Altered potassium channel distribution and composition in myelinated axons suppresses hyperexcitability following injury
Figures
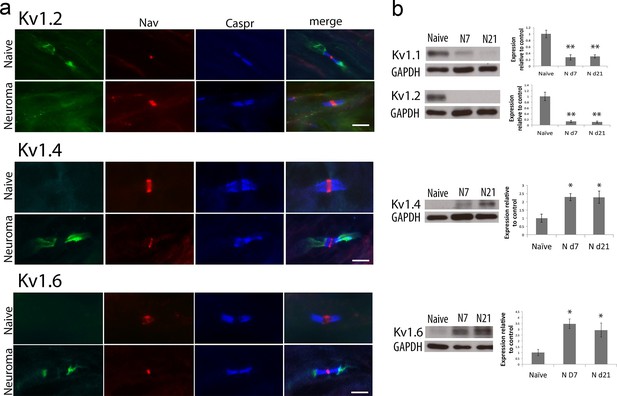
Kv1 channels expression in the naïve nerve and 21 days after sciatic nerve axotomy (note that the samples were collected from the neuroma site).
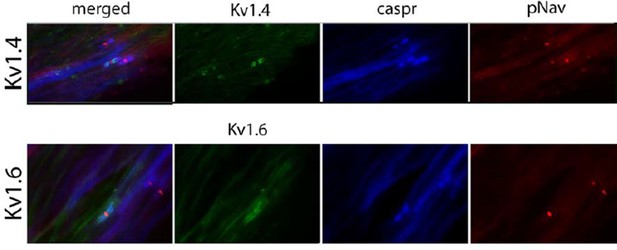
(a) Representative images of longitudinal nerve sections immunostained with Kv1 channels in green (Kv1.2, Kv1.4 and Kv1.6 respectively), a panNav antibody in red (to identify the node), and caspr in blue (to identify the paranode). Kv1.2 is expressed in the juxtaparanode in naïve nerves but it is not present at 21 days after nerve injury. Kv1.4 and kv1.6 are not present in uninjured nerve but are expressed after nerve injury. Note that when Kv1.4 and Kv1.6 are expressed, they are not confined to the juxtaparanode only but invade the paranode. (b) Western blots showing expression of Kv1 channels in the naïve nerve, at 7 and 21 days after axotomy. Kv1.1 and Kv1.2 are expressed in the naïve nerve and down-regulated after axotomy, while Kv1.4 and Kv1.6 have a low/null expression in the naïve nerve and are up-regulated after injury (*p<0.05, **p<0.001, one Way ANOVA, n=6 per group). Scale bars = 5 μm.
-
Figure 1—source data 1
Source data for Figure 1.
- https://doi.org/10.7554/eLife.12661.004
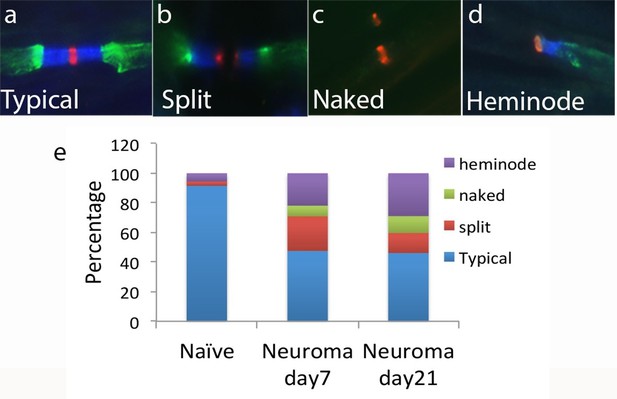
Nav channel expression.
Representative sections of longitudinal nerves immunostained with Kv1.2 in green, a panNav antibody in red (to identify the node), and caspr in blue (to identify the paranode) from neuroma day 21. (a) A typical pattern of Nav expression localized at the node of Ranvier and flanked by caspr staining is shown. The altered forms of Nav channel accumulations seen in the injured nerve included (b) split nodes: These were nodes that had two distinct Nav channels accumulations, separated by a gap in the Nav channels staining within the same fibre and with each Nav channels accumulation flanked on one side with caspr staining, or (c) naked nodes: those Nav channel accumulations that lacked an association with caspr (d) heminodes: nodes where the caspr staining was located on only one side of a contiguous Nav channel accumulation. (e) Quantification of different types of sodium cluster accumulation in the naïve state and after nerve injury is shown.
