Regulation of mRNA translation during mitosis
Figures
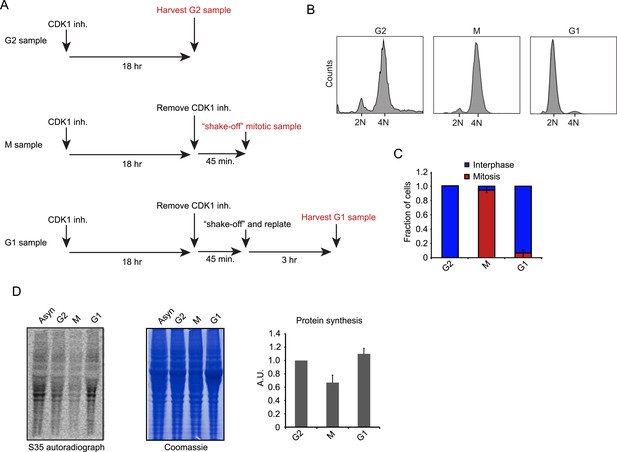
Cell synchronization and analysis of translation efficiency during the cell cycle.
(A) Schematic overview of RPE-1 cell synchronization protocol. RO-3306 (6 μM) was used as the CDK1 inhibitor. (B, C) G2, M and G1 samples were prepared as outlined in (A). (B) FACS analysis (Hoechst staining of DNA) reveals that the samples are effectively synchronized in the respective cell cycle phase. (C) The number of mitotic cells was scored by microscopy based on chromosome condensation (DNA stained with DAPI). Graph is average of 3 independent experiments with ∼50 cells scored per experiment. Error bars represent standard error of the mean (SEM). (D) RPE-1 cells were synchronized as described in (A). Before harvesting, cells were incubated with S35-methionine for 10 min to radioactively label newly synthesized proteins. The left panel shows the autoradiograph of newly synthesized proteins. The middle panel shows total protein content of the cells stained by Coomassie. Right panel shows quantification of autoradiographs of 3 independent experiments, normalized to total protein. Mean and standard deviation (SD) are shown.
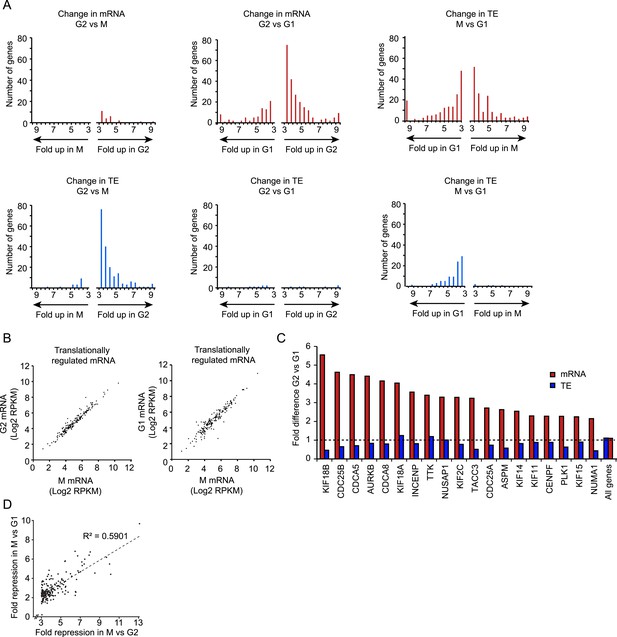
Transcriptional and translational regulation affect distinct cell cycle transitions.
(A) For each gene the ratio of mRNA levels (red bars) and translation efficiency (TE) (blue bars) was determined for G2 vs M (left) or G2 vs G1 (middle) and M vs G1 (right). The number of genes that showed changes in mRNA levels (>threefold difference, red bars) or changes in TE (or >threefold difference in TE combined with >twofold difference in the ribosome footprint value, blue bars) was plotted in a pair of histograms. (B) mRNA levels are plotted for all genes that are translationally regulated in M vs G2. The left graph shows the mRNA levels in M compared to G2, the right graph shows mRNA levels of M compared to G1. Note that mRNA levels of translationally regulated mRNAs are similar in G2, M and G1. (C) 19 well characterized cell cycle proteins that show strong cell cycle-dependent regulation of mRNA levels were manually selected. Fold difference between G2 and G1 in mRNA levels (red bars) and TE (blue bars) is shown. While there is a large change in mRNA levels, the TE is similar in G2 and G1 for the majority of these mRNAs. (D) The subset of genes that was translationally repressed in M compared to G2 (182 genes) was selected and the fold difference in TE for M vs G2 was plotted against the fold difference in TE for M vs G1. Results show that genes which are translationally repressed in M compared to G2, or repressed to similar levels in M when compared to G1.
