Localized epigenetic silencing of a damage-activated WNT enhancer limits regeneration in mature Drosophila imaginal discs
Figures
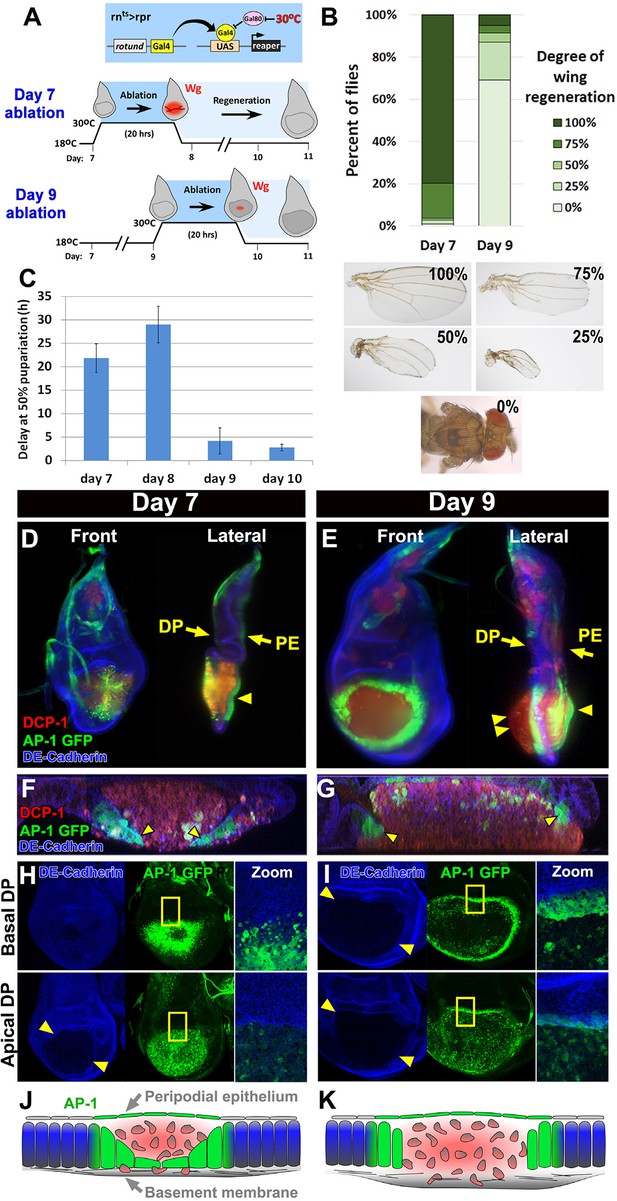
Cellular changes that accompany reduced regeneration in older discs.
(A) The rnts>rpr ablation system used to induce tissue damage and stimulate regeneration in L3 wing-imaginal discs on day 7 and day 9 of development. The molting from 2nd to 3rd larval instar occurs around day 6 at 18°C. Gal4 under the control of the rotund (rn) enhancer drives the pro-apoptotic gene rpr (or egr in rnts>egr) in the developing wing pouch. Ablation is repressed at 18°C by a ubiquitous temperature-sensitive Gal80 (Gal80ts), until the temperature is increased to 30°C to inactivate the Gal80ts and induce cell death. Ablation of the pouch is complete after 20 hr for rnts>rpr (or 40 hr for rnts>egr). The temperature is then reduced (downshift) to end ablation and allow regeneration. Wg (red) is observed in response to ablation in a day 7 disc, but this response is greatly diminished in a day 9 ablated disc, correlating with a loss of regenerative ability. (B) Distribution of adult wing sizes from rnts>rpr flies where a 20 hr ablation was initiated on day 7 or day 9 of larval development, showing a decrease of regeneration in mature discs. n > 400 flies per experiment. Examples of wing sizes scored are shown below. In almost all cases both wings of a single fly showed equivalent levels of regeneration. (C) Average delay measured as hours at which 50% of larvae have pupariated compared to unablated wild type. Ablation induces a developmental delay of 22.2 hr on day 7, which is progressively reduced to just 2.8 hr on day 10. Error bars are SD of at least 3 biological repeats. (D-I) Wing discs imaged following rpr ablation at the time of the downshift to 18°C. (D-E) Light sheet microscopy of wing discs following ablation with rnts>rpr on day 7 (D) and day 9 (E). Discs were embedded in agarose and imaged in 360°. Two orthogonal planes are shown. Abundant cellular debris is observed between the peripodial epithelium (PE) and the bulging basement membrane of the disc proper (DP) and is visualized with an antibody to cleaved Drosophila DCP1 (DCP1, red). In day 9 discs cell debris is seen outside of the DP, confined by the basement membrane (double arrowhead). Cells expressing the AP-1 reporter (AP-1, green) are found adjacent to the ablated region in the DP and in the intact PE overlying the ablated region (single arrowheads) on both day 7 and day 9 ablations. Blue: DE-cadherin. (F-G) Confocal Z-sections of the ablated pouch of a day 7 (F) and day 9 (G) ablation. There is a discontinuity in the DP following ablation. However following a day 7 ablation (F), but not following a day 9 ablation (G), AP-1 positive cells (arrowheads) appear to migrate inward to close the gap in the DP. (H-I) Expression of an AP-1 GFP reporter in discs ablated on day 7 (H) and day 9 (I), and imaged at basal (top panels) and apical (bottom panels) levels of the DP. Zoom panel shows enlargement of the area within the yellow rectangle. On both days 7 and 9, AP-1-GFP is also observed in the cells of the DP surrounding the ablated region and in the debris within the DP. The flattened cells that seem to be closing the ablated region on day 7 are observed at basal but not apical focal planes (H). The AP-1 positive cells adjacent to the ablation in day 9 discs retain their columnar morphology (G) and do not seem to cover the discontinuity in the DP caused by the ablation (I). (J-K) Drawings illustrating the contrasting cellular responses to ablations initiated on day 7 and day 9.
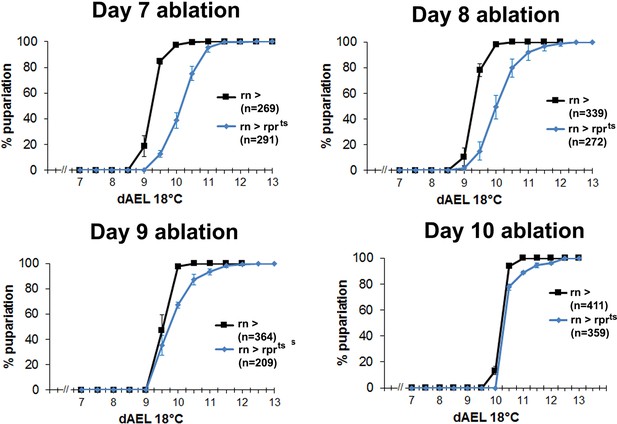
Developmental delay associated with damage declines in mature discs.
Graphs showing developmental timing of pupariation in wild type larvae following rnts>rpr ablation on day 7, 8, 9 and 10, compared to the undamaged control. Error bars are SD of at least 3 biological repeats, n is shown for each condition.
