Loss of the transcription factor Meis1 prevents sympathetic neurons target-field innervation and increases susceptibility to sudden cardiac death
Figures
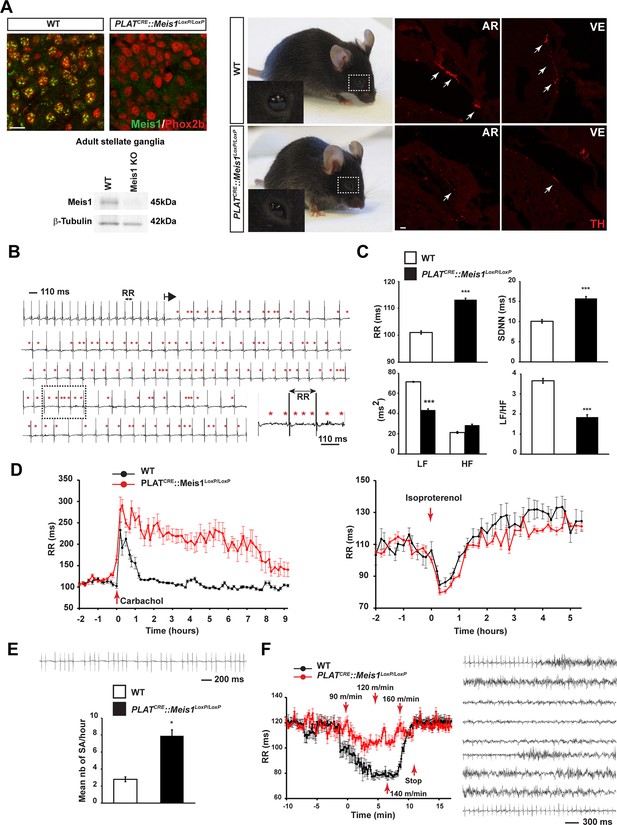
Specific Meis1 inactivation in the PNS results in sympatho-vagal deregulation of cardiac functions.
(A) Immunochemistry for Meis1 and the transcription factor Phox2b on sections of SCGs from E16.5 WT and PLATCRE::Meis1LoxP/LoxP mice, and western blot analysis for Meis1 from adult stellate ganglia. Surviving adult mutants exhibited blepharoptosis (white dotted square). Immunostaining for TH+ sympathetic fibers in the heart of newborn WT and PLATCRE::Meis1LoxP/LoxP mice. VE = Ventricle, AR = Atrium. White arrows point at TH+ sympathetic fibers. Scale bar = 20 µm. (B) Example of an ECG trace recorded in an adult male PLATCRE::Meis1LoxP/LoxP mouse. The black arrow indicates the start of a junctional bradycardia and the red asterisks mark abnormal P waves locations. Dotted black square delineates the power magnification of the ECG trace showed. (C) HRV analysis of the ECGs recordings of PLATCRE::Meis1LoxP/LoxP and WT mice. Graphical representations of the mean RR interval, the standard deviation of all normal to normal beats (SDNN), the power of low frequency band (LF), the power of the high frequency band (HF) and the LF/HF ratio. (D) Time course of the mean RR interval during isoproterenol or carbamoylcholine chloride challenges in WT and PLATCRE::Meis1LoxP/LoxP mice. (E) Typical sinus arrest (SA) trace obtained in a PLATCRE::Meis1LoxP/LoxP mouse and quantification of the mean number of sinus arrest per hour over a 12 hr ECG recording in PLATCRE::Meis1LoxP/LoxP vs WT mice. (F) Mean RR interval after treadmill exercise. Typical ECG trace during the recuperation period following treadmill exercise. Data are represented as mean +/- s.e.m. n = 8 in each group; *p<0.05, ***p<0.001. See also Figure 1—figure supplement 1 and 2 , Figure 1—source data 1.
-
Figure 1—source data 1
Morphologic and left ventricular function parameters assessed by Doppler echocardiography in WT and PLATCRE::Meis1LoxP/LoxP mice.
HR, heart rate; AWTd, anterior wall thickness during diastole; AWTs, anterior wall thickness during systole; LVIDd, left ventricular internal diameter during diastole; LVIDs, left ventricular internal diameter during systole; PWTd, posterior wall thickness during diastole; PWTs, posterior wall thickness during systole; EF, ejection fraction; FS, fractional shortening; FAC, fractional area change; AoVTI, aortic velocity time integral; E, mitral inflow early filling velocity; A, mitral inflow late filling velocity. Data are represented as mean ± SD. Abbreviations: HR: Heart Rate; AWTd: Anterior Wall Thickness during diastole; AWTs: Anterior Wall Thickness during systole; LVIDd: Left Ventricular Internal Diameter during diastole; LVIDs: Left Ventricular Internal Diameter during systole; PWTd: Posterior Wall Thickness during diastole; PWTs: Posterior Wall Thickness during systole; EF (%) M-mode: Ejection Fraction; FS (%) M-mode: Fractional Shortening; Ao VTI: Aortic flow Velocity Time Integral; E wave: Mitral inflow early filling velocity (passive inflow); A wave: Mitral inflow late filling velocity (active inflow); E/A: Ratio E wave / A wave FAC (%): Fractional area; EF (%) B-mode: Ejection Fraction measured on B-mode.
