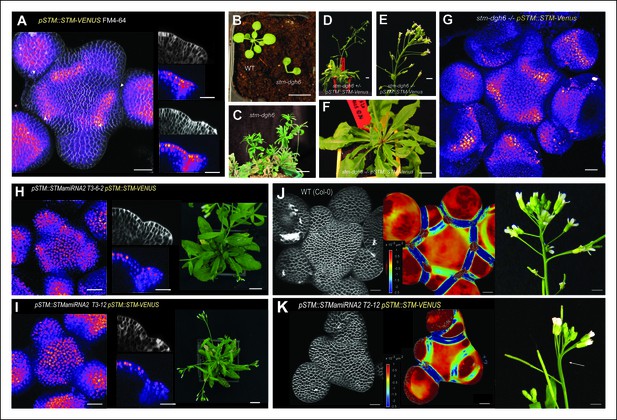
Organ separation requires STM expression at the boundary.
(A) Representative expression pattern of the translational fusion pSTM::STM-Venus in a FM4-64 stained meristem showing an increased signal intensity in boundaries. Scale bar, 20 μm. (B–F) The translational fusion pSTM::STM-Venus partially rescues the phenotype of the strong mutant allele stm-dgh6 (n = 5). (B) Phenotype of 3-week-old WT and stm-dgh6 plants. Note the absence of postembryonic organs in the mutant. Scale bars, 1 cm. Aerial phenotype of 2-month old stm-dgh6 plants. Scale bar, 1 cm. (D) Representative stm-dgh6 pSTM::STM-Venus plant. Scale bar, 1 cm. (E) Representative stm-dgh6 pSTM::STM-Venus inflorescence. Scale bar, 1 cm. (F) Representative stm-dgh6 pSTM::STM-Venus rosette. Scale bar, 1 cm. (G) Representative expression pattern of the translational fusion pSTM::STM-Venus in a stm-dgh6 (-/-) meristem showing a similar expression pattern as in the WT. Scale bar, 20 μm. (H, I) Homogeneous expression pattern of the translational fusion pSTM::STM-Venus in two independent pSTM::STMamiRNA lines. Scale bar (microscopy), 20 μm. Scale bar (whole plant), 1 cm. (J) FM4-64 stained WT meristem (ecotype Col-0), Gaussian curvature extracted using the level set method and MorphoGraphX, Scale bar (microscopy), 20 μm. Scale bar (inflorescences), 1 cm. (K) FM4-64 stained pSTM::STMamiRNA pSTM::STM-Venus meristem, Gaussian curvature extracted using the level set method and MorphoGraphX. Scale bar, 20 μm. Boundaries do not scale to the reduced meristem size; inflorescence phenotype with fusion events. Scale bar, 1 cm.

Molecular characterization of the stm-dgh6 pSTM::STM-Venus.
(A) 48 pSTM::STM-Venus plants segregating the stm-dgh6 mutation. (B) stm-dgh6 genotyping of the plants shown in A. No obvious difference can be detected between the different lines.
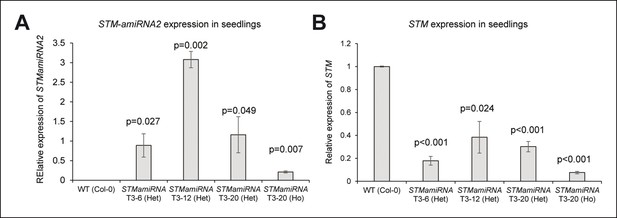
Molecular characterization of the pSTM::STMamiRNA lines.
(A) STMamiRNA expression in 2-week-old seedlings by qPCR in three independent pSTM::STMamiRNA lines (see Material s and methods). Values are displayed with a Student confidence interval (α = 0.05) and compared using a bilateral Student test. (B) STM expression in 2-weeks-old seedlings by qPCR in three independent pSTM::STMamiRNA lines, as in A.
