Digital signaling decouples activation probability and population heterogeneity
Figures
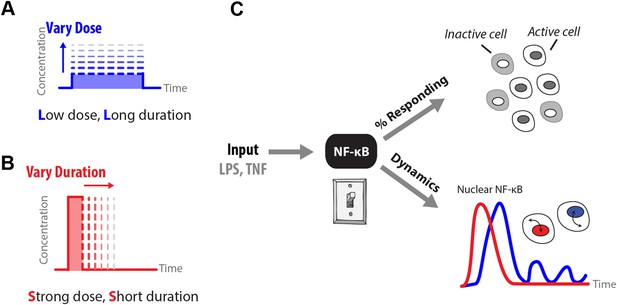
How does input profile determine digital signaling response?
Since the amplitude and time profile of input signals depends on biological context, such as distance to an infection site or pathogen loading, we use microfluidics to manipulate dose (A) and duration (B) of LPS and TNF input signals, which induces digital activation of NF-κB. (C) Switch-like digital NF-κB responses are analyzed in terms of fraction of cells that activate in the population and heterogeneity in the dynamic responses in activating cells.
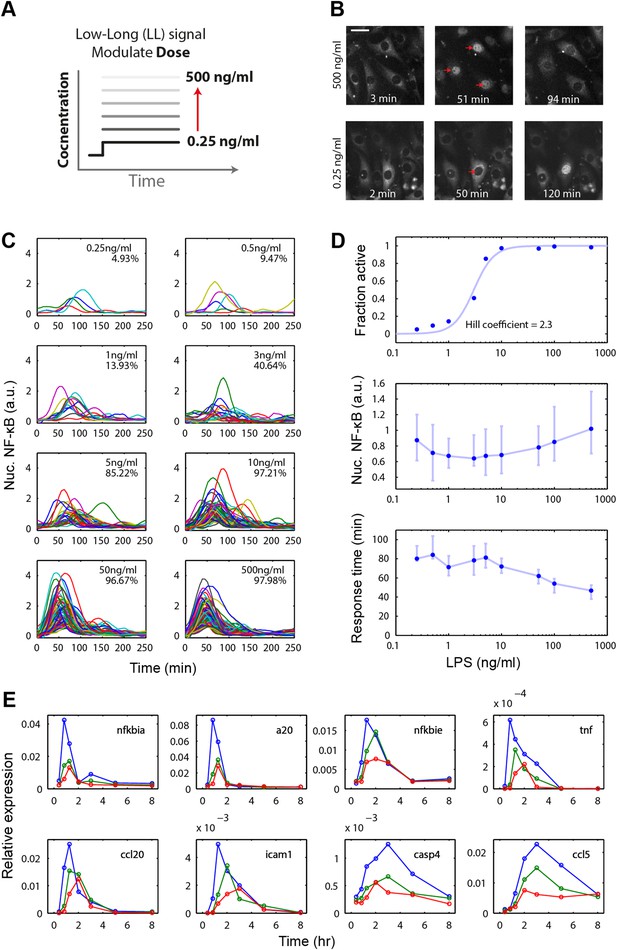
Digital, time-delayed NF-κB activation under varied LPS dose stimulation.
(A) Cells process pathogen signal dose and duration to dynamically activate NF-κB, which induces gene expression and coordinates the innate immune response. We test the role of pathogen load by varying LPS concentration from 0.25 ng/ml to 500 ng/ml using microfluidic cell culture. (B) Time series images of NF-κB activation following LPS treatment. Top row: high LPS dose causes nearly 100% of cells to respond synchronously. Bottom row: at low LPS concentration, less than 5% of cells respond and initiate NF-κB activation with variable, delayed timing. The cells respond digitally, with nearly all cytoplasmic NF-κB moving into the nucleus. The response amplitude (indicated by peak intensity of nuclear p65-dsRed fluorescence) depends on the initial NF-κB abundance in the nucleus and exhibits high variability across doses. (C) Trajectories of NF-κB activation (intensity of nuclear p65-dsRed) tracked in single cells over time for LPS doses ranging from 500 to 0.25 ng/ml. As the LPS dose decreases, response timing becomes delayed and variable, and the percent of responding cells in the population drops. (D) Across the LPS doses tested: top panel, dose-response curve of the fraction of active cells (plotted is the mean of two duplicate cell chambers in the chip for each condition), middle panel, the intensity of nuclear NF-κB at the peak of the response, and lower panel, time until the peak of the response. Peak nuclear NF-κB amplitude is highly variable across doses. The dose response shows a sharp drop in fraction of active cells between 1 and 5 ng/ml concentration, indicating that the activation threshold is within this range for most cells. With lower dose, the response time increases in both median duration and variability. In middle and lower panels, data points and error bars represent median and interquartile range, respectively. (E) NF-κB dependent gene expression dynamics under varied LPS concentrations (blue: 500 ng/ml, green: 100 ng/ml, red: 50 ng/ml). With lower LPS concentration, several genes show delayed induction. TNF dose-modulated expression of the same genes can be found in Figure 3 of Tay et al. (2010) and Figure 2A of Pękalski et al. (2013).
