EGF-dependent re-routing of vesicular recycling switches spontaneous phosphorylation suppression to EGFR signaling
Figures
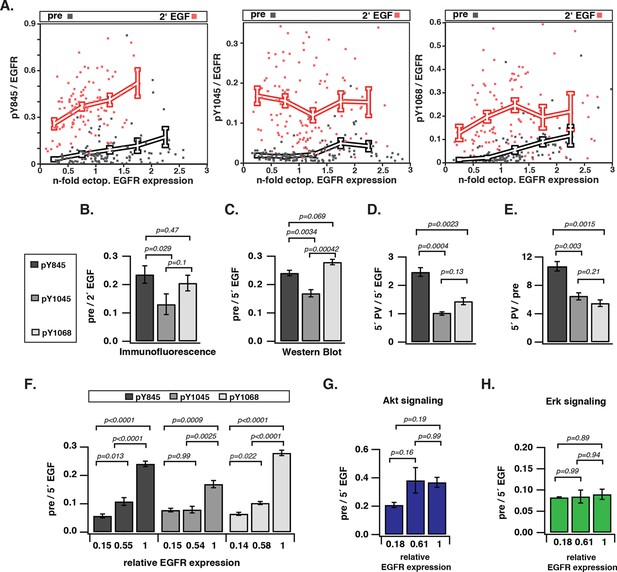
Spontaneous tyrosine phosphorylation in response to EGFR expression.
(A) Scatter plots show the relative phosphorylation (mean fluorescence intensity of pYN-antibody/EGFR-mCitrine) of three tyrosine residues (N= 845, 1045, or 1068) versus EGFR-mCitrine expression in multiples of mean endogenous EGFR levels (see Figure 1—figure supplement 1A,B). Points (black: pre-; red: 2-min post-stimulation with 100 ng/ml EGF) represent single COS-7 cells (see Figure 1—figure supplement 1C) and thick lines indicate mean values of binned data. (B,C) Spontaneous phosphorylation of Y845, Y1045 and Y1068. (B) Immunofluorescence data showing mean fluorescence intensity of pYN-antibody over EGFR-mCitrine (pre) normalized to mean fluorescence intensity of pYN-antibody over EGFR-mCitrine, 2-min post-stimulation with 100 ng/ml EGF (2’ EGF). YN(number of cells pre, 2’ EGF): Y845(119, 108); Y1045(145, 109); Y1068(127, 87). (C) Western blot analysis of COS-7 lysates probed for anti-GFP antibody and either anti-pY845 (n=5 blots), anti-pY1045 (n=3) or anti-pY1068 (n=3) show pYN-antibody band over anti-GFP band (pre) normalized to the same fraction 5-min post-stimulation with 100 ng/ml EGF (5’ EGF). Data correspond to the highest levels of expressed EGFR-mCitrine (3 µg cDNA, Figure 1—figure supplement 2). (D, E) Autonomous phosphorylation of Y845, Y1045, and Y1068 upon pervanadate (PV) treatment. Western blot analysis of COS-7 lysates probed for anti-pY845, anti-pY1045, anti-pY1068 and anti-GFP antibody (all n=3 blots). (D) pYN-antibody band over anti-GFP band 5 min post-addition of PV (0.33 mM) normalized to relative phosphorylation 5-min post-stimulation with 100 ng/ml EGF (Figure 1—figure supplement 3). (E) pYN-antibody band over anti-GFP band 5-min post-addition of PV (0.33 mM) normalized to relative phosphorylation before stimulation (pre). (F) Dependence of EGFR phosphorylation on its expression level. Western blot analysis of COS-7 lysates transfected with increasing amounts (0.5, 1.5, and 3 µg cDNA) of EGFR-mCitrine. Blots were probed for anti-pY845 (n=5 blots), anti-pY1045 (n=3), anti-pY1068 (n=3), and anti-GFP for EGFR-mCitrine (Figure 1—figure supplement 2). The ordinate displays the fraction of EGFR-band over tubulin band in each lane relative to 3 µg cDNA and bar diagram shows relative EGFR phosphorylation of the three tyrosine residues (see 'Materials and methods'). (G, H) Dependence of Akt and Erk activation on EGFR expression level. Western blots of COS-7 lysates transfected with increasing amounts (0.5, 1.5, and 3 µg cDNA) of EGFR-mCitrine and probed for phosphorylated Ser473 on Akt, total Akt, phosphorylated Thr202/Tyr204 on Erk1/2, and total Erk1/2 levels. Data represents 'ratio of fractions' of either Akt phosphorylation (E) or Erk phosphorylation as a function of EGFR-mCitrine expression level as described in (D) (n=3 blots, Figure 1—figure supplement 4). All error bars correspond to standard error of the mean. EGFR, epidermal growth factor receptor.
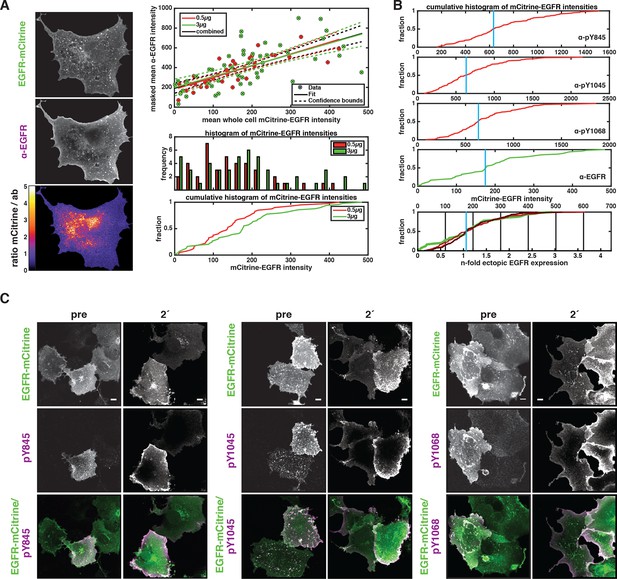
Relating ectopic EGFR-mCitrine expression with endogenous EGFR expression levels and dependency of autonomous EGFR activation on EGFR expression levels.
(A) Left panel: Exemplary COS-7 cell expressing EGFR-mCitrine (fluorescence intensity displayed in upper image) that was immunostained against EGFR (antibody fluorescence displayed in middle image). The background-corrected ratio of intensities (lower image) clearly shows areas of mismatch, where antibody staining failed to reach all available EGFR-mCitrine (red-yellow-white, ratio > 2). We therefore quantified the average anti-EGFR fluorescence intensity in the image masked with the thresholded ratio-image (0.15 < ratio mCitrine / anti-EGFR < 1.5). This average anti-EGFR fluorescence intensity is plotted against the average whole-cell EGFR-mCitrine intensity (right panel, upper graph) for transfection with 0.5 µg (red) and 3 µg (green) cDNA. To be able to relate mCitrine fluorescence intensity with endogenous EGFR expression levels, this data was fitted with a linear model (straight lines with dashed lines for 95% confidence intervals), yielding a slope of m=1.15 ± 0.13 and an abscissa intercept of b=190 ± 25 (all data). This intercept corresponds to the anti-EGFR fluorescence without ectopic EGFR-mCitrine expression and m/b is the factor that translates mCitrine intensity to n-fold ectopic expression over endogenous levels. This quantification is almost irrespective of the amount of cDNA used for EGFR-mCitrine transfection. Compared to transfection with 0.5 µg cDNA (red), the frequency of a higher average EGFR-mCitrine intensity occurring (right panel, lower graphs) clearly increases when 3 µg (green) of cDNA were used, however, not by a factor of 6 that could be expected. For one, a larger amount of cDNA not only increases the amount of cDNA per cell, but also the number of transfected cells (transfection efficiency). Secondly, selection of cells under the microscope suffers from a bias toward transfected cells. However, we used all cells in a field of view to also incorporate weakly expressing cells in our results. (B) For single-cell immunofluorescence experiments (Figure 1A) that were stained with antibodies against EGFR phosphorylation, we matched the cumulative EGFR-mCitrine fluorescence intensity histograms (red curves in upper three plots, for the phosphotyrosine-specific antibody indicated) by normalizing with a scalar to that of anti-EGFR immunostaining (green curve, same as green curve in A). Thereby, the median values of the mCitrine intensity distributions (blue vertical lines) coincide (lowest plot) and the n-fold ectopic EGFR expression over mean endogenous level can be calculated for each series of experiments. (C) Expression level dependence of EGFR-mCitrine phosphorylation at Y845 (left panel), Y1045 (middle panel), and Y1068 (right panel). Immunostaining with specific pYN-antibodies (second column) in COS-7 cells ectopically expressing EGFR-mCitrine (first column) and corresponding green/magenta overlay (third column) before or after 2-min stimulation with 100 ng/ml EGF (Figure 1A). All scale bars: 10 μm. EGFR, epidermal growth factor receptor.