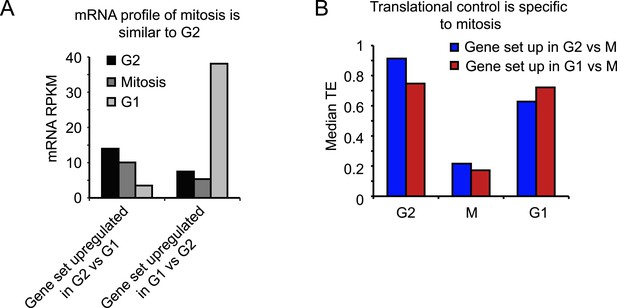
Transcriptional and translational regulation affect distinct cell cycle transitions.
(A) The median mRNA RPKM value was determined for the set of genes for which the mRNA was higher (>threefold) in G2 compared to G1 (left), or higher in G1 compared to G2 (right). Graphs show that mitotic mRNA levels are more similar to G2 than to G1. (B) The subset of genes that was translationally repressed in M compared to either G2 (182 genes, blue bars) or compared to G1 (86 genes, red bars) was selected. For both these gene sets, the median TE value was determined for G2, M and G1. Results show that genes which are translationally repressed upon mitotic entry, are re-activated upon mitotic exit.
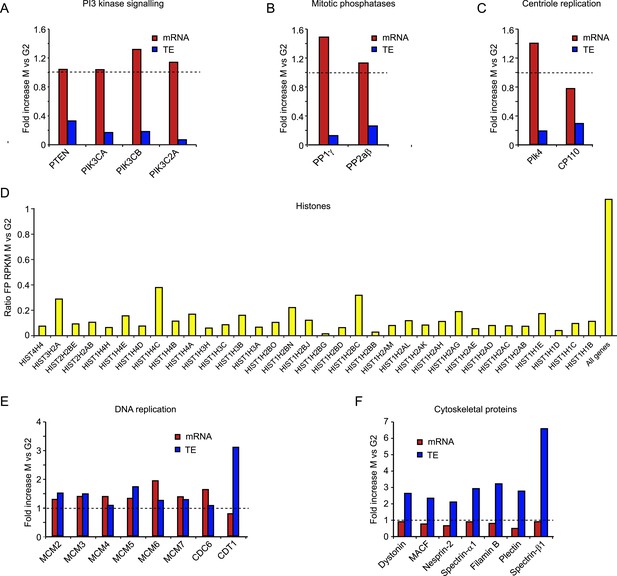
Translational regulation affects many cell cycle-dependent processes.
(A, B, C, E, F) mRNA levels (red bars) and TE (blue bars) were compared between M and G2 for multiple genes in the same biological pathway. (D) Since most histone mRNAs do not contain poly(A) tails and are thus not enriched in the total mRNA sample, the footprint RPKM values were compared between M and G2.
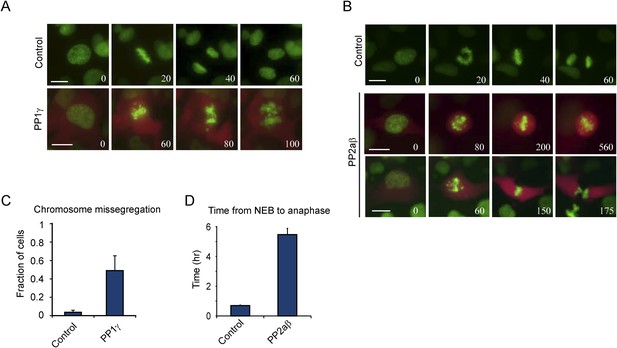
Excessive PP1γ and PP2aβ activity perturbs chromosome segregation.
(A–D) RPE-1 cells expressing either H2B-GFP alone or together with mCherry and PP1γ (A, C) or mCherry and PP2aβ (B, D) were analyzed by time-lapse microscopy. Simultaneous expression of mCherry and untagged PP1γ or PP2aβ was accomplished by inserting a P2A ribosome skipping sequence in between mCherry and the phosphatase sequence. (A) Stills from representative videos of control cell (upper panel) or PP1γ overexpressing cell (lower panel). Time is shown in min. (B) Stills from representative video of control cell (upper panel) or PP2aβ expressing cells (lower two panels). The top PP2aβ expressing cell shows a prometaphase arrest with misaligned chromosomes, while the bottom cell shows a cell with a prometaphase delay and subsequent cytokinesis without chromosome segregation, known as a ‘cut’ phenotype. Time is shown in min. (C) The fraction of cells in which one or more chromosomes mis-segregated was determined for control cells and cells expressing PP1γ. (D) shows the average time from NEB to anaphase for control cells and cells expressing PP2aβ. All graphs are the mean and SD of 3 independent experiments with 20–40 cells analyzed per experiment. Scale bars, 10 μm.
