Mechanical sensitivity of Piezo1 ion channels can be tuned by cellular membrane tension
Figures
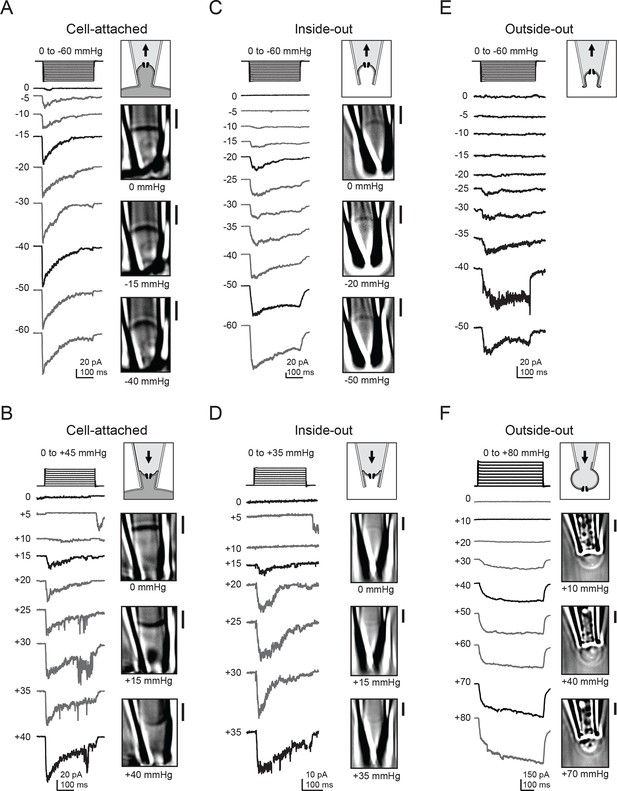
Electrophysiology and high-contrast imaging of Piezo-containing membranes.
Pressure-step protocol, representative currents and corresponding images from individual cell-attached patches from a HEK293t cell expressing mouse Piezo1-IRES-GFP, upon negative (A) and positive (B) pressure stimulation. Pressure-step protocol, respective representative currents and corresponding images from individual inside-out patches upon negative (C) and positive (D) pressure stimulation. Pressure-step protocol, respective representative currents and images from individual outside-out patches upon negative (E) and positive (F) pressure stimulation. All patches were held at −80 mV. Scale bars are 2 µm for all images.
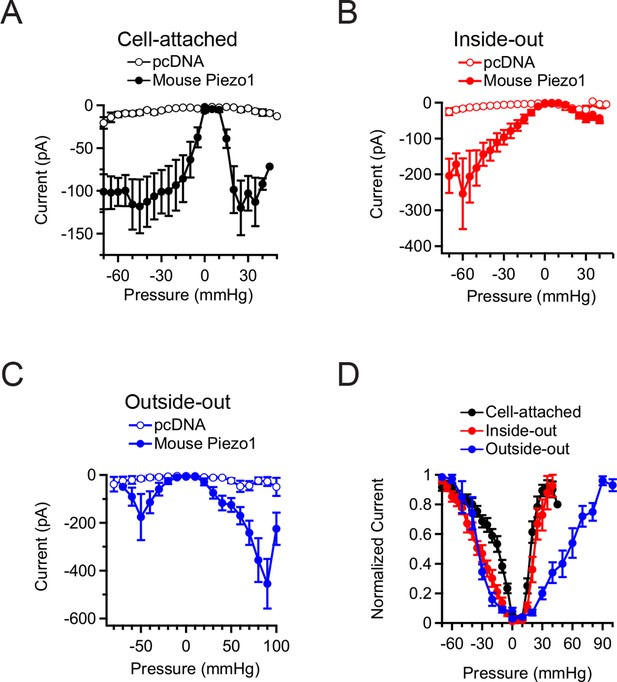
Mean Piezo1 current responses for all patch configurations upon positive and negative pressure stimulation.