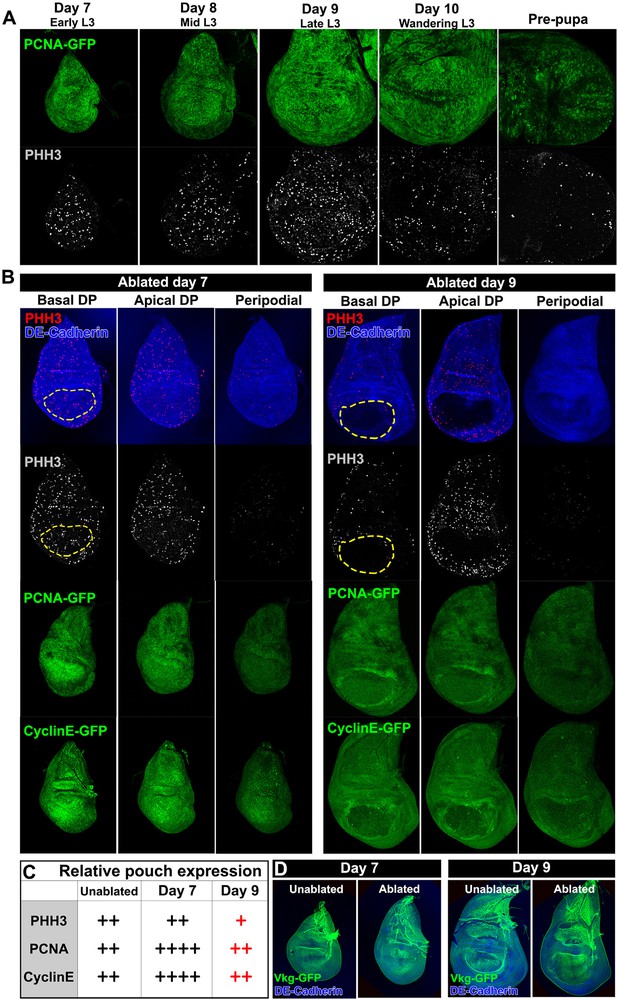
Cells in the wing disc are still proliferating at a time when they can no longer regenerate.
(A) Wild type discs at days 7 through 10 of L3, and at the prepupal stage, showing expression of the PCNA-GFP transgene as an indicator of E2F activity (green), and phospho-histone H3 (PHH3, gray) showing mitotic cells. Markers for proliferation are present throughout L3 development up to pupariation. (B) Discs ablated with rnts>rpr at day 7 (left) and day 9 (right), imaged at the time of downshift at basal DP, apical DP and peripodial levels, showing expression of PHH3 (red and gray), PCNA-GFP (green) and a GFP reporter of CyclinE (green). Structure of the discs is shown by DE-Cadherin (blue), and the approximate extent of the wing pouch is indicated by the dashed lines. There is an increased number of PHH3-positive proliferative cells within the ablated region on day 7 versus day 9, likely caused by the rapid migration of proliferating cells into the ablated region. (C) Summary of the gene expression observed in (B). Together the markers reveal that the proliferative response to damage declines in older discs. Note that cells in mitosis are only a subset of actively proliferating cells, and therefore the PHH3 marker is likely an underrepresentation of cell proliferation. (D) Expression of the basement membrane component Vkg-GFP in unablated and ablated day 7 and day 9 discs. Ablation does not affect the integrity of the basement membrane at either stage of development.
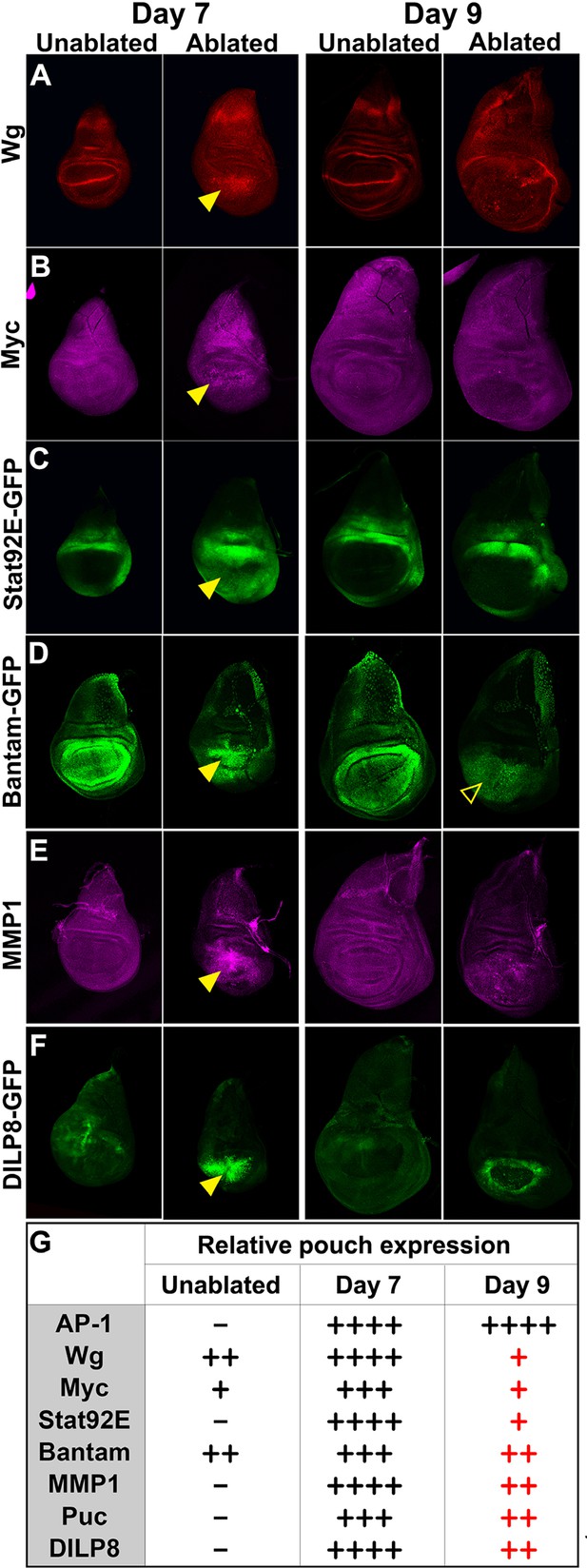
Changes in expression of key regulators of regeneration that accompany reduced regeneration in older discs.
(A-F) Basal confocal sections of discs ablated with rnts>rpr on day 7 or day 9 imaged at the downshift to 18°C, with equivalently staged unablated discs, detecting (A) Wg, (B) Myc, (C) Stat92E-GFP reporter (D) bantam-GFP reporter, (E) Mmp1 and (F) DILP8-GFP reporter. In each case the expression of the protein or reporter is increased by ablation on day 7 (arrowheads) when compared to identically stained unablated control discs. In discs ablated on day 9, there is no detectable upregulation of Wg (A) or Myc (B) in the pouch. Expression of the bantam-GFP (D), Mmp-1 (E) and DILP8-GFP (F) is consistently reduced when compared to a day 7 ablation and coincides mainly with cellular debris (for example, open arrowhead in D). (G) Summary of gene expression observed in this study in discs ablated on day 7 and day 9, compared to unablated discs. Results that change between day 7 and day 9 are indicated in red.
