Human blindsight is mediated by an intact geniculo-extrastriate pathway
Figures
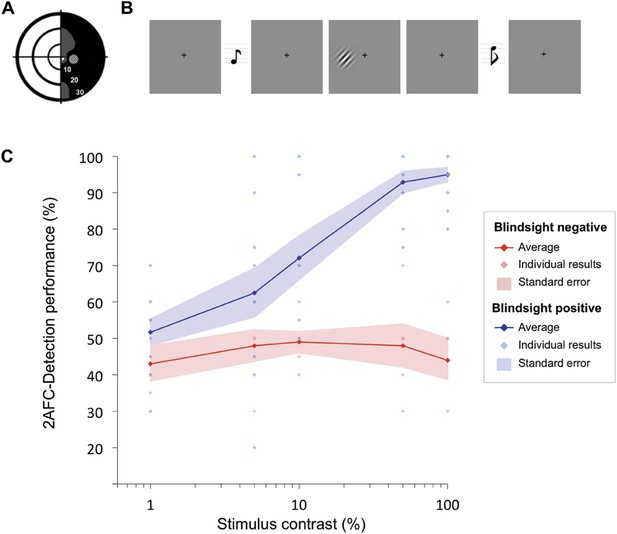
Psychophysics protocol and results.
(A) Example Humphrey visual field deficit drawn schematically, with the location of the target stimulus superimposed. Dense visual field loss is shown in black (<0.5%) and partial loss in grey (<2%). (B) Illustration of the 2AFC-temporal detection procedure. Participants fixated on a central cross, with the onset of each 1500ms interval alerted by a low (interval 1) or high pitch (interval 2) tone. The stimulus could appear in either interval, for a period of 500 ms. At the end of the trial, participants were instructed to decide in which interval the stimulus appeared. (C) Detection performance with increasing stimulus contrast, shown separately for blindsight positive (blue) and blindsight negative (red) patients. Individual results are also plotted for each patient. Chance level is 50%.
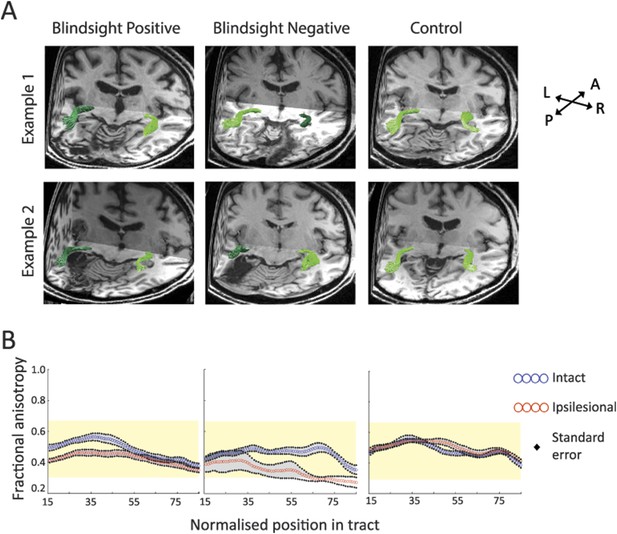
(A) 3-D representations of ipsilateral tracts between the LGN and hMT+.
Examples are shown for blindsight positive patients PB9 and PB8, blindsight negative patients PN2 and PN3 and control participants C8 and C4. Dark green tracts are in the ipsilesional damaged hemisphere, light green tracts are in the intact hemisphere and controls. Tracts are overlaid on a 3-D representation of participant's structural T1-weighted images. (B) Average FA along the ipsilateral geniculate-hMT+ pathways of blindsight positive patients, blindsight negative patients, and controls. Blindsight positive patients show a slight reduction in anisotropy over the proximal half of the ipsilesional pathway, although the distal half shows no notable difference to the intact hemisphere. Blindsight negative patients show a marked reduction in FA in the damaged hemisphere beyond the 35th node, continuing to the end of the tract. Control participants show similar results for both hemispheres (right hemisphere blue, left hemisphere red), with FA close to 0.5 throughout. The control range for this pathway is displayed in yellow on all charts.