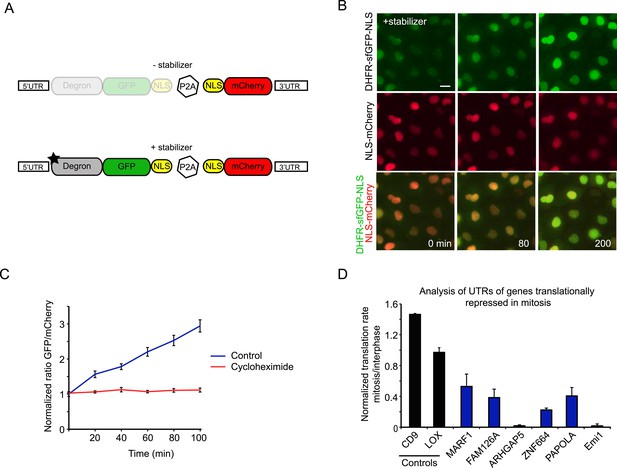
Analysis of translation efficiency in living cells using a fluorescence-based translation reporter.
(A) Schematic representation of the live-cell translation reporter. An inducible degron (DHFR-Y100I) fused to sfGFP and an NLS is separated from an NLS-mCherry protein by a P2A ribosome skipping sequence, which allows these two proteins to be synthesized as separate proteins from a single transcript. Upon addition of the small molecule stabilizer trimethoprim (TMP), newly synthesized DHFR-sfGFP-NLS is stabilized and GFP fluorescence increases over time due to new GFP protein synthesis. Thus, GFP fluorescence increase reports on translation efficiency. The mCherry signal is used to normalize for the plasmid copy number per cell. (B) RPE-1 cells stably expressing the reporter were treated with 50 μM TMP and followed by time-lapse microscopy. Scale bar, 20 μm. Time is indicated in min. (C) Quantification of GFP/Cherry ratio (mean and standard deviation, n = 8 cells) with or without cycloheximide treatment. (D) 5′ and 3′ UTRs from indicated genes were inserted in the reporter. Cells expressing the different reporters were blocked in mitosis with taxol, treated with TMP and imaged for 4 hr. To determine the translation rate, the GFP/mCherry ratio was calculated at the start and end of each video for both interphase and mitotic cells. The ratio of translation rates in mitosis and interphase for each reporter is shown. For Emi1, mitotic cells were compared with G2 phase cells only, as translation was also reduced in G1 (unpublished observation). Results are mean and SEM of 3 independent experiments with 10–20 cells analyzed per condition per experiment.
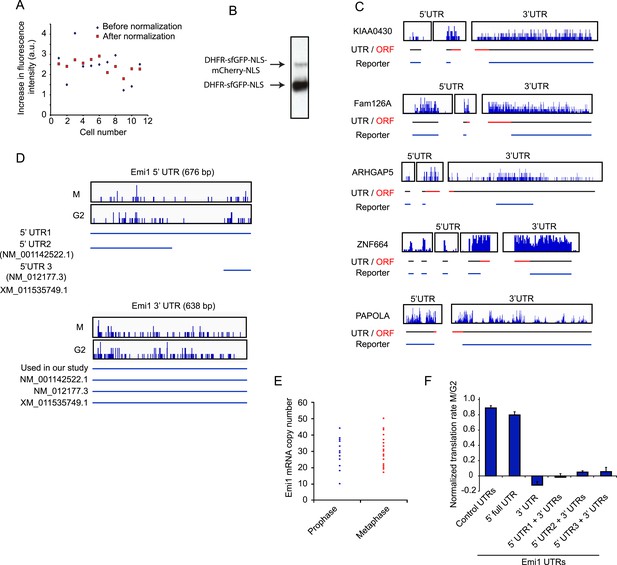
Analysis of mRNA sequences that confer translational regulation.