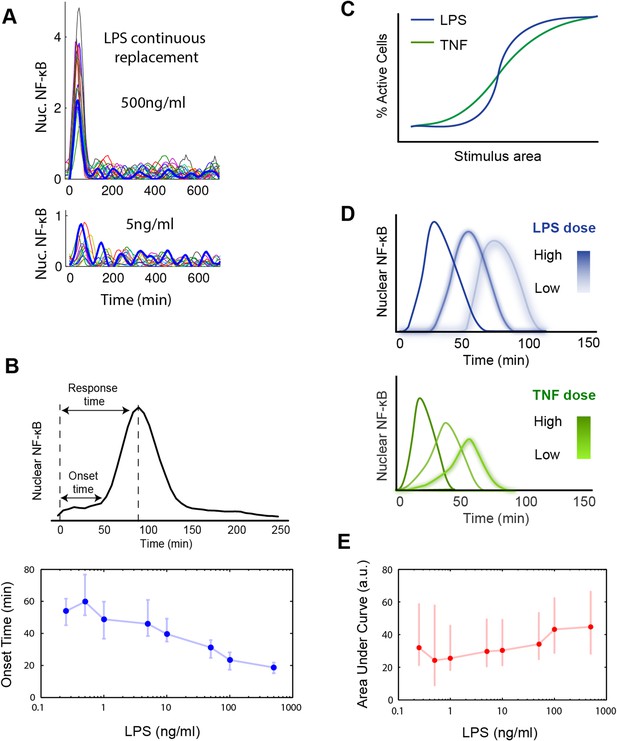
Digital, time-delayed NF-κB activation under continuous LPS stimulation.
(A) Sustained NF-κB oscillations for continuously perfused LPS achieving constant LPS concentration. Bolded blue line: example cell. (B) Analysis of the LPS induced NF-κB nuclear localization peak. Lower LPS doses induce a pronounced onset delay, but the peak shape is mostly conserved across different doses. Data points and error bars are median and interquartile range, respectively. (C) Cartoon illustrating activation curves for LPS vs TNF. (D) Cartoon illustrating first peak time profile for LPS vs TNF. LPS leads to greater dose-dependent delay than TNF and a more conserved peak shape. (E) Area under the NF-κB localization curve show little change across all doses tested. Data are plotted as median and interquartile range.

Statistical analysis for NF-κB peak amplitude and timing measurements under LPS dose modulation (corresponding to Figure 2D).
Each table contains p-values of two-sample T-test for each dose combination. Red denotes statistical significance (p < 0.05).
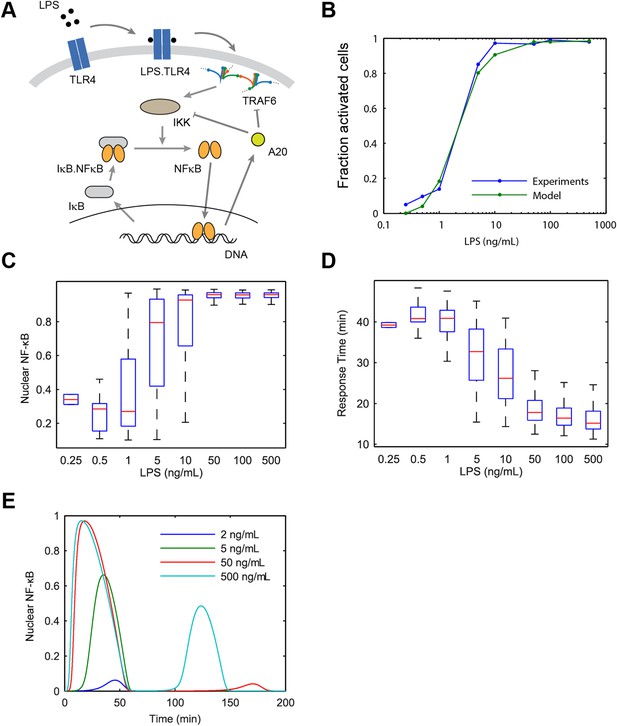
Model scheme and simulation of LPS dose modulation.
(A) The scheme of the model. LPS binds TLR4 leading to TRAF6 activation, which cooperatively activates IKK. Active IKK induces IκB degradation, which allows NF-κB to enter the nucleus and upregulate expression of IκB and A20. New IκB sequesters NF-κB in the cytoplasm and A20 inhibits upstream pathway activation by IKK and TRAF6. (B) Simulated versus experimental LPS dose response. (C) NF-κB peak intensities (expressed as proportion of total NF-κB molecules in the nucleus). (D) NF-κB response time as function of LPS dose. (E) Sample simulated curves of nuclear NF-κB fractions under LPS treatment. In box and whisker plots (C, D), the central red line is the median, the edges of the box are the 25th and 75th percentiles, and the whiskers extend to the most extreme data points.