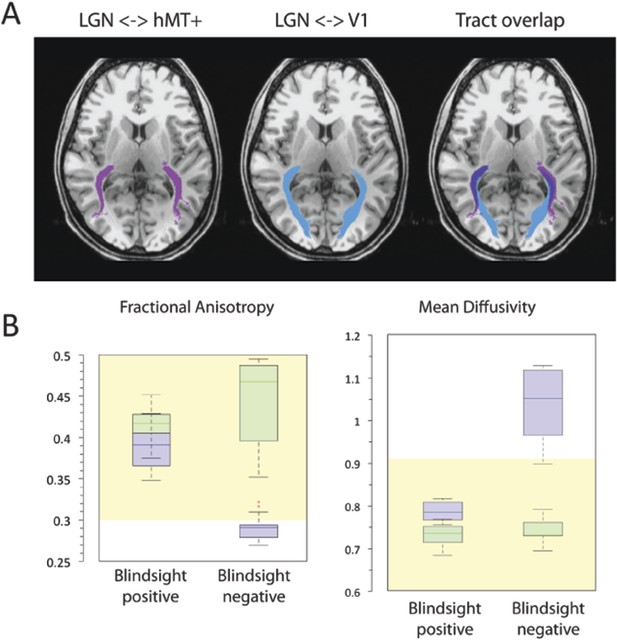
(A) Normal ipsilateral tracts between the LGN and hMT+, and the LGN and V1 demonstrate a proximal region of overlap.
Tracts are demonstrated in a control participant, C2, comparing ipsilateral connections between the LGN and hMT+ (pink) and the LGN and V1 (blue). When these pathways are superimposed, there is a significant region of overlap in the proximal portion of these pathways. In cases of V1 damage where there is retrograde degeneration, this overlapping region of the geniculate-hMT+ pathway may become contaminated by degenerated tracts in the V1 pathway. (B) Box plots comparing FA and MD in the distal portion of the geniculate-hMT+ pathway, in blindsight positive and negative patients. The ipsilesional hemisphere is shown in purple, and the intact hemisphere in green. Blindsight positive patients show significant overlap in the FA of the distal portion of this pathway in the damaged and sighted hemispheres. There is a slight increase in MD in the damaged hemisphere, however this is not marked and both measures fall within the control range. In comparison, blindsight negative patients show a marked difference in FA and MD for this pathway in the damaged and sighted hemispheres. The ipsilesional measures extend beyond the control range, implying that they are pathological and significantly impaired. Adjacent values are defined as the lowest and highest observations that are still inside the region defined by the following limits: Lower Limit = Q1 − 1.5 x IQR, Upper Limit = Q3 + 1.5 × IQR. The age-matched control FA and MD range for this pathway are displayed in yellow.
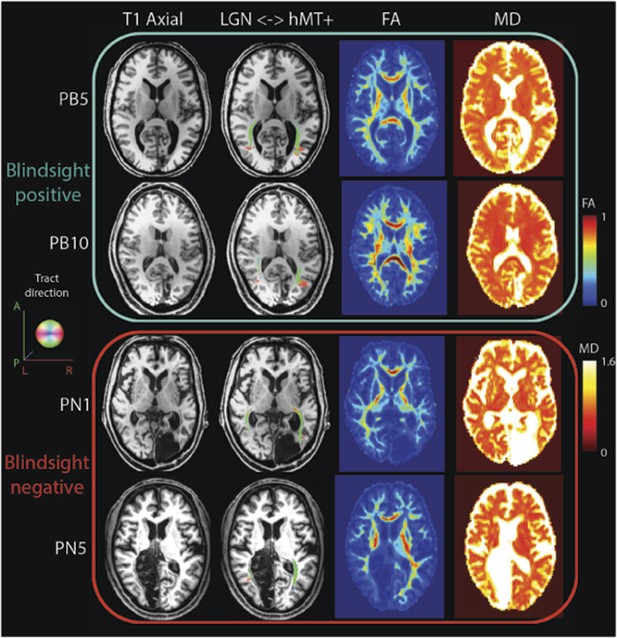
FA and MD maps in blindsight positive and negative patients, demonstrating the spatial relationship with the geniculate-hMT+ pathways.
