Akt-mTORC1 signaling regulates Acly to integrate metabolic input to control of macrophage activation
Figures
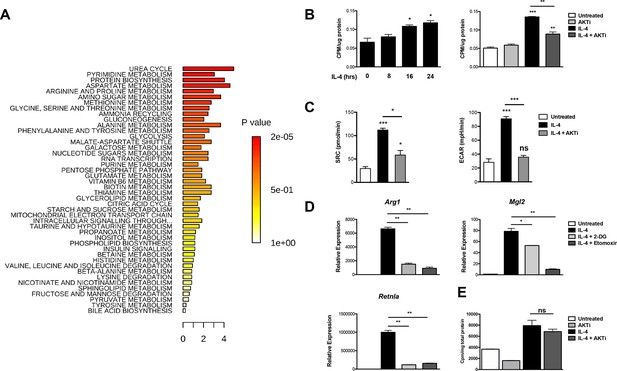
Akt regulates enhanced glucose utilization in M2 macrophages.
(A) Top metabolic pathways enriched in macrophages stimulated for 12 hr with IL-4 (relative to unstimulated macrophages) as identified by LC/MS-based metabolomics profiling. (B) M2 macrophages increase glucose uptake in an Akt-dependent manner. BMDMs were treated with IL-4 for the indicated time periods (left) or 16 hr +/- the Akt inhibitor MK2206 (Akti) (right), followed by analysis of uptake of 3H-deoxy-D-glucose. (C) Increased glucose utilization in M2 macrophages is associated with enhanced oxidative metabolism and glycolysis. BMDMs were treated with IL-4 for 20 hr +/- Akt inhibitor, followed by analysis of spare respiratory capacity (SRC) and aerobic glycolysis (ECAR) in extracellular flux analyses. (D) M2 gene induction is sensitive to the glycolysis inhibitor 2-deoxyglucose (2-DG). BMDMs were treated with IL-4 for 16 hr +/- 2-DG or the β-oxidation inhibitor etomoxir pretreatment, followed by analysis of M2 gene induction by qPCR. (E) Akt does not regulate β-oxidation in M2 macrophages. BMDMs stimulated for 36 hr with IL-4 +/- Akt inhibitor pretreatment were incubated for 3 hr with 3H-palmitate for analysis of β-oxidation. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
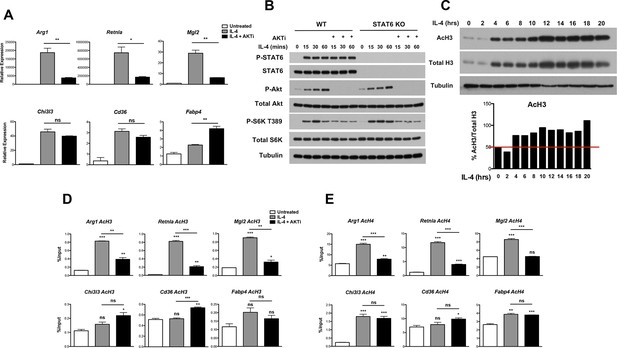
Akt regulates inducible histone acetylation at some M2 genes.
