Small molecule inhibition of Csk alters affinity recognition by T cells
Figures
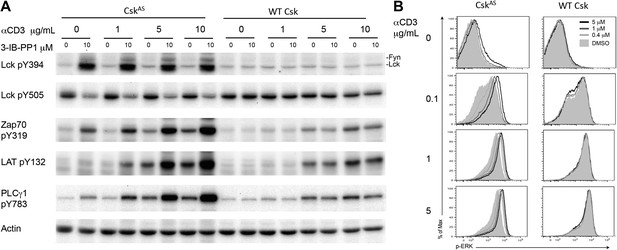
Inhibiting Csk increases the magnitude of ligand-induced TCR signaling and reduces the threshold for TCR activation.
(A) Purified CskAS (AS) or wildtype (WT) CD4+ T cells stimulated for 2 min with 1 μg/ml, 5 μg/ml or 10 μg/ml anti-CD3ε antibody in the presence of DMSO or 10 μM 3-IB-PP1 were analyzed by immunoblotting for the phosphorylation of the activation loop tyrosine of Lck and Fyn (Src pY416 antibody) and the inhibitory tyrosine of Lck (Lck pY505), phosphorylated ZAP-70 (ZAP70 pY319), LAT (LAT pY132) and PLC-γ1 (PLCγ1 pY783), as well as total actin (loading control). Data are representative of at least three independent experiments. (B) Purified total CskAS (AS) or wildtype (WT) T cells stimulated with the indicated dose of anti-CD3ε antibody for 2 min in the presence of DMSO or 5 μM, 1 μM or 0.4 μM 3-IB-PP1 were analyzed for phosphorylated ERK (p-ERK) by phosphoflow. Histograms were gated on CD4+ cells. Data are representative of at least three independent experiments for AS cells and two independent experiments for WT cells.
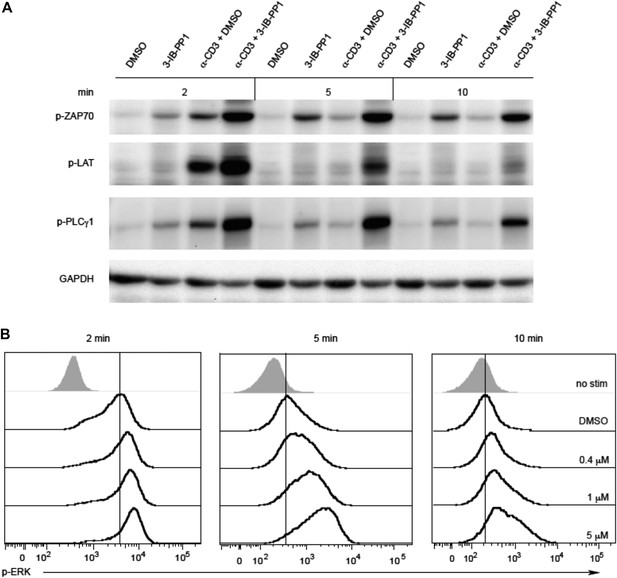
Inhibiting Csk during TCR stimulation prolongs TCR signals.
(A) Purified CskAS CD4+ T cells stimulated for 2, 5 or 10 min with anti-CD3ε antibody in the presence of DMSO or 10 μM 3-IB-PP1 were analyzed by immunoblotting for the phosphorylated ZAP-70, LAT and PLC-γ1 as well as total GAPDH (loading control). Data are representative of at least three independent experiments. (B) Purified total CskAS T cells stimulated with anti-CD3ε antibody for 2, 5 or 10 min in the presence of DMSO or 5 μM, 1 μM or 0.4 μM 3-IB-PP1 were analyzed for phosphorylated ERK (p-ERK) by phosphoflow. Histograms were gated on CD4+ cells. Data are representative of at least three independent experiments.
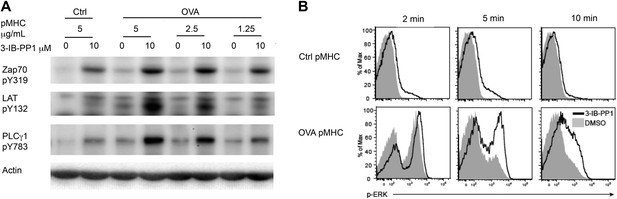
Inhibiting Csk reduces the threshold for TCR activation and prolongs signaling induced by pMHC engagement.