-
Figure 2—source data 1
Source data for Figure 2.
- https://doi.org/10.7554/eLife.12661.006
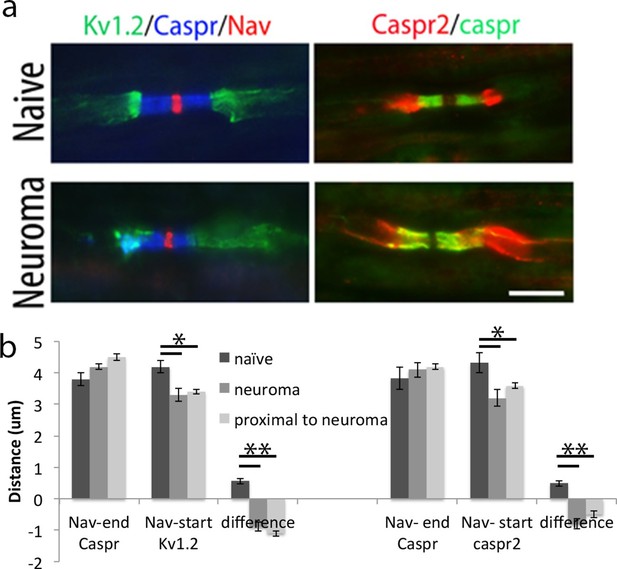
Relocalization ok Kv1.2 and caspr2 at 7 days after neuroma.
(a) Representative longitudinal sections of nerves immuno-stained with Kv1.2 in green, a panNav antibody in red (to identify the node) and caspr in blue (to identify the paranode). Kv1.2 is expressed in the juxtaparanode in naïve nerves but it also co-localized with caspr staining at 7 days after injury. Representative longitudinal sections of nerves immuno-stained with caspr2 in green and caspr in red. Caspr2 is confined to the juxtaparanode in naïve nerve but co-localized with caspr at 7 days after injury. (b) We quantified the distance between the sodium channel staining (Nav) and the end of the caspr staining, distance between the sodium channel staining (Nav) and the start of the Kv1.2/1caspr2 staining, and difference between these distances. A negative value represents an overlap of paranodal and juxtaparanodal proteins. Note that he distance between the sodium channel staining (Nav) and the end of the caspr staining remains unchanged after nerve injury, while the distance between the sodium channel staining (Nav) and the start of the Kv1.2/caspr2 staining is significantly shortened after nerve injury, indicating co-localization of Kv1.2 and caspr2 with caspr (n = 5 animals, 20–41 nodes per animal), p<0.001, one way ANOVA Tukey post hoc tests). We analyzed uninjured (naïve) nerve, nerve at the site of the neuroma (day 7), and nerve 1 cm proximal to the neuroma (day 7). The effect on Kv1.2 re-localization remains the same at the site far from the neuroma. The effect on caspr2 re-localization is slightly smaller at the site 1 cm proximal to the neuroma compared with the neuroma site, but it is still significantly different from the naïve **p<0.001, *p<0.05, PRN = paranode, JXP = juxtaparanode. Scale bars = 5 μm.
-
Figure 3—source data 1
Source data for Figure 3.
- https://doi.org/10.7554/eLife.12661.008
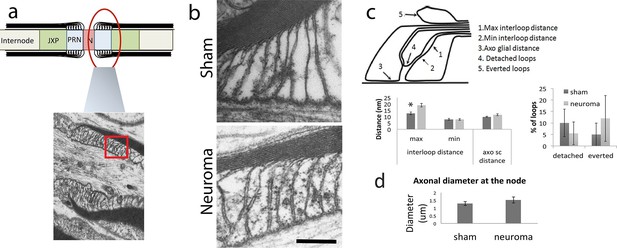
Ultrastructural anatomy of the node of Ranvier within the sciatic nerve following axotomy.
We used electron microscopy to look at the ultrastructure anatomy of the node. (a) Shows a diagram of the node, paranode and juxtaparanode and a low magnification section of this area in a sham-operated nerve. The red box denotes the area that was used for quantification as seen in b. (b) High magnification views of the paranodal loops are shown in the sham and 21 days following axotomy (magnification 135 000x) (c) We quantified different aspects of the attachment of the Schwann cell paranodal loops to the axon. This is illustrated in right panel which denotes the different parameters measured: The maximal and minimal distance between interloops, the distance between the glia and the axon, the number of detached loops and the number of everted loops. We found a significant increase in the maximal distance between loops in the neuroma compared to sham nerves (one way ANOVA, p = 0.005). There were no significant differences in any of the other measurements. (d) We quantified the diameter of the axons at the site of the node and found no difference between the uninjured and injured axons. Scare bars: 200 nm.
