Mapping quantal touch using 7 Tesla functional magnetic resonance imaging and single-unit intraneural microstimulation
Figures
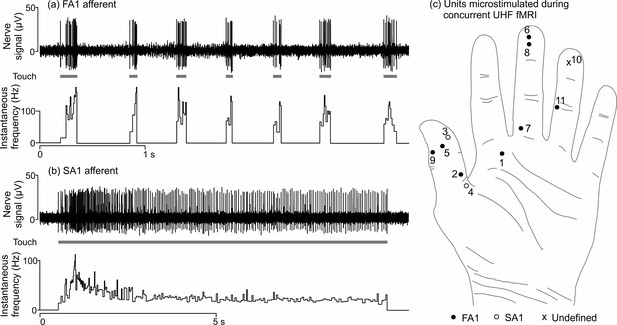
Physiological recordings from mechanoreceptive afferents and the location of afferents that were microstimulated during 7T fMRI.
Example microneurography recording (top) along with the instantaneous firing frequency (bottom) for (a) an FA1 afferent (U1; see Table 1) and (b) an SA1 afferent collected inside the 7T MR scanner environment. In (a), mechanical taps were delivered to the center of the FA1’s receptive field and (b) a long-lasting mechanical indentation was applied at the center of the SA1’s receptive field, using a wooden stick (see gray blocks). (c) Location of the afferents that were microstimulated during 7T fMRI (see Table 1). U9 was located on the right hand, but has been transposed onto the left hand for this schematic. The ‘undefined’ (x) afferent relates to a sensation that was felt as a line, which likely indicates two single afferents in close proximity being stimulated simultaneously.
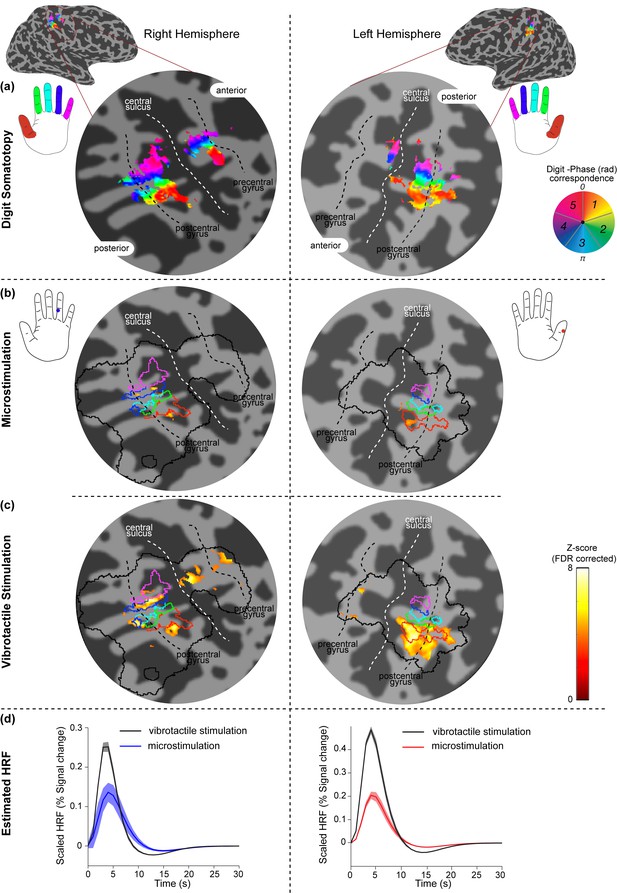
Spatial localization of INMS-induced versus vibrotactile-induced responses in contralateral S1.
Activation maps related to stimulation of two different afferents in Participant 4 are rendered onto a flattened cortical patch spanning the central sulcus of the right (left of figure) and left (right of figure) hemispheres. Dark gray represents the sulci and light gray the gyri. (a) Digit somatotopy, where phase values (in radians) and corresponding preferred stimulus location (fingertip) are shown. Orderly representation of the digits is found on the posterior bank of the central sulcus (white line) and the post-central gyrus (dashed black line), corresponding to S1. (b) Statistical maps (Z > 3.08, FDR-adjusted) from INMS of U11 (left) and U9 (right). BOLD activation is localized within the expected digit ROI identified from digit somatotopy, as shown by the blue (digit 4) and red (digit 1) lines, which denote phase values encoded by the blue (3.77–5.03 rad) and orange (0–1.26 rad) colors respectively. The solid black line indicates the SI hand mask (calculated by dilating the somatotopy map by 5 voxels) within which FDR correction was performed. (c) Statistical maps (Z > 3.08, FDR-adjusted) for vibrotactile stimulation of the corresponding receptive fields of U11 (top) and U9 (bottom). (d) HRF estimated from the GLM analysis for INMS and vibrotactile stimulation averaged across voxels of the ROI (U10, top; U9, bottom). Error bars show voxel-wise parameter standard errors averaged across voxels of the ROI.