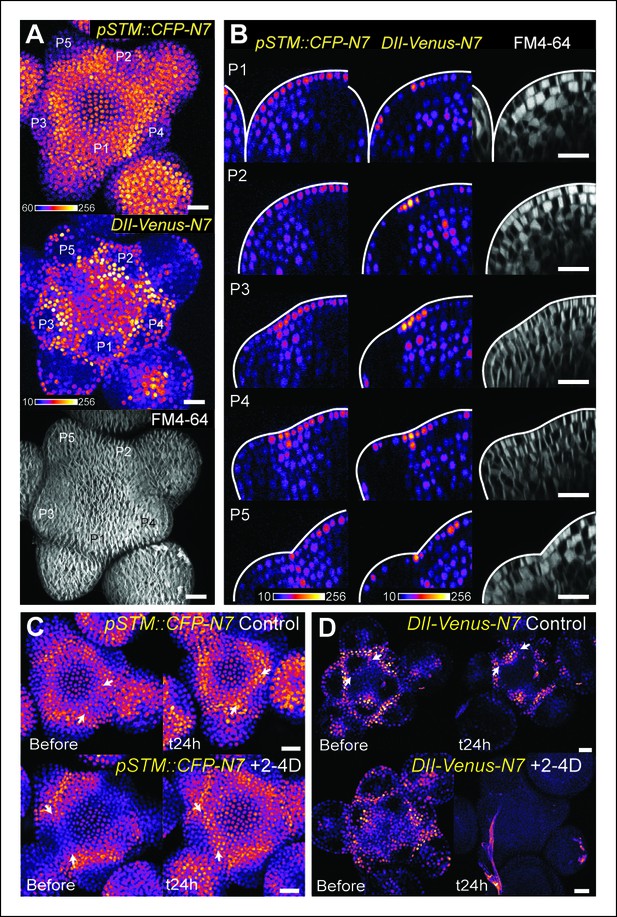
The DII-Venus and pSTM::CFP-N7 signals largely overlap and can be uncoupled.
(A) Projection of a representative meristem expressing both pSTM::CFP-N7 and DII-Venus-N7 and stained with FM4-64: both signals are induced in the boundary. (B) Orthogonal sections through the middle of the boundaries of the successive primordia of the SAM presented in showing an overlap of both signals, except in the L2 layer. (C, D) Overnight treatment with 10 μM of synthetic auxin 2,4-D on dissected meristems: (C) no effect on pSTM-CFP expression after 2,4-D application (Control: n = 3, 2-4D treatment: n = 3). White arrows point at new CFP signals in boundaries (C and D); (D) total degradation of DII-Venus after 2,4-D application (Control: n = 11, 2-4D treatment: n = 12). Scale bars, 20 µm.
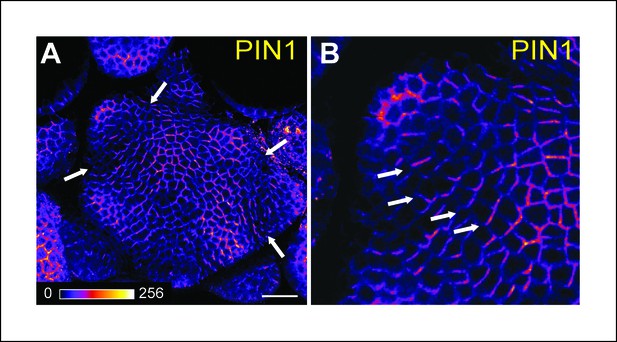
PIN1 localization in the SAM.
Whole-mount PIN1 immunolocalization in the SAM (n = 6). (B) close-up from (A). White arrows point at boundaries where the polarity of PIN1 is strengthened and predicts an auxin depletion in this domain. Scale bars, 20 µm.
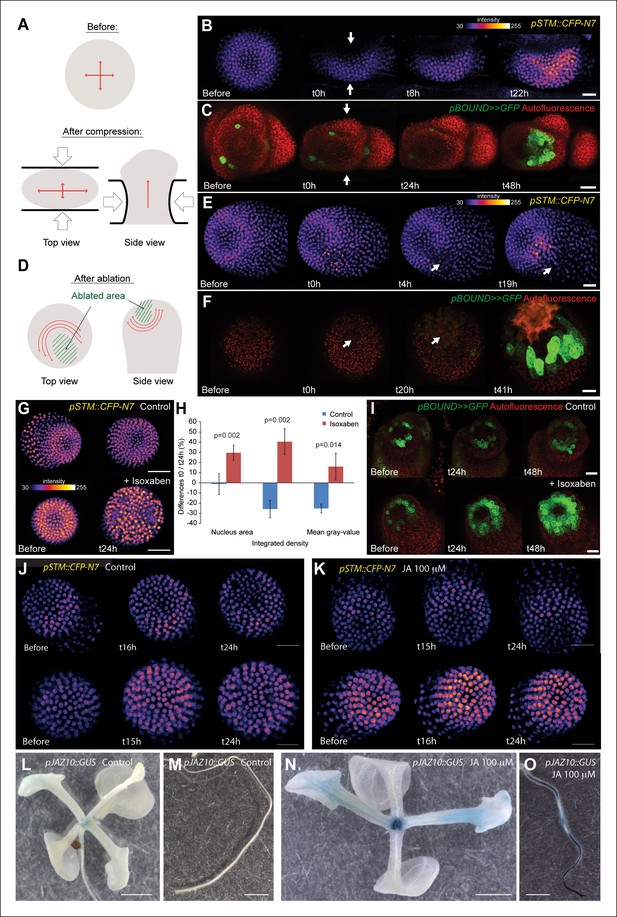
STM expression can be induced by mechanical perturbations.