-
Figure 4—source data 1
Source data for Figure 4.
- https://doi.org/10.7554/eLife.12661.010
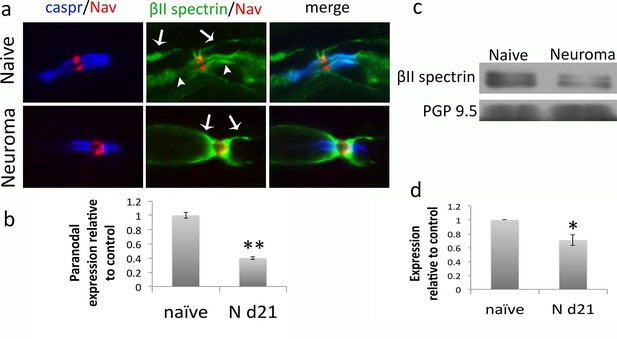
βII spectrin expression in naïve and neuroma nerves.
(a) Representative sections of longitudinal nerves immunostained with βII spectrin in green, a panNav antibody in red (to identify the node), and caspr in blue (to identify the paranode). βII spectrin is expressed both in the surface of Schwann cells (arrows) and in the axon at the paranodal and juxtaparanodal region (arrow heads) in naïve nerves. At 21 days after axotomy (neuroma), βII spectrin can be only seen in the Schwann cell (arrows) but not in the axonal domains. (b) Quantification of βII spectrin immunofluorescence in the paranode (identified by caspr staining) showing a significant reduction in neuroma versus naïve (immunofluorescence normalised to naïve: 0.4 ± 0.02, p<0.001, t-test, n = 50–83 heminodes). (c) Western blots showing expression of βII spectrin in the DRG of naïve and day 21 neuroma. (d) Quantification of WBs. Expression of βII spectrin in the DRG was reduced by 30% after nerve injury. PGP9.5 was used as a loading control (expression relative to naïve: 0.7 ± 0.08, p = 0.04, t-test). **p<0.001, *p<0.05. Error bars denote SEM.
-
Figure 5—source data 1
Source data for Figure 5.
- https://doi.org/10.7554/eLife.12661.012
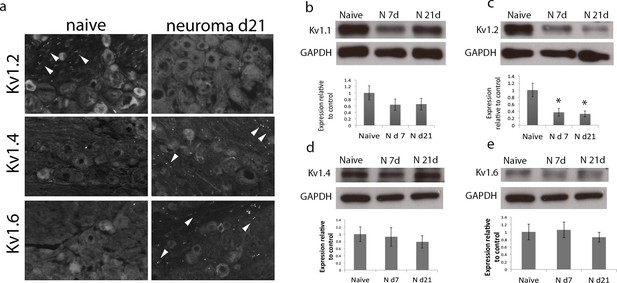
Expression of Kv1 channels in the DRG in the naïve state, 7 and 21 days after axotomy (neuroma).
(a) Representative sections of naive and neuroma day 21 DRG immunostained with Kv1.2, Kv1.4, and Kv1.6. Note that Kv1.2 expression in DRG cells and axonal juxtaparanodes (arrow heads) is reduced after injury, while Kv1.4 and Kv1.6 expression remains unchanged in DRG cells, and it is present in axonal juxtaparanodes (arrow heads) after injury. In each panel (b–e), a representative blot is shown for each time point with GAPDH as a loading control. Quantification of 6 animals per condition is shown below (b,d,e) Expression of Kv1.1, Kv1.4 and Kv1.6 within the DRG does not significantly change after axotomy. (c) Kv1.2 expression is significantly decreased after axotomy. (*p<0.05, one Way ANOVA, n = 6 per group).
-
Figure 6—source data 1
Source data for Figure 6.
