A simple retinal mechanism contributes to perceptual interactions between rod- and cone-mediated responses in primates
Figures
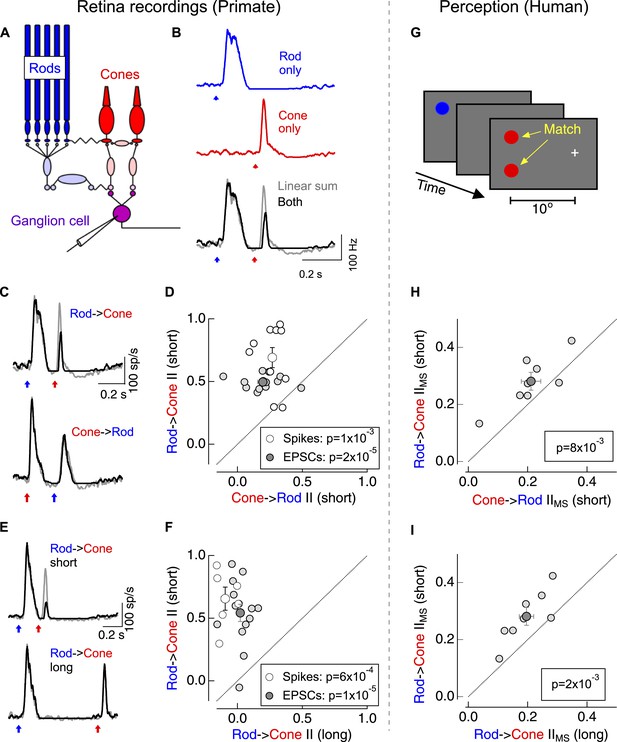
Asymmetric nonlinear rod-cone interactions.
(A) Diagram of the retinal circuits that convey rod- and cone-generated signals to the brain. Dim short-wavelength light preferentially activates rod photoreceptors whereas long-wavelength light preferentially activates long wavelength (L) cone photoreceptors (see Figure 1—figure supplement 2). (B) Protocol for testing for nonlinear rod-cone interactions electrophysiologically. Neural responses in ON parasol ganglion cells to rod (top) and cone (middle) flashes were recorded separately, and the sum of these responses (bottom, gray) was compared to trials in which the rod and cone flashes were delivered together (bottom, black). (C) Average spike responses from an example cell comparing rod → cone (top) and cone → rod (bottom) interactions. (D) Summary data across cells comparing rod → cone and cone → rod interaction indices for spikes and excitatory postsynaptic currents (EPSCs). (E) Average spike responses from an example cell comparing rod → cone interactions for short (0.2 s, top) and long (0.8 s, bottom) time offsets. (F) Summary data across cells: short intervals were 0.2–0.3 s and long intervals were ≥0.8 s. (G) Protocol used to test for nonlinear rod-cone interactions in human perception (also see ‘Materials and methods’ and Figure 1—figure supplement 3). Observers fixated on a cross while rod and cone flash sequences (similar to electrophysiology stimuli) were delivered to their peripheral retina (∼10° eccentricity). Observers compared the perceived brightness of two test flashes, one in the same location as the initial flash and the other spatially offset. (H) Comparison of perceptual rod → cone and cone → rod interactions for 8 human observers. (I) Comparison of perceptual rod → cone interactions for short and long time offsets. Each marker in panels D and F represents the average interaction for a single ganglion cell. Each marker in H and I represents the average monocular pathway-specific interaction for a single observer (see ‘Materials and methods’ and Figure 1—figure supplement 3). All retinal recordings (B–F) from whole mount retina.