(A) Purified CskAS;OTII CD4+ T cells stimulated for 3 min with 5 μg/ml bead-bound control pMHC tetramer or 5 μg/ml, 2.5 μg/ml or 1.25 μg/ml bead-bound OVA pMHC tetramer in the presence of DMSO or 10 μM 3-IB-PP1 were analyzed by immunoblotting for the phosphorylated ZAP-70, LAT and PLC-γ1, as well as total actin (loading control). Data are representative of three independent experiments. (B) Purified CskAS;OTII CD4+ T cells stimulated with 5 μg/ml bead-bound control-pMHC tetramer or 2.5 μg/ml bead-bound OVA pMHC tetramer for 2, 5 or 10 min in the presence of DMSO or 5 μM 3-IB-PP1 were analyzed for phosphorylated ERK (p-ERK) by phosphoflow. Histograms were gated on CD4+ Vα2+ cells. Data are representative of three independent experiments.
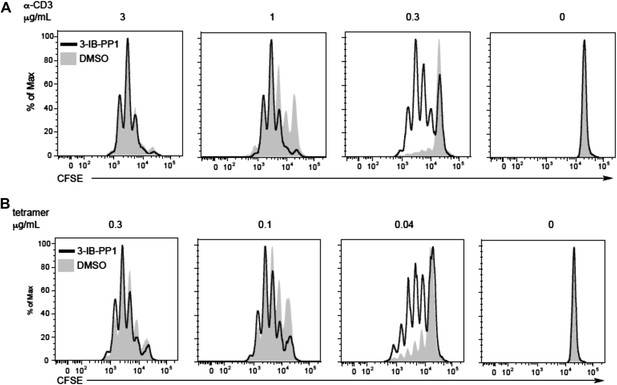
Csk inhibition shifts the threshold for cellular proliferation in response to anti-CD3 or p-MHC tetramer stimulation.
(A) Purified CD4+ T cells from CskAS mice were loaded with CFSE and stimulated with 3 μg/ml, 1 μg/ml or 0.3 μg/ml plate-bound anti-CD3ε and 1 μg/ml anti-CD28 for 72 hr in the presence or absence of 5 μM 3-IB-PP1. Data are representative of three independent experiments. (B) Purified CD4+ T cells from CskAS;OT-II mice were loaded with CFSE and stimulated with 0.3 μg/ml, 0.1 μg/ml or 0.04 μg/ml plate-bound OVA pMHC tetramer and 1 μg/ml anti-CD28 for 72 hr in the presence or absence of 5 μM 3-IB-PP1. (A, B) Cells were then analyzed by flow cytometry for CFSE intensity. Histograms were gated on CD4+ cells. Data are representative of three independent experiments.
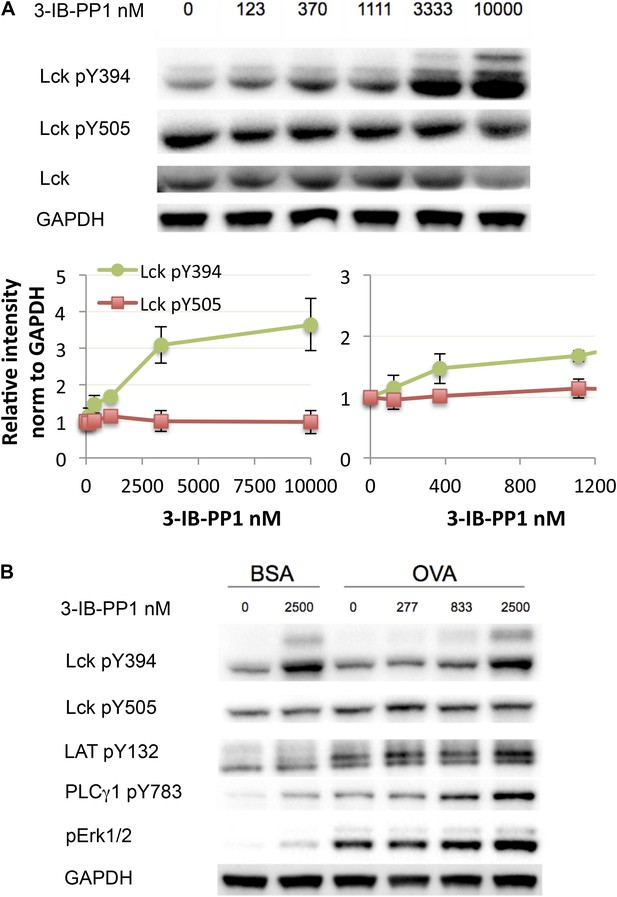
Weak Csk inhibition and Lck activation potentiate agonist signaling.