- https://doi.org/10.7554/eLife.12661.014
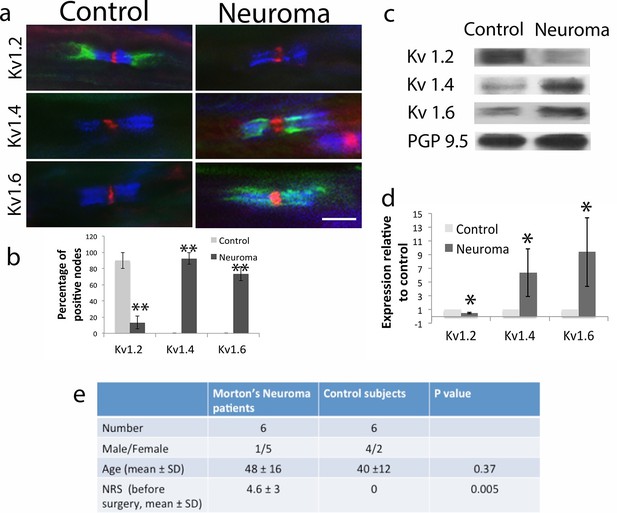
Kv1 channels expression in the sural nerve of healthy volunteers (control) and from patients with painful Morton neuroma.
(a) Representative sections of longitudinal nerves immunostained with Kv1 channels in green (Kv1.2, Kv1.4 and Kv1.6 respectively), a panNav antibody in red (to identify the node), and caspr in blue (to identify the paranode). Kv1.2 is expressed in the juxtaparanode in control nerves but it is not present in the injured nerve. Kv1.4 and kv1.6 are not present in control nerve but are expressed in neuroma within the juxtaparanode and encroaching on the paranode nodes. (b) Quantification of the percentage of Kv1.2, Kv1.4, and Kv1.6 positive nodes in control and neuroma nodes (n = 3 per group, one way ANOVA). (c) Western blots showing expression of Kv1 channels in control and neuroma nerve. (d) Quantification of WBs (n = 6 per group, one way ANOVA). Kv1.2 is expressed in the control nerve and down regulated after axotomy, while Kv1.4 and Kv1.6 have a low/null expression in the control nerve and are up-regulated in neuroma. PGP9.5 was used as a loading control. Error bars denote (e) Patients and control subjects demographic data. The female/male ratio is different in patients with Morton neuroma and controls (patients with traumatic lesion of the hand) reflecting the F/M ratio of these different conditions. The mean age of patients with Morton neuroma is slightly higher than in control subjects, although it is not significant (t-test). All patients with Morton neuroma presented with pain (mean NRS 4.6), while control presented no pain in the area innervated by the sural nerve (Mann Whitney test). NRS: numerical rate score. Error bars denote SEM. Scale bars = 5 μm, **p<0.001, *p<0.05.
-
Figure 7—source data 1
Source data for Figure 7.
- https://doi.org/10.7554/eLife.12661.016
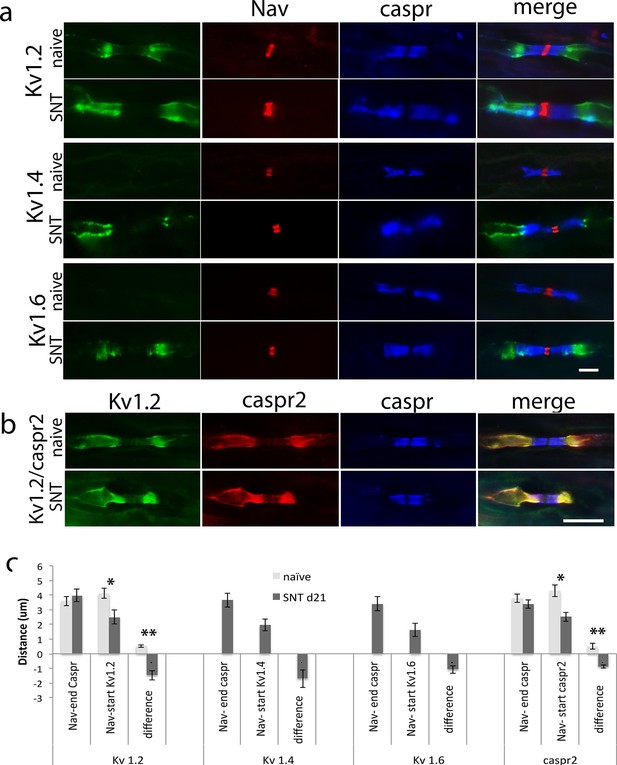
Kv1 channels expression in the dorsal roots of naïve animals and 21 days after spinal nerve transection (SNT).