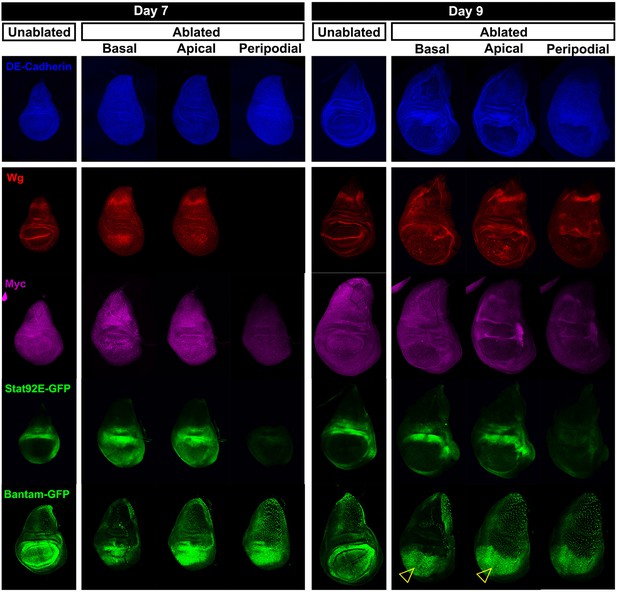
Expression of reporters shown at multiple sections in the epithelium.
Wing discs following ablation with rnts>rpr on day 7 and day 9 imaged at the downshift to 18°C. Optical sections at basal disc proper, apical disc proper and peripodial epithelium levels, with unablated control discs for comparison are shown. Genes studied include those involved in growth and patterning; Wg pathway (anti-Wg), Myc (anti-Myc), JAK/STAT (multimerized STAT binding site GFP reporter), Yorkie activity (a bantam-GFP reporter). Cellular debris is indicated by open arrowheads. DE-cadherin, blue.
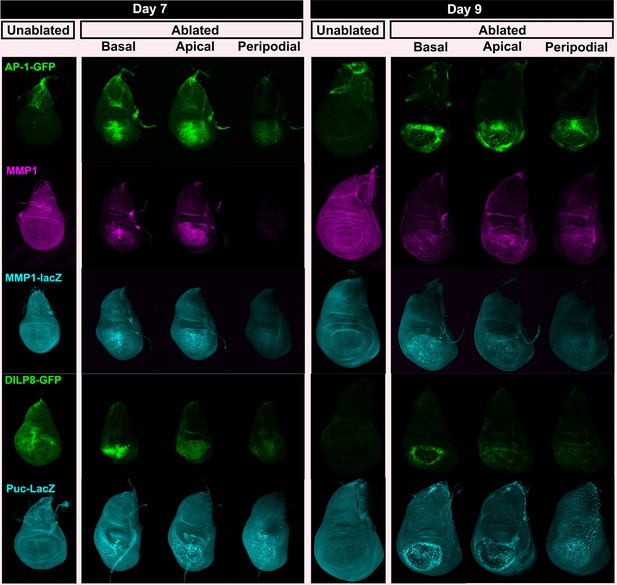
JNK pathway target gene expression shown at multiple levels.
Wing discs as in Figure 2—figure supplement 1, examining reporters and targets of the JNK pathway; AP-1 activity (multimerized AP-1 binding site GFP reporter), Mmp1 (anti-Mmp1 and an Mmp1-lacZ reporter), DILP8 (a DILP8-GFP gene trap) and Puckered (a puc-lacZ reporter).
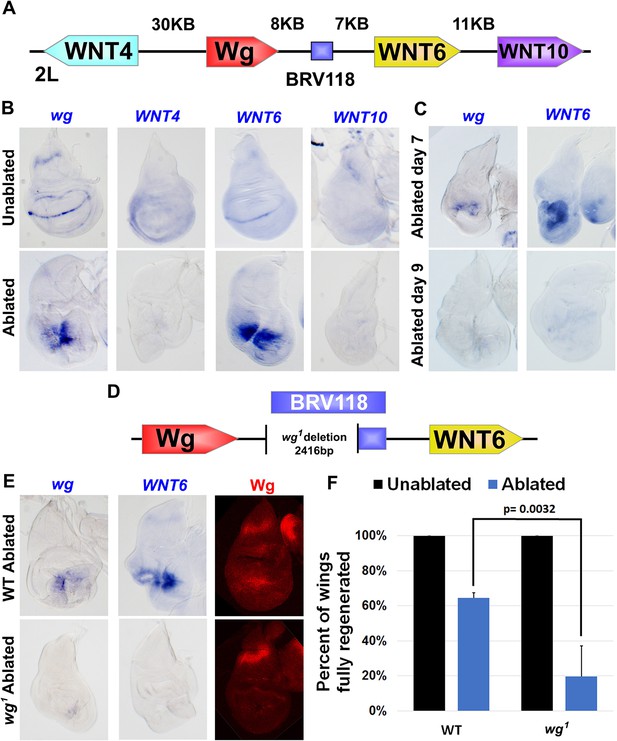
The BRV118 enhancer is necessary for wg and Wnt6 expression following tissue damage.
(A) Schematic of the major WNT locus showing the BRV118 enhancer (blue rectangle). Approximate intergenic distances are labeled. (B) RNA in situ hybridization to detect wg, Wnt4, Wnt6 and Wnt10 RNA in day 7 unablated (top row) and rnts>egr ablated (bottom row) wing discs. Only wg and Wnt6 demonstrate significant upregulation of RNA in response to damage. (C) RNA in situ hybridization to detect wg, and Wnt6 RNA in discs ablated with rnts>egr on day 7 and day 9. Transcription of both wg and Wnt6 in response to damage is absent in a day 9 disc. (D) Schematic of the lesion in the wg1 allele that deletes most of the BRV118 enhancer. (E) RNA in situ hybridization to detect wg and Wnt6 RNA and Wg protein, in wild type (top row) and homozygous wg1 (bottom row) day 7 rnts>egr ablated discs. The damage-specific expression of wg and Wnt6 RNA, and Wg protein levels, are reduced in wg1 discs compared to wild type. (F) Adult wing sizes following ablation with rnts>egr on day 7 in wild type and wg1 homozygotes, indicating the percentage of animals that eclose with fully regenerated wings. Regeneration of wg1 homozygous discs is significantly reduced compared to wild type. Error bars indicate SD of at least three independent experiments, scoring a total of >200 animals in each experiment. Only untransformed wg1 wings were scored (for explanation of untransformed see Figure 3—figure supplement 1).
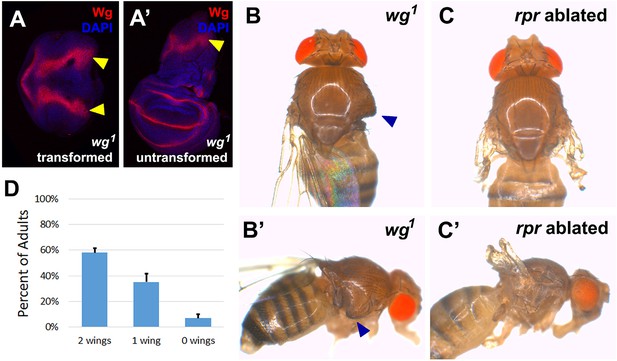
The wg1 allele causes transformation of wing to notum in a proportion of the population.