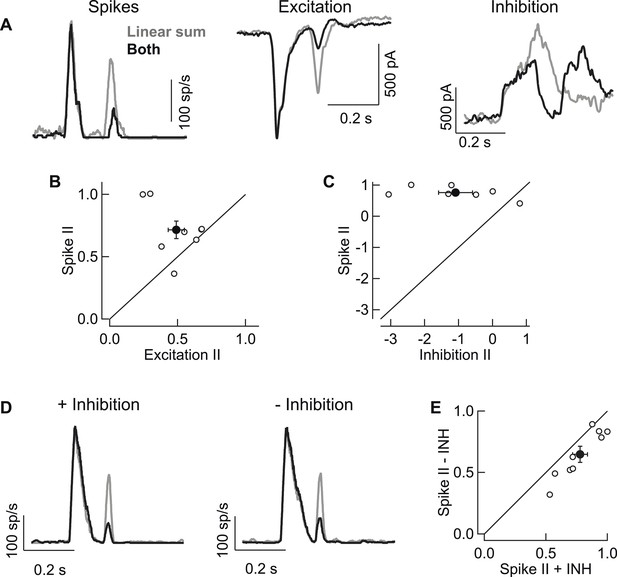
Rod → cone interactions in RGC spike output are largely dependent on interactions present in the RGC's excitatory synaptic inputs.
(A) Single cell example of rod → cone interactions in spikes (left), excitatory synaptic input (middle) and inhibitory synaptic input (right). (B) Interaction indices (II) for excitatory inputs resembled those observed in spikes. (C) Interactions in inhibitory inputs were highly variable and did not match spike interactions observed in the same cells. (D) Example cell from dynamic-clamp experiments to test contributions of inhibitory synaptic input to rod → cone interactions in spike output. Spike responses elicited by excitatory and inhibitory inputs (left) were compared to those elicited by excitatory inputs alone (right). (E) Population data plotting interaction indices from dynamic-clamp experiments in D. All recordings from whole mount retina.
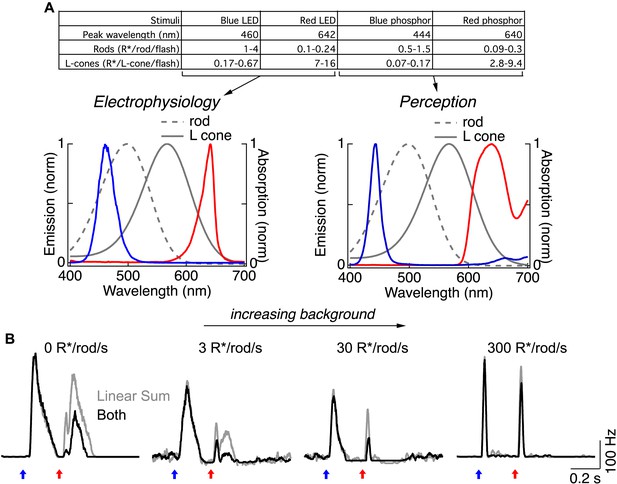
Rod → cone interactions and photoreceptor selectivity depend on mean luminance.
(A) Photoisomerization calibration values for flashes used in this study. (left) Emission spectra of the blue and red LEDs and absorption spectra for the rod and L-cone photoreceptors in primate (Baylor et al., 1984; Baylor et al., 1987) (gray). (right) Emission spectra of the filtered blue and red phosphors used in perceptual experiments. (B) (left) Spike responses of an ON parasol ganglion cell to short- and long-wavelength flashes in the dark. Under these conditions, the response to the long-wavelength flash has two kinetically-distinct components that correspond to activation of cones (fast initial component) and rods (prolonged component) (Dunn et al., 2007; Schwartz et al., 2013). Both components were suppressed by a prior short-wavelength flash. (middle) Increasing the mean luminance reduces the gain of the rods and the rod bipolar pathway. Under these conditions the long-wavelength flash elicited a clear cone response, however the rod component was suppressed at 3 R*/rod/s and absent at 30 R*/rod/s. Nonlinear rod-cone interactions were clear at these backgrounds. (right) At a mean luminance of 300 R*/rod/s ganglion cell responses were fast and transient regardless of which LED was used; retinal interactions between blue and red flashes were largely absent under these conditions. All recordings from whole mount retina.