(A) CskAS CD8+ T cells were rested and stimulated with indicated doses of 3-IB-PP1 for 3 min, lysed, and immunoblotted for activating (pY394) and inhibitory (pY505) site Lck phosphorylation. Below is quantification (± sem) of three independent experiments immunoblot. (B) CskAS;OTI naive CD8+ T cells were rested, stimulated with bead-bound BSA or 10 μg/ml pMHC-OVA for 3 min, lysed and assayed by immunoblot. Data are representative of at least two independent experiments.
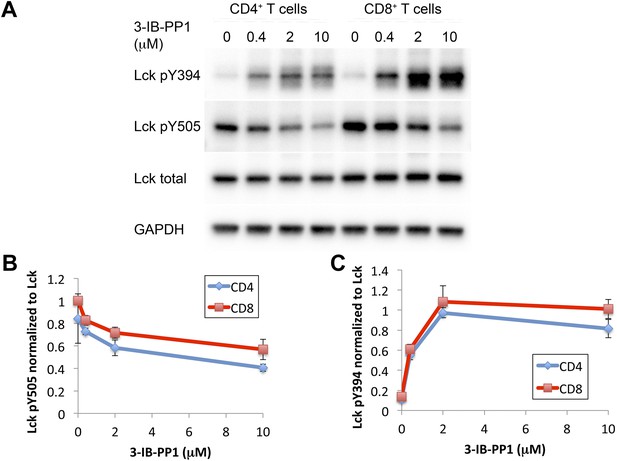
Comparison of Csk inhibition and Lck activation in CD4+ vs CD8+ T cells.
(A) CskAS CD4+ and CskAS CD8+ T cells were rested and stimulated with indicated doses of 3-IB-PP1 or DMSO for 2 min and then lysed. Lysates were immunoblotted for activating (pY394) and inhibitory (pY505) site Lck phosphorylation, total Lck, and GAPDH (loading control). Data are representative of three independent experiments. (B, C) Quantification of Lck pY505 and Lck pY394 of three independent experiments (as in A), with levels normalized to Lck levels per cell type. Error bars are standard deviation.
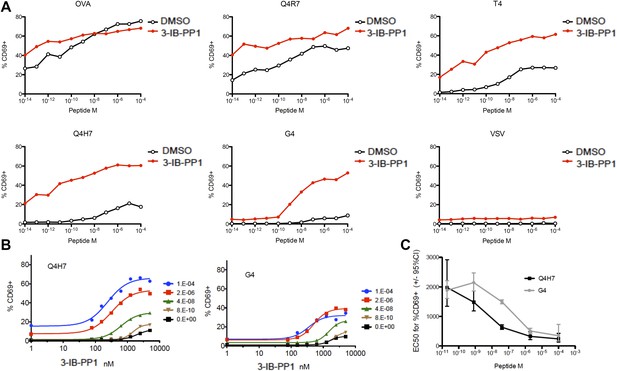
Csk inhibition potentiates response to weak agonists.
(A) CskAS;OTI naive CD8+ T cells were stimulated with MitoC-treated WT splenocytes, preloaded with indicated peptides, in presence of DMSO or 5 μM 3-IB-PP1 for 4 hr and CD69 upregulation was measured by flow cytometry. (B) CskAS;OTI naive CD8+ T cells were stimulated as in A with varying doses of 3-IB-PP1 with dose–response fit. (C) EC50 of 3-IB-PP1 (nM) for % CD69 positive cells for data in B. Data are representative of two independent experiments.
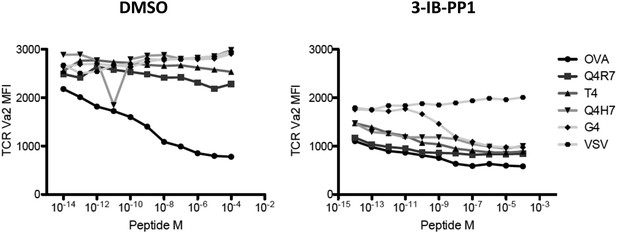
TCR Valpha2 downregulation in cells in Figure 4A.
Only OVA triggers peptide-dependent TCR downregulation. 5 μM 3-IB-PP1 induces ligand-independent downregulation of TCR, as seen in VSV peptide. In the presence of CskAS inhibition, all the other altered peptides induce further, dose-dependent loss of TCR.