- https://doi.org/10.7554/eLife.11627.004
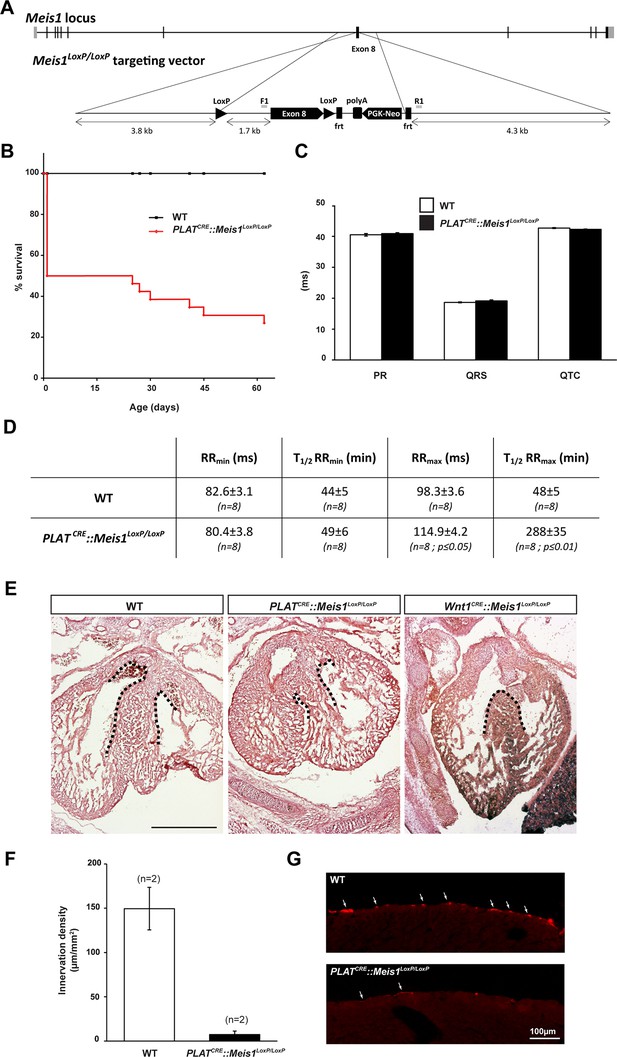
Sympatho-vagal deregulation of cardiac functions following Meis1 inactivation in the PNS.
(A) Targeting vector used for the generation of the Meis1 conditional knockout mouse strain (Meis1LoxP/LoxP). The exon 8 of the gene encoding the beginning of the homeodomain has been flanked with LoxP sites. (B) Kaplan-Meier survival curve showing the premature death of PLATCRE::Meis1LoxP/LoxP mice (in red) compared to WT mice (in black). (C) Measure of the PR, QRS and QTc intervals on a 24 hr ECG telemetric recording showed no statistically difference between groups, indicating that Meis1 invalidation in the PNS does not affect the intrinsic cardiac conduction properties. (D) Table recapitulating the minimum RR (RRmin), corresponding to the maximal effect of isoproterenol on heart rate and the time to lose half of the effect (T1/2RRmin), the maximum RR interval (RRmax), corresponding to the maximal effect of carbamoylcholine chloride and the duration to lose half of the effect (T1/2RRmax) in WT and PLATCRE::Meis1LoxP/LoxP mice. Data are represented as mean +/- s.e.m., n = 8 in all experiments. (E) Histologic section of E14.5 WT, PLATCRE::Meis1LoxP/LoxP and Wnt1CRE::Meis1LoxP/LoxP hearts embryos. Whereas the cardiac septum was identical to that of WT in 3/3 analyzed PLATCRE::Meis1LoxP/LoxP embryos, we observed a defect in the cardiac septum of 1/1 Wnt1CRE::Meis1LoxP/LoxP embryo. Scale bar = 500 µm. (F) Quantification of the density of TH+ sympathetic fibers on sections of the heart in PLATCRE::Meis1LoxP/LoxP and WT adult mice. (G) Representative images showing that the remaining fibers in PLATCRE::Meis1LoxP/LoxP are thinner than in WT mice. Data are represented as mean +/- s.e.m. n = 2. Scale bars = 100 µm.
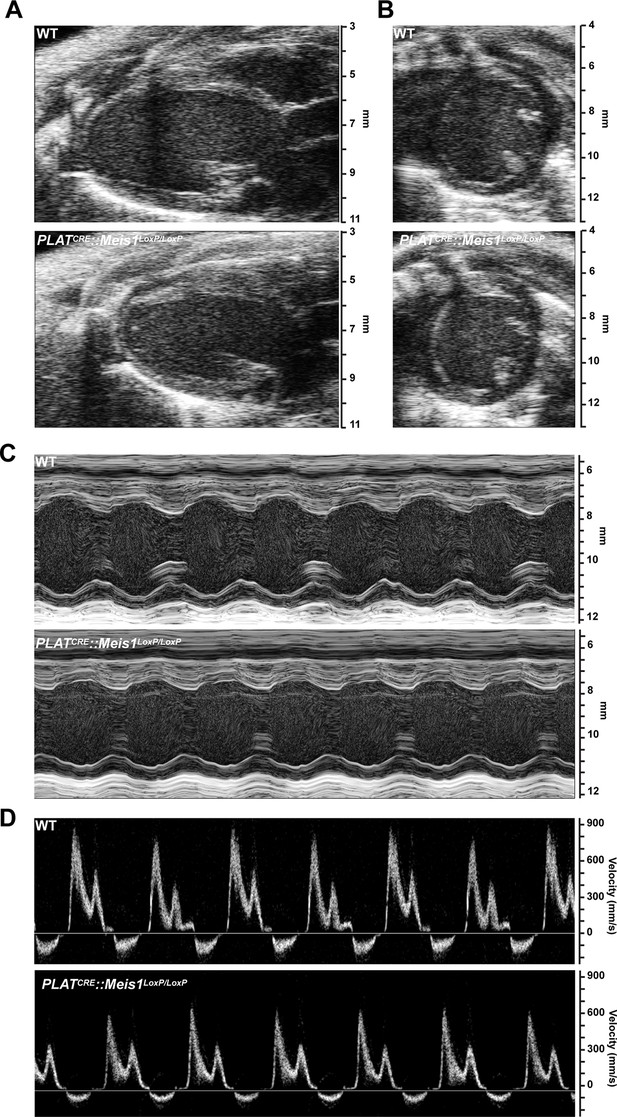
Cardiac echography analysis revealed no difference following Meis1 inactivation.
(A) Representative B-mode images of heart parasternal long axis view. (B) Representative B-mode images of heart short axis view. (C) Representative M-mode images of heart short axis view at the level of papillary muscle. (D) Representative mitral inflow velocity from the apical four view chamber. Evaluations of left ventricular morphology and function by Doppler echocardiography showed no difference between WT and PLATCRE::Meis1LoxP/LoxP mice.