(A to C) Global compression of meristems with a microvice lead to an increase in STM expression (arrows indicate the direction of the compression). (A) Predicted impact of compression on the mechanical stress pattern. (B) pSTM::CFP-N7 signal before and after compression in a representative meristem (n = 8). (C) pBOUND>>GFP signal before and after compression in a representative meristem (n = 11, red dots correspond to plast auto-fluorescence). (D to F) Ablation of a small number of cells leads to an increase in STM expression (white arrows indicate the site of ablation). (D) Predicted impact of a local ablation on the mechanical stress pattern. (E) pSTM::CFP-N7 signal before and after ablation with a needle (n > 30). (F) pBOUND>>GFP signal before and after ablation using a pulsed UV laser (n > 12, red dots correspond to plast auto-fluorescence). (G and H) Isoxaben treatment leads to an increase of pSTM::CFP-N7 signal. (G) Representative pSTM::CFP-N7 signal after overnight immersion in water with DMSO (upper panel) or in 10 μM isoxaben (lower panel). Note the increased nucleus size after isoxaben treatment, consistent with increased endoreduplication levels. (H) Quantifications: CFP signal intensity in 10 nuclei from the central zone of 6 isoxaben-treated meristems and 7 water-treated meristems. Values are displayed with a Student confidence interval (α = 0.05) and compared using a bilateral Student test. (I) Isoxaben treatment leads to an increase in pBOUND>>GFP signal (red dots correspond to plast auto-fluorescence). (I, left) Representative pBOUND>>GFP signal after overnight immersion in water (n = 10). (I, right) Representative pBOUND>>GFP signal after overnight immersion in 5 to 20 μM isoxaben (n = 20). Scale bars, 20 µm. (J, K) Jasmonate does not enhance STM promoter activity (J) pSTM::CFP-N7 signal after prolonged incubation in water supplemented with 1/1000 V/V ethanol. (K) pSTM::CFP-N7 signal after prolonged incubation in water supplemented with 100 µM jasmonate diluted in ethanol (1/1000 V/V). Scale bars, 20 µm. (L-O) Jasmonate enhances pJAZ10 promoter activity. (L, M) Aerial part (L) and root (M) of 3 week old NPA grown seedlings. pJAZ10::GUS staining after overnight incubation in water supplemented with 1/1000 V/V ethanol (n = 8). (N,O) Aerial part (N) and root (O) of 3 week old NPA grown seedlings. pJAZ10::GUS staining after overnight incubation in water supplemented with 100 µM jasmonate diluted in ethanol (1/1000 V/V) (n = 14). Scale bars, 0.5 cm.
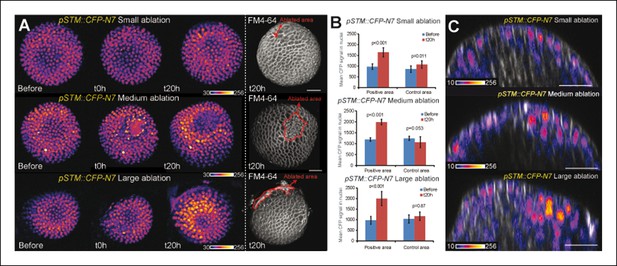
pSTM::CFP-N7 induction after ablations of different sizes.
(A) Z-projections of meristems expressing pSTM::CFP-N7 and labeled with FM4-64 (white) before and after ablations of different sizes. (B) Quantification of the CFP signal intensity in 10 nuclei around the ablation site (positive area) or in the opposite side of the meristem (control area) of the three meristems presented in A. Values are displayed with a Student confidence interval (α = 0.05) and compared using a bilateral Student test. (C) Longitudinal sections (5 µm thick maximal projections) of the ablated meristems presented in A, 20 hr after the ablations revealing that the induction of pSTM::CFP-N7 roughly scales to the ablation size. Scale bars, 20 µm.