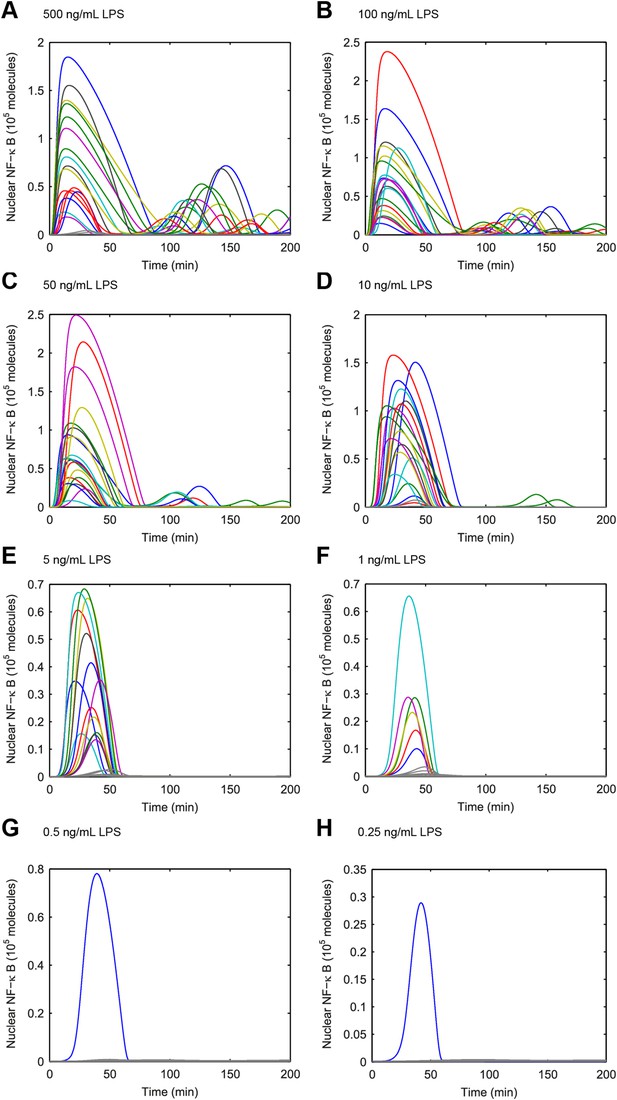
Simulated NF-κB trajectories for various doses of LPS treatment.
(A–H) LPS concentration decreases over time due to cellular internalization in sealed microfluidic chambers, leading to damped oscillations. (Note: y-scale changes between plots.)
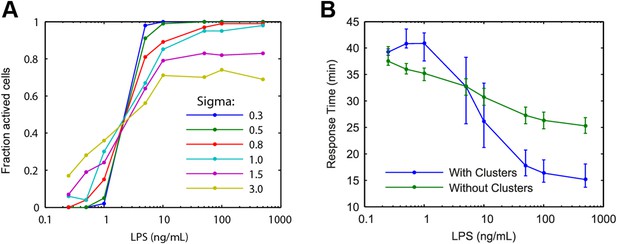
Modelling predictions for the LPS pathway.
(A) Extrinsic noise is generated by selecting the number of TLR4 molecule for each cell from a lognormal distribution. Fraction of active cells was analyzed for increased TLR4 variability (parameter Sigma). With higher TLR4 number variability, the change in fraction of active cells becomes more gradual with changing LPS concentration. (B) Simulation response delay under varied LPS dose with and without clustering. Clustering mediating cooperative IKK activation is required to reproduce the experimentally observed response delay with decreasing LPS dose. Central point and error bars represent median and interquartile range, respectively.
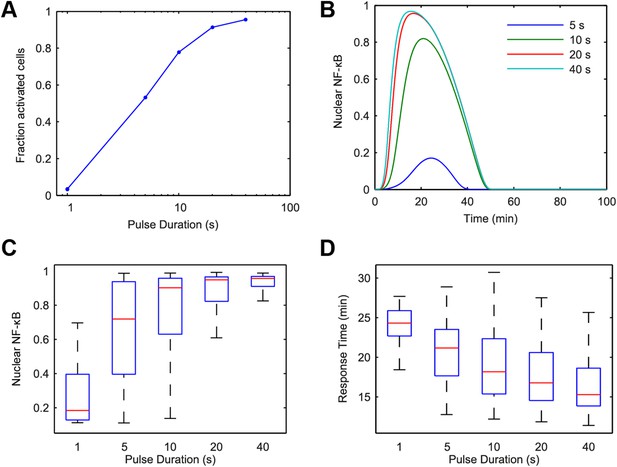
Model simulation predicts that stimulus duration controls fraction of activating cells and response timing variability.
(A) Simulated fractions of activated cells under increasing duration of 500 ng/ml LPS pulse. (B) Sample simulated curves of NF-κB under 5- to 40-s duration LPS (500 ng/ml) pulse. (C) Distributions of nuclear NF-κB amplitude and (D) response times of activating cells under various durations of LPS treatment. In box and whisker plots (C, D), the central red line is the median, the edges of the box are the 25th and 75th percentiles, and the whiskers extend to the most extreme data points.