(A) Pressure-evoked currents from cell-attached patches from HEK293t cells expressing empty vector (pcDNA; open circles) or Mouse Piezo1-IRES-GFP (closed circles). Separate patches were tested for positive and negative pressure stimulation. N = 7 for pcDNA at negative pressure, N = 6 for pcDNA at positive pressure, N = 15 for Piezo1 at negative pressure and N = 12 for Piezo1 at positive pressure. (B) Pressure-evoked currents from inside-out patches from HEK293t cells expressing empty vector (pcDNA; open circles) or Mouse Piezo1-IRES-GFP (closed circles). Separate patches were tested for positive and negative pressure stimulation. N = 4 for pcDNA at negative pressure, N = 3 for pcDNA at positive pressure, N = 10 for Piezo1 at negative pressure and N = 7 for Piezo1 at positive pressure. (C) Pressure-evoked currents from outside-out patches from HEK293t cells expressing empty vector (pcDNA; open circles) or Mouse Piezo1-IRES-GFP (closed circles). Separate patches were tested for positive and negative pressure stimulation. N = 3 for pcDNA at negative pressure, N = 7 for pcDNA at positive pressure, N = 6 for Piezo1 at negative pressure and N = 11 for Piezo1 at positive pressure. (D) Normalized mean current-pressure relations for all six configurations. For each individual patch currents were normalized to the peak current for that patch. All data points are mean ± s.e.m.
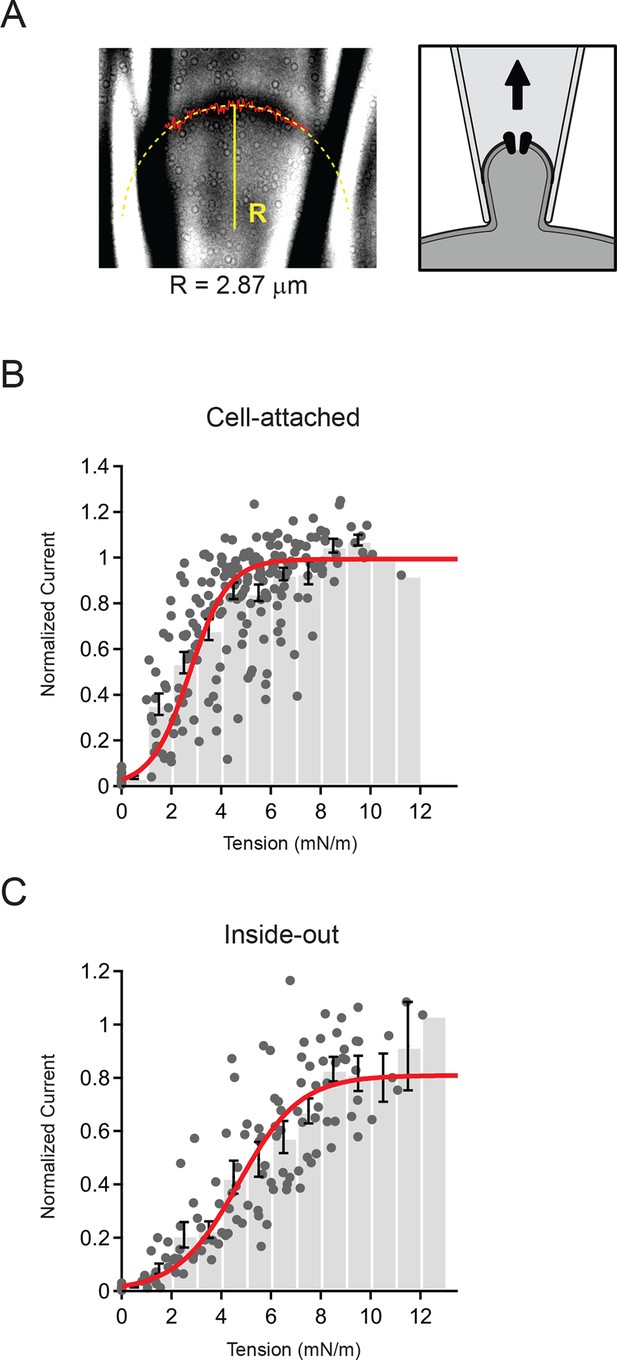
Measurement of membrane curvature and quantification of membrane tension.