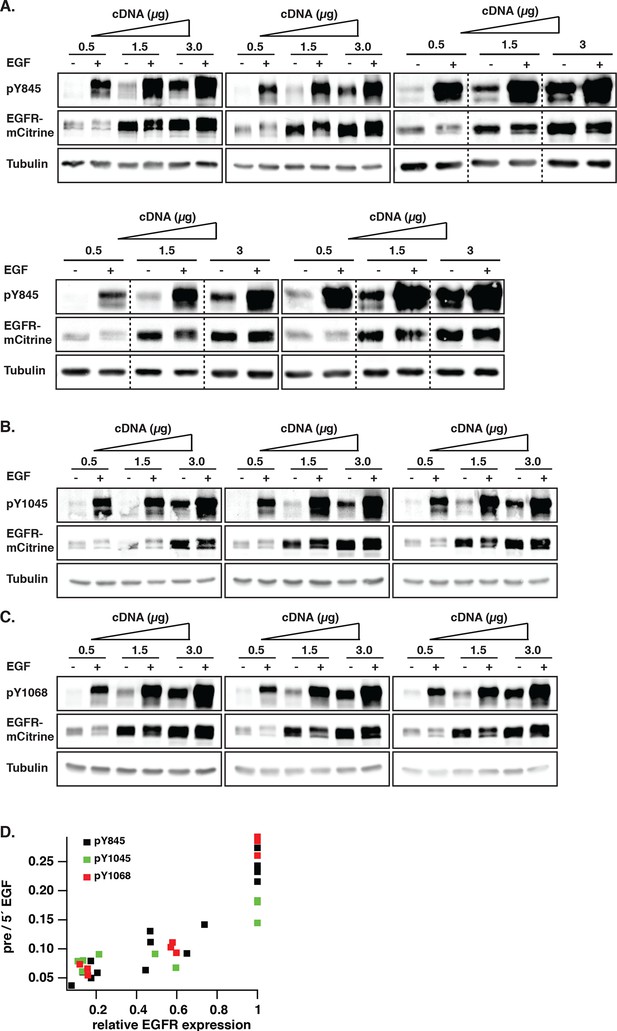
Dependency of autonomous EGFR activation on EGFR expression levels.
(A-–C) COS–7 cells were transfected with increasing amounts of EGFR-mCitrine cDNA (0.5, 1.5, and 3 µg). Lysates were collected pre or 5-min post-stimulation with 100 ng/ml EGF and used in Western blot analysis and phosphorylation of Y845 (A), Y1045 (B), and Y1068 (C) was determined with specific pYN-antibodies. EGFR-mCitrine was detected with an anti-GFP antibody. Anti-tubulin was used as a loading control. (D) Scatter plots shows pYN-antibody band over anti-GFP band (pre) normalized to the same fraction 5-min post-stimulation with 100 ng/ml EGF for individual bands versus the relative EGFR expression level normalized to the tubulin band relative to 3 µg cDNA (see 'Materials and methods'). EGFR, epidermal growth factor receptor.
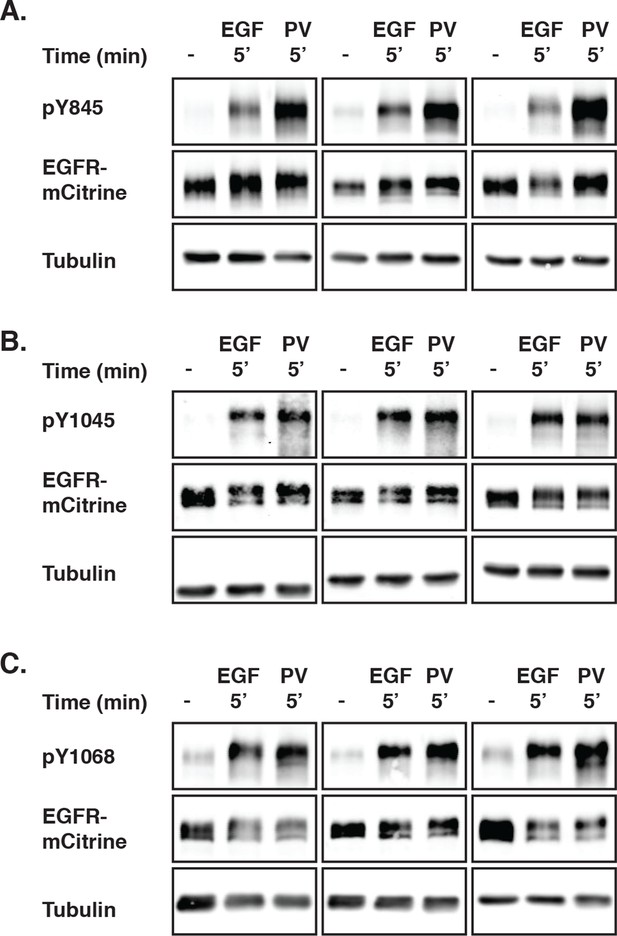
EGFR phosphorylation induced by PV-mediated PTP inhibition and EGF stimulation.
(A-(–C) COS–7 cells expressing EGFR-mCitrine were stimulated with EGF (100 ng/ml) or treated with PV (0.33 mM) for 5 min. The collected lysates were used in Western blot analysis, and the phosphorylation of Y845 (A), Y1045 (B), and Y1068 (C) was determined by using specific pYN-antibodies. EGFR-mCitrine was detected with an anti-GFP antibody. Anti-tubulin was used as a loading control. EGFR, epidermal growth factor receptor; PV, pervanadate; PTP, protein tyrosine phosphatase.
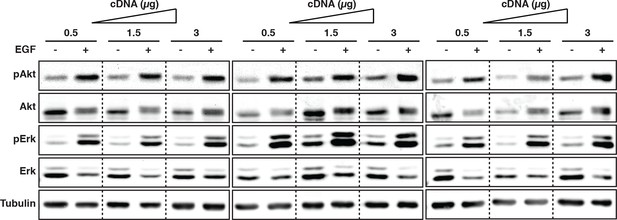
Dependency of downstream EGFR signaling on EGFR expression levels.