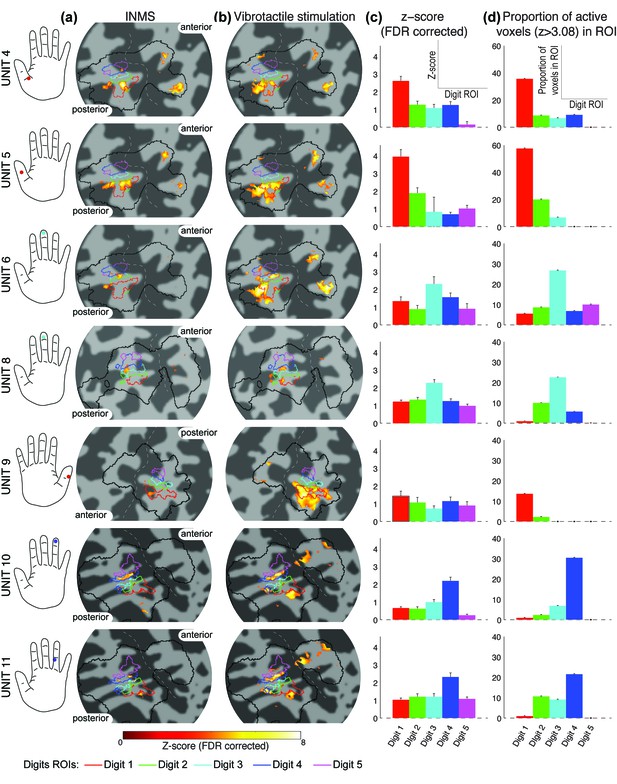
Spread of activation across the digit ROIs identified from the somatotopy.
(a) Statistical maps (Z > 3.08, FDR-adjusted) from INMS of seven single units in participants 2, 3 and 4. In each case the activation map is rendered onto a flattened cortical patch spanning the central sulcus of the right hemisphere. Dark gray represents the sulci and light gray the gyri. The solid black line indicates the SI hand mask (calculated by dilating the somatotopy map by 5 voxels) within which FDR correction was performed. Activation is localized within the expected digit ROI (black line) identified from the digit somatotopy (see color legend). (b) Statistical maps (Z > 3.08, FDR-adjusted) for vibrotactile stimulation of the corresponding receptive field of units. (c) Z-scores (FDR-corrected) of the INMS BOLD response averaged across voxels for each of the digit ROIs identified from the traveling-wave analysis. Error bars indicate standard error across voxels in ROI. (d) Proportion of voxels activated by the INMS paradigm at Z>3.08 (FDR-corrected) for each digit ROI. The source data for plots in panels (c) and (d) are available in the Figure 3—source data 1.
-
Figure 3—source data 1
Source files for plots of Z-score and Proportion of active voxels in each Digit ROI.
This matlab file contains variables for each individual unit (U4, U5, U6, U8, U9, U11) with fields ‘micro_stats’ and ‘vibro_stats’ containing a structure with the results for single unit INMS and vibrotactile stimulation of the unit’s receptive field, respectively. Each structure has the following fields: ‘zetaMean’, ‘betaSem’: (5 digits x 1)-vector containing mean Z-score (FDR-corrected) and standard error across voxels for each Digit ROI; ‘PropActVox’: (5 digits x 1)-vector containing proportion of active voxels (Z>3.08, FDR-corrected) in each Digit ROI; and ‘betaMean’, ‘betaSem’: (5 digits x 1)-vector containing mean GLM parameter estimate and standard error across voxels for each Digit ROI. GLM parameter estimates are not plot in Figure 3 but are used for subsequent group analysis.