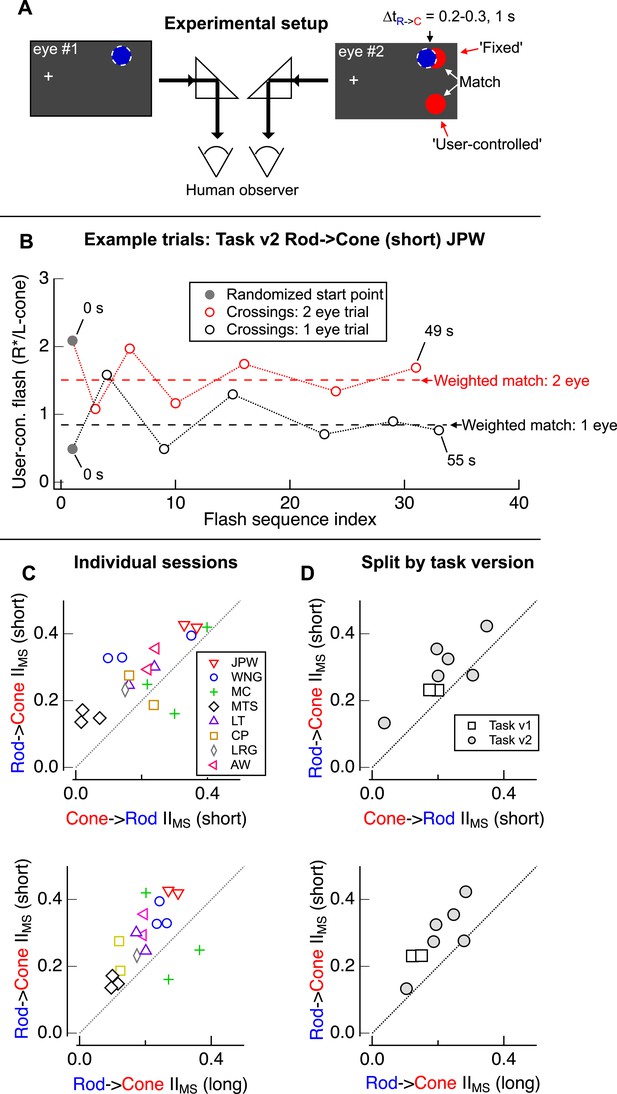
Design of psychophysical experiments presented in Figure 1.
(A) Assuming that retinal and cortical gains are multiplicative, retinal interactions were isolated by comparing the brightness matches from trials in which the adapt and test flashes were delivered to the same eye (e.g., blue and red stimuli delivered to eye #2) to those obtained from trials in which flashes were delivered to separate eyes (e.g., blue stimulus delivered to eye #1 and red stimuli to eye #2). (B) Two example trials from a naive observer's session (Rod-Cone (short) Task v2: same eye trial in black, separate eye trial in red). Observers adjusted the intensity of the bottom test flash (‘y-axis’; flash position spatially offset from that of adapt flash) to match their perception of the top test flash (same location as adapt flash). After completing a total of 6 crossings, users were automatically advanced to the next trial. When test and adapt flashes were delivered to the same eye, interactions were stronger than when flashes were delivered to separate eyes. (C) Individual sessions for each observer. (top) Comparison of rod → cone (short) and cone → rod (short) sessions. (bottom) Comparison of rod → cone (short) and rod → cone (long) sessions. (D) Mean interactions (Figure 1F, I) split by task version: Task v1 (squares) and Task v2 (circles). (top) Comparison of mean rod → cone (short) and cone → rod (short) interactions. (bottom) Comparison of mean rod → cone (short) and rod → cone (long) interactions. Each marker represents average data from a single observer.
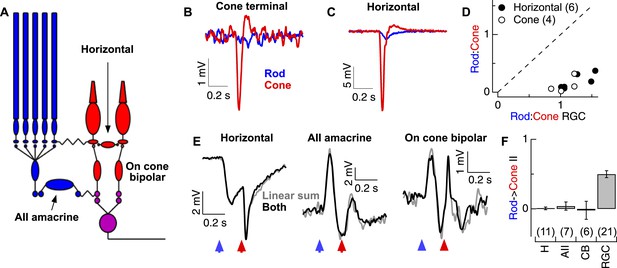
Rod and cone-mediated signals are largely independent upstream of the cone bipolar → RGC synapse.