(A-A’) Wing discs dissected from day 7 wg1 homozygous larvae and stained for Wg (red) and DAPI (blue), showing a transformed disc (A), in which presumptive wing tissue is replaced by notum, indicated by the duplicated wg notum stripe (arrowheads), and an untransformed wing disc (A’) that is morphologically normal and has wild type wg expression. Transformation is thought to occur at L2 when the wing pouch is specified by wg expression (Sharma and Chopra, 1976; Whitworth and Russell, 2003), and thus ablation of the pouch by rnts>rpr or rnts>egr does not occur in these discs, (B-B’) A wg1 homozygous adult with one untransformed left wing and a transformed right wing. Wing tissue is replaced by additional notum and associated bristles following transformation (arrowheads). (C-C’) For comparison, a wild type adult with two non-regenerated wings resulting from rnts>rpr ablation. The presence of hinge tissue and normal bristle formation allows clear distinction between ablated versus transformed wings; only untransformed wings were scored in our assays. (D) Quantification of the wing to notum transformation frequency in the wg1 homozygous stock used in our experiments. Error bars are SD, n>300 flies. On average 58% of animals eclose with two untransformed wings. The frequency of the variably penetrant transformation phenotype can be shifted by selective breeding of two wing, one wing or no wing flies, but not eliminated (Sharma and Chopra, 1976).
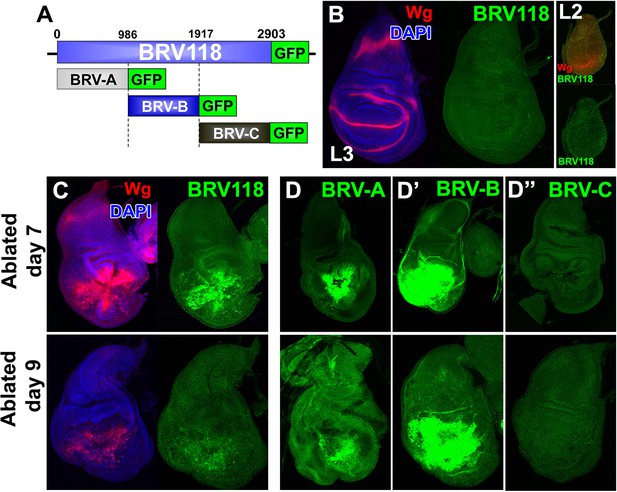
BRV118 is a damage-responsive WNT enhancer.
(A) Schematic of the 2.9 kb BRV118-GFP reporter transgene, and the subdivisions used to generate the ~1 kb BRV-A, BRV-B and BRV-C reporters. All transgenes were inserted into the same genomic landing site to enable direct comparison. Numbers indicate nucleotide positions in BRV118. (B) Unablated day 7 disc bearing the BRV118-GFP reporter, stained for Wg (red) and DAPI (blue). No GFP is detected during normal development throughout L3 (green) or L2 (insets). Discs were staged using the Wg expression patterns: diffuse Wg in the primordial pouch tissue to indicate L2 and the defined D-V stripe and "hinge circles" to indicate L3 (red). (C, D-D’’) Discs bearing the BRV118-GFP (C), BRV-A (D), BRV-B (D’) or BRV-C (D’’) reporters ablated with rnts>egr on day 7 (top row) or day 9 (bottom row). The BRV118-GFP reporter (green) is expressed in a pattern closely resembling that of damage-induced Wg (red), on both day 7 and day 9. The BRV-A reporter has weaker activation than the full enhancer on day 7 and on day 9. The BRV-B reporter has much stronger activation than the full BRV118 and is not attenuated with age. BRV-C is not expressed following ablation.
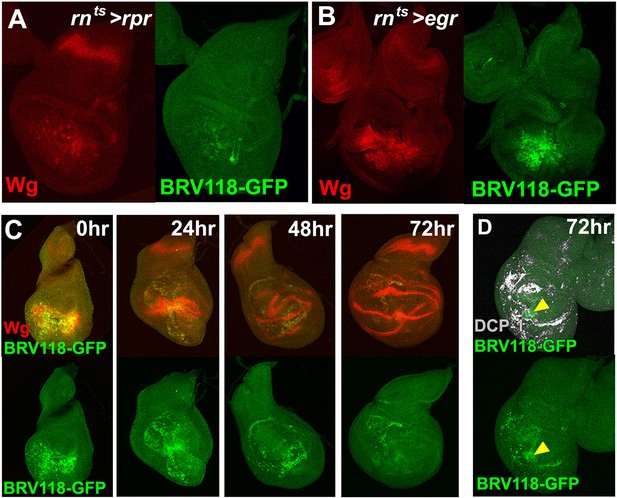
The BRV118 enhancer reporter demonstrates damage-responsive but not developmentally-regulated wg expression.
(A-B) Discs bearing the BRV118-GFP reporter ablated with rnts>rpr(A) and rnts>egr(B) on day 7, stained for GFP (green) and Wg (red), showing the enhancer responds to rpr induced cell death at a lower level than egr, similar to wg expression. (C) Discs bearing the BRV118-GFP enhancer following ablation with rnts>egr on day 7 and imaged at 0 hr (immediately at temperature downshift), 24 hr, 48 hr and 72 hr of regeneration. Discs are stained for GFP (green) and Wg (red), showing the initial overlap of expression patterns between Wg and the enhancer reporter (at 0 hr and 24 hr), followed by divergence of the expression domains, where Wg regains its developmental expression pattern in the hinge and D-V boundary, while GFP expression is maintained in the blastema of the regenerating pouch (48 hr and 72 hr). This GFP is distinct from cell debris, indicated by DCP-1 staining (gray, D).
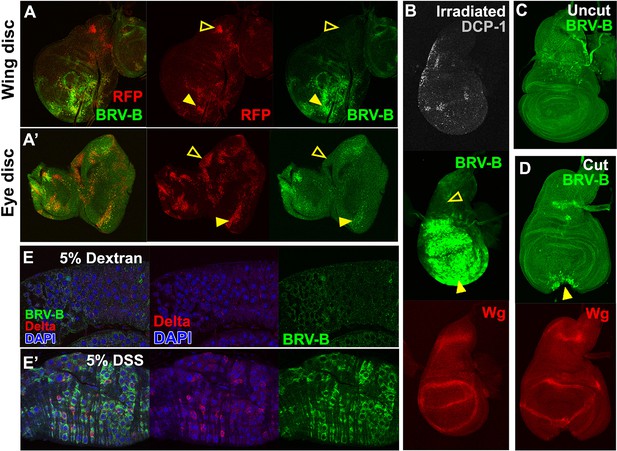
BRV-B is a damage-responsive enhancer in multiple contexts.