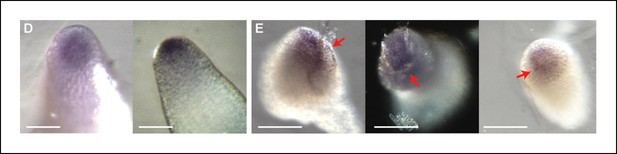
STM mRNA distribution after ablation in the SAM.
(A) Whole mount in situ hybridizations using a STM probe in a SAM from WT NPA grown plants. (B) Whole mount in situ hybridizations using a STM probe in a SAM from WT NPA grown plants 24 hr (right and left panels) and 48 h (central panel) after ablation. Red arrows point at the ablation sites. Scale bars, 100 µm.
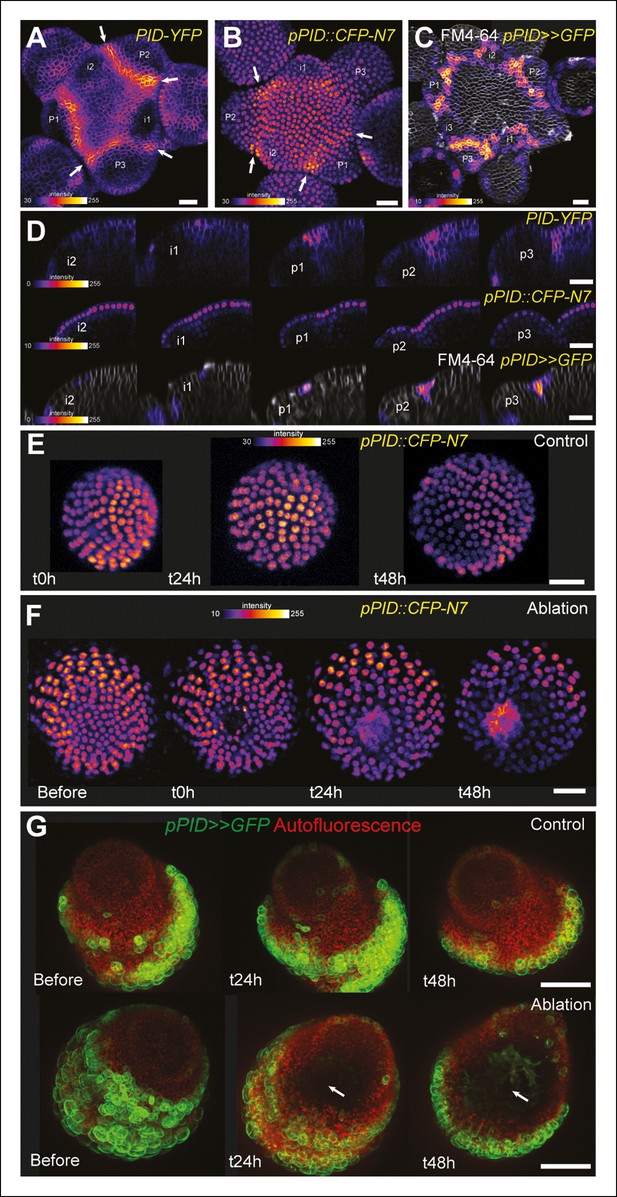
PINOID promoter activity is not affected by mechanical perturbations.
(A–C) PINOID expression pattern in representative meristems: a higher expression of PINOID is observed in boundaries. (A) Expression pattern of the translational fusion pPID::PID-YFP. (B) Expression pattern of the transcriptional reporters pPID::CFP-N7. (C)Expression pattern of pPID>>GFP. (D) Orthogonal sections through the middle of the boundaries of the meristems presented in (A–C). (E–F) Time lapse of a representative meristems showing the absence of response of pPID::CFP-N7 (F, n = 13) after ablation when compared to the control (E, n = 6). (G) Time lapse of a representative meristems showing the absence of response of pBOUND>>GFP after ablations (control n = 14, ablation n = 7). Scale bars, 20 µm.