(A) Retinal circuit diagram. (B–C) Rod and cone flashes that produced near-equal amplitude responses in RGCs elicited strong cone responses and weak rod responses in the axon terminals of L-cones (B) and in horizontal cells (C). (D) Summary across cells. Ratios of the responses to rod- and cone-preferring stimuli in horizontal (H) and L-cone terminals plotted vs the response ratios in RGCs to the same flashes from the same slices. (E) Rod → cone interactions were absent in circuit elements upstream of the RGC: (left) horizontal cells, (middle) AII amacrine cells and (right) cone bipolar cells. (F) Mean rod → cone interaction indices for all horizontal, AII amacrine, cone bipolar and retinal ganglion cells (mean ± SEM, number of cells in parenthesis). Interaction indices for horizontal cells (p = 0.49), AIIs (p = 0.45) and cone bipolars (p = 0.49) did not differ significantly from 0, while those of RGCs did (p < 10−7). All recordings except the RGCs reported in F (whole mount) from retinal slices.
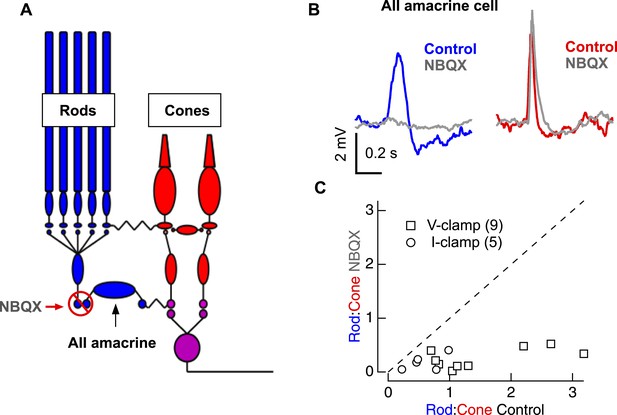
Pharmacological evidence that rod- and cone-mediated signals traverse the retina through largely distinct circuitry.
(A) Circuit diagram illustrating the logic for using NBQX, an AMPAR antagonist. (B) Rod- and cone-preferring stimuli were adjusted to produce clear responses in AII amacrine cells. Bath application of NBQX selectively eliminated input from the rod bipolar pathway. (C) Population data for experiments in B; response ratios in NBQX are plotted vs response ratios in control conditions. If the majority of the rod-mediated signal traversed the retina through cone bipolar cells, the rod-mediated response should be largely insensitive to NBQX and the data would lie near the dashed line. Instead, the greater sensitivity of the rod-mediated response to NBQX indicates that most of this response is conveyed through the rod bipolar cell. Each round markers represents an AII amacrine cell recorded in the current-clamp configuration (with corresponding K-based internal solution—see ‘Materials and methods’) whereas each square marker represents an AII amacrine cell recorded in the voltage-clamp configuration (with corresponding Cs-based internal solution—see ‘Materials and methods’). Similar results were obtained regardless of recording configuration. All voltage-clamp recordings from retinal slices, and current-clamp recordings (including B) from whole mount retina.