COS–7 cells were transfected with increasing amounts of EGFR-mCitrine cDNA (0.5, 1.5, and 3 µg). Lysates were collected pre or 5-min post-stimulation with 100 ng/ml EGF and used in Western blot analysis. Blots were probed with phospho-specific antibodies for Akt and Erk and total Akt and total Erk antibodies. Anti-tubulin was used as a loading control. EGFR, epidermal growth factor receptor.
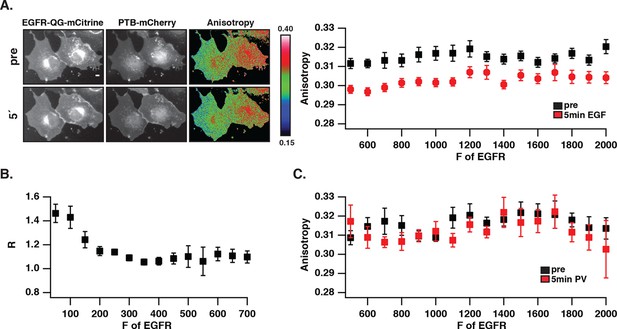
Autonomously and ligand-activated EGFR are different molecular states.
(A) EGFR-QG-mCitrine anisotropy in COS-7 cells. Representative fluorescence images of EGFR-QG-mCitrine (first column), PTB-mCherry (second column), and anisotropy of EGFR-QG-mCitrine (third column) upon EGF stimulation for the indicated time. Scale bar: 10 μm. Graph shows the anisotropy of EGFR-QG-mCitrine versus its binned mean fluorescence intensity (F of EGFR) per pixel (black: pre-, red: 5-min post-stimulation with 100 ng/ml EGF). (B) Corresponding phosphorylation of EGFR-QG-mCitrine. Graph shows the recruitment (R, see 'Materials and methods') of PTB to EGFR versus the binned mean fluorescence intensity (F of EGFR) of EGFR-QG-mCitrine per pixel. (C) Anisotropy of spontaneously activated EGFR-QG-mCitrine upon PV-mediated PTP inhibition. Graph shows the anisotropy of EGFR-QG-mCitrine versus its binned mean fluorescence intensity (F of EGFR) per pixel (black: pre-, red: 5-min post-treatment with PV 0.33 mM; see Figure 2—figure supplement 1B). All error bars correspond to (standard error of the mean. EGFR, epidermal growth factor; PTB, phosphotyrosine-binding domain; PV, pervanadate.
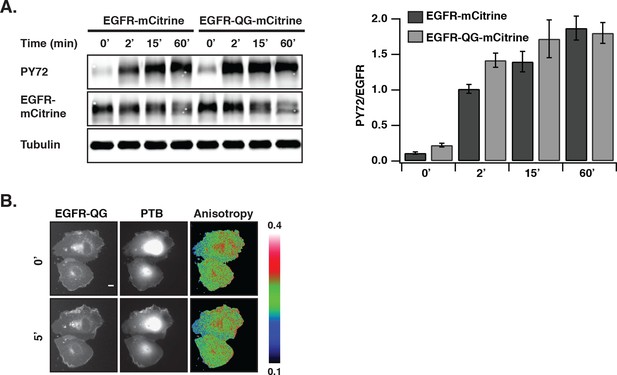
EGF-induced EGFR-QG-mCitrine phosphorylation and anisotropy upon PTP inhibition by PV.
(A) COS–7 cells expressing EGFR-mCitrine or EGFR-QG-mCitrine were stimulated with EGF (100 ng/ml). The collected lysates were used in western blot analysis and probed with the generic phospho tyrosine antibody PY72 and anti-GFP (EGFR-mCitrine). Anti-tubulin was used as a loading control. The bar diagram shows the relative phosphorylation of EGFR-mCitrine and EGFR-QG-mCitrine for the indicated time points (mean ± SEM, n=3). (B) EGFR-QG-mCitrine anisotropy upon PV treatment. Representative fluorescence images of COS–7 cells co-expressing EGFR-QG-mCitrine and PTB-mCherry and the corresponding anisotropy images of EGFR-QG-mCitrine. Upper row: Pre-stimulation, lower row: 5-min post-stimulation with 0.33 mM PV. Scale bar: 10 μm. EGFR, epidermal growth factor receptor; PTB, phosphotyrosine-binding domain PV,pervanadate; SEM, standard error of the mean.
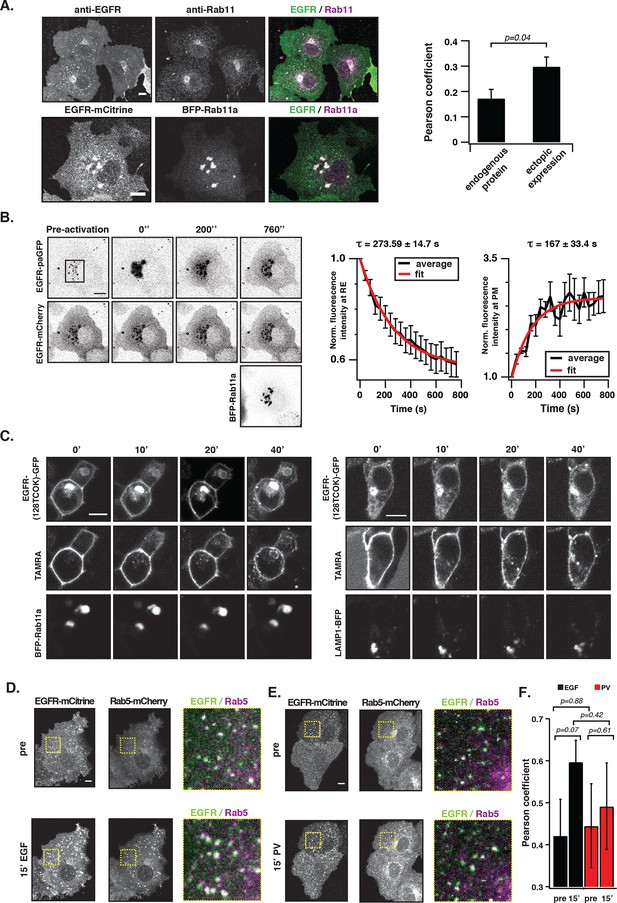
EGFR continuously recycles through the RE.
