The Melanocortin Receptor Accessory Protein 2 promotes food intake through inhibition of the Prokineticin Receptor-1
Figures
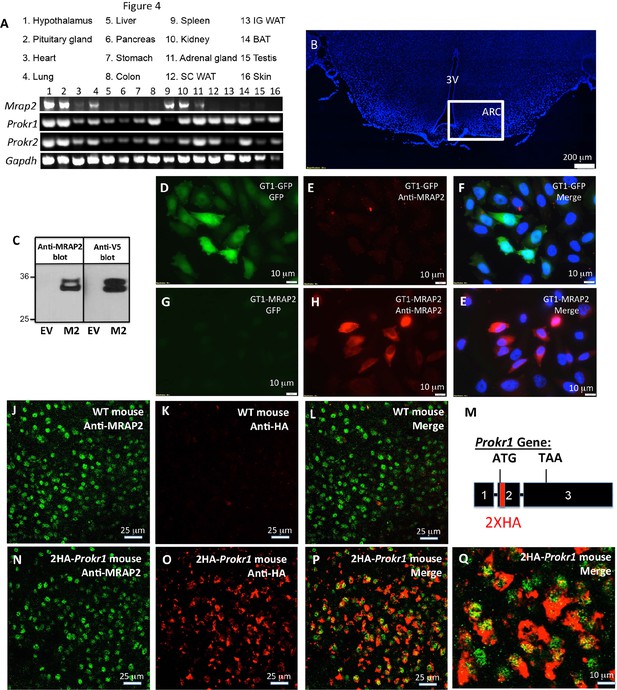
Tissue distribution of PKRs and MRAP2.
(A) Expression of MRAP2, PKR1 and PKR2 mRNA, measured by RT-PCR in tissues harvested from male mice. (B) DAPI-stained mouse brain section containing the hypothalamus. Square depicts the position of the arcuate nucleus. (C) Validation of anti-MRAP2 antibody by western blot on lysates from CHO cells transfected with empty vector (EV) or mouse MRAP2 (M2). (D-I) Validation of the MRAP2 antibody by immunofluorescence using GT1-1 cells stably expressing GFP (D,E and F) or MRAP2 (G,H and E). (J–L) Confocal images of immunofluorescence detecting MRAP2 (in green) or HA (in red) in the arcuate nucleus of a wild type mouse. (M) Schematic representation of the insertion of 2XHA tags after the start codon of the PKR1 gene in mice using CRISPR/Cas9 technology. A more detailed description is depicted in Figure 1—figure supplement 1. (N-Q) Confocal images of immunofluorescence detecting MRAP2 (in green) or HA-PKR1 (in red) in the arcuate nucleus of the 2HA-Prokr1 mouse.
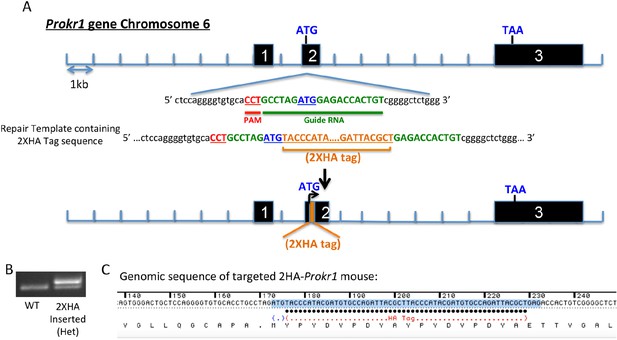
Generation of the 2HA-Prokr1 mouse model.
(A) Schematic representation of the CRISPR/Cas9 strategy targeting the Prokr1 gene for inserting the HA tags. (B) Genotyping of the 2HA- Prokr1 mice. The higher band represents the region around the start codon with the 2HA insertion. (C) Sequencing result of the Prokr1 gene with the 2XHA insert in the successfully targeted mouse.