(A) RPE-1 cells expressing the fluorescent reporter with control UTRs were treated with TMP for 3 hr and changes in GFP and mCherry signals were followed by time-lapse microscopy. The total increase in GFP fluorescence was measured per cell and after background subtraction was plotted in the graph (blue diamonds, each diamond represents 1 cell). mCherry signal was measured at the start of the video and the absolute increase in GFP fluorescence was divided by the initial mCherry signal to normalize for reporter copy number per cell (red squares). Note that the spread after normalization is smaller. (B) Western blot of RPE1 cells expressing the translational reporter and treated with TMP for 24 hr probed for GFP. Note that two bands are visible; the bottom bands represents the protein product after successful ribosome skipping at the P2A site, while the weak upper band is a fusion protein that is generated after failure of ribosome skipping at the P2A site. (C, D) Visual representation of RNA-seq data of the 5′ and 3′ UTRs of indicated genes. Individual sequencing reads are mapped onto the UTR sequences, in which the 5′ nucleotide of each sequencing read is shown. RNA-seq data are shown for G2 samples (C) or for both both G2 and M cells (D). (E) Untreated RPE-1 cells were fixed and Emi1 mRNA was detected by single molecule FISH. The number of mRNAs was counted in either prophase or metaphase (as determined by DNA condensation and chromosome alignment). (F) Either control UTRs or indicated Emi1 UTRs were inserted in the fluorescence translation reporter (note: 5′ UTR numbering refers to numbered isoforms shown in [D]). Cells expressing the different reporters were left unsynchronized or blocked in mitosis with taxol, treated with TMP and imaged for 8 hr. Cells were defined as G2 cells if they entered mitosis within 5 hr. To determine the translation rate, the GFP/mCherry ratio was calculated at the start and end of a 3 hr interval. The ratio of translation rates in M and G2 for each reporter is shown. Results are mean and SD of 3 independent experiments with 10 cells analyzed per condition per experiment. Note that complete translational inhibition is expected to result in a slightly negative value for the normalized translation rate, because GFP levels will actually be lower at the end of the 3 hr measurement interval if no new GFP is synthesized, due to the decay of existing GFP protein.
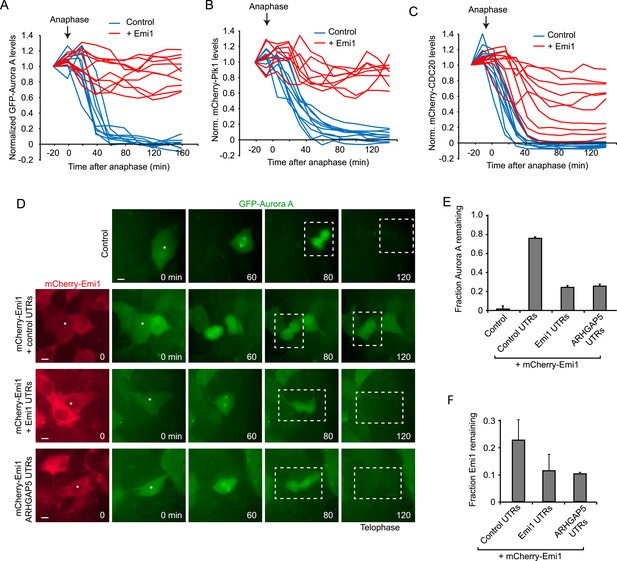
Translational inhibition of Emi1 during mitosis promotes APC/Cdh1 activation in telophase.
(A–C) RPE-1 cells expressing Aurora A-GFP (A), mCherry-Plk1 (B) or mCherry-CDC20 (C) alone or combined with fluorescently tagged Emi1 were analyzed by time-lapse microscopy and the protein degradation rates were assayed through quantification of fluorescence intensities over time as cells progressed through mitosis. (D–F) RPE-1 cells stably expressing Aurora-GFP and, where indicated, mCherry-Emi1 with indicated UTRs, were analyzed be time-lapse microscopy. Representative images (D) and quantification of Aurora A-GFP levels (E) and mCherry-Emi1 levels (F) are shown. Asterisks and dotted boxes mark dividing cells. Degradation of Aurora A normally occurs between anaphase onset and telophase. For quantification only cells were included that had very low mCherry-Emi1 fluorescence to ensure low expression level of exogenous Emi1 (see also Figure4—figure supplement 1C,D). Scale bar, 10 μm. Mean and standard error of 3 independent experiments, with ∼10 cells analyzed per experimental condition per experiment.
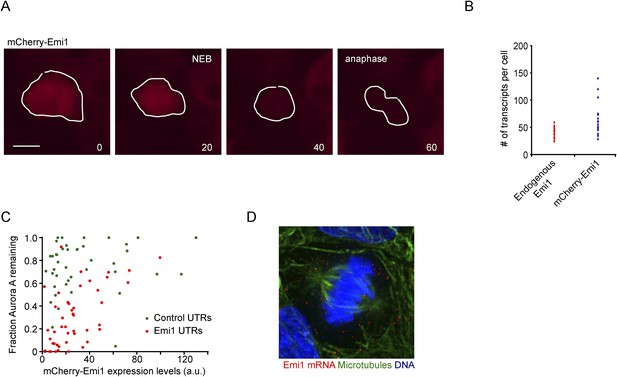
Translational repression of Emi1 by its UTRs facilitates APC activation.