(A) Co-localization of EGFR and Rab11. Left panel, first row: Immunostaining of endogenous EGFR (left), Rab11 (middle) and corresponding green/magenta overlay (right) in fixed COS-7 cells. Left panel, second row: Fluorescence images of EGFR-mCitrine (left), BFP-Rab11a (middle), and corresponding green/magenta overlay (right) in living COS-7 cells. Right panel: Quantification of co-localization between EGFR and Rab11 in A (n=15 cells), B (n=12) by Pearson’s correlation coefficient. (B) Fluorescence redistribution after photoactivation of EGFR-paGFP on the recycling endosome. First row: EGFR-paGFP fluorescence at the indicated time in seconds (photoactivation area: black rectangle in the pre-activation image), second row: corresponding EGFR-mCherry fluorescence and image in third row: BFP-Rab11a fluorescence. Right graphs: Loss of EGFR-paGFP fluorescence at the RE normalized to EGFR-mCherry fluorescence (left) and corresponding gain of EGFR-paGFP fluorescence at the PM (right), normalized to EGFR-mCherry fluorescence. Average trace (black ± SEM.; n=6 cells) was fitted to an exponential function (red) to retrieve time constants (τ) of EGFR fluorescence intensity lossat the RE and its gain at the PM. (C) Fluorescence redistribution of TAMRA-labeled-EGFR in HEK293 cells. Representative fluorescence images of EGFR-(128TCOK)-GFP (first row), TAMRA-labeled EGFR-(128TCOK)-GFP (second row), and BFP-Rab11a (third row, left panel) or LAMP1-BFP (third row, right panel) for the indicated time (min) in the respective columns (see 'Materials and methods'). (D) Co-localization of ligand-activated EGFR and Rab5 in COS-7 cells. Representative fluorescence images of EGFR-mCitrine (first column), Rab5-mCherry (second column), and green/magenta overlay of selected ROIs (third column), pre- and 15-min post- stimulation with EGF (100 ng/ml). (E) Co-localization of spontaneously activated EGFR and Rab5 in COS-7 cells. Representative fluorescence images of EGFR-mCitrine (first column), Rab5-mCherry (second column), and green/magenta overlay of selected ROIs (third column), pre- and 15-min post- treatment with 0.33 mM PV. (F) Quantification of co-localization between EGFR-mCitrine and Rab5-mCherry with Pearson’s correlation coefficient upon EGF stimulation (100 ng/ml, n=5 cells) or PV treatment (0.33 mM, n=7 cells) for the indicated time points. All scale bars: 10 μm. EGFR, epidermal growth factor receptor; PM, plasma membrane; PV, pervanadate; RE, recycling endosome; SEM, standard error of the mean.
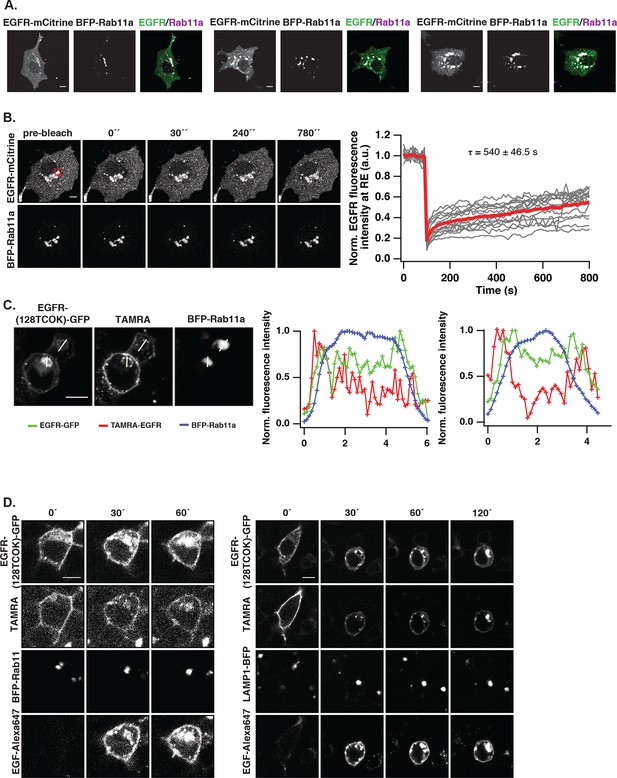
Vesicular trafficking of autonomously and ligand-activated EGFR.
(A) Co-localization of EGFR and Rab11 upon inhibition of protein synthesis. Representative fluorescence images of three individual cells co-expressing of EGFR-mCitrine (first row) and BFP-Rab11a (second row), and corresponding green/magenta overlay (third row) after treatment with cyclohexamide (10 μg/ml) for app. 20 hr. (B) Fluorescence recovery after photobleaching EGFR-mCitrine in COS–7 cells co-expressing BFP-Rab11a. Representative fluorescence images of EGFR-mCitrine and BFP-Rab11a for the indicated time points (left panel). EGFR-mCitrine was photobleached in the indicated region at the RE (red square). The normalized mean fluorescence intensity of the bleached area is plotted as a function of time (red line: average curve, grey lines: individual recovery curves, n=16 cells, right panel). (C) EGFR trafficking from the PM to the RE. Fluorescence images of EGFR-TCOK(128)-GFP, TAMRA-EGFR, and BFP-Rab11a with white lines for the profile analysis. Line plots show the normalized fluorescence intensity of BFP-Rab11a (blue), TAMRA-EGFR (red), and EGFR-GFP for the upper (middle panel) and lower cell (right panel). The highest intensity of each channel was normalized to 1. (D) Fluorescence redistribution of TAMRA-labeled EGFR upon EGF stimulation. Representative fluorescence images of HEK293 cells ectopically expressing EGFR-(128TCOK)-GFP and BFP-Rab11a (left panel) or LAMP1-BFP (right panel) for the indicated time points. EGFR-(128TCOK)-GFP was selectively labeled on the plasma membrane using TAMRA and simultaneously stimulated with 100 ng/ml EG(F). All scale bars: 10 μm. EGFR, epidermal growth factor receptor; PM, plasma membrane; RE, recycling endosome.
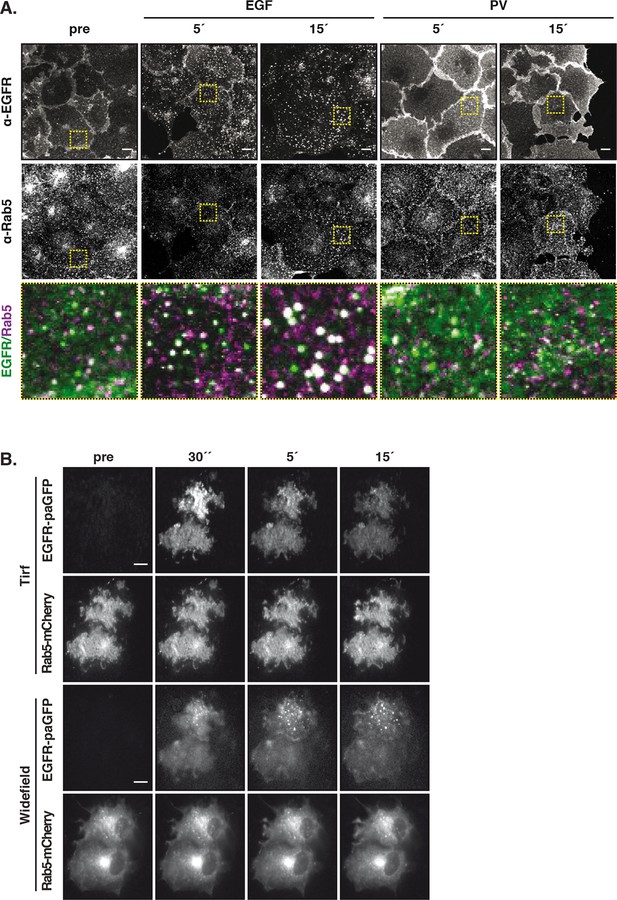
Vesicular trafficking of autonomously-activated EGFR.
(A) Immunostaining of endogenous EGFR (upper row), Rab5 (middle row) and green/magenta overlay of selected ROIs (lower row) in fixed COS–7 cells after stimulation with EGF (100 ng/ml) or treatment with PV (0.33 mM) for the indicated time. (B) Fluorescence redistribution after photoactivation of EGFR-paGFP. Representative fluorescence images of COS-7 cells co-expressing EGFR-paGFP and Rab5-mCherry for the indicated time points. EGFR-paGFP was photoactivated at the basal membrane in TIRF microscopy and its redistribution and co-localization with Rab5-mCherry was followed over time in TIRF and widefield microscopy. All scale bars: 10 μm. EGFR, epidermal growth factor receptor; PV, pervanadate.