- https://doi.org/10.7554/eLife.12812.007
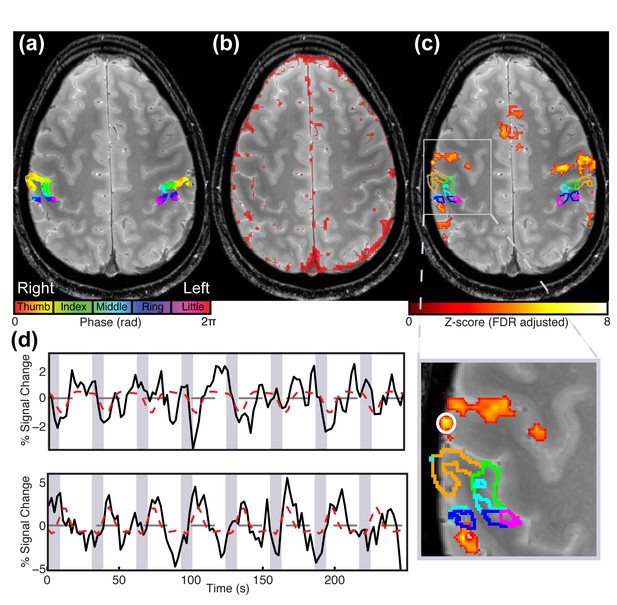
Comparison of contralateral S1 responses to different paradigms for Participant 1.
Statistical maps overlaid on a high resolution T2*-weighted structural image. (a) Digit somatotopic maps obtained with the traveling-wave paradigm for both hands, showing the location of the maps in the posterior bank of the central sulcus. (b) Map of veins identified using T2*-weighted magnitude and phase images. Phase images are unwrapped and high-pass filtered. A map of veins is approximated by thresholding the unwrapped, filtered phase image and convolving the identified voxels with a 2 mm kernel. (c) Statistical maps (Z > 3.08, FDR-adjusted) for INMS of U1. Note, there is no activation in the S1 hand area, as shown by the ROIs delineating each of the digits. (d) Time series of the BOLD response to INMS of U1 for the digit 2 ROI, denoted by the green line in image (upper panel) and of a region of activation co-localized with a vein as indicated by the white circle (lower panel).
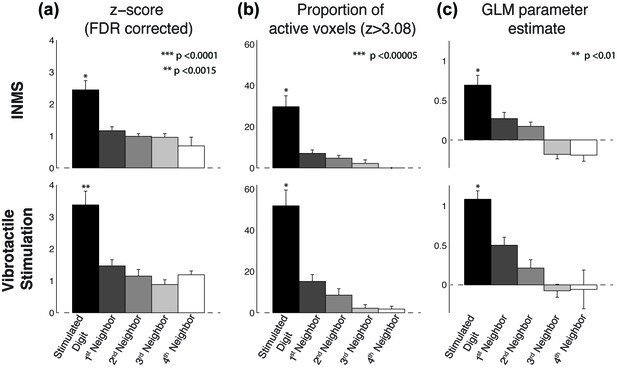
Group analysis (N = 7 units) of the BOLD response to INMS and vibrotactile stimulation of the unit’s receptive field, showing the stimulated digit compared to the neighboring digits.
(a) Z-scores (FDR-corrected) of INMS response in digit ROIs (defined from digit somatotopy) averaged across ROIs for the stimulated digit (N = 7) compared to neighboring digits (1st degree neighbors, N = 11; 2nd degree neighbors, N = 9, 3rd degree neighbors, N = 5, 4th degree neighbors, N = 3). The z-score for the stimulated digit was significantly different to that of neighboring digits. ***p<0.0001, **p<0.005, statistical significance corrected for multiple comparison using Bonferroni correction. (b) Proportion of voxels activated by the INMS (top) and vibrotactile (bottom) paradigm at Z>3.08 (FDR-corrected) for the stimulated digit compared to the neighboring digits. Mean and standard error across ROIs. The proportion of active voxels in the stimulated digit ROI was significantly different to that of neighboring digits. ***p<0.00005, statistical significance corrected for multiple comparison using Bonferroni procedure. (c) GLM parameter estimates of the INMS (top) and vibrotactile (bottom) paradigm for the stimulated digit compared to the neighboring digits. The parameter estimate in the stimulated digit ROI was significantly higher than that of neighboring digits. **p<0.01, statistical significance corrected for multiple comparison using Bonferroni procedure. For all plots (a–c) the mean and standard error across N measures is shown. The source data used for the ANOVA tests are available in the Figure 4—source data 1.