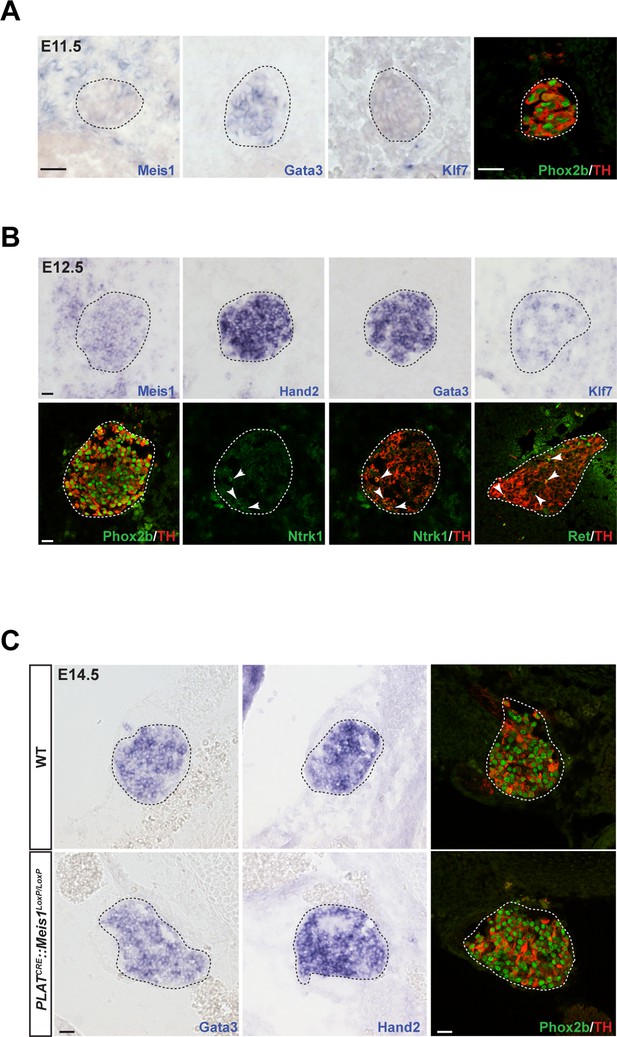
Meis1 expression in sympathetic neurons is incompatible with early sympathetic specification but coincides with target-field innervation onset.
(A) ISH for Meis1, Gata3 and Klf7, and immunochemistry for Phox2b and TH in WT E11.5 embryonic SCGs. (B) ISH for Meis1, Hand2, Gata3 and Klf7 and immunochemistry for Phox2b, TH, Ntrk1 and Ret in WT E12.5 embryonic SCGs. White arrows point at Ntrk1 or Ret neurons. (C) ISH for Gata3 and Hand2 and immunochemistry for Phox2b and TH on WT and PLATCRE::Meis1LoxP/LoxP E14.5 embryonic SCGs. White and black dotted lines delineate the SCGs. Scale bar = 20 µm. See also Figure 2—figure supplement 1.
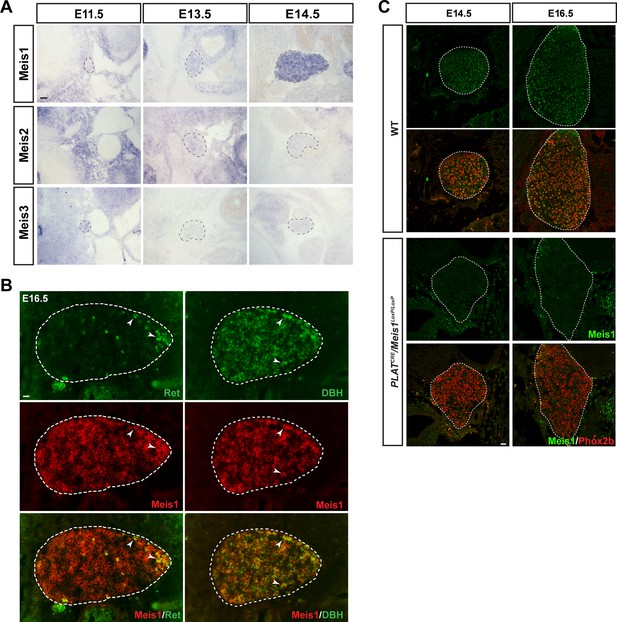
Meis1 is the only member of the Meis family to be expressed by embryonic sympathetic neurons and is indifferently expressed by noradrenergic and cholinergic sympathetic neurons.
(A) In situ hybridization (ISH) for Meis1, Meis2 and Meis3 on E11.5, E13.5 and E14.5 WT embryos showed that Meis1 is the only member of the family to be expressed in sympathetic ganglia. Black dashed lines encircles the SCGs. (B) Double ISH for Meis1 (pseudo color in red) with Dopamine β-Hydroxylase (DBH, pseudo color in green) or with Ret (pseudo color in red) indicated that both major subpopulations (noradrenergic and presumptive cholinergic) of sympathetic neurons expressed Meis1 at E16.5. White arrowheads point at double positive neurons. White dotted lines encircle the SCGs. (C) Immunochemistry for Phox2b and Meis1 on SCGs from E14.5 and E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos confirmed the loss of Meis1 protein at these stages. Scale bars = 20 µm.