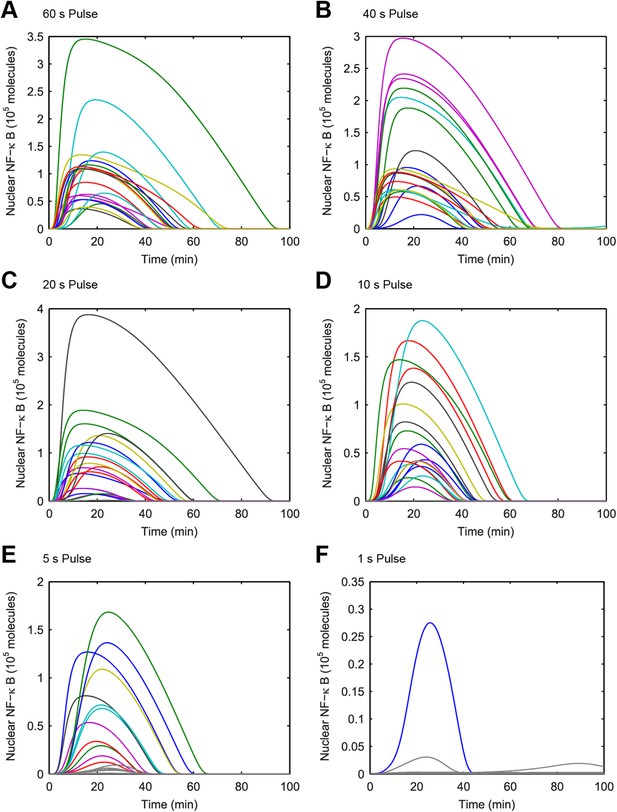
(A–F) Simulated NF-κB single-cell trajectories with randomly sampled numbers of TLR4 and NF-κB for 1- to 60-s durations of LPS exposure.
The concentration of LPS is 500 ng/ml. Color: activated cells. Gray: inactivated cells. (Note: y-scale changes between plots.)
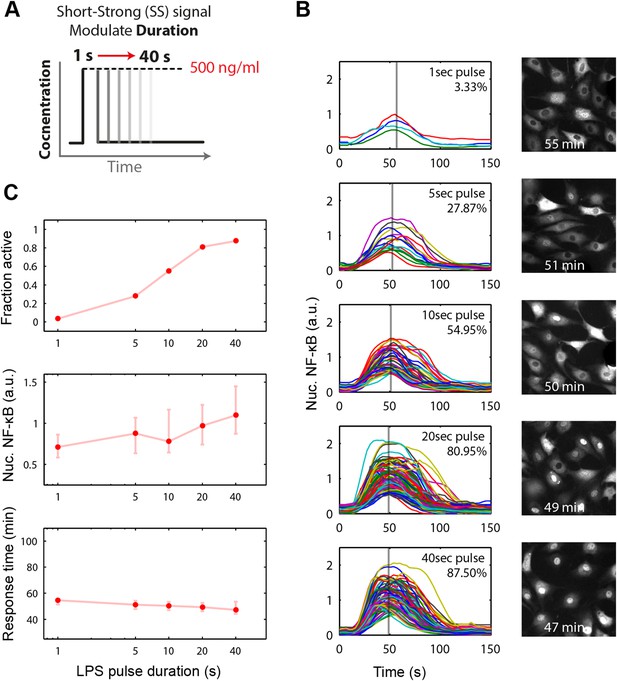
Short duration LPS pulse stimulation modulates responding cell fraction and fast, uniformly timed response.
(A) LPS duration is manipulated using microfluidic cell culture in the range of 1–40 s. Dose is held constant at 500 ng/ml. (B) Single-cell NF-κB trajectories for 1- to 40-s duration LPS pulse stimulation. Short pulse LPS reduces variation in timing in the start of NF-κB activation. (C) Top panel: fraction of active cells as a function of LPS pulse duration. (Plotted points are the mean of two duplicate chambers in chip.) Middle panel: NF-κB nuclear response intensity as a function of LPS pulse duration. Lower panel: time of the NF-κB response peak as a function of LPS pulse duration. Timing variability is dramatically reduced under short-pulsed stimulation (see Figure 2D for comparison to constant stimulation). In middle and lower panels, data points and error bars represent median and interquartile range, respectively.