-
Figure 4—source data 1
Source files for ANOVA tests.
This matlab file contains the 2D-matrices (11 x 5), related to each panel in Figure 4, that were used for the 1-way analysis of variance (performed using the ‘anova1’ matlab command). Each matrix row contains data for each of the 7 units (there are up to eleven 1st degree neighboring digit ROIs) and each matrix columns represents the ‘proximity’ to the stimulated digit ROI (stimulated digit ROI, 1st degree, 2nd degree, 3rd degree and 4th degree neighboring digit ROIs). ‘Zeta_micro’ and ‘Zeta_vibro’ are the matrices containing the Z-score (FDR-corrected) values, ‘PerVox_micro’ and ‘PerVox_vibro’ contain the proportion of active voxels (Z>3.08, FDR-corrected) and’ Beta_micro’ and ‘Beta_vibro’ contain the GLM parameter estimates for INMS and vibrotactile stimulation respectively. ANOVA results show a significant difference in mean Z-score (F4,30=14.08, p<10-5; F4,30=12.97, p<10-5), proportion of active voxels (F4,30=16.12, p<10-6; F4,30=17.64, p<10-6) and GLM parameter estimates (F4,30=13.52, p<10-5; F4,30=14.1, p<10-5) across the stimulated and neighboring digit classification (INMS; vibrotactile).
- https://doi.org/10.7554/eLife.12812.010
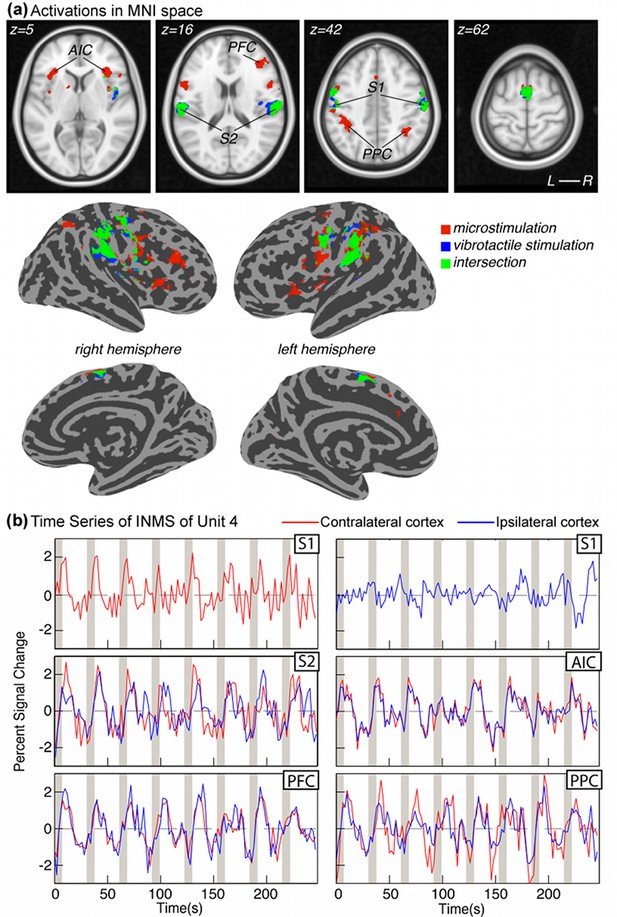
fMRI activation patterns and time courses in cortical areas.
(a) Cortical activation patterns in MNI space. Transverse slices and surface reconstructions showing areas of activation in response to INMS (red clusters) and mechanical vibrotactile stimulation applied directly to the respective unit’s receptive field (blue clusters), as well as areas of overlap (green clusters). Clusters represent common regions of significant activation from all single FA1 units on the left hand (U1, U4, U6, U8, and U11). Individual statistical maps for each afferent were thresholded at Z < 3.08 after correcting for multiple comparisons (FDR) and cluster-corrected at p=0.01, prior to forming the conjunction map. (b) BOLD time courses due to INMS for U4 in different cortical areas. Responses contralateral (right) to the hand stimulation site are shown in red and ipsilateral (left) responses are shown in blue.