(A) Akt activity stimulates induction of a subset of M2 genes. BMDMs were stimulated with IL-4 for 16 hr +/- the Akt inhibitor MK2206 (Akti) pretreatment, followed by analysis of M2 gene induction by qPCR. (B) The Jak-Stat and Akt-mTORC1 pathways are activated independently downstream of the IL-4R. WT and Stat6 KO BMDMs were stimulated with IL-4 +/- Akt inhibitor as indicated. Analysis of Stat6, Akt, and mTORC1 activation was assessed by western blotting. (C) IL-4 induces a global increase in histone H3 acetylation. BMDMs were stimulated with IL-4 over the time course indicated, followed by analysis of histone H3 acetylation by western blotting. Bottom, quantitation of acetylated H3 over total H3. (D, E) Akt regulates inducible H3 (D) and H4 (E) acetylation at some M2 genes. BMDMs stimulated with IL-4 for 16 hr +/- Akt inhibitor pretreatment were subject to ChIP analysis using antibodies to acetylated H3 or acetylated H4. Enrichment of the indicated M2 gene promoters was assessed by qRT-PCR. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
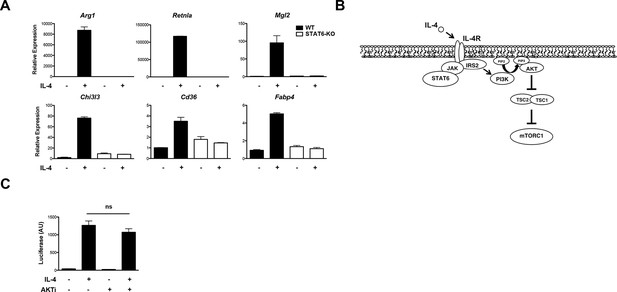
Stat6 and Akt-mTORC1 pathways are independent signaling branches downstream of the IL-4R.
(A) M2 gene induction is dependent on Stat6. WT and Stat6 KO BMDMs were stimulated with IL-4 followed by analysis of M2 gene induction by qPCR. (B) IL-4 signaling activates the Jak-Stat and Akt-mTORC1 pathways. Receptor ligation activates the latent activity of Jak1 and Jak3 kinases, leading to phosphorylation and activation of Stat6. Jak-mediated phosphorylation of the IL-4R also allows engagement of the adaptor protein IRS2. IRS2 recruits PI3K, which generates PIP3 from PIP2 at the plasma membrane, followed by recruitment, phosphorylation, and activation of Akt. Activated Akt phosphorylates and inactivates the TSC complex, a negative regulator of mTORC1, to activate mTORC1. (C) Stat6 transcriptional activity is not affected by block of Akt activity. BMDMs were transfected with a Stat6-dependent luciferase reporter, followed by 16 hr IL-4 stimulation +/- Akt inhibitor pretreatment. Stat6 activity was assessed by a luciferase assay. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
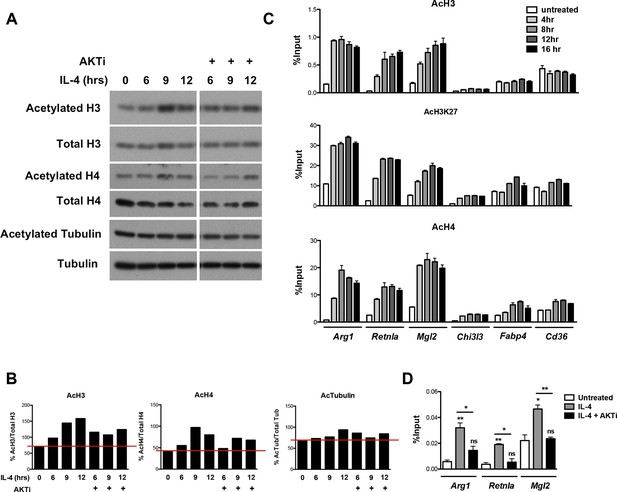
Akt regulates inducible histone acetylation at some M2 genes.
(A) IL-4 increases global histone acetylation in an Akt-dependent manner. BMDMs stimulated as indicated were analyzed by western blotting for H3 and H4 acetylation. (B) Quantitation of the western blotting data in A. (C) IL-4 induces an increase in H3, H3K27, and H4 acetylation at promoters of M2 genes. BMDMs were stimulated with IL-4 over the time course indicated, followed by ChIP analysis to examine H3, H3K27, and H4 acetylation at M2 gene promoters. (D) Akt regulates Pol II recruitment at some M2 genes. BMDMs stimulated for 16 hr with IL-4 +/- Akt inhibitor pretreatment were subject to ChIP analysis using antibodies to Pol II. Enrichment of the indicated M2 gene promoters was assessed by qRT-PCR. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
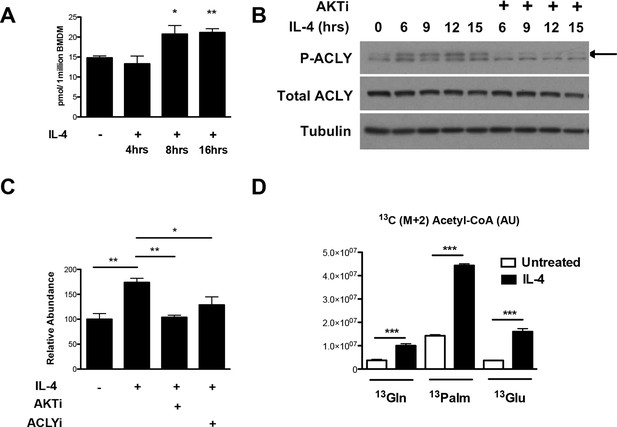
The Akt-Acly axis regulates inducible Ac-CoA production in M2 macrophages.