(A-A’) egr-expressing clones activate BRV-B in some but not all regions of wing and eye discs. Heat-shock induced FLP-out clones expressing Gal4 (marked by RFP, red) were generated on day 4 and 5 of development in larvae bearing the BRV-B reporter. Clones were allowed to grow, and subsequently Gal4 was activated by inactivation of Gal80ts to express egr for 24 hr on day 8. GFP is expressed in and surrounding large dying clones (closed arrowheads) in wing discs (A) and eye/antennal discs (A’), but is absent from egr-expressing clones in various parts of each disc (open arrowheads), including the notum of the wing disc. (B) Disc from larva bearing the BRV-B reporter irradiated with 45 Gy on day 7, dissected after 16 hr and stained for cell death marker DCP-1 (gray), Wg (red) and GFP (green). The BRV-B reporter is strongly activated throughout the wing pouch and hinge (arrowhead), but not in the presumptive notum (open arrowhead). (C-D) Day 7 discs bearing the BRV-B reporter, physically cut (D, arrowhead indicates cut site) or left intact (C), and cultured in Schneider’s medium for 16 hr, followed by staining to detect GFP and Wg. Reporter activation is detected specifically along the cut site. (E-E’) Confocal sections through midguts of adult flies bearing the BRV-B reporter following 2 days of feeding 5% dextran solution as control animals, (E) or gut-damaging 5% DSS solution (E’). Guts were stained with anti-Delta (red) to show the intestinal stem cell (ISC) population, and GFP to detect reporter activity. Damaged gut tissue in DSS fed animals increases ISC number. Strong induction of the BRV-B reporter (green) is observed but not in the population that expresses Delta.
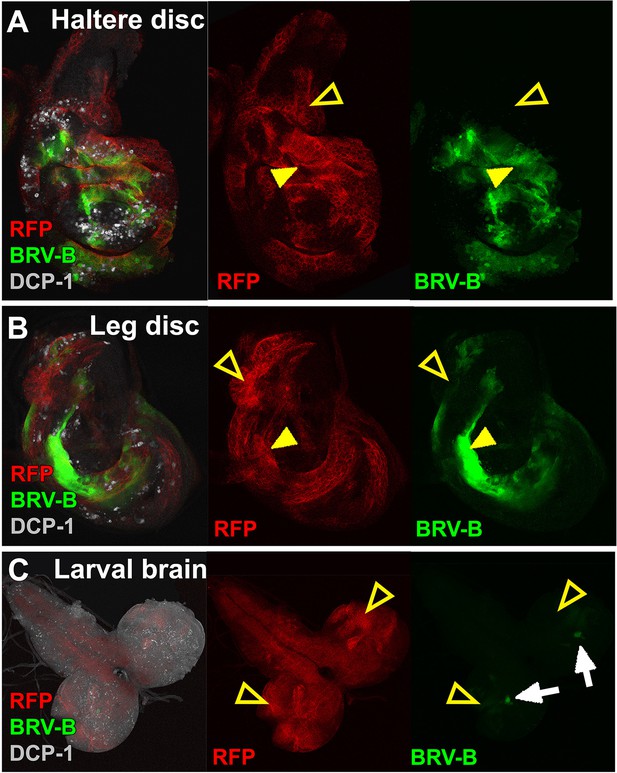
The BRV-B enhancer reporter is activated by genetic ablation in different tissues.
(A-C) Temporally controlled expression of egr in clones generated within haltere (A) and leg (B) discs of larvae bearing the BRV-B reporter, as in Figure 5A–A’. Expression of egr in clones of day 8 discs results in activation of the reporter in and around dying clones (closed arrowheads). Some areas of the discs are refractory to reporter activation despite egr expression (open arrowheads). Similar expression of egr in clones in the non-regenerating tissue of the larval brain (C) does not result in BRV-B activation (open arrowheads). Low level expression of the reporter can be seen in unidentified neurons within of the optic lobe, which is unrelated to ablation (white arrows).
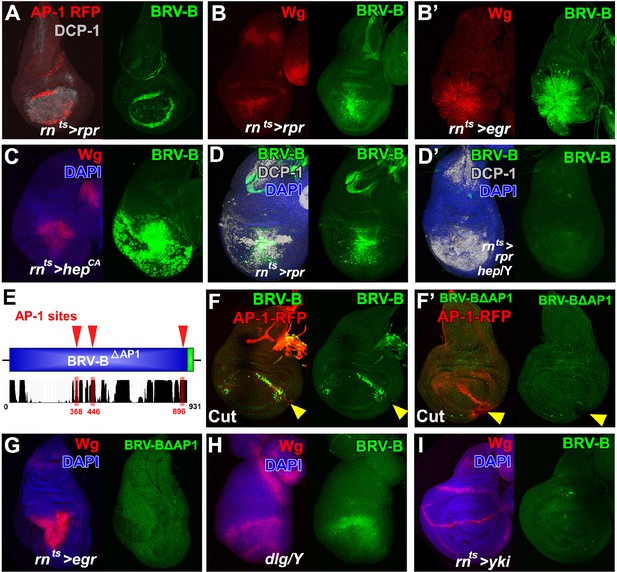
BRV-B reporter activation requires the JNK/AP-1 pathway.
(A) Basal section of a day 9 disc bearing the AP-1 RFP reporter shows JNK pathway activity (red) and the BRV-B GFP reporter (green) following ablation by rnts>rpr, imaged at the time of downshift to 18°C. Reporter expression overlaps in wound edge cells, distinct from dead cells (DCP-1, gray), (B-B’) Day 7 discs bearing the BRV-B reporter, ablated using rnts>rpr (B) or rnts>egr (B’) and imaged at the time of downshift to 18°C, demonstrating stronger expression of both Wg (red) and BRV-B GFP (green) following ablation with egr compared to rpr. (C) A day 7 disc bearing the BRV-B reporter expressing rnts>hepCA for 20 hr to activate JNK signaling in the wing pouch. Both Wg (red) and the BRV-B GFP (green) are strongly expressed. (D-D’) Day 7 discs bearing the BRV-B reporter ablated with rnts>rpr in a wild type (D) or hep- hemizygote (D’), imaged at the time of downshift to 18oC. GFP expression is abolished in the hep- mutant following ablation, while cell death is unaffected (DCP-1 staining, gray). (E) Schematic showing three predicted AP-1 binding sites (red) in the BRV-B enhancer fragment. The black histogram below shows a measure of the evolutionary conservation of the BRV-B DNA sequence in twelve Drosophila species, mosquito, honeybee and red flour beetle, based on a phylogenetic hidden Markov model (https://genome.ucsc.edu/). Numbers indicate nucleotide position in the BRV-B element. (F-F’) Day 7 discs bearing an AP-1-RFP reporter and BRV-B (F) or BRV-B with AP-1 sites deleted (BRV-BΔAP-1, F’) physically cut and cultured for 16 hr. Activation of BRV-B (green) occurs along the edges of the cut site (arrowheads), coincident with AP-1-RFP expression (red) (F). Conversely the BRV-BΔAP-1 reporter is not activated by physical wounding (F’). (G) Basal section of a day 7 disc bearing BRV-BΔAP-1, following rnts>egr ablation, imaged at the time of downshift to 18oC. Loss of the predicted AP-1 sites abolishes activation of the reporter (green). (H) Maximum projection Z-stack of a dlg hemizygous mutant disc from early L3 bearing the BRV-B enhancer. BRV-B reporter expression is maximal in the dorsal hinge region coinciding with the site most prone to neoplastic overgrowth (Khan et al., 2013). (I) Maximum projection Z-stack of a day 7 disc bearing the BRV-B enhancer following expression of rnts>yki for 20 hr. Hyperplastic growth resulting from yki expression fails to activate BRV-B GFP expression.