Individual results are shown for two blindsight positive patients PB5, and PB10 and two blindsight negative patients, PN1 and PN5. All four patients showed bilateral ipsilateral fascicles between the LGN and hMT+, including the damaged hemisphere (column two). In the damaged hemisphere of blindsight positive patients the region directly underlying tracts corresponds to relatively intact MD and FA measures, not notably different from the intact hemisphere. However, both blindsight negative patients have tracts in the damaged hemisphere that traverse a region of tissue with markedly abnormal FA and MD values (columns three and four).
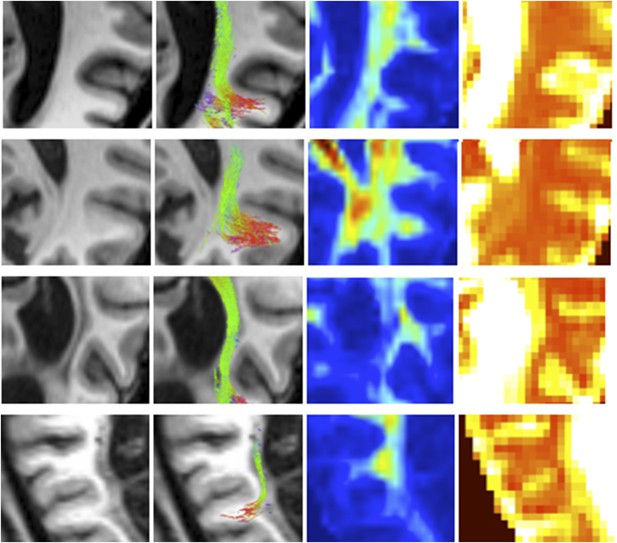
Zoomed in view demonstrating ipsilesional geniculate-hMT+ tracts with the corresponding T1-weighted structural, FA and MD maps.
Blindsight positive patients are shown in A and B with blindsight negative patients in C and D.
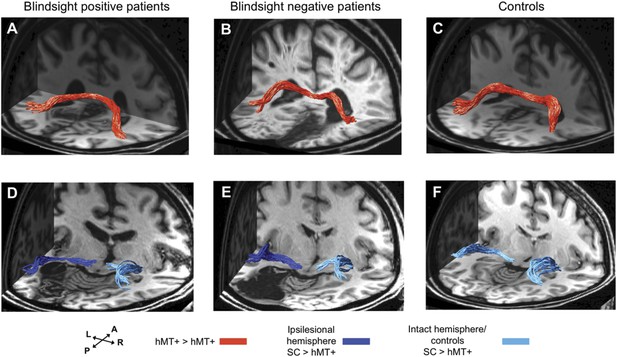
3-D representations of interhemispheric tracts between hMT+ bilaterally and ipsilateral tracts between SC and hMT+.
(A–C) Interhemispheric hMT+ tracts in blindsight positive patient, PB3, a blindsight negative patient, PN2 and a control participant, C9. (D–F) Ipsilateral collicular-hMT+ tracts in blindsight positive patient, PB8, a blindsight negative patient, PN3 and a control participant, C2. Red tracts represent crossing, interhemispheric connections between hMT+ bilaterally. Dark blue tracts are connections between SC and hMT+ in the ipsilesional damaged hemisphere, light blue tracts show the same collicular-hMT+ pathway in the intact hemisphere, and in controls. Tracts are overlaid on a 3-D representation of participant's structural T1-weighted images.