(A) IL-4 treatment increases Ac-CoA production. BMDMs were stimulated for the indicated time periods with IL-4, followed by analysis of Ac-CoA levels by LC-MS. (B) Akt regulates IL-4-inducible Acly phosphorylation. BMDMs were stimulated as indicated, followed by analysis of Acly phosphorylation by western blotting. Arrow indicates phospho-Acly. (C) Akt and Acly regulate IL-4-inducible production of Ac-CoA. BMDMs stimulated for 16 hr with IL-4 +/- inhibitor pretreatment were analyzed for levels of Ac-CoA by LC-MS. (D) BMDMs were stimulated or not for 12 hr with IL-4, followed by a 2 hr incubation with 13C6-glucose, 13C16-palmitate, or 13C5-glutamine. Carbon tracing into Ac-CoA was assessed by LC-MS. Data shows arbitrary units of labeled 13C (M+2) in the different conditions. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
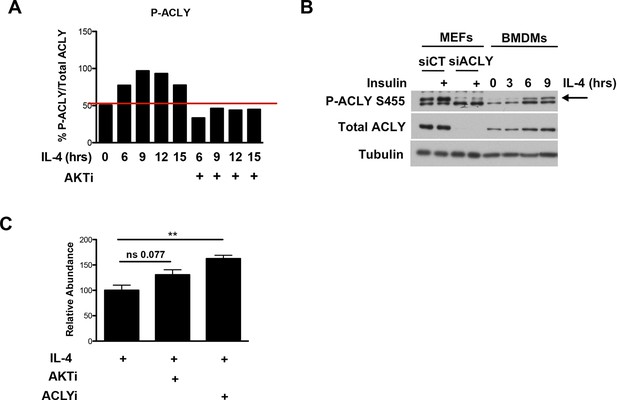
Akt regulates Acly to control inducible Ac-CoA production in M2 macrophages.
(A) Quantitation of the Acly phosphorylation from Figure 3B. (B) Knockdown of Acly in MEFs. Lysates from MEFs transfected with control siRNA (siCT) or siRNA to Acly (siAcly) were run next to BMDM lysates to unequivocally identify the bands corresponding to p-Acly and total Acly. (C) Citrate accumulates upon block of Akt or Acly activity. Citrate levels in BMDMs from the steady state metabolomics experiment in Figure 1A. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
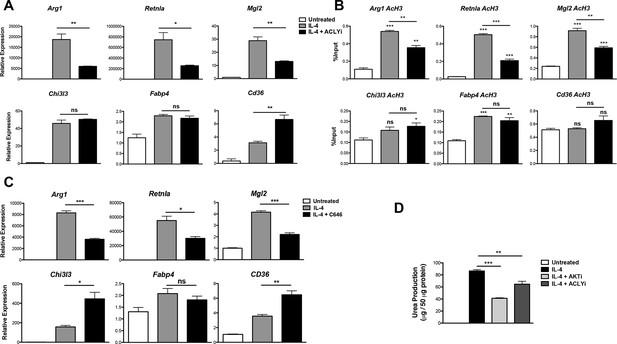
Acly controls inducible histone acetylation at some M2 genes.