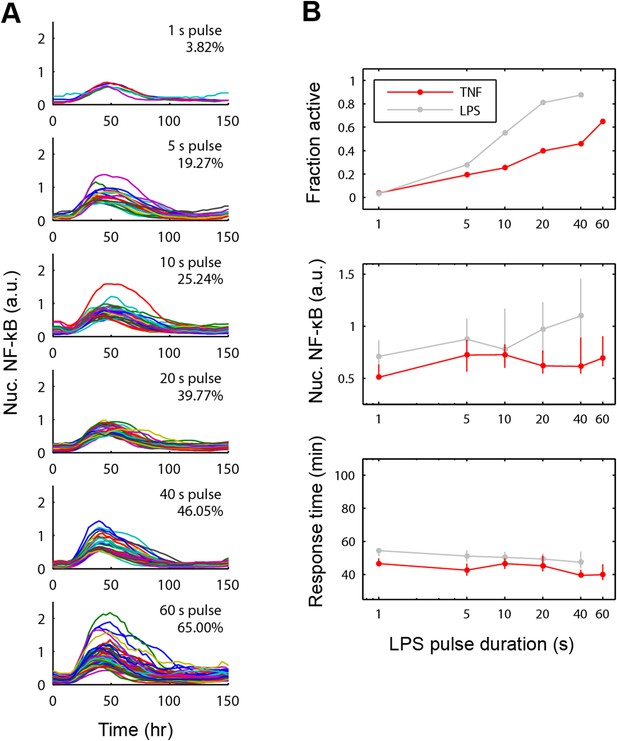
NF-κB dynamics under pulsed stimulation with TNF at 10 ng/ml concentration.
(A) Single-cell NF-κB trajectories for TNF pulse durations of 1, 5, 10, 20, 40, and 60 s. The number and percent of activated cells is indicated in the plot. (B) Comparison of active NF-κB cell fraction (top), response amplitude (middle), and response time (bottom) under LPS vs TNF pulses. The fraction-active curve is less steep under TNF than LPS. Fraction of active cells is plotted as mean for two duplicate cell chambers for each condition. In middle and lower panels, data points and error bars represent median and interquartile range, respectively. LPS data are duplicated from main Figure 5C for comparison.
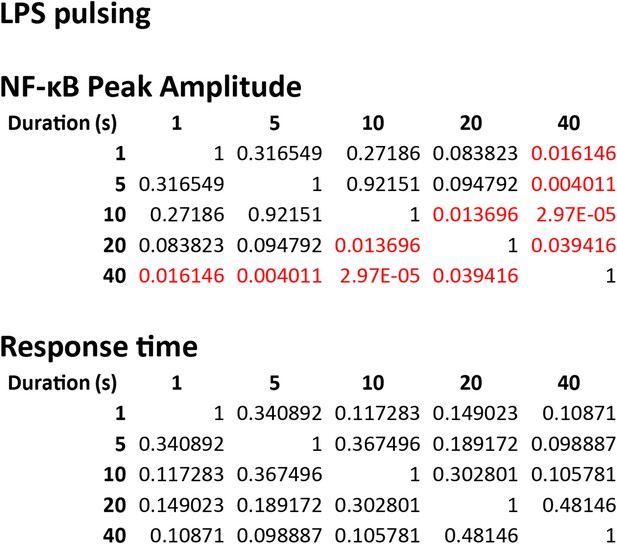
Statistical analysis for NF-κB peak amplitude and timing measurements under LPS duration modulation (corresponding to Figure 5B).
Each table contains p-values of two-sample T-test comparing each duration combination. Red denotes statistical significance (p < 0.05).
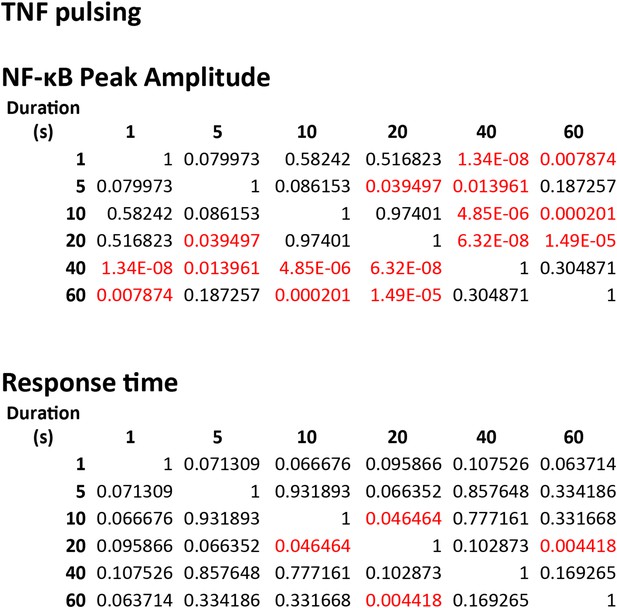
Statistical analysis for NF-κB peak amplitude and timing measurements under TNF (10 ng/ml) duration modulation (corresponding to Figure 5—figure supplement 1).
Each table contains p-values of two-sample T-tests comparing each duration combination. Overall, amplitude is affected to a greater extent than response time under changing TNF duration. Red denotes statistical significance (p < 0.05).