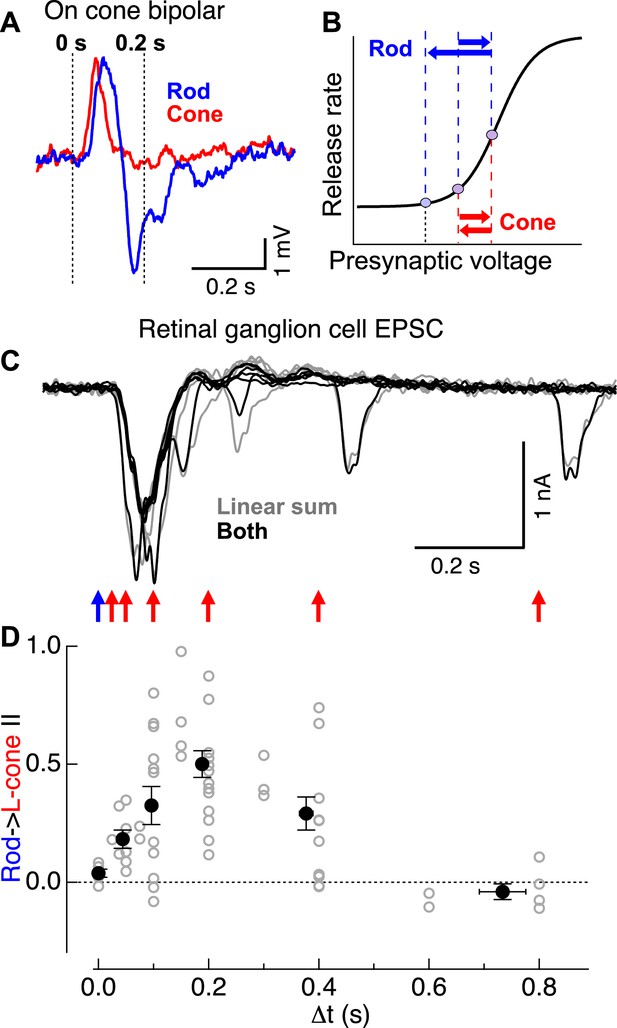
Rod → cone interactions exhibit a similar time course as the observed overshoot in cone bipolar cell voltage following a rod flash.
(A) Cone bipolar voltage responses to rod and cone flashes exhibited distinct kinetics. (B) Illustration of impact of kinetic differences on cone bipolar synaptic output. Rightward blue and red arrows indicate depolarizing components of rod- and cone-mediated responses, while leftward arrows indicate hyperpolarizing components. (C) Time course of rod → cone interactions probed by delivering paired flashes across a range of temporal offsets. (D) Summary of time dependence of rod → cone interactions (individual cells in gray, population averages binned by Δt in black). Similar to the overshoot of the rod-mediated response in cone bipolar cells (A), rod → cone interactions were maximal when flashes were offset by 0.1–0.3 s. On cone bipolar cell recordings from retinal slices, and On parasol recordings from whole mount retina.
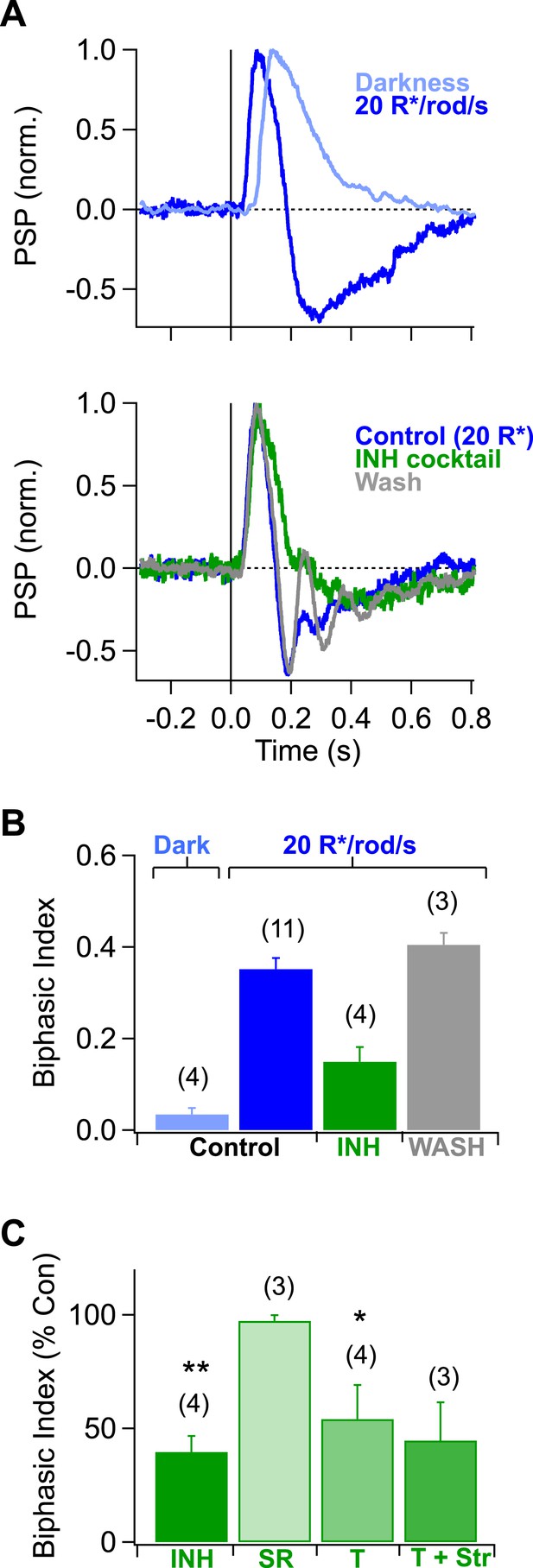
Inhibition creates a rapid overshoot in rod-mediated signals at mean light levels where both rods and cones are active.
(A) top Rod-mediated responses in AII amacrine cells were monophonic in darkness and became biphasic as mean luminance was increased to 20 R*/rod/s. bottom Inhibitory blockers significantly reduced the fast (∼200 ms after the flash) component of the overshoot, likely reflecting suppression of presynaptic inhibition to the rod bipolar cell terminal. After returning to control solution for >8 min the fast overshoot recovered. (B) Population data. The inhibitory cocktail (INH, green) contained 2 μM Strychnine (Str), 25 μM