(A) Representative image of cell-attached patch and schematic showing orientation of membrane. The solid red line marks the measured position of the membrane and the dashed yellow line is a circular fit to this position. Both steps were performed using a script written in Igor Pro (see Materials and methods). For this representative patch the radius R from the fit (solid yellow line) was 2.87 µm. (B) Current-tension histogram for Piezo1 responses to negative pressure in cell-attached patches from HEK293t cells. For each cell, current-pressure curves were fit with a sigmoid, and each response normalized to the plateau from the fit. Tension was calculated using the measured membrane curvature from the corresponding image for each response and normalized current plotted against tension (gray circles). Data were binned (bin width 1 mN/m) and pooled (black bars; mean ± s.e.m). Binned data were fit with a Boltzmann function: Imax/(1+exp(-(T-T50])/k])) where Imax is the maximal normalized current, T is tension, T50 is the tension of half-maximal activation, and k is the slope factor. The standard deviation of the normalized amplitude for each bin was used to weight the fit. Fit parameters Imax = 0.99±0.01, T50 = 2.7±0.1 mN/m, k = 0.8±0.1. N = 15 cells and 218 responses. (C) Current-tension histogram for Piezo1 responses to negative pressure in inside-out patches from HEK293t cells. Plot was generated as described in (B). Fit parameters: Imax = 0.81±0.04, T50 = 4.7±0.3 mN/m, k = 1.2±0.1. N = 10 cells and 123 responses.
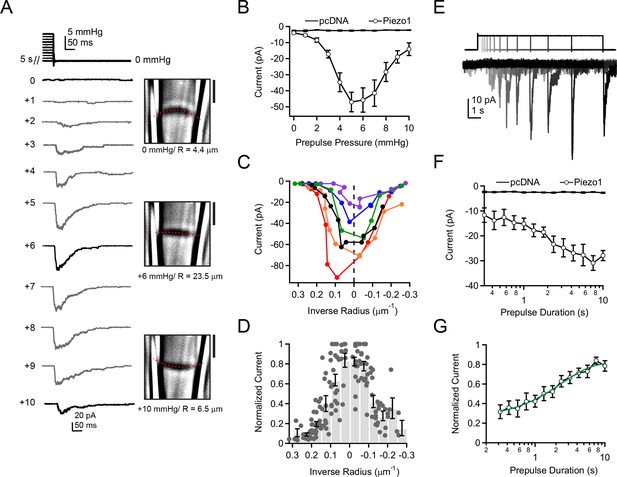
Activation of Piezo1 currents upon release of pressure stimulation.
(A) Left, pressure stimulus protocol and representative currents showing activation of Piezo1 ion channels in a cell-attached patch upon release of a 5 s positive pressure stimulus. Holding potential was −80 mV. Right, corresponding images for 0, +6, and +10 mmHg pressure steps with membrane patch radius R fit superimposed (red dashes) and calculated radius indicated below. (B) Mean peak current upon release of a 5 s positive pressure pulse (0 to +10 mmHg) for cells transfected with empty vector (pcDNA; N = 9 cells) and with mouse Piezo1 (N = 14 cells). (C) Current-radius relationships for six representative measurements performed as shown in (A). The solid black line is showing the measurement in (A). (D) Normalized current-radius relationship for all measurements. For each individual patch currents were normalized to the maximal response from that patch and plotted versus inverse radius. Data were binned (bin width 0.05 µm-1); bars represent mean normalized amplitude ± s.e.m. for each bin. N = 14 cell-attached patches and 148 responses. (E) Pressure-stimulus protocol and representative currents showing the time course of current increase with longer prepulse duration in a patch expressing mouse Piezo1. (F) Mean peak current as a function of prepulse duration for cells transfected with pcDNA or Piezo1 (N = 9 and N = 11, respectively). For each Piezo1 patch, the prepulse amplitude that caused maximal current for that cell (determined with protocol in (A)) was used. For our patch pipette sizes this was typically +5 or +6 mmHg; +5 mmHg was used for all pcDNA patches. (G) Normalized mean peak current as a function of prepulse duration for cells transfected with Piezo1. For each individual patch, currents were normalized to maximal response from that patch. Mean data were fit with an exponential function I=Imax + A*exp(-t-t0)/tau. Fit parameters Imax = 0.82±0.02, A = 0.49±0.02, tau = 2.4±0.3 ms. N = 11 cells. All data points are mean ± s.e.m.