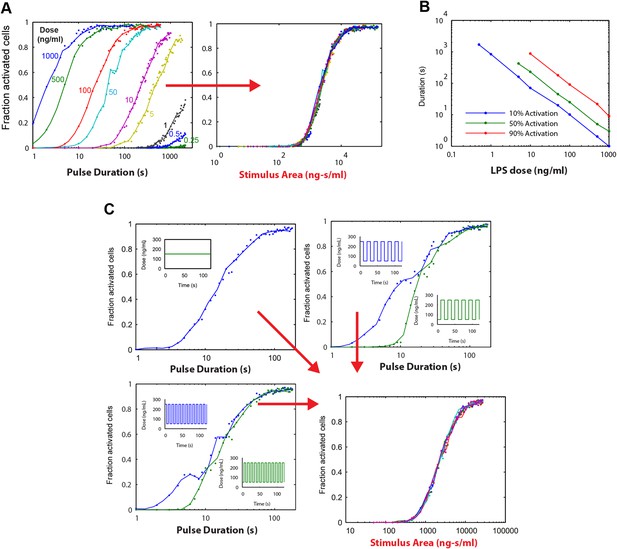
Simulations demonstrating an ‘Area Rule’, that is, the relationship between LPS stimulus area and fraction of active cells.
(A) The simulated fractions of activated cells for pulsed inputs of LPS with various doses and durations. Each fraction is estimated by 500 independent simulations. When the points are plotted as a function of stimulus area (rather than duration), all points fall on the same curve, indicating that stimulus area tightly controls the fraction of active cells. (B) The minimal duration for certain fractions of activation as a function of dose. The minimal duration is determined by searching for the first tested time point where the estimated fractions of activation are above the threshold. The doses for which the threshold level cannot be achieved are not shown in the figure. Blue: 10% activation, green: 50% activation, red: 90% activation. (C) Further verification of the relationship between stimulus area and active cell fraction using square wave input profiles. Equal area input was generated using either a single pulse (top left), square wave with 10-s period (lower left), or square wave with 20-s period (top right). Regardless of input shape, all simulated points fall on the same curve when plotted as a function of stimulus area. For square wave inputs, one input begins high (blue) while another input (green) begins low. Note that the curves intersect for durations 10 s, 20 s, 30 s (or 20 s, 40 s, 0 s, … for the input with 20-s period) when area under the two signals is the same.
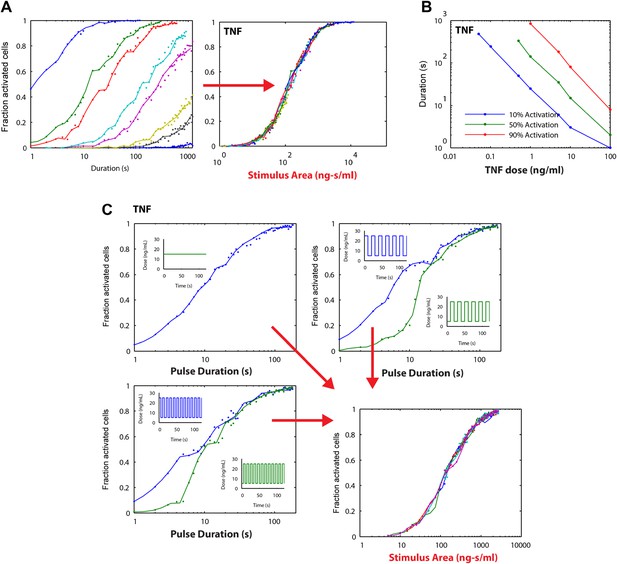
Stimulus area simulation using a TNF model (Tay et al., 2010) revealed that the ‘Area Rule’ holds also for TNF.
(A) Simulated fractions of activated cells for pulsed inputs of TNF with various doses and durations. (B) The minimal duration for certain fractions of activation as a function of dose. (C) Verification of the relationship between stimulus area and active cell fraction using square wave input profiles. Equal area input was generated using either a single pulse (top left), square wave with 10-s period (lower left), or square wave with 20-s period (top right).
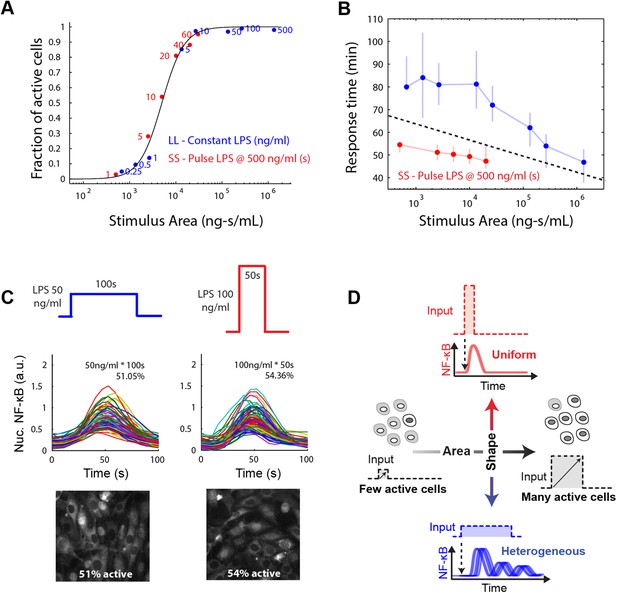
Stimulus area determines NF-κB population response.