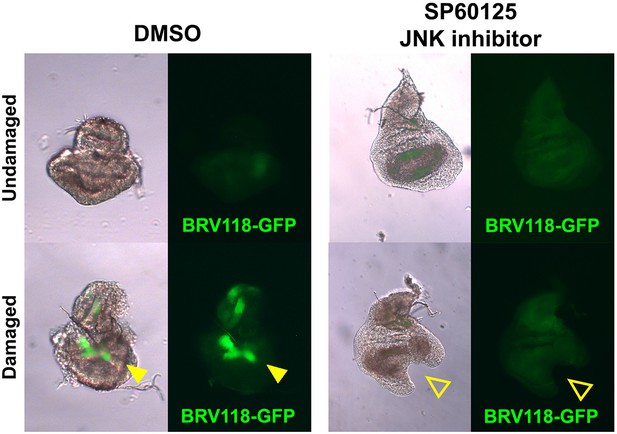
The BRV-B reporter is activated by physical damage, and is dependent on JNK signaling.
Day 7 discs bearing the BRV118-GFP reporter (green), physically fragmented with tungsten wire (yellow arrowheads), and cultured in Robb’s media for 16 hr, in the presence of 1% DMSO vehicle (left) or 10 μM of the small molecule JNK pathway inhibitor SP600125 in 1% DMSO (right). Addition of the inhibitor prevents activation of the reporter following damage (open arrowheads), and also constrains the morphological changes (folding and curling of the disc epithelium) associated with culturing conditions.
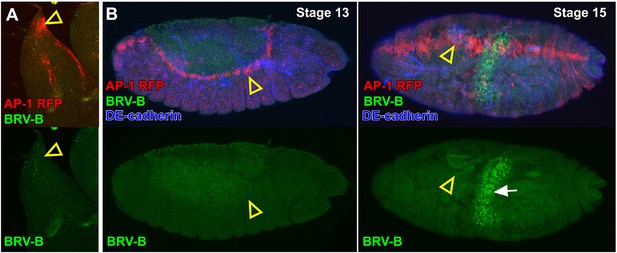
Activation of the BRV-B reporter coincides with damage-induced, but not developmentally regulated JNK pathway activity.
(A) Dorsal-most portion of a late larval wing disc bearing the AP-1 RFP reporter (red) and BRV-B GFP reporter (green), showing activation of the JNK pathway in the peripodial cells of the apical notum that will participate in JNK-dependent thorax closure after puparium formation (open arrowhead), but absence of BRV-B expression. (B) Stage 13 embryo (left) and stage 15 embryo (right) bearing the AP-1 RFP reporter (red) and the BRV-B GFP reporter (green) undergoing dorsal closure, showing activation of the JNK pathway in leading edge cells (open arrowheads), but absence of BRV-B expression. GFP autofluorescence of the gut is visible in the stage 15 embryo (white arrow). DE-cadherin, blue.
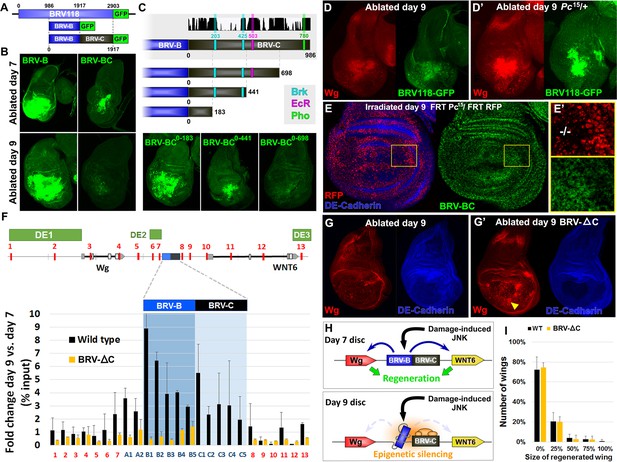
Polycomb-mediated epigenetic silencing of the BRV118 enhancer limits damage-responsive wg expression in mature discs.
(A) Schematic of the BRV-BC reporter transgene in relation to BRV118 and BRV-B. Numbers indicate nucleotide positions. (B) Basal sections of day 7 discs bearing the BRV-B (left panels) or BRV-BC (right panels) reporters, ablated by rnts>egr on day 7 (top row) or day 9 (bottom row). Addition of the BRV-C fragment reduces expression on day 7 and almost completely abolishes expression on day 9. (C) Schematic of the deletion series used to test silencing of BRV-B by BRV-C fragments (top). Predicted binding sites for Brinker (Brk), Ecdysone Receptor (EcR) and Pleiohomeotic (Pho) are shown as colored bars. The black histogram shows a measure of the evolutionary conservation in BRV-C, as in Figure 6E. Confocal sections of day 7 discs ablated by rnts>egr bearing each BRV-BC derivative (bottom) shows that multiple regions across BRV-C are required for silencing of BRV-B. (D-D’) Day 9 discs bearing the BRV-BC reporter following rnts>egr ablation in wild type (D) and Pc15/+ heterozygotes (D’). Both damage-induced Wg (red) and reporter GFP expression (green) is elevated in Pc15/+ discs compared to wild type. (E-E’) Heat shock induced Pc15 mutant clones (marked by the absence of RFP) in a wandering stage larval disc bearing the BRV-BC reporter (green). Following clone induction, discs were irradiated with 45 Gy and dissected after 16 hr. BRV-BC reporter expression is elevated in Pc15 clones (E’, -/- homozygous Pc15) but significantly lower in the wild-type twin spot (E’, red). (F) Chromatin immunoprecipitation by anti-H3K27me3 followed by qPCR to detect regions surrounding wg and Wnt6 (numbered primer sets 1–13) and the BRV118 region (lettered primers sets). ChIP-qPCR was performed in wild type and homozygous BRV-ΔC day 7 and day 9 discs, and shown as the fold change between the two days. Schematic above shows numbered amplicon positions in the genome, WNT gene loci and previously identified developmental WNT enhancers (green, DE1: Wing disc hinge and embryonic enhancers, (Neumann and Cohen, 1996; Von Ohlen and Hooper, 1997), DE2: Notum and leg/antennal disc enhancer (Pereira et al., 2006), DE3: Eye/antennal disc and leg enhancer (Koshikawa et al., 2015)). In wild type discs (black bars) epigenetic silencing marks increase at the BRV118 locus in day 9 discs versus day 7, while levels at the wg and Wnt6 coding sequences, and surrounding developmental wg enhancers, remain largely unchanged. Deletion of the BRV-C region of the enhancer from the genome abolishes the increase of H3K27me3 at the enhancer (yellow bars). Error bars are SD of repeats from 3 independent ChIP experiments. (G-G’) Day 9 wild type discs (G) or homozygous for BRV-ΔC (G’) following ablation by rnts>rpr. Absence of the BRV-C enhancer fragment produces significantly more Wg in response to damage (G’, arrowhead). (H) Model of damage response regulation in a day 7 (top) and a day 9 (bottom) disc. JNK signaling activates wg and Wnt6 expression in response to damage via the BRV-B region, but epigenetic silencing of the enhancer, nucleated by BRV-C, in a day 9 disc overrides this activation. (I) Assay of adult wing sizes that develop from homozygous BRV-ΔC discs ablated with rnts>rpr on day 9. Absence of the BRV-C enhancer fragment does not alter regeneration of ablated disc. Error bars are SD of 3 biological repeats, n>200 flies per repeat.