(a) Representative sections of longitudinal dorsal roots immunostained with Kv1 channels in green (Kv1.2, Kv1.4 and Kv1.6, respectively), a panNav antibody in red (to identify the node), and caspr in blue (to identify the paranode). Kv1.2 is expressed only in the juxtaparanode in naïve nerves but after injury it invades the paranode. Kv1.4 and kv1.6 are not present in uninjured nerve but are expressed within the juxtaparanode after nerve injury and also invade the paranode. (b) Re-localization of caspr2 at 21 days after spinal nerve transection (SNT). Representative sections of longitudinal L5 dorsal roots immuno-stained with caspr2 in red, Kv1.2 in green, and caspr in blue. Kv1.2 and caspr2 are expressed in the juxtaparanode in naïve nerves but co-localized with caspr staining at 21 days after injury. (c) Quantification of: distance between the sodium channel staining (Nav) and the end of the caspr staining, distance between the sodium channel staining (Nav) and the start of the Kv1.2/1.4/1.6/caspr2 staining, and difference between these distances. A negative value in this difference represents an overlap of paranodal and juxtaparanodal proteins. Note that the distance between the sodium channel staining (Nav) and the end of the caspr staining remains unchanged after nerve injury, while the distance between the sodium channel staining (Nav) and the start of the Kv1.2/1.4/1.6/caspr2 staining is significantly shortened after nerve injury. Kv1.4 and Kv1.6 were absent in naive (n = 5 animals/4 sections per animal, *p<0.05, **p<0.001). Scale bars = 5 μm.
-
Figure 8—source data 1
Source data for Figure 8.
- https://doi.org/10.7554/eLife.12661.018
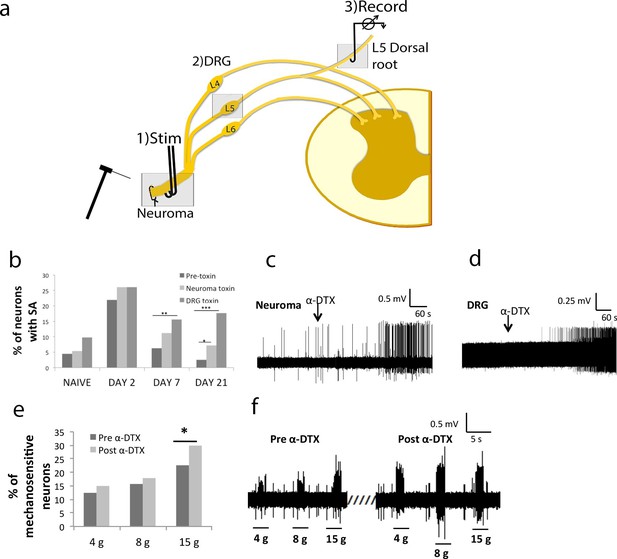
Local application of α-DTX reinstates primary afferent hyperexcitability at later time points following nerve injury.
(a) Schematic illustration of 3-chamber recording system. 1) Recording chamber, 2) middle chamber, 3) stimulating chamber. The toxin was applied in chambers 1 or 2, respectively. (b) Following sciatic nerve transection, there is a large increase in the proportion of primary afferents demonstrating spontaneous activity at day 2, which is suppressed at days 7 and 21 post injury; Local application of α-DTX to the neuroma and L5 DRG at these later time points (days 7 and 21) significantly increases the proportion of afferents, which are spontaneously active (total proportions per group, chi-square tests, *p<0.05, **p<0.01, ***p<0.001) (c) neuroma application and (d) DRG application of α-DTX. Both recordings were carried out 21 days post-surgery. (e) In the presence of α-DTX, significantly more neurons respond to mechanical stimulation at the neuroma site using a 15 g von Frey filament (*p<0.05; total proportions per group, chi-square tests, all neuroma day 21). (f) Representative traces showing greater responsiveness to mechanical stimulation with von Frey filaments after local α-DTX application.
-
Figure 9—source data 1
Source data for Figure 9.
- https://doi.org/10.7554/eLife.12661.020
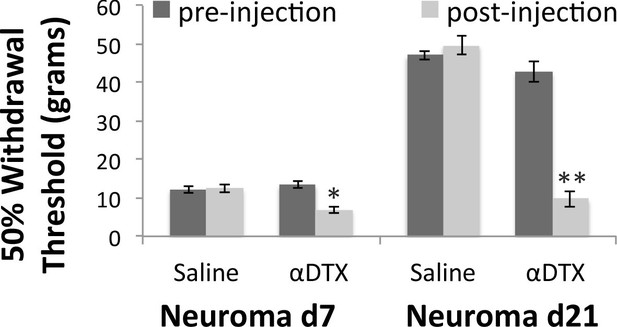
Mechanical hypersensitivity is restored by blocking Kv1 channels.