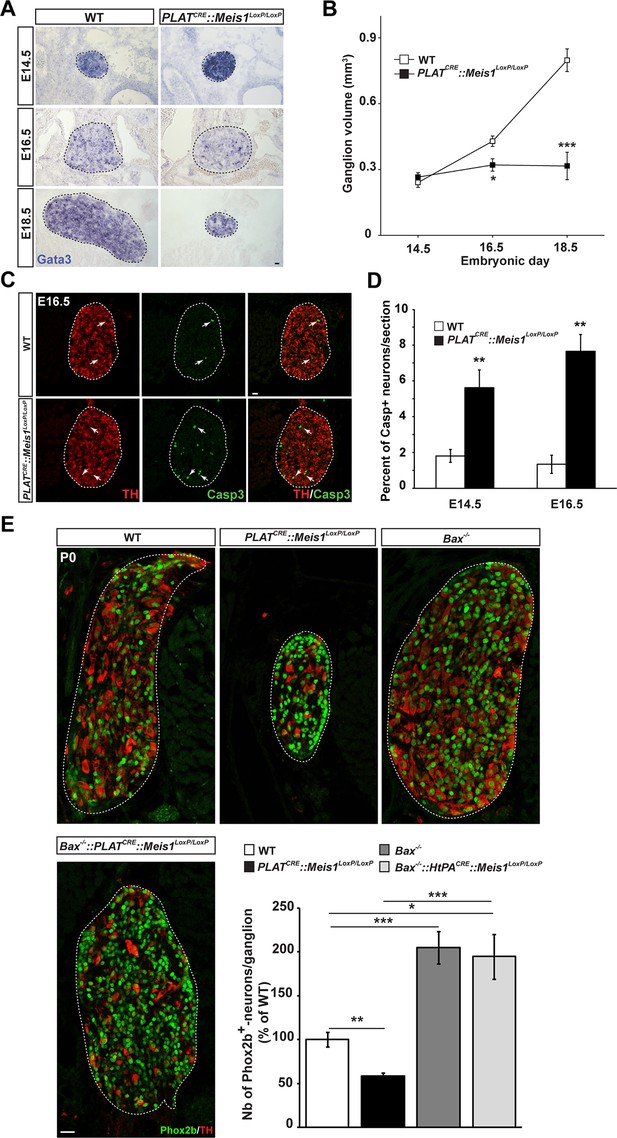
Exacerbated neuronal apoptosis in Meis1 mutant sympathetic neurons.
(A) ISH for Gata3 on SCGs of PLATCRE::Meis1LoxP/LoxP and WT embryos at E14.5, 16.5 and E18.5. (B) Quantification of the volume of the SCGs in WT vs PLATCRE::Meis1LoxP/LoxP mice at E14.5, E16.5 and E18.5. (C) Representative images of the SCGs visualized by immunochemistry for activated Caspase-3 (Casp3) and TH at E16.5 in WT and PLATCRE::Meis1LoxP/LoxP mice. Arrows point at Casp3+/TH+ neurons. (D) Quantification of the average number of Casp3+/TH+ neurons per section in the SCGs of PLATCRE::Meis1LoxP/LoxP and WT embryos at E14.5 and E16.5. (E) Immunochemistry for Phox2b and TH on SCGs of P0 WT, Bax-/-, PLATCRE::Meis1LoxP/LoxP and PLATCRE::Meis1LoxP/LoxP::Bax-/- newborn pups, and quantification of the number of Phox2b+ neurons per SCG in P0 WT, Bax-/-, PLATCRE::Meis1LoxP/LoxP and Bax-/-::PLATCRE::Meis1LoxP/LoxP mice. Data are represented as mean +/- s.e.m. n = 3; *p≤0.05; **p≤0.01; ***p≤0.005. Scale bar = 20 µm. See also Figure 3—figure supplement 1 and 2.
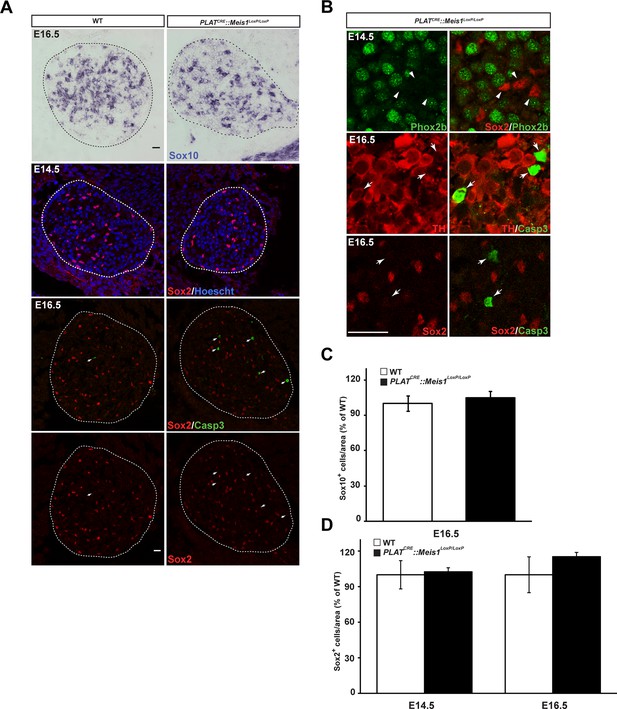
Survival of glial progenitor cells is not affected following Meis1 inactivation.
(A) ISH for Sox10 and immunochemistry for Sox2 and Casp3 on E14.5 and E16.5 SCGs from WT and PLATCRE::Meis1LoxP/LoxP embryos. White arrows point at Casp3+ cells. (B) Immunochemistry for Sox2 and Phox2b on E14.5 SCGs showed that their expressions are mutually exclusive. Immunochemistry for Sox2 or TH with Casp3 showed that Casp3 immunoreactivity was present in TH+ neurons but never in Sox2+ glial progenitor cells. (C) Quantification of the number of Sox10+ cells in E16.5 WT and PLATCRE::Meis1LoxP/LoxP SCGs. (D) Quantification of the number of Sox2+ cells in E14.5 and E16.5 WT and PLATCRE::Meis1LoxP/LoxP SCGs. Data are represented as mean +/- s.e.m. n = 3; Scale bars = 20 µm in A and B.
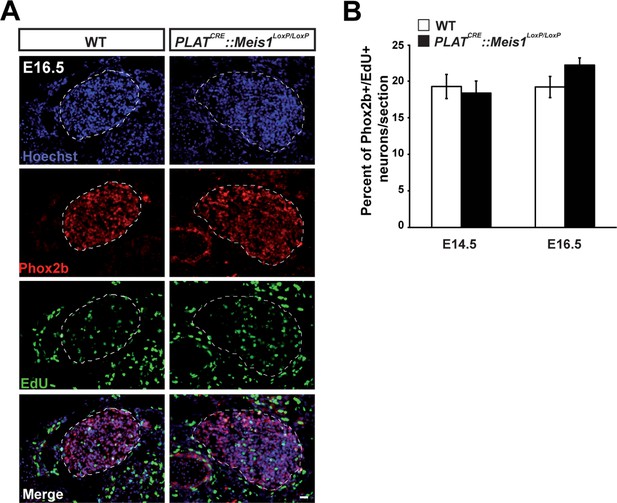
Meis1 inactivation does not affect early proliferation of sympathetic neurons.