(A) Stimulus area determines fraction of active cells. The experimentally tested dose and duration inputs fall on the same hill-like activation curve when plotted as a function of stimulus area, as predicted by model simulations, indicating that total integrated ligand concentration (stimulus area) controls the probability of cell activation. These results show that pathogen load (i.e., LPS dose) and duration of exposure (i.e., LPS pulse duration) are integrated by NF-κB system and together determine the population response. (B) Response time discriminates between sustained, low intensity (blue) and transient, high intensity (red) stimulus. Data points and error bars represent median and interquartile range, respectively. (C) Experimental verification that stimulus integral over time determines the fraction of active cells. A pulse of either LPS 50 ng/ml for 100-s duration or 100 ng/ml for 50-s duration generated approximately the same responding cell percentage as a, 51% and 54% for the two inputs, respectively. (D) Input profile controls digital responses along two axes: integral over stimulus (area) controls the fraction of activated cells in the population. Input temporal profile (shape) controls dynamic heterogeneity in responding cells. In (B), data points and error bars represent median and interquartile range.
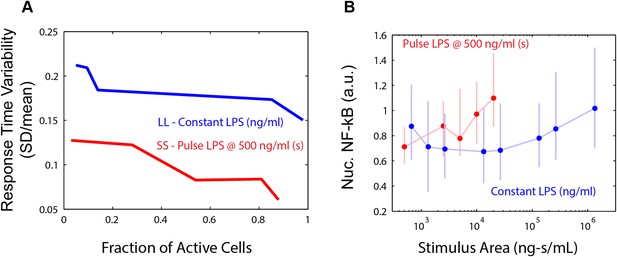
LPS response heterogeneity under pulsed and continuous input.
(A) Comparison of cell-to-cell dynamic heterogeneity for different fractions of active cells for either constant or pulsed input (Figures 2 and 5). Pulsed (duration-modulated) LPS input (red) achieves lower response time variability than constant (dose-modulated) input for the same fraction of active cells. (B) Unlike response delay, response intensity does not provide sufficient information to distinguish between pulsed LPS and constant LPS signals. Data points and error bars represent median and interquartile range, respectively.
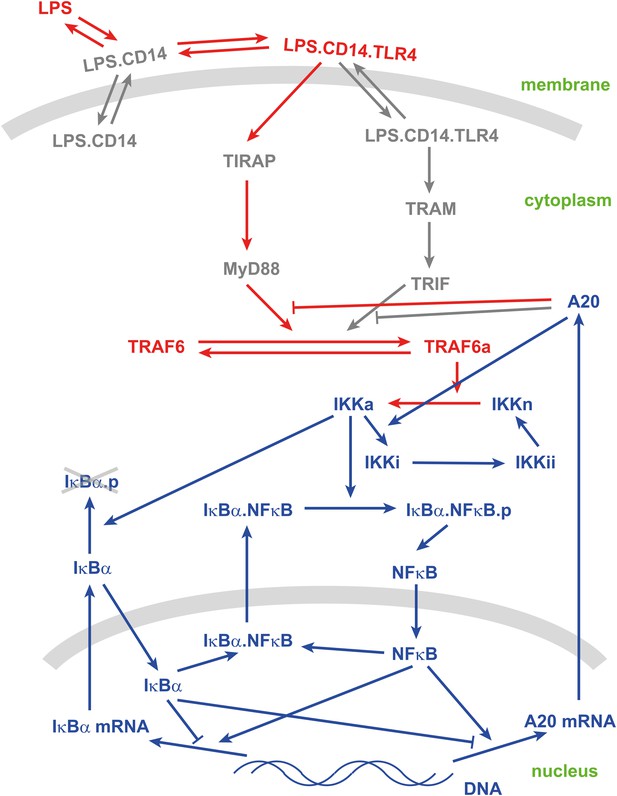
Schematic view of the LPS-mediated NF-κB pathway.
Text: species; arrows: reactions. Blue: existing species and reactions in the model in Tay et al. (2010); Red: newly introduced species and reactions; Gray: neglected species and reactions.
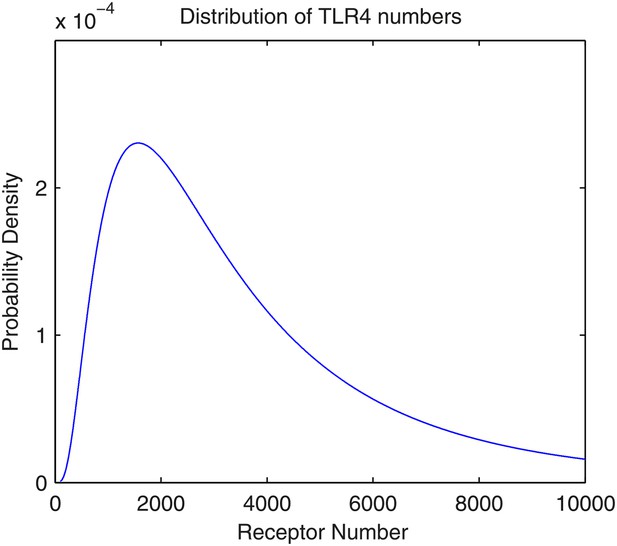
The distribution of the number of TLR4 receptors in the model.