Mechanical withdrawal thresholds were assessed by applying a range of Von Frey hairs to the skin over the neuroma site (labelled with a suture). Animals were randomised to receive either subcutaneous αDTX or saline 30 min before testing. Hypersensitivity after nerve injury is very pronounced until day 7, when it slowly starts recovering. At this time point, perineuromal application of αDTX reversed this early recovery. At 3 weeks after nerve injury hypersensitivity is much recovered and perineuromal injection of αDTX restored mechanical hypersensitivity to levels seen acutely after injury (RM two way ANOVA, *p<0.05, **p<0.001).
-
Figure 10—source data 1
Source data for Figure 10.
- https://doi.org/10.7554/eLife.12661.022
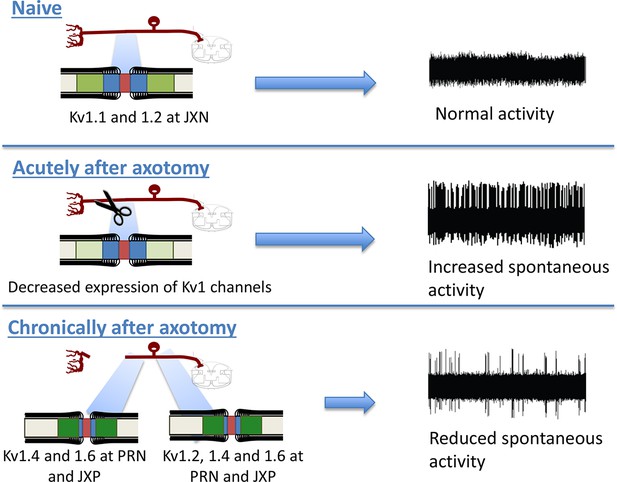
Schematic illustration of the changes in Kv1 channels subunit composition and distribution within the nodal complex and the relationship to hyperexcitability.
In the naïve state, Kv1 channels (Kv1.1 and 1.2 shown in green) are localised to the juxtaparanode and separated from voltage-gated sodium channels at the node (red) by the paranode (blue). Acutely following axotomy myelinated primary afferents show a marked increase in spontaneous activity as a consequence of complex changes in increased pro-excitatory drive (for instance from voltage-gated sodium channels) as well as reduced 'brakes' on excitability At later stages within the neuroma, although there is less expression of Kv1.1 and Kv1.2, the expression of Kv1.4 and 1.6 increases. Remote from the injury within the dorsal root expression of Kv1.1 and 1.2 is maintained and the expression of Kv1.4 and Kv1.4 also increases. Furthermore Kv1 channels are redistributed to the paranode as well as being expressed within the juxtaparanode. These changes are associated with a suppression of hyperexcitability.

Tables
Different antibodies used in the study.
| Antibody | Concentration used IHC WB | Company |
|---|---|---|
| Rabbit anti Pan voltage gated sodium channel (Cat No. S6936) | 1:1000 | Sigma-Aldrich |
| Mouse anti Kv1.2 (K14/16.2) | 1:100 1:500 | UC Davis/NIH NeuroMab Facility |
| Mouse anti Kv1.1 (K36/15.1) | 1:100 1:200 | UC Davis/NIH NeuroMab Facility |
| Mouse anti Kv1.4 (K13/31) | 1:100 1:200 | UC Davis/NIH NeuroMab Facility |
| Mouse anti Kv1.6 (K19/36) | 1:100 1:500 | UC Davis/NIH NeuroMab Facility |
| Guinea Pig anti Caspr | 1:1000 1:1000 | From Dr Manzoor Bhat - UT Health Science Center San Antonio (Bhat et al., 2001) |
| Rabbit anti Caspr2 (ab105581) | 1:500 1:400 | Abcam |
| Rabbit anti Pan Neurofascin | 1:500 | Gift from Prof Peter Brophy- University of Edinburgh (Pomicter et al., 2010) |
| Mouse anti βII spectrin (Clone 42) | 1:500 1:1000 | BD Bioscience |
| GAPDH | 1:10000 | Abcam |
| PGP 9.5 | 1:5000 | Ultraclone |
-
IHC: Immunohistochemistry; WB: Western Blot analysis.