(A) Pregnant mice were injected with 0.7 mg 5-ethynyl-2´-deoxyuridine (EdU) at 14 and 16 days post-coitum. 3 hr following injection, pregnant mice were sacrificed and embryos were dissected, fixed in PFA and crysosectionned. Chemical revelation of EdU incorporation combined with immunochemistry for Phox2b on E16.5 SCGs from WT and PLATCRE::Meis1LoxP/LoxP embryos showed no difference between the two genotypes. (B) Quantification of the number of Phox2b+ neurons that incorporated EdU per section in SCGs from E14.5 and E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos showed that about 18 to 22% of sympathetic neurons divided within the two last hours. No significant difference was observed between WT and PLATCRE::Meis1LoxP/LoxP embryos. Data are represented as mean +/- s.e.m. n = 3. Scale bar = 20 µm.
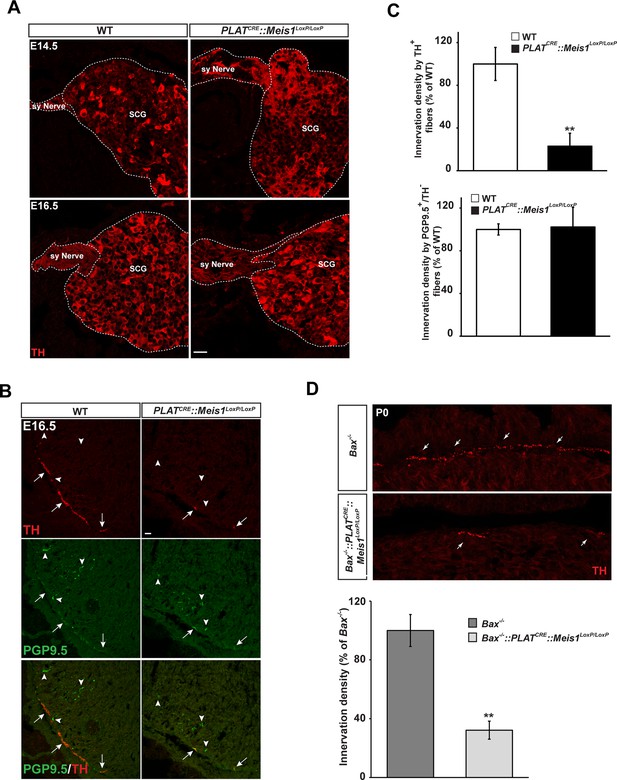
Lack of terminal sympathetic target-field innervation in Meis1 mutant embryos.
(A) Immunochemistry for TH on SCGs from E14.5 and E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos showing that proximal axonal growth of sympathetic neurons is not affected. Sy Nerve = sympathetic nerve. (B) Visualization of TH+ sympathetic fibers and others PGP9.5+/TH- peripheral neurons projections in the heart of PLATCRE::Meis1LoxP/LoxP and WT embryos at E16.5. Arrows point at TH+ sympathetic fibers and arrowheads point at PGP9.5+/TH- nerves. (C) Quantification of the density of TH+/PGP9.5+ sympathetic and TH-/PGP9.5+ sensory fibers on heart sections of PLATCRE::Meis1LoxP/LoxP and WT embryos at E16.5. (D) Representative images and quantification of TH+ sympathetic fibers in the heart of Bax-/- and PLATCRE::Meis1LoxP/LoxP::Bax-/- newborn pups. Data are represented as mean +/- s.e.m. n = 3; **p≤0.01. Scale bar = 20 µm. See also Figure 4—figure supplement 1.
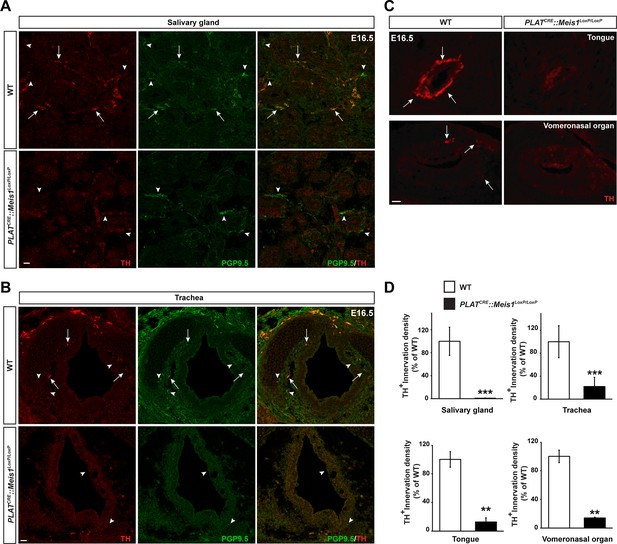
Sympathetic target-field innervation of peripheral organs is compromised following Meis1 loss of function.
(A and B) Visualization of TH+ sympathetic fibers (in red) and others PGP9.5+/TH- peripheral nervous projections (in green) by immunochemistry in the salivary glands (A) and the trachea (B) of PLATCRE::Meis1LoxP/LoxP and WT embryos at E16.5. Only the TH+ sympathetic innervation is affected in the mutant. Arrows point at TH+ sympathetic fibers and arrowheads point at PGP9.5+/TH- nerves. (C) Visualization by immunochemistry of TH+ sympathetic fibers (in red) in the tongue and the vomeronasal organ of PLATCRE::Meis1LoxP/LoxP and WT embryos at E16.5. Arrows point at TH+ sympathetic fibers. (D) Quantification of the density of TH+ sympathetic fibers on sections of the salivary glands, the vomeronasal organ, the tongue and the trachea in PLATCRE::Meis1LoxP/LoxP and WT embryos at E16.5 revealed that SCG neurons failed to establish contact with their peripheral targets during embryogenesis. Data are represented as mean +/- s.e.m. n = 3; ** p≤0.01; *** p≤0.005. Scale bars = 20 µm.