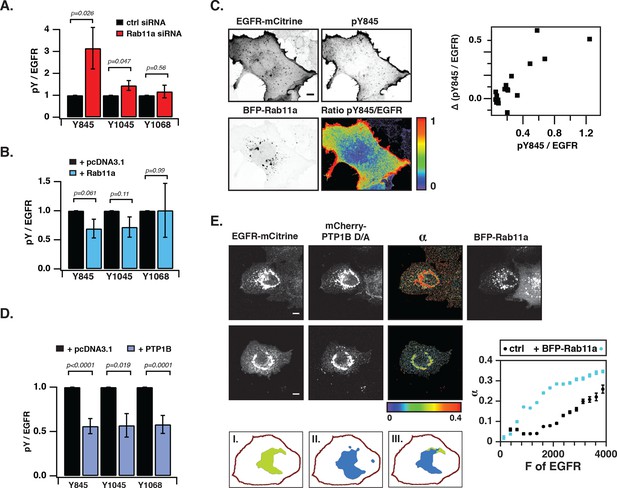
Suppression of spontaneous autocatalytic EGFR activation by vesicular recycling.
(A) Effect of Rab11a knockdown on EGFR-mCitrine phosphorylation in COS-7 cells. The bar diagram shows the relative phosphorylation (pY/EGFR) of EGFR-mCitrine on Y845, Y1045, and Y1068 upon siRNA-mediated Rab11a knockdown normalized to pY/EGFR for cells transfected with non-targeting siRNA. For non-targeting siRNA: Y845 (n=69 cells), Y1045 (n=82), Y1068 (n=75). For Rab11a siRNA: Y845 (n=75 cells), Y1045 (n=74), Y1068 (n=83) (see Figure 4—figure supplement 1A,B). (B) Effect of BFP-Rab11a expression on EGFR-mCitrine phosphorylation in COS-7 cells. The bar diagram shows the relative phosphorylation (pY/EGFR) of Y845, Y1045 and Y1068 on EGFR-mCitrine upon ectopic expression of BFP-Rab11a normalized to pY/EGFR in the presence of empty pcDNA 3.1. For BFP-Rab11a ectopic expression: Y845 (n=52 cells), Y1045 (n=34), Y1068 (n=38) and for pcDNA 3.1: Y845 (n=125 cells), Y1045 (n=110), Y1068 (n=138) (see Figure 4—figure supplement 2A). (C) Spatial distribution of spontaneously phosphorylated Y845 in COS-7 cells. Left panel: Representative fluorescence images of EGFR-mCitrine (top left), immunostaining of pY845 (top right), BFP-Rab11a (bottom left), and a ratio image of pY845/EGFR-mCitrine (bottom right). Right panel: Graph shows the difference in Y845 auto-phosphorylation between the PM and RE (Δ pY845 over EGFR, see materials and methods) as a function of overall EGFR phosphorylation level in individual cells (pY845/EGFR). (D) Effect of ectopically expressed BFP-PTP1B on EGFR-mCitrine phosphorylation in COS-7 cells. The bar diagram shows the relative phosphorylation (pY/EGFR) of EGFR-mCitrine on Y845, Y1045, and Y1068 upon ectopic expression of BFP-PTP1B normalized to pY/EGFR upon transfection with empty pcDNA 3.1. For BFP-PTP1B ectopic expression: Y845 (n=69 cells), Y1045 (n=59), Y1068 (n=61) and for pcDNA 3.1: Y845 (n=54 cells), Y1045 (n=57), Y1068 (n=51) (see Figure 4—figure supplement 2B). (E) Spatial distribution of the interacting fraction (α, third column) of EGFR-mCitrine (first column) with mCherry-PTP1B D/A (second column) as detected by FLIM-FRET, with (upper row) or without (lower row) ectopic expression of BFP-Rab11a (4th column). Graph shows average α in regions of high EGFR-mCitrine intensity as a function of mean fluorescence (F of EGFR) with (blue, n=28 cells) or without ectopic expression of BFP-Rab11a (black, n=20). Lower row: percentage of EGFR/PTP1B D/A interactions in the vicinity of the RE was retrieved from the overlap (III) between areas with high α values (I) and areas with high intensity of BFP-Rab11a fluorescence (II) (see 'Materials and methods' and Figure 4—figure supplement 3) All scale bars: 10 μm. All error bars correspond to standard error of the mean. EGFR, epidermal growth factor receptor.
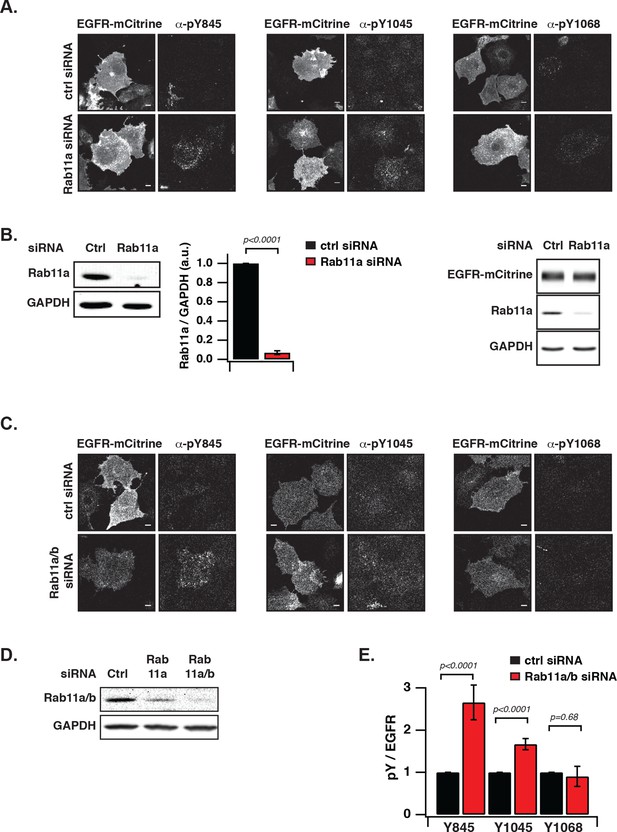
Suppression of autonomous EGFR activation by recycling through perinuclear membranes.
(A) Effect of Rab11a knockdown on EGFR phosphorylation at Y845, Y1045, and Y1068. COS-7 cells expressing EGFR-mCitrine were immunostained with specific pYN-antibodies to detect phosphorylation of Y845, Y1045, and Y1068 upon transfection with non-targeting siRNA (Ctrl, upper columns) or upon siRNA-mediated Rab11a knockdown (Rab11a, lower columns). (B) Left: Western blot of COS-7 lysates 72 hr post-transfection with non-targeting siRNA (Ctrl, first column) or Rab11a siRNA (siRab11a, second column). Blots were probed for Rab11a and GAPD(H) Middle: Bar diagram represents the ratio of Rab11a to GAPDH (n=5; mean ± SEM). Right: EGFR-mCitrine expression upon Rab11a knockdown. Western blot of COS-7 cells expressing EGFR-mCitrine after transfection with non-targeting siRNA (Ctrl, first column) or Rab11a siRNA (siRab11a, second column). Blot was probed for Rab11a and GAPDH and an anti-GFP antibody to detect EGFR-mCitrine. (C) Effect of Rab11a/b knockdown on EGFR phosphorylation at Y845, Y1045 and Y1068. COS-7 cells expressing EGFR-mCitrine were immunostained with specific pYN-antibodies to detect phosphorylation of Y845, Y1045, and Y1068 upon transfection with non-targeting siRNA (Ctrl, upper columns) or upon siRNA-mediated Rab11a/b knockdown (Rab11a/b, lower columns). (D) Western blot of COS-7 lysates 72 hr post-transfection with non-targeting siRNA (Ctrl, first column) or Rab11a siRNA (siRab11a, second column) or Rab11a/b siRNA (siRab11a/b, third column). Blots were probed for, Rab11a/b and GAPDH. (E) Effect of Rab11a/b knockdown on EGFR-mCitrine phosphorylation. The bar diagram shows the relative phosphorylation pY/EGFR of EGFR-mCitrine on Y845, Y1045, and Y1068 upon transfection with non-targeting siRNA or upon siRNA-mediated Rab11a/b knockdown. For non-targeting siRNA:Y845 (n=95 cells), Y1045 (n=97) Y1068 (n= 84). For Rab11a/b siRNA: Y845 (n=147), Y1045 (n=96), Y1068 (n=88). All scale bars: 10 μm. All error bars correspond to standard error of the mean. EGFR, epidermal growth factor receptor.