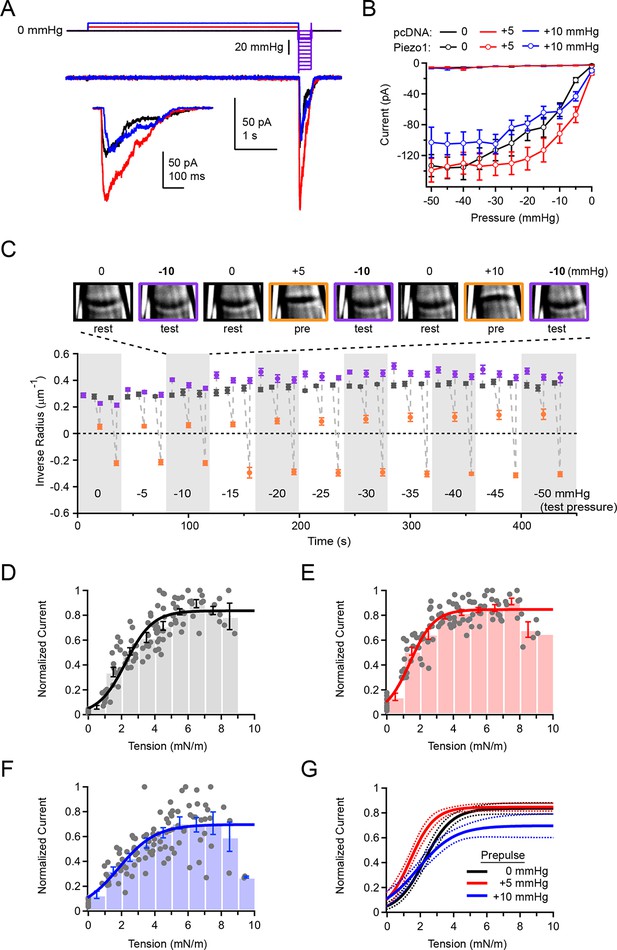
Overall Piezo1 sensitivity is regulated by resting membrane tension.
(A) Stimulus protocol and representative currents from a cell-attached HEK293t cell patch expressing mouse Piezo1-IRES-GFP. The test pulse for these currents was −10 mmHg (thick purple line); holding potential was −80 mV. Inset shows test currents at magnified scale. (B) Peak current-pressure relationships for test pulses (0 to −50mmHg, Δ5 mmHg) following 5 s 0 mmHg, +5 mmHg and +10 mmHg prepulses.. All data points are mean ± s.e.m. N = 8 cell-attached patches (pcDNA) and 11 cell-attached patches (Piezo1). (C) Mean patch curvature as a function of time during protocol performed shown in (A). Representative images of one individual patch are shown above. Each patch was tested with no prepulse (0 mmHg), a +5 mmHg prepulse, and a +10 mmHg prepulse at each test pressure before advancing to the next test pressure. Grey markers show inverse radius during rest periods (0 mmHg, between stimuli), purple markers show inverse radius during 300 ms test pulses (0 to −50 mmHg, Δ5 mmHg), orange markers show inverse radius during +5 mmHg or +10 mmHg prepulse. All data points are mean ± s.e.m. N = 11 for cell-attached patches. (D–F) Normalized current-tension relationships obtained from protocol shown in (A) using no prepulse (0 mmHg) (D), +5 mmHg prepulse (E) and +10 mmHg prepulse (F). Currents from individual patches are normalized to the maximal response for each patch. Data were pooled and binned (bin width 1 mN/m); bars represent mean ± s.e.m. N = 11 patches. Binned data were fit with a Boltzmann function I = Imax/(1+exp(-(T-T50/k)) where I is normalized current, Imax is the plateau, T is tension, T50 is the tension of half-maximal activation, and k is the slope factor. The standard deviation of the normalized amplitude for each bin was used to weight the fit. Fit parameters for no prepulse (0 mmHg): Imax = 0.84±0.02, T50 = 2.2±0.1 mN/m, k = 0.8±0.1. For +5 mmHg prepulse: Imax = 0.85±0.01, T50 = 1.4±0.1 mN/m, k = 0.7±0.1. For +10 mmHg prepulse: Imax = 0.70±0.04, T50 = 1.8±0.2 mN/m, k = 1.1±0.2. (G) Fits from D-F overlayed (solid line) with 95% confidence intervals (dashed lines).
Videos
Response of cell-attached HEK293t patch to stimulation with negative pressure in −5 mmHg increments; acquired at 7.9 frames/s, played at 50 frames/s.
Video corresponds to cell in Figure 1A.