(A) RPE-1 cells stably expressing mCherry-Emi1 were followed by time-lapse microscopy. Representative images are shown of a cell expressing mCherry-Emi1 undergoing cell division when Emi1 protein is degraded. (B) Either control RPE-1 cells or RPE-1 cells stably expression mCherry-Emi1 were fixed and mRNA was visualized by single molecule FISH. The number of endogenous Emi1 mRNA molecules or exogenous mCherry-Emi1 mRNA molecules was quantified in G2 phase of the cell cycle. (C) RPE-1 cells stably expressing Aurora A-GFP together with mCherry-Emi1 with either control UTRs or Emi1 UTRs were followed by time-lapse microscopy. Fluorescence intensities of Aurora A-GFP in prophase and telophase/early G1 were measured, and the fraction of GFP fluorescence remaining in telophase/early G1 compared to prophase was plotted on the y-axis. The fluorescence intensity of mCherry-Emi1 was also measured in prophase (before Emi1 degradation) and was plotted on the x-axis. Each dot represents a single cell. Graph shows that at similar mCherry-Emi1 protein expression Aurora A is degraded more efficiently when Emi1 is under control of its own UTRs. (D) Untreated RPE-1 cells were fixed and Emi1 mRNA was visualized by single molecule FISH. Microtubules and DNA were stained with an anti-α-tubulin antibody and DAPI, respectively. Scale bars, 10 μm.
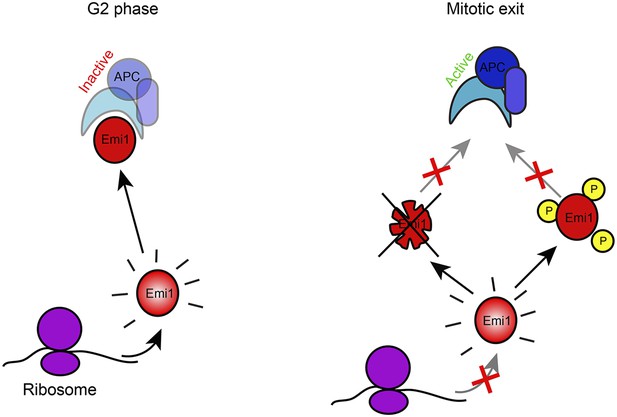
Model of Emi1 regulation in mitosis.
Model of Emi1 regulation in G2 and M. In G2, new Emi1 protein is continuously synthesized and existing Emi1 protein is stable allowing robust buildup of Emi1 protein levels and inhibition of APC/Cdh1 (left). In mitosis, Emi1 synthesis is repressed and pre-existing Emi1 protein is inactivated through protein degradation and CDK1-dependent phosphorylation, resulting in full inhibition of Emi1 activity, allowing APC/Cdh1 activation in telophase (middle) (Note- additional Emi1-independent mechanisms (not depicted) keep APC/Cdh1 inactive in (pro)metaphase).
Videos
Video of two RPE-1 cells going through cell division.
The first cell to divide expresses both Aurora A-GFP and mCherry-Emi1, while the second cell only expresses Aurora A-GFP Note that upon division, Aurora A-GFP degradation is perturbed in the cell expressing mCherry-Emi1. Time interval between images is 20 min.
Additional files
-
Supplementary file 1
Quality control of the deep sequencing data set. (A) The number of reads for the two biological replicates of the G2 FP sample was plotted on a log2 scale. Only genes with an average of at least 100 reads per replicate where selected for subsequent analysis. The box marked off by the two black lines indicates the genes that were excluded from the analysis, as they had less than 200 reads total. (B) After filtering the data (see ‘Materials and methods’) the ratio of the RPKM values was determined for all genes of each set of replicates. The standard deviation was determined for each set of replicates. The data sets of the two replicates were also fit and the R2 for the fit was determined.
- https://doi.org/10.7554/eLife.07957.014
-
Supplementary file 2
Genome-wide mRNA levels in G1, G2 and M phase cells.
- https://doi.org/10.7554/eLife.07957.015
-
Supplementary file 3
Ribosome profiling dataset.
- https://doi.org/10.7554/eLife.07957.016