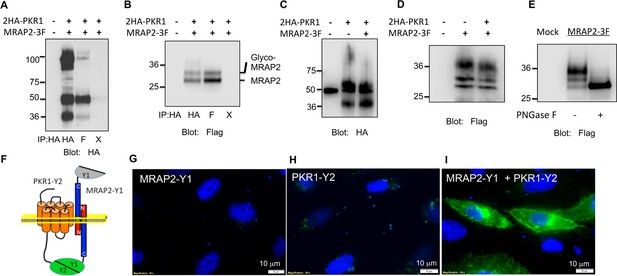
PKR1 and MRAP2 coimmunoprecipitation and localization in live cells.
(A) and (B) Co-immunoprecipitation of 2HA-PKR1 and MRAP2-3Flag from transfected CHO cells. PKR1 was detected using rabbit anti-HA antibody (A), and MRAP2 was detected using anti-Flag antibody (B). X= beads only, no antibody was used for the IP. (C) and (D) Immunoprecipitation of PKR1 (C) or MRAP2 (D) from cells expressing PKR1 alone, MRAP2 alone or both. PKR1 was immunoprecipitated and detected with mouse anti-HA and MRAP2 was immunoprecipitated and detected with mouse anti-Flag. (E) Western blot detecting MRAP2 treated or not with PNGase F. (F) Schematic representation of bimolecular fluorescence complementation (BiFC) between YFP fragments fused to PKR1 and MRAP2. (G, H and E) CHO cells were transfected with MRAP2-Y1 (G), PKR1-Y2 (H) or both (E) Nuclei stained with Hoechst 33,342 are shown in blue and YFP fluorescence in green.
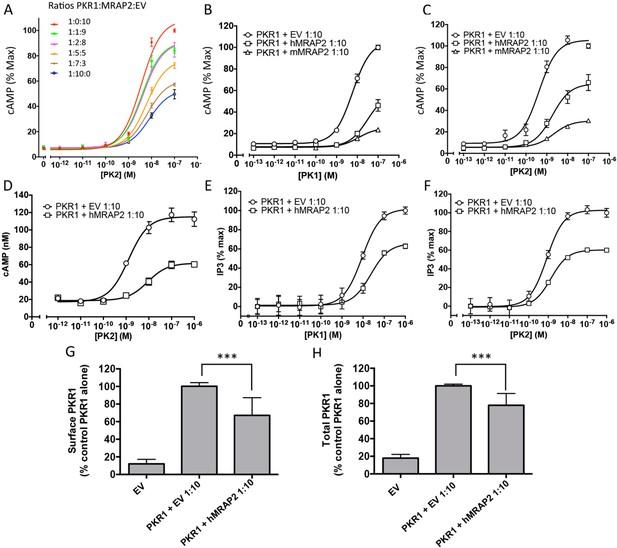
MRAP2 inhibits PKR1 signaling.