(A) Acly regulates induction of some M2 genes. BMDMs stimulated for 16 hr with IL-4 +/- Acly inhibitor pretreatment were analyzed for M2 gene induction by qRT-PCR. (B) Acly regulates inducible H3 acetylation at some M2 genes. BMDMs stimulated for 16 hr with IL-4 +/- Acly inhibitor pretreatment were subject to ChIP analysis using antibodies to acetylated H3. Enrichment of the indicated M2 gene promoters was assessed by qRT-PCR. (C) The p300 inhibitor C646 reduces induction of some M2 genes. BMDMs stimulated for 16 hr with IL-4 +/- C646 pretreatment were analyzed for M2 gene induction by qRT-PCR. (D) Akt and Acly control IL-4-inducible arginase activity. BMDMs were stimulated for IL-4 for 24 hr +/- inhibitor pretreatment, followed by analysis of arginase activity in cellular lysates as assessed by urea production. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
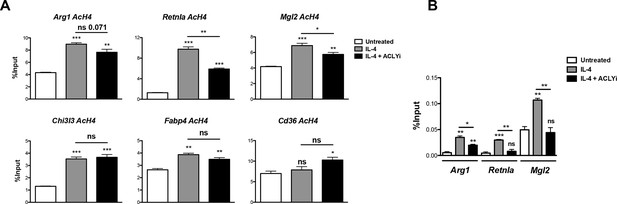
Acly controls inducible histone acetylation at some M2 genes.
(A) Acly regulates inducible H4 acetylation at some M2 genes. BMDMs stimulated for 16 hr with IL-4 +/- Acly inhibitor pretreatment were subject to ChIP analysis using antibodies to acetylated H4. Enrichment of the indicated M2 gene promoters was assessed by qRT-PCR. (B) Acly regulates Pol II recruitment at some M2 genes. BMDMs stimulated for 16 hr with IL-4 +/- Acly inhibitor pretreatment were subject to ChIP analysis using antibodies to Pol II. Enrichment of the indicated M2 gene promoters was assessed by qRT-PCR. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
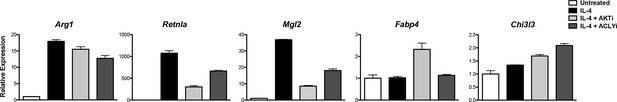
Akt-Acly signaling regulates M2 activation in peritoneal macrophages.
Elicited peritoneal macrophages were treated for 16 hr with IL-4 +/- the indicated inhibitors, followed by analysis of M2 gene induction by qRT-PCR.
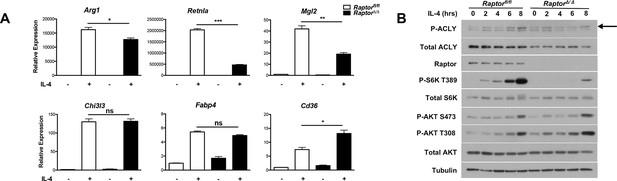
mTORC1 controls Acly protein levels to regulate M2 activation.
(A) mTORC1 regulates M2 activation. M2 gene expression in Raptorfl/fl and Raptor△/△ BMDMs stimulated with IL-4 for 16 hr as assessed by qRT-PCR. (B) mTORC1 regulates Acly protein levels. Acly protein expression in Raptorfl/fl and Raptor△/△ BMDMs stimulated as indicated.
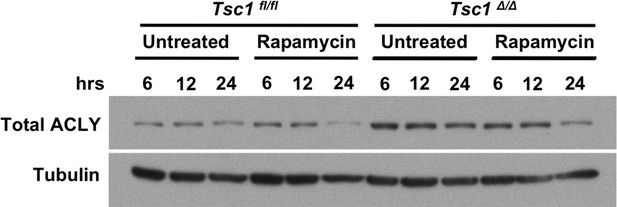
mTORC1 activity regulates levels of Acly.
Tsc1fl/fl and Tsc1△/△ BMDMs were treated with rapamycin or not for the indicated time periods, followed by analysis of levels of Acly by western blotting.