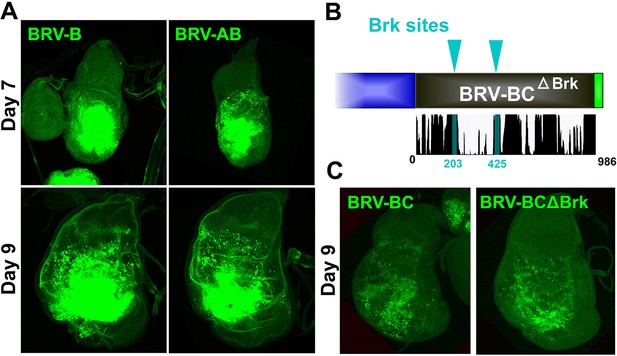
Additional characterization of the silencing activity in BRV118.
(A) Wing discs bearing the BRV-B (left) and BRV-AB (right) reporters following ablation with rnts>egr on day 7 (top) and day 9 (bottom), stained for GFP (green). Addition of the BRV-A fragment does not reduce expression of the BRV-B reporter following ablation on either day. (B) Diagram of predicted Brk binding sites that are deleted in the BRV-BC reporter, yielding the BRV-BCΔBrk reporter transgene. (C) Wing discs bearing the BRV-BC and BRV-BCΔBrk reporter transgenes (green) following ablation with rnts>egr on day 9, stained for GFP (green). Loss of the Brk sites does not alleviate silencing by the BRV-C fragment.
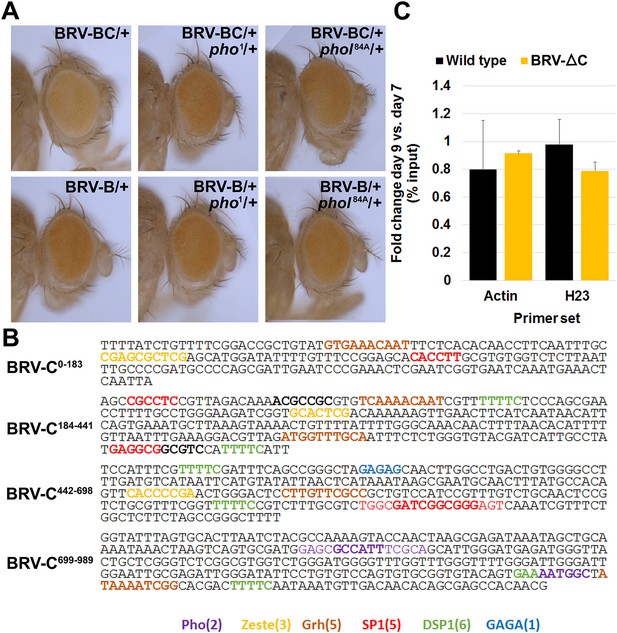
Additional data on PcG-mediated repression by BRV-C.
(A) Eye colors resulting from expression of the w+ marker gene associated with the BRV-B (top row) or BRV-BC (bottom row) reporter transgenes, in wild type (left panels), a pho null heterozygous mutant (middle panels) or a phol heterozygote (right panels). Presence of the BRV-C fragment reduces expression of the w+, resulting in lighter eyes, while this difference is lost in the pho/+ and phol/+ mutants. (B) Sequences that match consensus binding sites for PcG proteins within the BRV-C genomic region. Motifs for the following are highlighted: Pleiohomeotic (Pho, purple, [Mihaly et al., 1998; Fritsch et al., 1999; Ringrose et al., 2003]) Zeste (yellow, (Mihaly et al., 1998), Grainyhead (Grh, brown, [Mace et al., 2005]), SP1 (red, [Brown et al., 2005]), Dorsal Switch Protein 1 (DSP1, green, [Déjardin and Cavalli, 2005]) and GAGA factor (GAGA, blue, [Strutt et al., 1997; Ringrose et al., 2003; Zhu et al., 2011]). (C) Control regions for the H3K27me3 ChIP of wing discs. Graph shows level of H3K27me3 binding at the act5c transcribed region (open chromatin) and the heterochromatin H23 loci (closed chromatin) (Zhang et al., 2008) in wild type and BRV-ΔC discs, shown as the ratio of binding in day 9 versus day 7 discs. Level of H3K27me3 binding does not change at either locus from day 7 to day 9. Error bars are SD of repeats from 3 independent ChIP experiments.
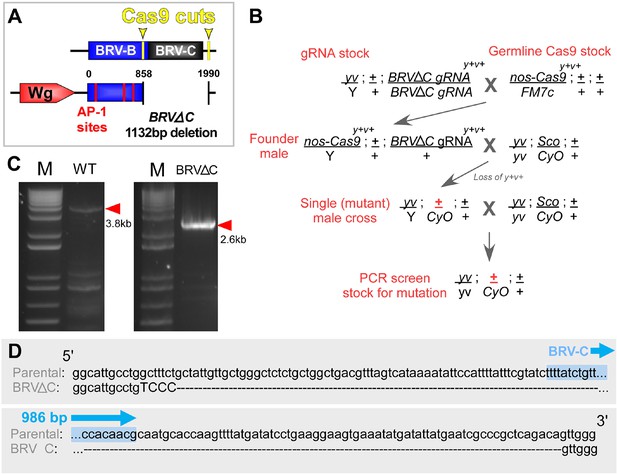
Cas9-mediated deletion of the BRV-C genomic region.
(A) Schematic of the CRISPR mediated BRV-C genomic deletion used to generate the BRV-ΔC allele. Numbers indicate nucleotide positions relative to BRV-BC. (B) Crossing scheme used to generate the BRV-ΔC allele. Over 100 lines were screened via genomic PCR, yielding a single deletion line. (C) PCR showing amplicon of BRV-C genomic region from the parental line (left) and BRV-ΔC line (right). (D) Sequences of the parental chromosome used to generate the BRV-ΔC allele, and the deletion in relation to the 986 bp BRV-C sequence used in the enhancer reporter transgenes (shown in blue).
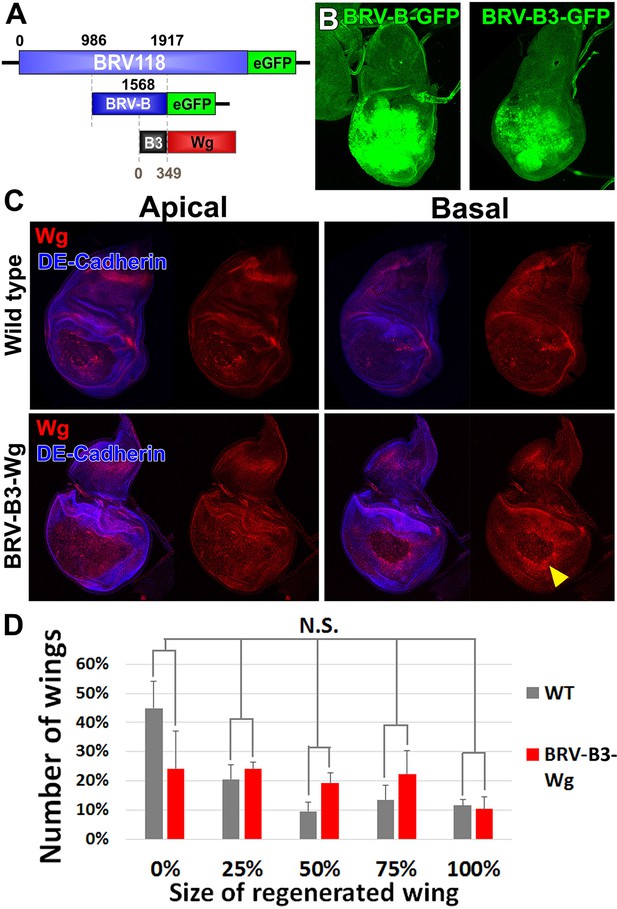
A fragment of BRV-B driving wg rescues damage-induced wg expression in late stage discs, but does not improve regeneration.