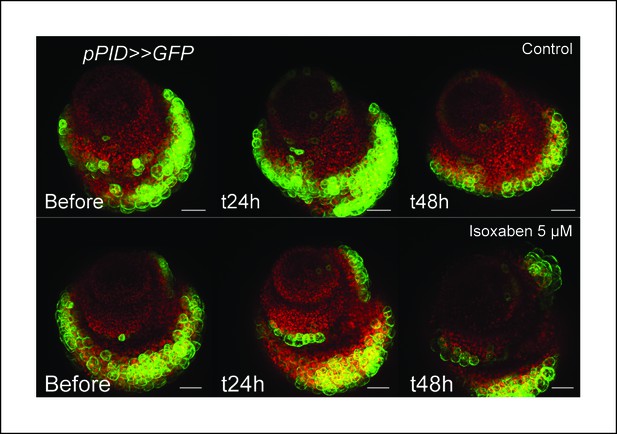
pPID is not significantly induced by isoxaben treatment.
Overnight treatment with isoxaben does not lead to an induction of pPID::GFP expression, even 48 h after the first exposition to isoxaben. (Control, n = 14; isoxaben treatment, n = 11). Scale bars, 20 µm.
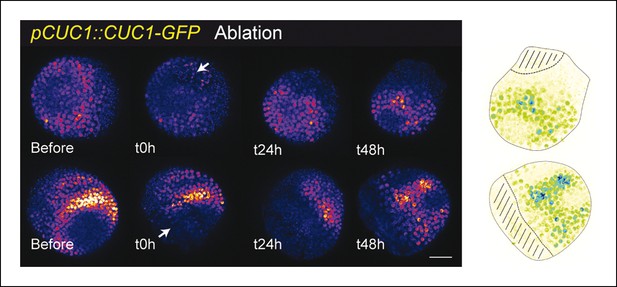
pCUC1 is not significantly perturbed after an ablation in the SAM.
pCUC1::CUC1-GFP expression after an ablation (arrow). No major changes are induced, and the pattern follows the organogenetic pattern instead of consolidating around the ablation site (n = 11). Scale bar, 20 µm. Drawings illustrate the last time points, with the hatched zone corresponding to the ablated zone.
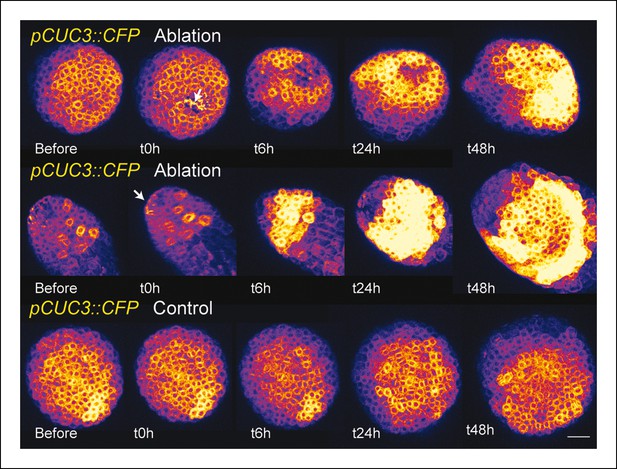
pCUC3 is induced after an ablation in the SAM.
pCUC3::CFP expression after an ablation. Note the steady induction, when compared to control (bottom line, n = 20). Scale bar, 20 µm.
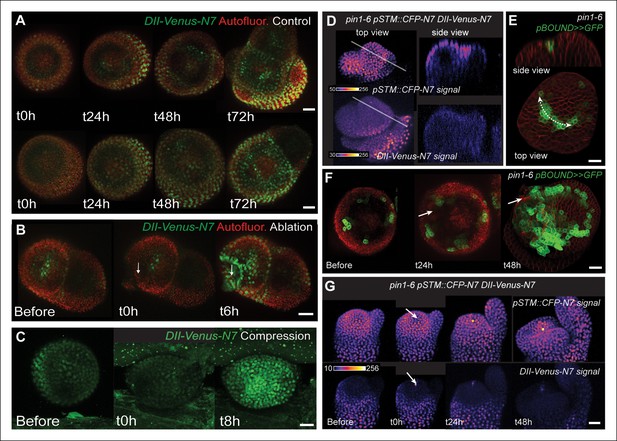
STM response to mechanical perturbations can be uncoupled from PIN1-dependent auxin distribution.