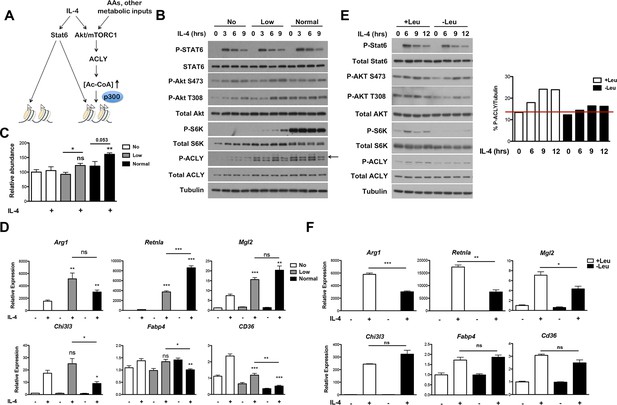
The Akt-mTORC1-Acly axis links metabolic input to control of M2 activation.
(A) Proposed model for how Akt-mTORC1-Acly signaling exerts gene-specific control of M2 activation. Akt-TORC1-Acly signaling integrates metabolic input to control levels of Ac-CoA production, which modulates histone acetylation and gene induction at some M2 genes by HATs such as p300. (B) Amino acid levels modulate the activity of the Akt-mTORC1-Acly axis. BMDMs cultured in media containing varying levels of amino acids (normal, low, or no) were stimulated with IL-4 for the indicated time periods, followed by analysis of Akt, mTORC1, and Acly activity by western blotting. (C) Amino acid levels modulate Ac-CoA production. BMDMs stimulated as in B. were harvested for LC-MS analysis of Ac-CoA levels after 12 hr IL-4 stimulation. (D) Amino acid levels modulate induction of some M2 genes. BMDMs stimulated as in B. were harvested for qRT-PCR analysis of M2 gene induction after 9 hr IL-4 stimulation. (E) Leucine deficiency attenuates the activity of the Akt-mTORC1-Acly axis. BMDMs cultured in leucine-replete or leucine-deficient media were stimulated with IL-4 for the indicated time periods, followed by analysis of Akt, mTORC1, and Acly activity by western blotting. Right, quantitation of Acly phosphorylation. (F) Leucine deficiency reduces induction of some M2 genes. BMDMs stimulated as in E. were harvested for qRT-PCR analysis of M2 gene induction after 16 hr IL-4 stimulation. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
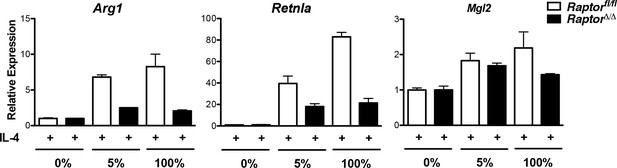
Amino acid levels modulate M2 gene expression in part through Raptor.
Raptorfl/fl and Raptor△/△ BMDMs were stimulated as in the experiment shown in Figure 6D, followed by analysis of M2 gene expression by qRT-PCR. IL-4-stimulated gene expression in the absence of amino acids is arbitrary set at 1.
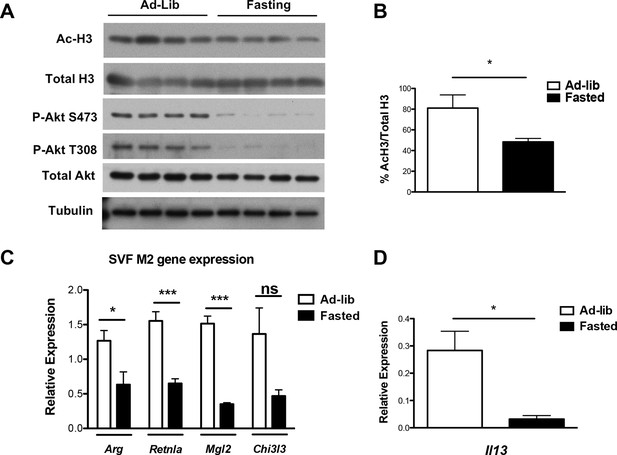
Feeding and fasting regulate M2 polarization of adipose tissue macrophages.