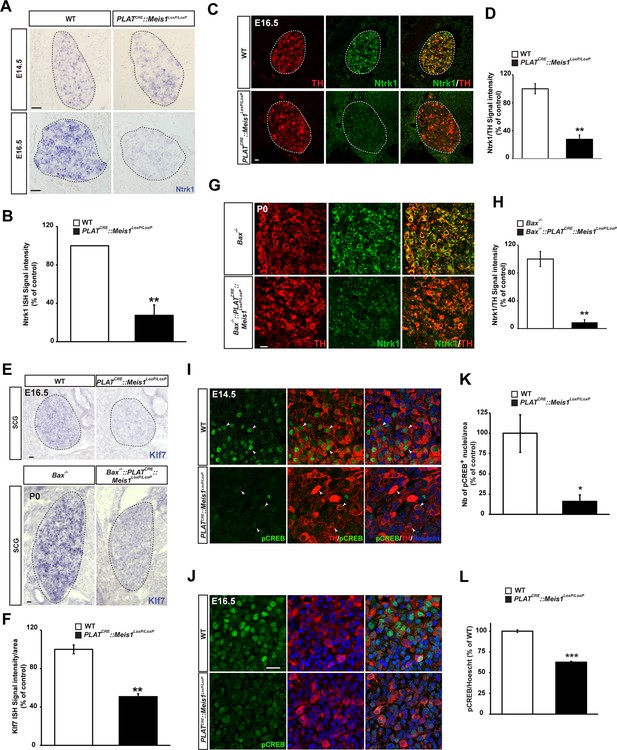
Target-field innervation signaling pathways in Meis1-inactivated sympathetic neurons.
(A) ISH for the neurotrophins high affinity receptor Ntrk1 in E14.5 and E16.5 WT and PLATCRE::Meis1LoxP/LoxP SCGs. (B) Quantification of Ntrk1 ISH signal intensity in E16.5 WT and PLATCRE::Meis1LoxP/LoxP SCGs. (C) Immunochemistry for TH and Ntrk1 on E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. White dotted lines encircle the SCGs. (D) Quantification of the intensity of immuno-fluorescence for Ntrk1 in the SCGs of E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. (E) Representative images of ISH for Klf7 on SCGs of E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos and on SCGs of P0 Bax-/- and PLATCRE::Bax-/-::Meis1LoxP/LoxP mice. (F) Quantification of Klf7 ISH signal intensity in SCGs of E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. (G) Immunochemistry for TH and Ntrk1 on P0 Bax-/- and PLATCRE::Meis1LoxP/LoxP::Bax-/- mice. (H) Quantification of the intensity of Ntrk1 immuno-fluorescence in the SCGs of P0 Bax-/- and PLATCRE::Meis1LoxP/LoxP::Bax-/- mice. (I) Immunochemistry for pCREB and TH on E14.5 SCGs from WT and PLATCRE::Meis1LoxP/LoxP embryos and quantification of the number of pCREB+ nuclei. (J) Immunochemistry for pCREB and TH on E16.5 SCGs from WT and PLATCRE::Meis1LoxP/LoxP embryos and quantification of pCREB signal intensity in neuronal nuclei. Data are represented as mean +/- s.e.m. n = 3; *p≤0.05; **p≤0.01. Scale bar = 20 µm. See also Figure 5—figure supplement 1 .
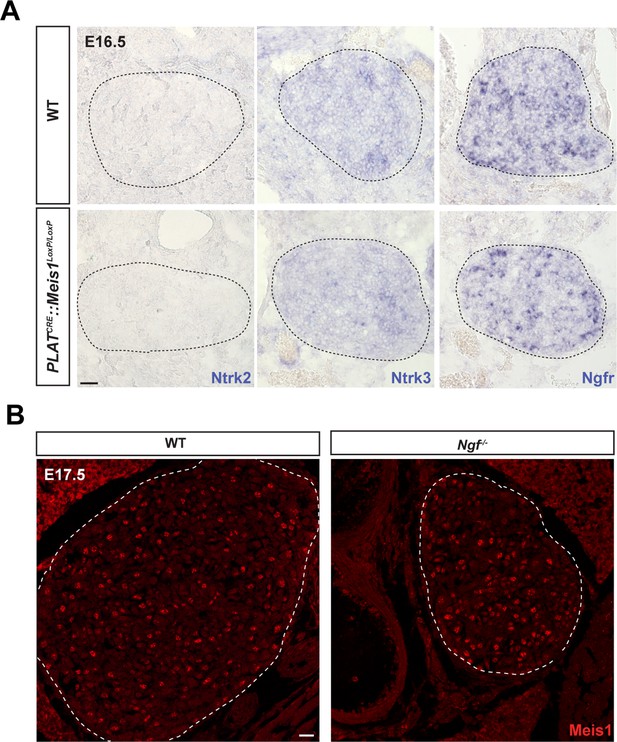
Meis1 loss of function does not interfere with the expression of other neurotrophins receptors than Ntrk1 and its expression is not dependent on Ngf/Ntrk1 signaling.
ISH for Ntrk2 and Ntrk3, the high-affinity receptors for Ntf2 and Ntf3 respectively and for Ngfr, the low affinity receptor for all members of the neurotrophins family on WT and PLATCRE::Meis1LoxP/LoxP SCGs in E16.5. Whereas Ntrk3 and Ngfr mRNAs are expressed by sympathetic neurons at this stage, Ntrk2 is not detected. No variation in the expression of Ntrk3 or Ngfr mRNAs could be detected in PLATCRE::Meis1LoxP/LoxP vs WT embryos. Dashed black lines delineate the SCGs. (B) Immunochemistry for Meis1 on SCGs from E17.5 WT and Ngf-/- embryos showed that Meis1 expression does not depend on Ngf/Ntrk1 signaling. Scale bar = 20 µm.