The number follows lognormal distribution with parameters μ = 8.0 and σ = 0.8.
Videos
Digital NF-κB response unders 1 second duration LPS exposure (500 ng/ml).
This video shows fibroblast cells expressing NF-κB p65-dsRed responding in a digital fashion to brief (1-s duration) stimulus in a microfluidic chamber. Only 3–4% of cells show a response.
Digital NF-κB response under 10 second duration LPS exposure (500 ng/ml).
This video shows fibroblast cells expressing NF-κB p65-dsRed responding in a digital fashion to 20-s duration stimulus in a microfluidic chamber, activating more than half (∼55%) of cells in the population.
Tables
The reactions in the TLR4-MyD88-dependent branch
| Reaction/Description | Rate laws |
|---|---|
| LPS → ∅ | ld[LPS] |
| Degradation of LPS | |
| LPS + CD14 → LPS.CD14 | lb,CD14[LPS][CD14] |
| Association of LPS and CD14 | |
| LPS.CD14 → LPS + CD14 | lf,CD14[LPS.CD14] |
| Dissociation of LPS.CD14 | |
| LPS.CD14 → LPS.CD14.int | lin,CD14[LPS.CD14] |
| Internalization of LPS.CD14 | |
| LPS.CD14.int → LPS.CD14 | lout,CD14[LPS.CD14.int] |
| Externalization of LPS.CD14.int | |
| LPS.CD14 + TLR4 → LPS.CD14.TLR4 | lb[LPS.CD14][TLR4] |
| Association of LPS.CD14 and TLR4 | |
| LPS.CD14.TLR4 → LPS.CD14 + TLR4 | lf[LPS.CD14.TLR4] |
| Dissociation of LPS.CD14.TLR4 | |
| TRAF6 → TRAF6a | |
| Activation of TRAF6 by LPS.CD14.TLR4 | |
| TRAF6a → TRAF6 | li[TRAF6a] |
| Inactivation of TRAF6 | |
| IKKn → IKKa | |
| Activation of IKK by TRAF6a | |
The additional reactions in the TLR4-TRIF-dependent branch
| Reaction/Description | Rate laws |
|---|---|
| LPS.CD14.TLR4 → LPS.CD14.TLR4.int | lin[LPS.CD14.TLR4] |
| Internalization of LPS.CD14.TLR4 | |
| LPS.CD14.TLR4.int → LPS.CD14.TLR4 | lout[LPS.CD14.TLR4.int] |
| Externalization of LPS.CD14.TLR4.int | |
| TRAF6 → TRAF6a | |
| Activation of TRAF6 by LPS.CD14.TLR4.int | |
The reactions upstream of IKK activation in the final model
| Reaction/Description | Rate laws |
|---|---|
| LPS → ∅ | ld[LPS] |
| Degradation of LPS | |
| LPS + TLR4 → LPS.TLR4 | lb[LPS][TLR4] |
| Association of LPS and TLR4 | |
| LPS.TLR4 → LPS + TLR4 | lf[LPS.TLR4] |
| Dissociation of LPS and TLR4 | |
| TRAF6 → TRAF6a | |
| Activation of TRAF6 by LPS.TLR4 | |
| TRAF6a → TRAF6 | li[TRAF6a] |
| Inactivation of TRAF6a | |
| IKKn → IKKa | |
| Activation of IKKn by TRAF6a | |
The conversion table of LPS between concentrations and molecular numbers
| Concentrations () | Molecular numbers |
|---|---|
| 500 | 1.2 × 107 |
| 100 | 2.4 × 106 |
| 50 | 1.2 × 106 |
| 10 | 2.4 × 105 |
| 5 | 1.2 × 105 |
| 1 | 2.4 × 104 |
| 0.5 | 1.2 × 104 |
| 0.25 | 6.0 × 103 |
The parameter values used in the model
| Parameters | Biological meanings | Values | Remarks |
|---|---|---|---|
| ld | Degradation rate of LPS | 5 × 10−4 | Fitted |
| lb | Binding rate of LPS and TLR4 | 5 × 10−9 | Fitted |
| lf | Unbinding rate of LPS.TLR4 | 4.5 × 10−4 | Fitted |
| la | Activation rate of TRAF6 | 1 × 10−7 | Fitted |
| kM,A20 | Constant for repressed TRAF6 activation | 1 × 105 | Assume same as Tay et al. (2010) |
| li | Inactivation rate of TRAF6a | 1 × 10−2 | Assume same as Tay et al. (2010) |
| c1 | Activation rate of IKK | 2 × 10−2 | Fitted |
| KIKKK | Dissociation constant for IKK activation | 3.5 × 103 | Fitted |
| n | Hill coefficient | 4 | Fitted, with support from Yin et al. (2009) |
| MIKKK | Total number of TRAF6 | 105 | Assume same as Tay et al. (2010) |