CHO cells in a 10 cm dish were transfected with 2.5 μg CRE-luciferase, 225 ng PKR1 and a total of 2.25 μg of plasmids encoding MRAP2 and/or empty vector in the ratios shown. After 24 hr cells were subcultured for use as follows: (A) Cells expressing PKR1 and different amounts of hMRAP2 and/or empty vector were stimulated with PK2 and cAMP responses measured with the CRE-luciferase cAMP assay. (B and C) Cells expressing PKR1 with empty vector, mouse MRAP2 (mMRAP2) or human MRAP2 (hMRAP2) at a 1:10 ratio were stimulated with (B) PK1 or (C) PK2 and cAMP responses measured with CRE-luciferase. (D). Cells expressing PKR1 with empty vector or hMRAP2 were stimulated with PK2 in the presence of 0.1 mM isobutylmethylxanthine and cAMP concentrations measured with the LANCE cAMP assay. (E and F) Cells expressing PKR1 with empty vector or mMRAP2 were stimulated with (E) PK1 or (F) PK2 in the presence of LiCl and IP3 production measured with the IP-One assay. (G) Surface or (H) total expression of PKR1 in cells transfected with empty vector, 2HA-PKR1 and empty vector, or 2HA-PKR1 and hMRAP2 using cell ELISA assays. One-way ANOVA with Tukey post test *p<0.05, **p<0.01, ***p<0.001
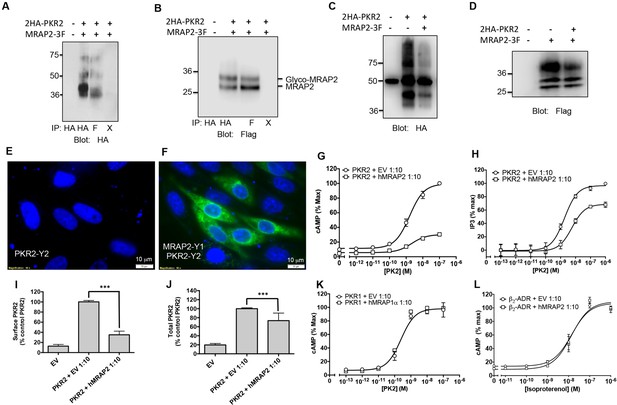
Specificity of MRAP2 regulation.
(A and B) Co-immunoprecipitation of 2HA-PKR2 and MRAP2-3Flag from transfected CHO cells. PKR2 was detected using rabbit anti-HA antibody (A), and MRAP2 was detected using mouse anti-Flag antibody (B). X= beads only, no antibody was used for the IP. (C and D) Immunoprecipitation of PKR2 (C) or MRAP2 (D) from cells expressing PKR2 alone, MRAP2 alone or both. PKR2 was immunoprecipitated and detected using mouse anti-HA antibody. MRAP2 was immunoprecipitated and detected with mouse anti-Flag). (E and F) CHO cells were transfected with PKR2-Y2 (E) or with PKR2-Y2 and MRAP2-Y1 (F). Nuclei stained with Hoechst 33,342 are shown in blue and YFP fluorescence in green. (G and H) cAMP production stimulated by (G) PK1 or (H) PK2 in cells expressing PKR2 with empty vector or hMRAP2. (I) Surface and (J) total expression of PKR2 in cells transfected with empty vector, 2HA-PKR2 alone or 2HA-PKR2 and MRAP2 using cell ELISA assays. (K) PK2-stimulatedcAMP in cells expressing PKR1 with empty vector or hMRAP1α. (L) Isoproterenol-stimulatedcAMP in cells expressing the β2-ADR with empty vector or MRAP2. cAMP responses were measured with the CRE-luciferase assay. One-way ANOVA with Tukey post test *p<0.05, **p<0.01, ***p<0.001
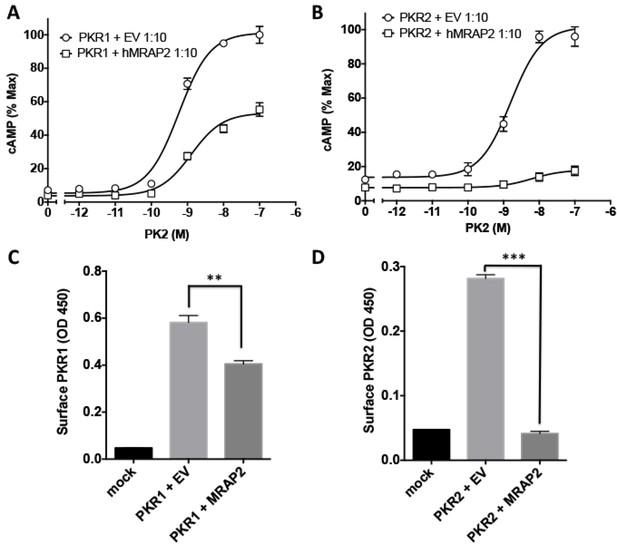
Regulation of PKR1 and PKR2 signaling and trafficking in GT1-1 cells.