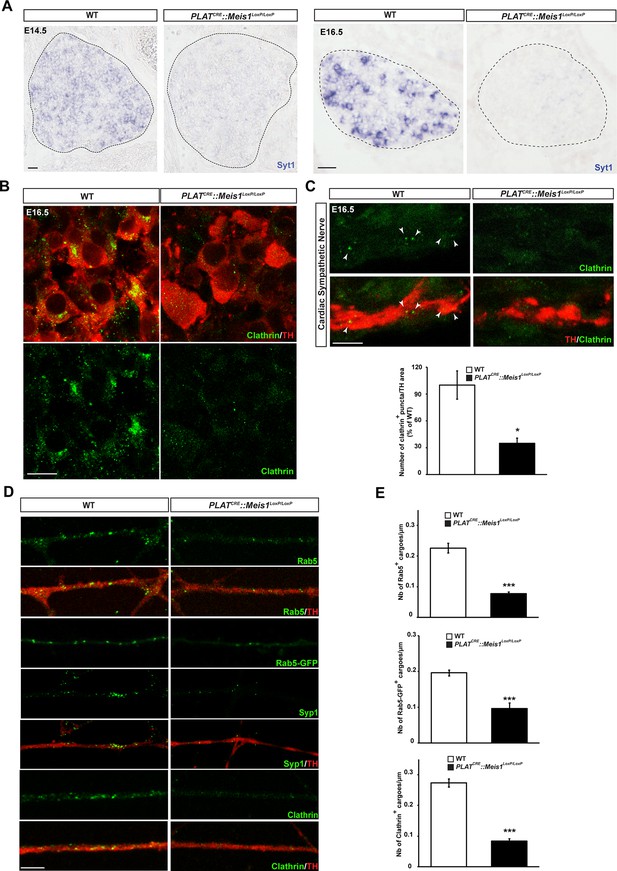
Meis1 target genes encode proteins necessary for early endosomes formation.
(A) ISH for Syt1 on SCGs of E14.5 and E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. (B) Immunochemistry for clathrin and TH on SCGs of E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. (C) Representative images and quantification of immunochemistry for clathrin and TH on the sympathetic nerves within the heart of E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. White arrowheads point at clathrin-coated pits and cargoes. (D) Immunochemistry for clathrin, TH, Syp1 and Rab5 and overexpression of a Rab5-GFP construct in cultured explants of SCGs from WT and PLATCRE::Meis1LoxP/LoxP embryos. (E) Quantification of the numbers of Rab5, Rab5-GFP and clathrin+ cargoes in cultured explants of SCGs. Dotted lines encircle the SCGs. Scale bar = 20 µm in A, 10 µm in B and 5 µm in C and D. Data are represented as mean +/- s.e.m; n = 3; *p≤0.05; *** p≤0.005. See also Figure 6—figure supplement 1 and 2, and Figure 6—source data 1–3 .
-
Figure 6—source data 1
Meis1 target genes in embryonic sympathetic neurons.
To identify Meis1 target genes, a ChIP-seq assay was conducted on E16.5 WT SCGs. The table includes the precipitated sequence ID (column A), the chromosome where the precipitated sequences match (column B), the start (column C) and the end (column D) of the precipitated sequences, the strand that the precipitated sequences recognize (column E), the name of the gene located within the 20kb of the precipitated sequence (column F), the gene accession number (column G), the strand where the 5’–3’ genes is located (column H) and the fold enrichment vs control experiment (column I).
- https://doi.org/10.7554/eLife.11627.017
-
Figure 6—source data 2
Bibliographical-based classification of Meis1 potential target genes.
The table indicates the precipitated sequence ID (column A) and the chromosome where the precipitated sequences match (column B). Bibliographical search for each identified Mei1 potential target genes revealed a strong enrichment in genes involved in synapse formation, function and architecture (column C), in target-field innervation (column D) and/or cardiac functions and/or pathologies (column E).
- https://doi.org/10.7554/eLife.11627.018
-
Figure 6—source data 3
Bibliographical classification of Meis1 potential target genes according to their function in endocytosis, exocytosis and vesicles transport.
The table indicates the genes names with one major reference corresponding to their implication in synaptic machinery (column 1), their ability to bind the clathrin and/or the dynein complexes (column 2), their involvement in cargoes traffic (column 3), in microtubule-associated axonal growth (column 4), in the regulation of endocytosis (column 5) and others genes indirectly implicated in these functions (column 6). Importantly, because of their wide expression in different vesicles types, many of these genes such as Lnp, Syt1, Htt, Nav1 or Stab2 can be classified in more than one column. Genes highlighted in yellow were also identified by Wilson et al. (Wilson et al., 2010).
- https://doi.org/10.7554/eLife.11627.019
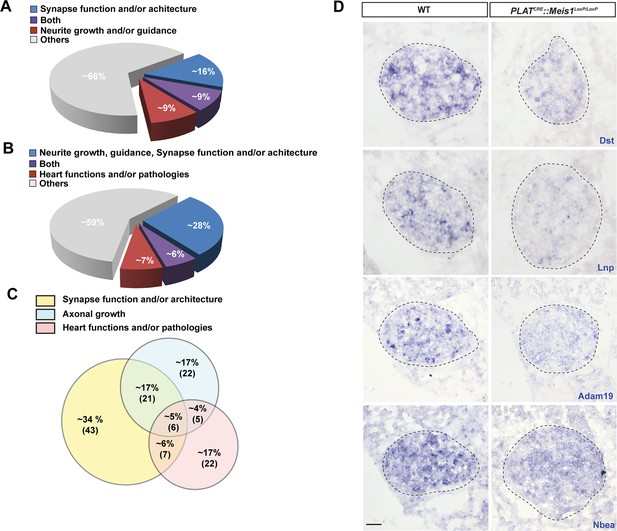
Bibliographical classification of Meis1 potential target genes.