(A) Schematic of the BRV-B3-wg transgene. The B3 fragment is a subdivision of the full BRV-B enhancer. Numbers indicate nucleotide positions. (B) Day 7 discs bearing BRV-B and BRV-B3 GFP reporters following ablation by rnts>egr, imaged at the time of downshift. BRV-B3 yields slightly reduced levels of expression compared to BRV-B. (C) Apical and basal sections of disc proper showing Wg (red) following rnts>rpr ablation on day 9 in wild type (top) and BRV-B3-wg bearing discs (bottom). The BRV-B3-wg transgene causes expression of Wg in the basal wound periphery cells (arrowhead), which is absent in the wild type disc. (D) Assay of adult wing sizes that develop following ablation with rnts>rpr on day 9 in wild type and BRV-B3-wg discs. Mean differences between wild type and BRV-B3-wg data sets is not statistically significant (N.S.), calculated by two way ANOVA. Error bars are SEM of 3 biological repeats, n>100 flies for each genotype/condition.
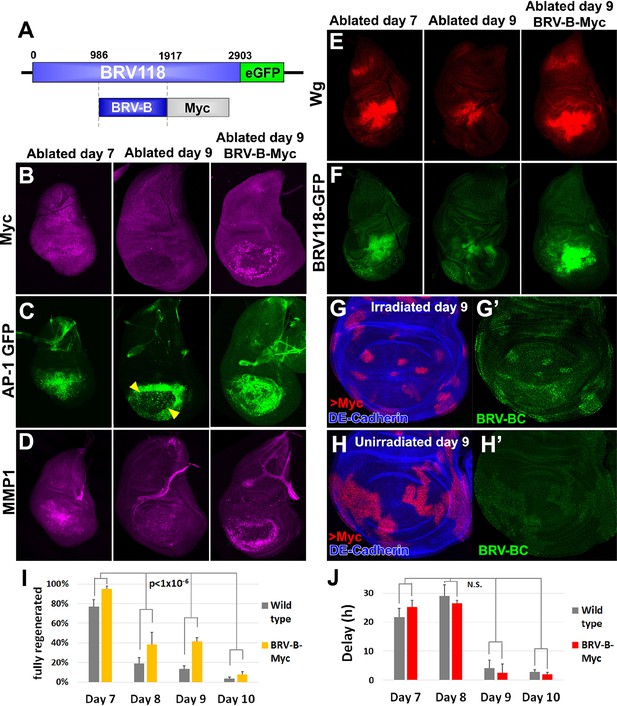
Circumventing the age-related repression of Myc improves regeneration.
(A) Schematic of the BRV-B-Myc transgene. Numbers indicate nucleotide positions. (B-D) Basal sections of discs following rnts>rpr ablation on day 7 (left panel) and day 9 (center panel), and on day 9 in discs bearing BRV-B-Myc transgene (right panel), imaged at the time of downshift. (B) Myc expression following ablation. Myc is expressed in basal wound periphery cells of a day 7 disc, but is absent in a day 9 ablated disc. The BRV-B-Myc transgene causes expression of Myc protein in an older disc. (C- D) Discs bearing the AP-1 GFP reporter (C) or stained with anti-Mmp1 (D), and imaged as in (B). AP-1 reporter expression is strongly expressed in basal cells of the pouch in day 7 ablated discs, coincident with Mmp1 expression. AP-1 reporter expression is found in the ring of wound periphery cells in day 9 ablated discs (arrowheads), while Mmp1 staining is reduced. Day 9 ablated discs bearing BRV-B-Myc have AP-1 GFP expressing cells covering the basal wound surface, resembling the expression pattern of day 7 discs, while Mmp1 expression is stronger than a wild type day 9 disc. (E-F) Discs stained for Wg (E) or bearing the BRV118-GFP reporter (F) following ablation with rnts>egr, and imaged as in (B-D). Declining expression of the reporter on day 9 is rescued in discs bearing the BRV-B-Myc transgene. Similarly, wg expression is stronger on a day 9 bearing the BRV-B-Myc transgene, comparable to expression level in an ablated day 7 disc. (G-G’) A day 9 disc bearing the BRV-BC enhancer with heat shock induced flip-out clones (marked by RFP, red) expressing Myc, irradiated with 45 Gy and dissected 16 hr later. Clones expressing Myc respond to damage by activating the BRV-BC enhancer (G’, green), while the enhancer remains inactive in neighboring damaged tissue not expressing Myc. (H-H’) Day 9 discs as in (G-G’), in the absence of irradiation. Expression of Myc in clones (red) causes low level of BRV-BC enhancer activity (H’, green), even in the absence of damage. (I) Assay of adult wing sizes that develop following ablation with rnts>rpr on day 7, 8, 9 and 10 in wild type and BRV-B-Myc discs, quantifying animals that eclose with fully regenerated wings. The BRV-B-Myc expressing animals consistently eclose with more of the full regenerated wings compared to wild type. Mean differences between wild type and BRV-B-Myc data sets is statistically significant, (p-value calculated by two way ANOVA). Error bars are SEM of at least 3 biological repeats, scoring a total of >200 animals in each condition. (J) Developmental timing of wild type and BRV-B-Myc expressing larvae following rnts>rpr ablation on day 7, 8, 9 and 10. Delay is measured as hours at which 50% of larvae have pupariated compared to unablated wild type. The BRV-B-Myc transgene (red bars) does not alter developmental timing following ablation compared to wild type (gray bars) on any day ablated. Mean differences between wild type and BRV-B-Myc data sets is not statistically significant (N.S.), calculated by two way ANOVA. Error bars are SEM of 3 biological repeats, n>100 flies for each genotype/condition.
Videos
The morphological response of a day 7 disc to ablation.
Three dimensional imaging using light sheet microscopy of discs ablated with rnts>rpr on day 7, with AP-1-GFP (green) and stained for DCP-1 (red), and DE-Cadherin (blue). A 360° rotation of the disc is shown, and the AP-1-GFP channel is shown separately.
The morphological response of a day 9 disc to ablation.
Three dimensional imaging of a rnts>rpr ablated day 9 disc, imaged as in Video 1.
Additional files
-
Supplementary file 1
Cloning and qPCR primers tables.
(A) Primers used for cloning transgenic reporters of the BRV118 enhancer and all derived constructs. (B) Primers used for qPCR analysis of ChIP DNA (Figure 7F and Figure 7—figure supplement 2C).
- https://doi.org/10.7554/eLife.11588.026