SR95531 (SR) and 50 μM TPMPA (T) to block glycine, GABAa- and GABAc- receptors, respectively. Number of recordings are indicated in parenthesis. Bars represent the mean biphasic index across recordings and error bars indicate the SEM. (C) Population data for pharmacological effects on the biphasic index. Bars represent the mean percentage of the biphasic index remaining in drug across cells; error bars indicate the SEM. Paired, two-tailed Student's t-tests were used to test significance; * and ** correspond to p-values less that 0.05 and 0.01, respectively. Current-clamp recordings from AII amacrine cells in whole mount retina.
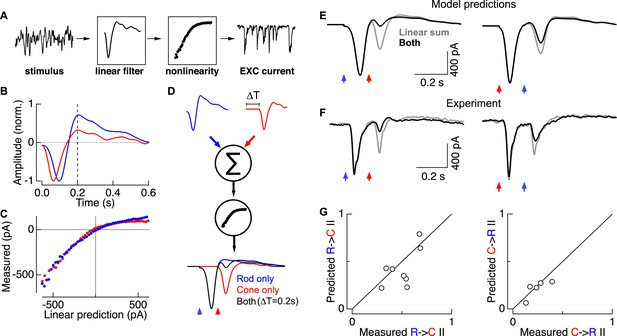
Linear summation followed by a rectifying nonlinearity can account for rod-cone interactions.
(A) Linear-nonlinear (LN) model construction. A time-varying rod- or cone-preferring stimulus and the resulting RGC excitatory synaptic inputs were used to derive the linear filter and static nonlinearity that relate the stimulus to the response. (B) Normalized linear filters for rod and cone stimuli. (C) Nonlinearities for rod and cone stimuli. (D) Rod-cone interactions were predicted by summing scaled and temporally-offset (i.e., 0.2 s) rod- and cone-preferring filters and passing the result through a common nonlinearity (see ‘Materials and methods’). (E) Predicted rod → cone (left) and cone → rod (right) interactions. (F) Measured rod → cone and cone → rod interactions. (G) Population data comparing predictions from the LN models to experimental observations. Each point represents a single cell in which LN model components and rod-cone interactions were measured. All recordings from whole mount retina.