(A) Time lapse of representative meristems from a NPA-grown plant and expressing DII-Venus. From t = h, the plants are not exposed to NPA anymore. (B) DII-Venus-N7 signal increases after ablation: Time lapse of a representative meristems from a NPA-grown plant and expressing DII-Venus as in C, after ablation (Control n = 15, Ablation n = 21). (C) Representative DII-Venus signal before and after compression. An increased signal is usually detected after 4 to 8 hr after compression in the overall meristem (n = 10). (D) Representative pin1-6 meristem expressing DII-Venus-N7 and pSTM::CFP-N7: the presence of CFP signal at the pseudo-boundary does not correlate with DII-Venus-N7 signal anymore (n = 6). (E) Representative pin1-6 meristem expressing pBOUND>>GFP showing the presence of GFP signal in a pseudo-boundary (n = 7). (F) Representative pin1-6 pBOUND>>GFP meristem after ablation: pBOUND>>GFP is induced around the site of ablation (n = 14). (G) Representative pin1-6 DII-Venus-N7 pSTM::CFP-N7 meristem after ablation: pSTM::CFP-N7 is induced around the site of ablation but DII-Venus-N7 is not (n = 10). Scale bars, 20 µm.
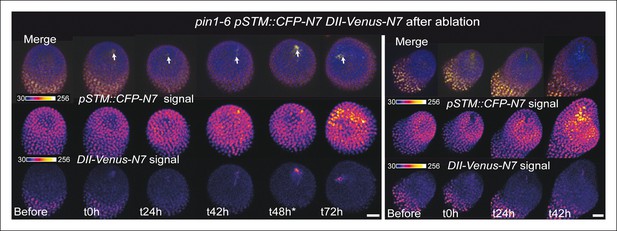
pin1-6 DII-Venus-N7 pSTM::CFP-N7 meristem after an ablation in the SAM.
Two representative pin1-6 DII-Venus-N7 pSTM::CFP-N7 meristem after ablation (time-lapse): pSTM::CFP-N7 is induced around the site of ablation but not DII-Venus-N7 (n = 10).
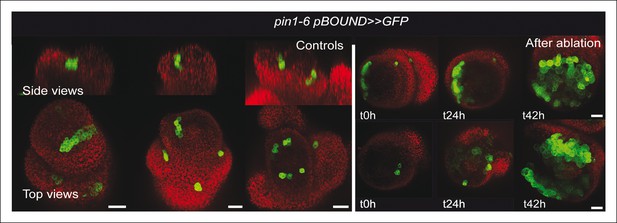
pin1-6 pBOUND>>GFP meristem after an ablation in the SAM.
Representative pin1-6 meristems expressing pBOUND>>GFP showing the presence of GFP signal in a pseudo-boundary (n = 7) and an induction of GFP signal after ablation (n = 14).
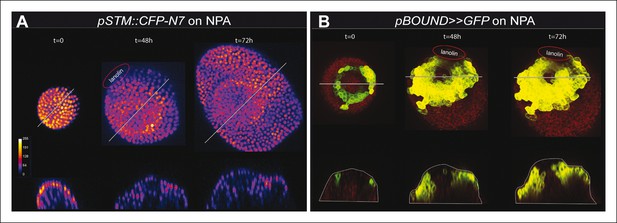
STM promoter activity during oryzalin-induced tissue folding in the presence of NPA.
(A) Top and side views of a pSTM::CFP-N7 meristem grown on NPA and maintained on NPA after the local application of the microtubule depolymerizing drug oryzalin. A bump is induced and a local increase in CFP signal is detected at the pseudo-boundary (n = 17/22). (B) Top and side views of a pBOUND>>GFP meristem grown on NPA and maintained on NPA after the local application of the microtubule depolymerizing drug oryzalin. A bump is induced and a local increase in GFP signal is detected at the pseudo-boundary (n = 20/21). Scale bars, 20 µm.
Videos
pSTM::CFP-N7 after compression (relates to Figure 5B).
https://doi.org/10.7554/eLife.07811.018pBOUND>>GFP after compression (relates to Figure 5C).
https://doi.org/10.7554/eLife.07811.019pSTM::CFP-N7 after an ablation (relates to Figure 5E).
https://doi.org/10.7554/eLife.07811.020pBOUND>>GFP after an ablation (relates to Figure 5F).
https://doi.org/10.7554/eLife.07811.021DII-Venus-N7 after an ablation (relates to Figure 7B).
https://doi.org/10.7554/eLife.07811.030Additional files
-
Supplementary file 1
Summary of the number of replicates for each mechanical test.
- https://doi.org/10.7554/eLife.07811.031