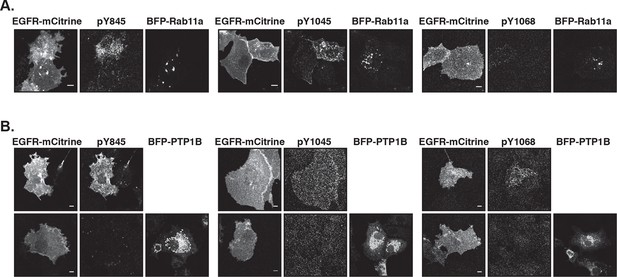
Suppression of autonomous EGFR activation by recycling through perinuclear membranes.
(A) Effect of ectopic BFP-Rab11a expression on EGFR phosphorylation at Y845, Y1045, and Y1068. COS-7 cells expressing EGFR-mCitrine were immunostained with specific pYN-antibodies to detect phosphorylation of Y845, Y1045, and Y1068 in the presence of empty pcDNA 3.1 or BFP-Rab11a. (B) Effect of ectopic BFP-PTP1B expression on EGFR phosphorylation at Y845, Y1045 and Y1068. COS-7 cells expressing EGFR-mCitrine were immunostained with specific pYN-antibodies to detect phosphorylation of Y845, Y1045, and Y1068 in the presence of empty pcDNA 3.1 (upper columns) or presence of BFP-PTP1B (lower columns). All scale bars: 10 μm. EGFR, epidermal growth factor receptor.
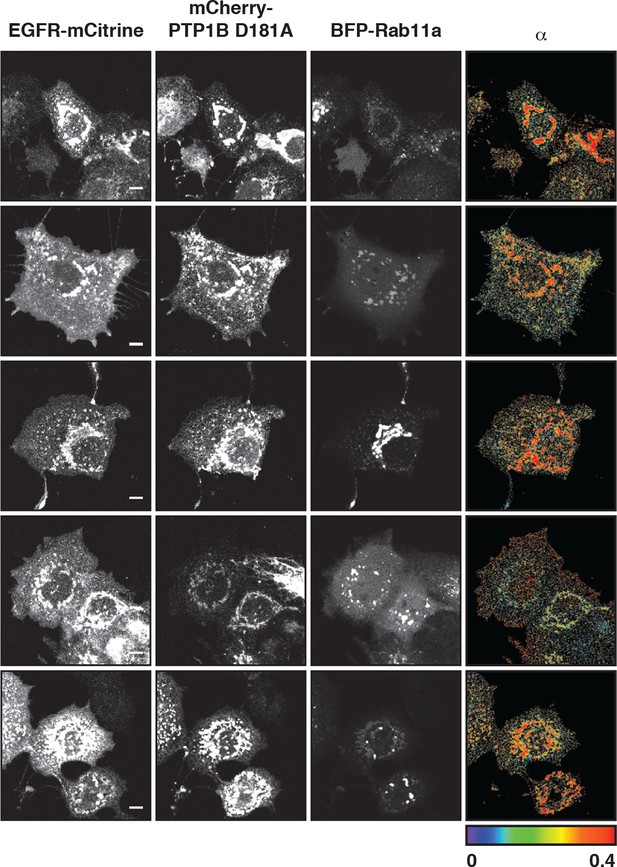
Interaction of EGFR and PTP1B D181A in perinuclear areas.
Representative fluorescence images of living COS–7 cells co-expressing EGFR-mCitrine, PTP1B D181A-mCherry, BFP-Rab11a and α map showing the fraction of interaction between EGFR-mCitrine and PTP1B D181A-mCherry. Scale bars: 10 μm. EGFR, epidermal growth factor receptor.
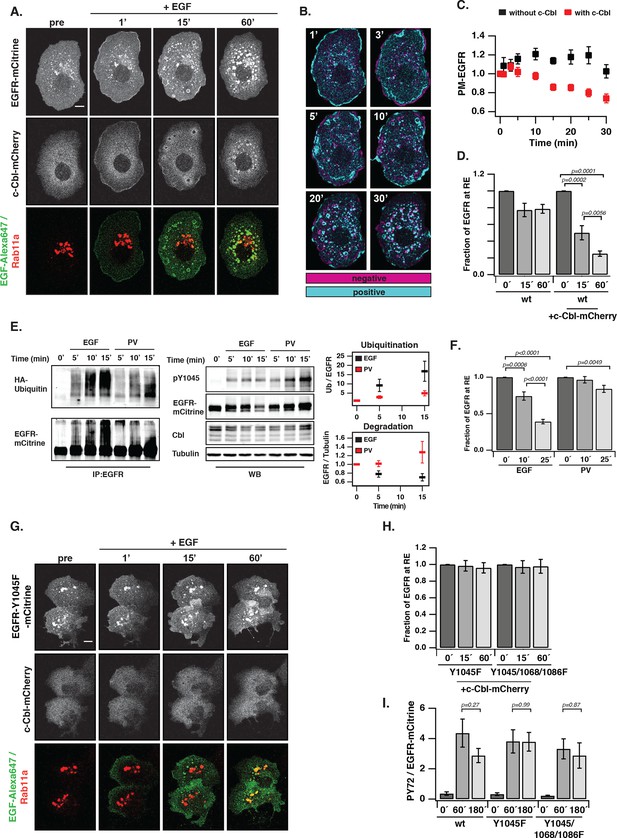
Ubiquitin-mediated switch in ligand-activated EGFR trafficking.