(A) GT1-1cells expressing CRE-Luciferase, 2HA-PKR1 and either empty vector or hMRAP2 were stimulated with PK2 and cAMP responses measured with the CRE-luciferase cAMP assay. (B) Cells expressing CRE-Luciferase, 2HA-PKR2 and either empty vector or were stimulated with PK2 and cAMP responses measured with CRE-luciferase. (C and D) ELISA measuring the surface expression of PKR1 (C) or PKR2 (D) in GT1-1 cells in the presence or absence of MRAP2. One-way ANOVA with Tukey post test *p<0.05, **p<0.01, ***p<0.001
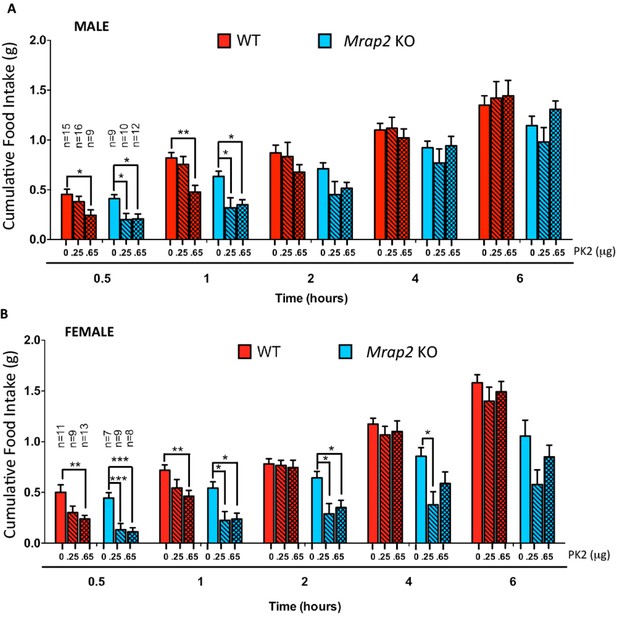
MRAP2 mediated regulation of PKR1 anorexigenic effect.
Cumulative food intake in male (A) and female (B) WT and Mrap2 KO mice after overnight fast and ICV injection of saline or the indicated dose of PK2. One-way ANOVA with Tukey post test *p<0.05, **p<0.01, ***p<0.001.
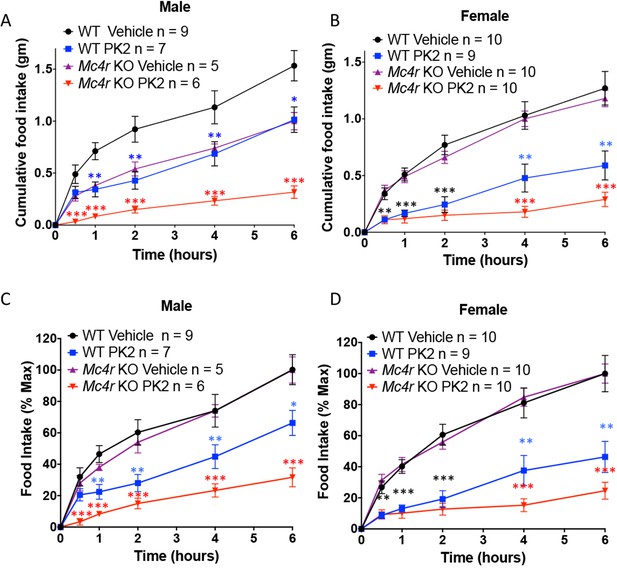
The Anorexigenic effect of PKR1 does not require MC4R.
Cumulative food intake in male (A) and female (B) WT and Mc4r KO mice after overnight fast and ICV injection of saline or the indicated dose of PK2. (C and D) Same results as those depicted in A and B but normalized to the food intake of the 'Vehicle' injected mice of the same genotype at the 6 hr time point. T-test *p<0.05, **p<0.01, ***p<0.001.