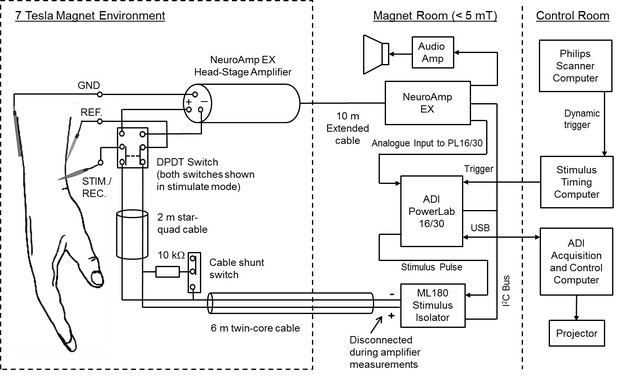
Figure of the experimental setup.
The PowerLab, NeuroAmp EX and ML180 stimulator were placed just inside the magnet room at a field strength not exceeding 5 mT. Placement of the interface equipment within the magnet room was preferred for safety reasons, as isolated cables connected to the participant did not then pass into the control room. The USB interface and trigger cables were passed through the radio frequency shield via a waveguide aperture. An amplifier and loudspeaker was driven from the NeuroAmp EX audio output to give audio feedback to the microneurographer. In addition, a projection of the computer screen could be viewed for visual confirmation of nerve signals. A switch was used to connect the electrodes to either the stimulator or the NeuroAmp head-stage pre-amplifier. In addition, a resistive shunt was placed across the stimulation leads to remove any build-up of charge before connecting or disconnecting the stimulator. Disconnection of the stimulator was necessary because of the high level of noise introduced when it was connected. Star-quad cable was used within the magnet environment to reduce the likelihood of induced currents due to scanner operation affecting the stimulus presentation.
Tables
Mechanoreceptive afferent units in which INMS was performed during 7T fMRI. The table details the unit type and location, as well as the frequency and perception of applied INMS. All units were located on the left hand unless stated.
| Participant | Unit | Type | Location | Physiology | Sensation | Frequency |
|---|---|---|---|---|---|---|
| 1 | 1 | FA1 | Palm | Yes | Buzzing | 30 Hz |
| 2 | 2 | FA1 | Base of digit 1 | Yes | Small dots | 60 Hz |
| 3 | SA1 | Middle of digit 1 | Yes | Pulling | 30 Hz | |
| 4 | SA1 | Base of digit 1 | Yes | Pulling | 30 Hz 60 Hz | |
| 5 | FA1 | Middle of digit 1 | Yes | Vibration | 60 Hz | |
| 6 | FA1 | Digit 3 fingertip | No | Tapping, vibration | 30 Hz 60 Hz 90 Hz | |
| 3 | 7 | FA1 | Base of digit 3 | Yes | Small, round point of tingle sensation | 30 Hz |
| 8 | FA1 | Digit 3 fingertip | No | Small, round point of tingle sensation | 30 Hz 60 Hz 90 Hz | |
| 4 | 9 | FA1 | Middle of digit 1 (right hand) | No | Prickle, flutter | 30 Hz |
| 10 | Undefined | Digit 4 fingertip | No | Small line* | 30 Hz | |
| 11 | FA1 | Middle of digit 4 | No | Flutter | 30 Hz |
-
*A small line sensation is indicative of the simultaneous stimulation of two afferents that are in close proximity.
Cortical areas showing significant activation to INMS of single mechanoreceptive afferents and the corresponding vibrotactile stimulation. Results show the mean and standard error across the five FA1 mechanoreceptive afferents subject to INMS at 30 Hz and corresponding vibrotactile stimulation of the perceived sensation, showing the number of units showing significant activation, MNI coordinates, beta values, Z-score and number of voxels in ROI. R=contralateral, L=ipsilateral. Source files for Table 2—source data 1 and Table 2—source data 2 contain single unit INMS and vibrotactile stimulation results, respectively, for each of the 5 (U1, U4, U6, U8, U11) individual units.