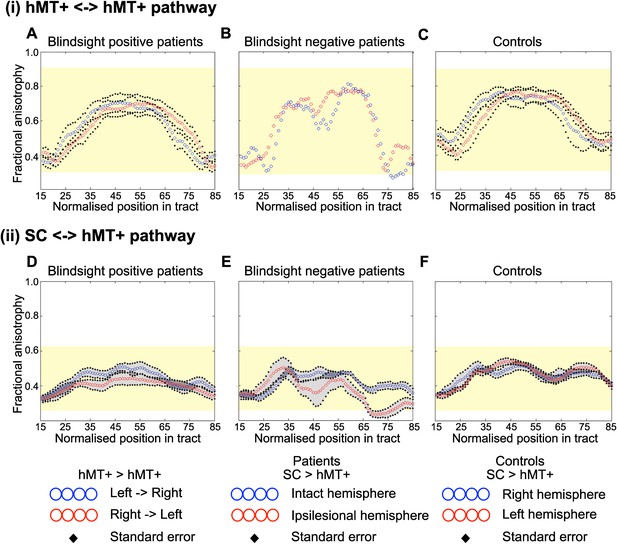
Average fractional anisotropy along the Interhemispheric hMT+ pathway and ipsilateral pathway between SC and hMT+.
(A) Blindsight positive patients show a similar FA to controls along the length of interhemispheric hMT+ pathways. (B) Blindsight negative patient, PN2, also shows a similar FA to controls along the length of this pathway. (C) Control participants show a normal peak in FA at the centre of the interhemispheric hMT+ pathway, representing the high degree of anisotropy at the corpus callosum. (D) Blindsight positive patients show a similar FA in the ipsilesional collicular-hMT+ pathway as the intact hemisphere and controls. (E) Blindsight negative patients show a slight drop in mean FA in the distal third of the ipsilesional collicular-hMT+ pathway. (F) Control participants show a fairly constant FA along the length of the collicular-hMT+ pathway, around 0.4. The control range for each pathway is displayed in yellow.
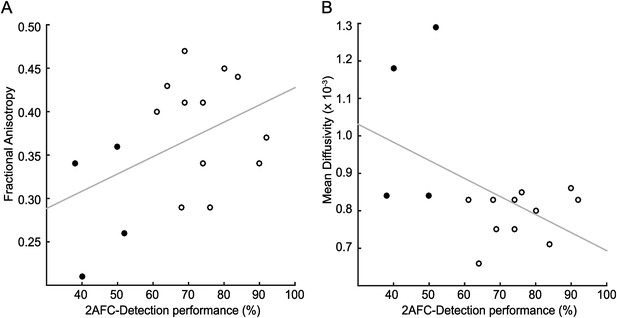
Correlation of tract microstructure in the distal region of the geniculate-hMT+ pathway with behavioural performance on the contrast detection task.
In both plots the filled symbols represent the values for the blindsight negative patients while the open symbols are from the blindsight positive patients. (A) shows the data for the FA values (r = 0.43; p = 0.09) and (B) shows the corresponding values for MD (r = −0.48; p = 0.06).
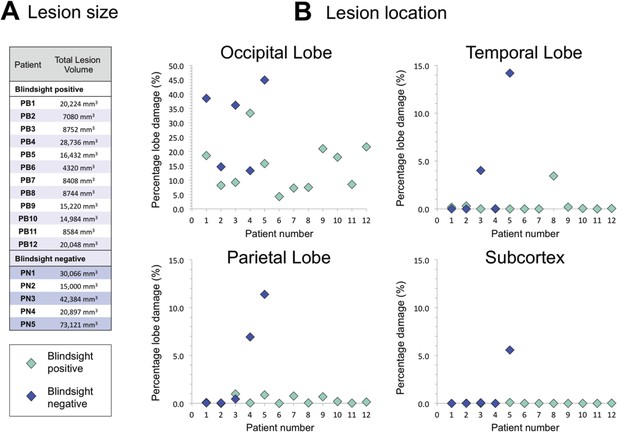
Comparison of lesion size and location in blindsight positive and negative patients.