(A) Time-lapse of EGFR and Rab11a co-localization after EGF stimulation of COS-7 cells with Alexa647-labeled EGF (5 ng/ml). Representative fluorescence images of EGFR-mCitrine (first row), c-Cbl-mCherry (second row), and BFP-Rab11a merge with Alexa647-labeled EGF (third row) at the indicated time in minutes. (B) Change in spatial distribution of EGFR-mCitrine upon EGF stimulation calculated as the difference between each time point and the one acquired prior to it, indicates areas with increased (cyan) or decreased (magenta) fluorescence. (C) Plot shows the normalized average fraction ± SEM of EGFR-mCitrine at the PM (PM-EGFR over total EGFR) in the presence (red, n=10 cells) or absence (black, n=6 cells) of c-Cbl-mCherry over time (see 'Materials and methods'). (D) Fraction of EGFR-mCitrine fluorescence at the RE with (n=10 cells) and without (n=6 cells) ectopic expression of c-Cbl-mCherry upon EGF stimulation for the indicated time. (E) Differential ubiquitination of ligand- and autonomously-activated EGFR. COS–7 lysates immunoprecipitated with anti-EGFR (left panel) or blotted for total proteins (middle panel). IP was probed with anti-HA (HA-ubiquitin) and anti-GFP (EGFR-mCitrine) and total lysates were probed with anti-GFP (EGFR-mCitrine), anti-Cbl (c-Cbl-mCherry), anti-tubulin (Tubulin), and anti-pY1045. Right panel: Quantification shows relative EGFR ubiquitination (Ub over EGFR) in the upper graph and degradation (EGFR over Tubulin) in the lower graph upon EGF stimulation (100 ng/ml) or PV treatment (0.33 mM) for the indicated time in minutes (see Figure 5—figure supplement 1A,B). (F) Fraction of EGFR-mCitrine fluorescence at the RE upon stimulation with 100 ng/ml EGF (n=12 cells) or 0.33 mM PV (n=14 cells) for the indicated time (see Figure 5—figure supplement 2). (G) Time-lapse of EGFR-Y1045F-mCitrine and BFP-Rab11a co-localization after EGF stimulation of COS-7 cells with Alexa647-labeled EGF (5 ng/ml). Representative fluorescence images of EGFR-Y1045F-mCitrine (first row), c-Cbl-mCherry (second row), and BFP-Rab11a merge with Alexa647-labeled EGF (third row) at the indicated time in minutes. (H) Fraction of EGFR-Y1045F-mCitrine (n=9 cells) or EGFR-Y1045/1068/1086F-mCitrine (n=9 cells) fluorescence at the RE upon EGF stimulation for the indicated time (min) in the presence of ectopically expressed c-Cbl-mCherry (see Figure 5—figure supplement 3A). (I) Sustained phosphorylation of EGFR-Y1045F-mCitrine and EGFR-Y1045/1068/1086F-mCitrine as compared to EGFR-mCitrine upon EGF stimulation. COS-7 lysates were probed with generic phosphotyrosine (PY72) and anti-GFP (EGFR-mCitrine) and bar diagram shows relative EGFR phosphorylation (PY72/EGFR) upon EGF stimulation (100 ng/ml) for the indicated time (see Figure 5—figure supplement 3B). All blots are n=3 (mean ± SEM). All scale bars: 10 μm. EGFR, epidermal growth factor receptor; SEM, standard error of the mean.
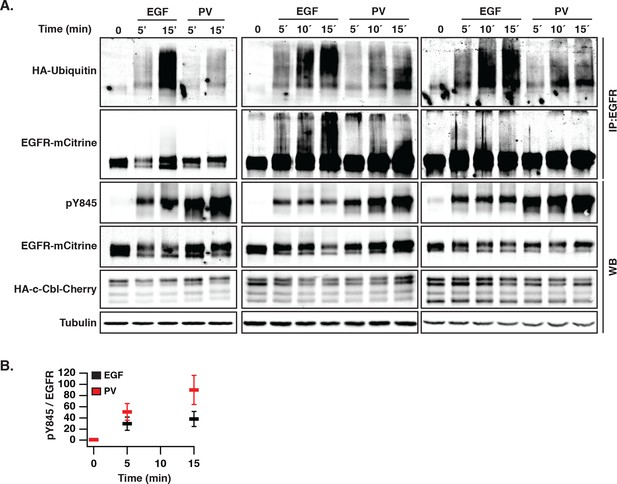
EGF-induced vesicular trafficking of wild-type EGFR-mCitrine and differential ubiquitination of autonomously and ligand-activated EGFR.
(A) Ubiquitination of EGFR. Western blot of COS–7 total cell lysates (total) (WB) and immunoprecipitated EGFR (IP:EGFR) (middle panel) showing time course of ubiquitination and phosphorylation of Y845 after incubation with EGF (100 ng/ml) or PV (0.33 mM). HA-ubiquitin and HA-c-Cbl-mCherry were co-expressed. Total lysates were probed with anti-GFP (EGFR-mCitrine), anti-Cbl (c-Cbl-mCherry), anti-tubulin (Tubulin), and anti-phospho-tyrosine 845 (pY845). IP was probed with anti-HA (HA-ubiquitin) and anti-GFP (EGFR-mCitrine). (B) Quantification shows the relative phosphorylation (mean ± SEM) on Y845 (pY845/EGFR) upon EGF stimulation and PV treatment for the indicated time points. EGFR, epidermal growth factor receptor; PV, pervanadate; SEM, standard error of the mean.
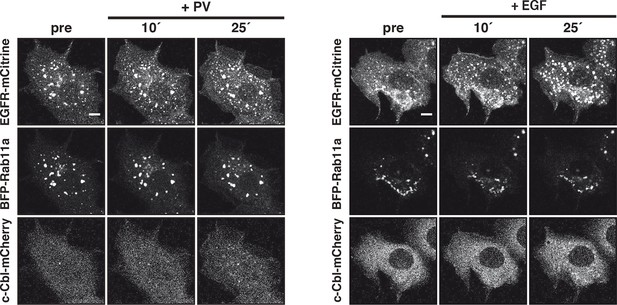
Vesicular trafficking of autonomously versus ligand-activated EGFR to the RE.
Representative fluorescence images of COS–7 cells co-expressing EGFR-mCitrine (upper rows), BFP-Rab11a (middle rows) and c-Cbl-mCherry (lower rows) upon 0.33 mM PV treatment (left panel) or 100 ng/ml EGF stimulation (right panel) for the indicated time in minutes. Scale bars: 10 μm. EGFR, epidermal growth factor receptor; PV, pervanadate; RE, recycling endosome.
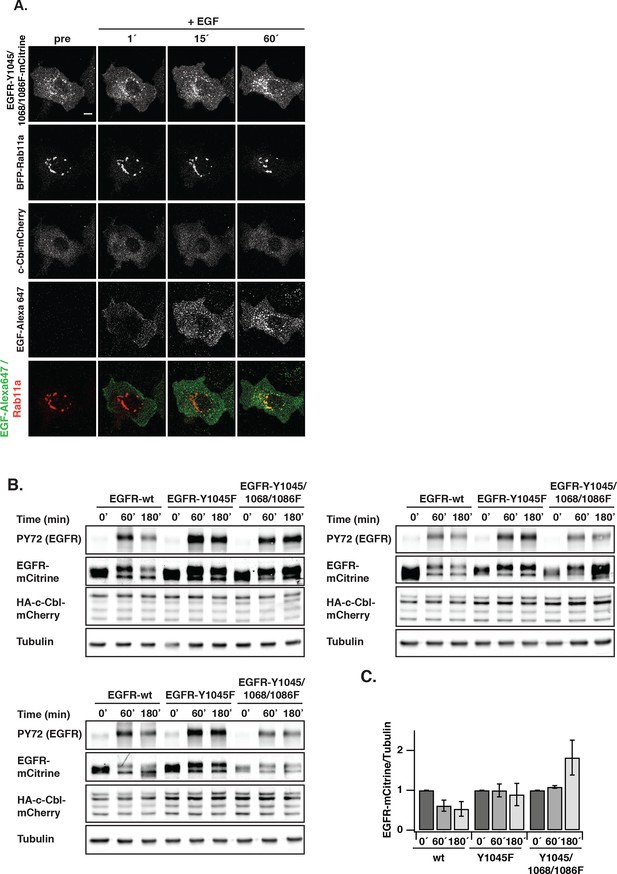
EGF-induced vesicular trafficking of EGFR-Y1045/Y1068F/Y1086F-mCitrine and c-Cbl-mediated degradation profiles of EGFR upon EGF stimulation.