| Single unit INMS | Vibrotactile stimulation | |||||||
|---|---|---|---|---|---|---|---|---|
| ROI | No. Units | x, y, z MNI co-ordinates | Beta | Z | Voxels | Beta | Z | Voxels |
| SI R SI L | 4 3 | 54, -12, 46 -52, -12, 44 | 1.4 ± 0.2 1.2 ± 0.2 | 5.9 ± 0.5 5.6 ± 0.8 | 38 ± 7 20 ± 9 | 1.3 ± 0.3 1.6 ± 0.3 | 5.4 ± 0.3 5.2 ± 0.2 | 41 ± 12 19 ± 1 |
| BA 40 R BA 40 L | 5 4 | 60, -22, 16 -60, -22, 16 | 1.4 ± 0.2 1.5 ± 0.4 | 4.9 ± 0.2 5.3 ± 0.2 | 56 ± 5 73 ± 5 | 1.4 ± 0.1 1.4 ± 0.2 | 4.8 ± 0.2 5.0 ± 0.1 | 54 ± 7 72 ± 12 |
| BA 43 R BA 43 L | 2 3 | 60, -4, 10 -58, -12, 14 | 1.1 ± 0.4 1.0 ± 0.4 | 5.4 ± 0.1 4.8 ± 0.3 | 45 ± 6 33 ± 8 | 1.2 ± 0.4 1.7 ± 0.3 | 4.4 ± 0.2 4.2 ± 0.2 | 30 ± 20 26 ± 11 |
| SMA R SMA L | 5 5 | 4, 0, 60 -2, 0, 60 | 1.2 ± 0.2 1.2 ± 0.2 | 4.8 ± 0.3 4.5 ± 0.3 | 93 ± 27 66 ± 19 | 1.3 ± 0.2 1.2 ± 0.1 | 4.8 ± 0.2 4.5 ± 0.3 | 43 ± 21 29 ± 6 |
| PMC R PMC L | 4 5 | 54, 0, 50 -52, -2, 50 | 0.8 ± 0.2 1.1 ± 0.1 | 4.7 ± 0.2 5.5 ± 0.3 | 36 ± 11 37 ± 7 | 1.1 ± 0.2 1.2 ± 0.1 | 5.0 ± 0.2 4.3 ± 0.1 | 46 ± 9 20 ± 8 |
| M1 R M1 L | 3 2 | 54, -6, 48 -52, -6, 48 | 0.9 ± 0.2 1.5 ± 0.2 | 5.2 ± 0.5 6.3 ± 0.1 | 51 ± 20 66 ± 36 | 0.8 ± 0.2 1.3 ± 0.1 | 5.0 ± 0.7 5.3 ± 0.5 | 31 ± 10 21 ± 3 |
| PIC R PIC L | 5 5 | 46, -2, 10 -42, -2, 10 | 0.8 ± 0.2 0.8 ± 0.1 | 4.2 ± 0.2 4.4 ± 0.2 | 45 ± 12 38 ± 14 | 0.8 ± 0.2 - | 4.7 ± 0.2 - | 27 ± 3 - |
| AIC R AIC L | 4 4 | 34, 26, 4 -32, 26, 4 | 1.2 ± 0.1 1.1 ± 0.1 | 4.7 ± 0.2 4.4 ± 0.2 | 146 ± 20 106 ± 21 | - - | - - | - - |
| PPC R PPC L | 4 5 | 38, -48, 50 -38, -48, 56 | 1.2 ± 0.1 1.0 ± 0.1 | 4.4 ± 0.3 4.4 ± 0.3 | 168 ± 44 172 ± 43 | - - | - - | - - |
| PFC R | 4 | 42, 34,18 | 1.2 ± 0.2 | 4.5 ± 0.3 | 78 ± 22 | - | - | - |
-
Table 2—source data 1
Source files for single unit INMS.
This matlab file contains 2D-matrices (19x5) with the results for single unit INMS for each of the 5 individual units (U1, U4, U6, U8, U11) in each of the 19 ROIs. 'BetaValues’ contains mean across voxels of the beta values, ‘Z-score’ contains the mean Z_score (FDR- corrected) across voxels and ‘NumberVoxels’ contains the number of significant active voxels (Z > 3.08, FDR-corrected) in the ROI. Table 2 summarizes the results by showing the mean and standard error across the 5 units.
- https://doi.org/10.7554/eLife.12812.013
-
Table 2—source data 2
Source files for vibrotactile stimulation.
This matlab file contains 2D-matrices (19 ROIs x 5 units) with the results for vibrotactile stimulation applied to the receptive field for each of the 5 individual units (U1, U4, U6, U8, U11) in each ROI. ‘BetaValues ‘contains mean across voxels of the beta values, ‘Z_score’ contains the mean Z-score (FDR- corrected) across voxels and ‘NumberVoxels’ contains the number of significant active voxels (Z > 3.08, FDR-corrected) in the ROI. Table 2 summarizes the results by showing the mean and standard error across the 5 units.
- https://doi.org/10.7554/eLife.12812.014