(A) Lesion size is given for each patient, and demonstrates a wide range of volumes in both patient groups. (B) Lesion location shows the proportion of lobe damage in each patient, within the occipital, temporal, and parietal lobes, as well as the subcortex. Subcortex incorporates the thalamus (including LGN and pulvinar), striatum, and superior colliculi, with an approximate unilateral volume of 50,000 mm3. Only one patient, PN5, demonstrated some involvement of this region, including the ipsilesional LGN and pulvinar, but not the superior colliculi.
Tables
Number of cleaned fascicles for the three pathways of interest in patients and control participants
| Subject | LGN ↔ hMT+ | Crossing hMT+ | SC ↔ hMT+ | |||
|---|---|---|---|---|---|---|
| Ipsi-lesional | Contra-lesional | Left → right | Right → left | Ipsi-lesional | Contra-lesional | |
| Blindsight positive patients | ||||||
| PB1 | 75 | 115 | 9 | no | 12 | 12 |
| PB2 | 19 | 196 | 6 | no | no | 18 |
| PB3 | 50 | 67 | 24 | 24 | 17 | 17 |
| PB4 | 19 | 315 | no | no | no | 15 |
| PB5 | 93 | 83 | 7 | 19 | 14 | 14 |
| PB6 | 12 | 64 | 13 | 15 | no | 17 |
| PB7 | 397 | 17 | no | no | 16 | 8 |
| PB8 | 87 | 37 | 12 | 16 | 20 | 17 |
| PB9 | 635 | 53 | no | no | 16 | no |
| PB10 | 32 | 29 | no | no | no | no |
| PB11 | 291 | 47 | 9 | 18 | 12 | no |
| PB12 | 194 | 17 | 17 | 13 | 15 | no |
| Blindsight negative patients | ||||||
| PN1 | 157 | 19 | no | no | 15 | 8 |
| PN2 | 17 | 226 | 13 | 15 | 7 | 21 |
| PN3 | 351 | 89 | no | no | 19 | 16 |
| PN4 | no | 101 | no | no | no | 19 |
| PN5 | 15 | 122 | no | no | no | 13 |
| Controls | ||||||
| C1 | 308 | 339 | 19 | 14 | 14 | 18 |
| C2 | 619 | 269 | no | no | 39 | 17 |
| C3 | 57 | 59 | 8 | 16 | 18 | no |
| C4 | 176 | 114 | 8 | 6 | 18 | 16 |
| C5 | 84 | 30 | 5 | 8 | 17 | 16 |
| C6 | 57 | 19 | no | no | 15 | 16 |
| C7 | 78 | 46 | no | no | 9 | no |
| C8 | 498 | 182 | 19 | 14 | 35 | 14 |
| C9 | 653 | 62 | 57 | 52 | 31 | 9 |
-
(1) Ipsilateral pathway between LGN and hMT+ (2) Pathway between hMT+ bilaterally via the corpus callosum (3) Ipsilateral pathway between SC and hMT+. Results are shown separately for the intact and damaged ‘ipsi-lesion’ hemispheres (right and left for control participants). ‘no’ = zero fascicles survived the cleaning process.
Additional files
-
Supplementary file 1
Clinical characteristics of patients.
- https://doi.org/10.7554/eLife.08935.013
-
Supplementary file 2
Number of uncleaned fascicles for the three pathways of interest in patients and control participants: (1) Ipsilateral LGN and hMT+ (2) hMT+ bilaterally via the corpus callosum (3) Ipsilateral SC and hMT+. Results are shown separately for the intact and damaged ‘ipsi-lesion’ hemispheres (right and left for control participants).
- https://doi.org/10.7554/eLife.08935.014