(A) Representative fluorescence images of COS–7 cells co-expressing the ubiquitination-impaired mutant EGFR-Y1045/1068/1086F-mCitrine, BFP-Rab11a, and c-Cbl-mCherry upon stimulation with EGF-Alexa647 (5 ng/ml). Merged images of EGF-Alexa647 (green) and BFP-Rab11a (red) are shown. Scale bar: 10 μm. (B) Lysates of COS–7 cells co-expressing EGFR-mCitrine or EGFR-Y1045F-mCitrine or EGFR-Y1045/1068/1086F-mCitrine and c-Cbl-mCherry and stimulated with EGF (100 ng/ml) for the indicated time points were used in western blot analysis. Blots were probed with the generic phospho tyrosine antibody PY72, anti-GFP (EGFR-mCitrine) and anti-c-Cbl. Anti-tubulin was used as a loading control. (C) Ratiometric quantification of total EGFR-mCitrine/tubulin for EGFR wild type and the ubiquitination-impaired EGFR mutants for the indicated time points (mean ± SEM, n=3 blots). EGFR, epidermal growth factor receptor; SEM, standard error of the mean.
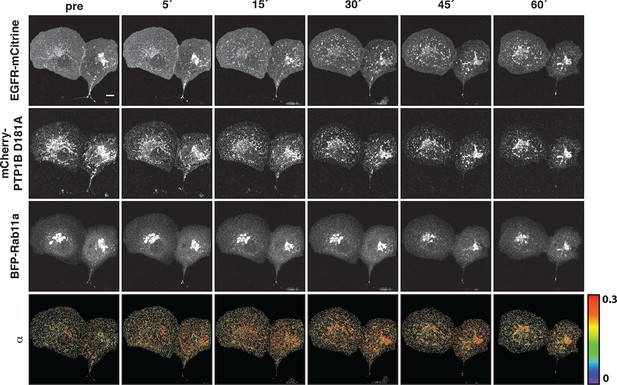
Interaction of EGFR and PTP1B D181A in perinuclear areas upon EGF stimulation.
Representative fluorescence images of living COS–7 cells co-expressing EGFR-mCitrine, PTP1B D181A-mCherry, BFP-Rab11a and α map showing the fraction of interaction between EGFR-mCitrine and PTP1B D181A-mCherry upon EGF stimulation (100 ng/ml) for the indicated time (min). Scale bar: 10 μm. EGFR, epidermal growth factor receptor.
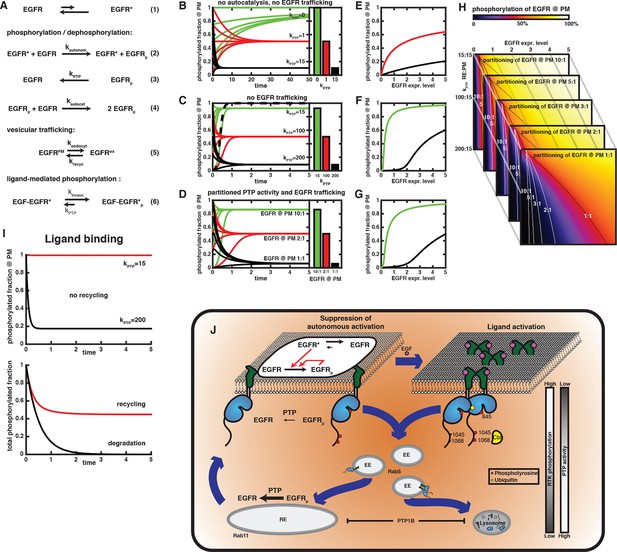
Compartmental model for EGFR spontaneous autocatalytic activation supression.
(A) The set of conversion reactions based on which ordinary differential equations were numerically integrated in Mathematica (see 'Materials and methods'). EGFR fluctuates between an inactive and an active conformation – denoted by star (1). The active conformation phosphorylates EGFR with the rate constant kautonom that encompasses the fraction of active conformation (2). Here, only phosphorylation of an autocatalytic tyrosine is considered. Phosphorylated EGFR is dephosphorylated (kPTP; 3) and can autocatalytically phosphorylate EGFR (kautocat; 4) similar to (2). All species of EGFR exchange between two compartments (from PM to internal – kendocyt – and back – krecyc; 5). kPTP may vary between both compartments. Ligand-binding locks EGFR in the active conformation, irrespective of its phosphorylation state (kkinase over kPTP). (B-–D) Time traces of EGFR phosphorylation and bar diagrams of the corresponding phospho-EGFR steady states at the PM for different parameters (green – phospho-EGFR >80%, red– phospho-EGFR ~50%, black – phospho-EGFR <10%): (B) without autocatalytic feedback or recycling and increasing values of kPTP; (C) with autocatalysis, but no recycling and increasing values of kPTP; and (D) autocatalysis, recycling and kPTP=15 at the PM versus kPTP=200 for the internal EGFR fraction with increasing steady state partitioning of EGFR towards the internal fraction. Dashed line in (C) is in the absence of phosphatase activity (phospho-EGFR rapidly approaches 100%). (E-–G) Steady state phosphorylation of EGFR at the PM for the situations displayed in (B–D) upon varying the total amount of EGFR per cell (expression level). Colors (green, red, black) correspond to the parameters in (B–D). (H) 2D plots of EGFR phosphorylation (color code displayed on top) analogous to the last line plot in (E) for different expression levels of EGFR (x-axis) versus increasing phosphatase activity acting on the internal EGFR fraction (y-axis). Each image corresponds to a different steady state partitioning of EGFR towards the PM (10:1, 5:1, 3:1, 2:1, 1:1); lines in each image mark 50% EGFR phosphorylation (red color) for the corresponding steady state EGFR partitioning. (I) Simulation of EGF-induced activation of EGFR. In the absence of recycling (upper time traces), zeroth order phosphorylation rate of liganded EGFR locked in the active conformation results in a stable phosphorylation of all EGFR for low PTP activity (red curve) or an extremely rapid dephosphorylation for high PTP activity (black curve). Recycling in the presence of partitioned PTP activity (as in (D), kPTP=15 at the PM and kPTP=200 on endomembranes) results in 100% phosphorylation at the PM and low phosphorylation of EGFR on endomembranes. The total phosphorylation of EGFR in the cell (abscissa) is then determined by steady state EGFR partitioning (~50% for equipartitioning, red curve). Only degradation of the internal fraction with ongoing endocytosis allows finite signaling whose duration is determined by the kinetics of endocytosis (black curve). (J) Schematic of switch in EGFR vesicular traffic. Vesicular recycling (thick blue arrows) of EGFR monomers from the PM through the pericentriolar RE within areas of high PTP activity and back continuously suppresses autonomous receptor phosphorylation. Horizontal black arrows: Chemical conversions, vertical red arrows: Causalities (left). Following EGF-binding to the receptor, fully active and phosphorylated dimers share the same entry into early endosomes as the monomer. However, ligand-induced ubiquitination of EGFR clusters mediates a switch in the endocytic trafficking from continuous recycling to unidirectional trafficking. By depleting the pool of monomeric EGFR at the RE, this results in a transient translocation of the receptors to the PM reinforcing activation, and a subsequent depletion from the PM by delayed endocytosis. During endosomal transit from the PM to LEs, receptors signal in the cytoplasm until their dephosphorylation in perinuclear areas with high PTP activity (right) and subsequent degradation in lysosomes.