(A) Diagram representing, among all identified Meis1 potential target genes, the percentage of genes with known functions in synapse machinery and target-field innervation. A significant proportion of these genes participate in both functions. (B) Diagram representing similarly the percentage of genes involved in target-field innervation, synaptogenesis and cardiac functions or pathologies with part of them involved in several of these processes. (C) Diagram showing, among selected target genes with known functions in synapse, target-field innervation and heart physiology and/or pathophysiology, the percentage of genes with function in both or all processes. Numbers in bracket indicate the absolute number of genes. (D) ISH for some of the Meis1 target genes identified by ChIP-seq and involved in cargoes formation, fusion or trafficking including Dst, Lnp, Adam19 and Nbea on SCGs of E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos illustrating the deregulation of their mRNAs expressions in the mutants. Dotted lines encircle the SCGs. Scale bars = 20 µm.
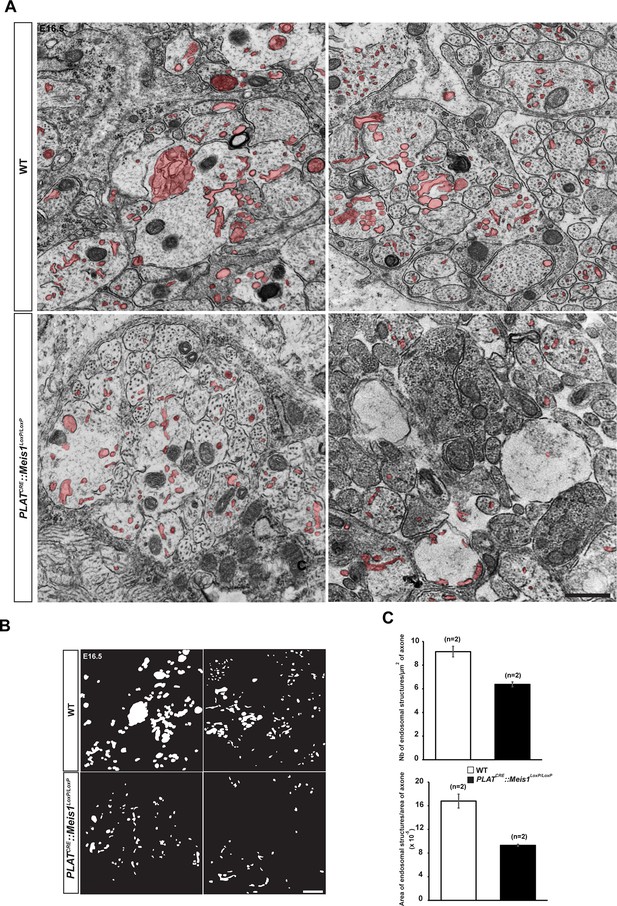
Analysis of endocytic ultrastructures in sympathetic mutant fibers.
(A) Representative electron microscopy images of sympathetic nerves exiting the SCG dissected from E16.5 WT and PLATCRE::Meis1LoxP/LoxP embryos. Note that WT axons contain more endocytic structures (colored in red) than mutant axons. Two representative pictures are shown for each genotype. (B) Camera lucida of the endocytic structures shown in (A). (C) Quantification of the number of endocytic structure per area in WT and PLATCRE::Meis1LoxP/LoxP axons section, and ratio between the area of endocytic structures and the area of axons in WT and PLATCRE::Meis1LoxP/LoxP nerves section. Data are represented as mean +/- s.e.m. n = 2–3. Scale bars = 500 nm.
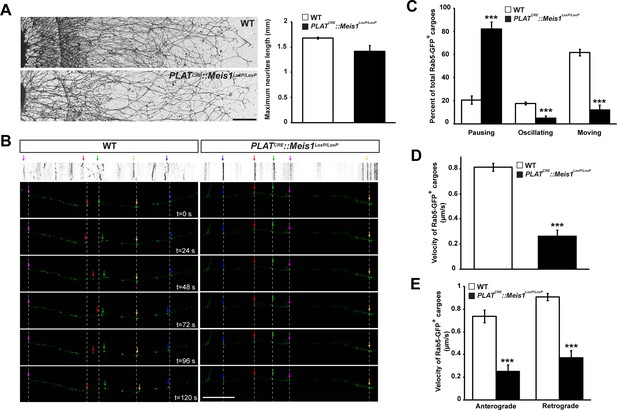
Meis1 is necessary for early endosomes trafficking.
(A) Camera lucida of TH-immunochemistry and quantification of neurites length on SCGs explants form WT and PLATCRE::Meis1LoxP/LoxP embryos. (B) Individual frames and corresponding kymograph of a 2 min time lapse video showing the movement of Rab5-GFP+ endosomes in WT (Video 1) and PLATCRE::Meis1LoxP/LoxP (Video 3) cultured SCGs explants. (C) Analysis of the percentage of the number of Rab5-GFP+ endosomes that are pausing, moving or oscillating in WT and PLATCRE::Meis1LoxP/LoxP cultured SCGs explants. (D) Measure of the velocity of Rab5-GFP+ endosomes that are moving in WT and mutant conditions. (E) Measure of the velocity of Rab5-GFP+ endosomes that are moving retrogradely and anterogradely in WT and PLATCRE::Meis1LoxP/LoxP cultured SCGs explants. Data are represented as mean +/- s.e.m; n = 3; ***p≤0.005. Scale bar = 200 µm in A, 20 µm in B. See also Videos 1–4.
Videos
Traffic of Rab5-GFP+ endosomes in WT neurons.
Representative videos showing 2 min acquisition of Rab5-GFP+ endosomes trafficking in the axons of cultured WT sympathetic neurons. See also Figure 7.
Impaired traffic of Rab5-GFP+ endosomes in Meis1 inactivated neurons.
Representative videos showing 2 min acquisition of Rab5-GFP+ endosomes trafficking in the axons of cultured PLATCRE::Meis1LoxP/LoxP sympathetic neurons. See also Figure 7.
Traffic of Rab5-GFP+ endosomes in WT neurons.
Representative videos showing 2 min acquisition of Rab5-GFP+ endosomes trafficking in the axons of cultured WT sympathetic neurons. See also Figure 7.
Impaired traffic of Rab5-GFP+ endosomes in Meis1 inactivated neurons.
Representative videos showing 2 min acquisition of Rab5-GFP+ endosomes trafficking in the axons of cultured PLATCRE::Meis1LoxP/LoxP sympathetic neurons. See also Figure 7.