Analysis of ATM M2 activation in fed and fasted mice. Mice (n=4/group) were allowed to feed ad-lib or fasted O/N prior to their sacrifice. The ATM-containing stromal vascular fraction of perigonadal white adipose tissue was obtained for analysis of Akt activity by western blotting (A); H3 acetylation by western blotting (A, B); expression of M2 genes by qRT-PCR (C); and expression of IL-13 by qRT-PCR (D). The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
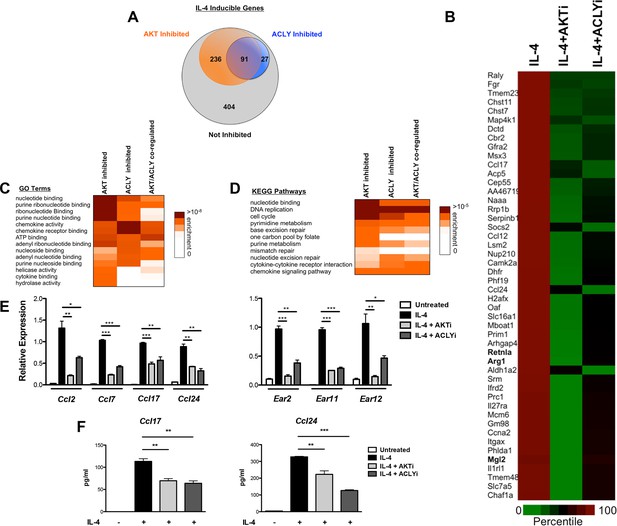
The Akt-Acly axis controls functional subsets of the M2 program.
(A) Venn diagram depicting the number of IL-4-inducible genes regulated by Akt and/or Acly signaling. (B) Heatmap of normalized rank ordered Log2 RPKM values of top 50 IL-4 response genes co-regulated by Akt and Acly. (C) Heatmap of enriched KEGG pathways within the cohort of IL-4-inducible genes. (D) Heatmap of enriched Gene Ontology terms within the cohort of IL-4-inducible genes. (E) qPCR analysis validates regulation of chemokine and Ear genes by Akt-Acly signaling. BMDMs were stimulated with IL-4 for 16 hr +/- Akt or Acly inhibitor. (F) ELISA analysis indicates that Akt-Acly signaling regulates production of CCL17 and CCL24. BMDMs were stimulated with IL-4 for 36 hr +/- Akt or Acly inhibitor. The student’s t-test was used to determine statistical significance, defined as *P<0.05, **P<0.01, and ***P<0.001.
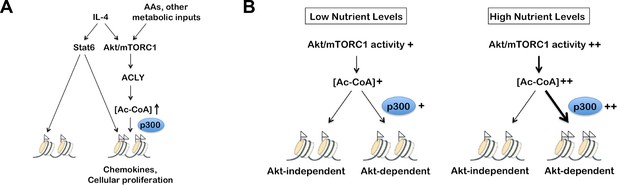
Working models.
(A) The Akt-mTORC1-Acly pathway couples metabolic input to control of particular subsets of the M2 program, including chemokines and cellular proliferation. (B) p300 links metabolic status and Akt-mTORC1 activity in the form of Ac-CoA levels to M2 gene induction. p300 has a high Km and is responsive to changes in the levels of its substrate, allowing it to couple increased Ac-CoA levels to enhanced histone acetylation at some Akt-dependent M2 genes. In contrast, HATs at Akt-independent M2 genes (not shown) have low Km and are relatively insensitive to metabolic status and changing Ac-CoA levels.
Additional files
-
Supplementary file 1
LC-MS peak areas and P-values for experiment described in Figure 1A (ranked by -P-values of pairwise comparison).
- https://doi.org/10.7554/eLife.11612.019
-
Supplementary file 2
Sequences of ChIP primers, as well as their positions relative to the transcriptional start site (TSS).
- https://doi.org/10.7554/eLife.11612.020





