Targeting senescent cells enhances adipogenesis and metabolic function in old age
Figures
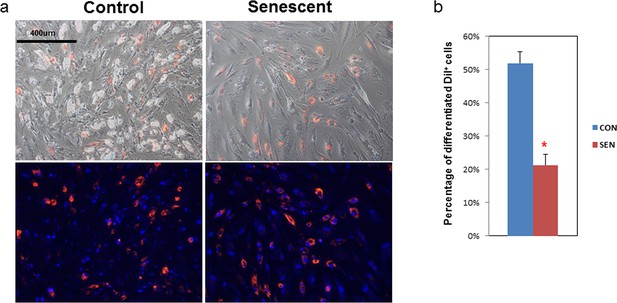
Adipogenesis in human fat progenitors is impeded by co-culture with senescent cells.
Primary subcutaneous human fat progenitors were labelled with DiI and seeded into wells containing either control or radiation-induced senescent preadipocytes. (a) Photographs were taken 15 days after initiating differentiation. Representative images are shown. DiI-positive cells are red and DAPI staining is blue. (b) Number of differentiated DiI positive cells as a percentage of total DiI positive cells is expressed as mean ± s.e.m. *p<0.00001. Results were obtained using separate strains of fat progenitors harvested from 6 healthy human subjects during surgery to donate a kidney (N=6). Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 1—source data 1
Adipogenesis in human fat progenitors is impeded by co-culture with senescent cells.
- https://doi.org/10.7554/eLife.12997.004
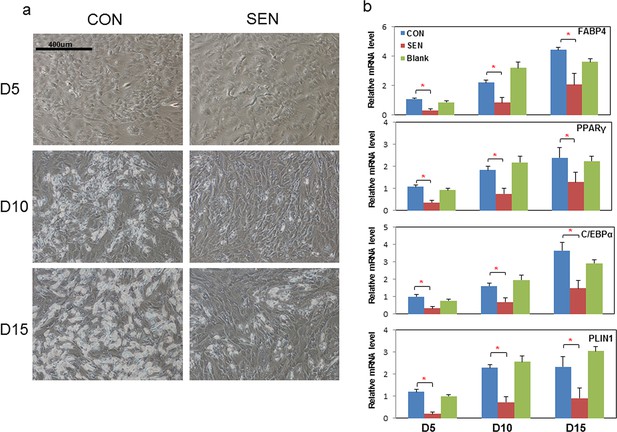
Conditioned medium from senescent cells impedes adipogenesis in human progenitors.
Conditioned medium (CM) was collected from a flask with no cells present (Blank), control non-senescent (CON), and senescent (SEN) fat progenitor cultures. Pooled human progenitors from subcutaneous fat of 5 healthy subjects were treated with 50:50% CM:differentiation medium (DM) for 15 days. (a) Representative images are shown at day 5, 10, and 15 of exposure to CM + DM. (b) Gene expression was analyzed by real-time PCR at day 5, 10, and 15 of exposure to CM + DM. Results are shown as fold change relative to the CON group at day 5. Results were obtained using CM from 5 strains of human primary fat progenitors from different subjects and expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 2—source data 1
Conditioned medium from senescent cells impedes adipogenesis in human progenitors.
- https://doi.org/10.7554/eLife.12997.006
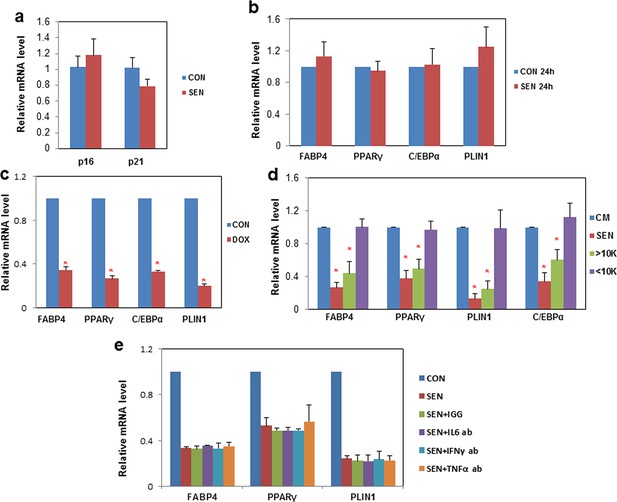
Senescent cells impede adipogenesis in fat progenitors.
(a) CM was collected from control non-senescent (CON) and senescent (SEN) fat progenitor cultures. Pooled human progenitors from subcutaneous fat of 5 healthy subjects were treated with 50:50% CM:DM for 15 days. Gene expression was analyzed by real-time PCR. Results were obtained using CM from 4 strains of human primary fat progenitors and expressed as mean ± s.e.m. (b) Pooled fat progenitors were pre-treated with CM collected from control (CON 24h) and senescent (SEN 24h) cells for 24 hours. Then they were treated with DM for 15 days. Gene expression was analyzed by real-time PCR. Results were obtained from 4 strains of human primary fat progenitors and expressed as mean ± s.e.m. (c) CM was collected from non-senescent (CON) and doxorubicin-induced senescent (DOX) fat progenitor cultures. Pooled human progenitors were treated with 50:50% CM:DM for 15 days. Gene expression was analyzed by real-time PCR. Results were obtained using CM from 3 strains of human primary fat progenitors and expressed as mean ± s.e.m. *p<0.05 (d) CM was collected from non-senescent (CON) and senescent (SEN) fat progenitor cultures. CM from SEN was separated into two fractions using molecular size filters with a cutoff at ~10 kd. The volumes of the fraction larger than ~10kd (>10k) and the fraction smaller than ~10kd (<10k) were matched to CM from SEN using blank CM. Pooled human fat progenitors were treated with 50:50% CM:DM for 10 days. Gene expression was analyzed by real-time PCR. Results were obtained using CM from 3 strains of human primary fat progenitors and expressed as mean ± s.e.m. *p<0.05. (e) CM was collected from control non-senescent (CON) and senescent (SEN) fat progenitor cultures. Pooled human progenitors from subcutaneous fat of 5 healthy subjects were treated with 50:50% CM:DM for 5 days in presence of 20μg/ml of IGG (SEN+IGG), IL6 antibody (SEN+IL6 ab), IFNγ antibody (SEN+ IFNγ ab) or TNFα antibody (SEN+TNFα ab). Gene expression was analyzed by real-time PCR. Results were obtained using CM from 2 strains of human primary fat progenitors and expressed as mean ± s.e.m. Two-tailed Student's t tests were used to determine statistical significance.
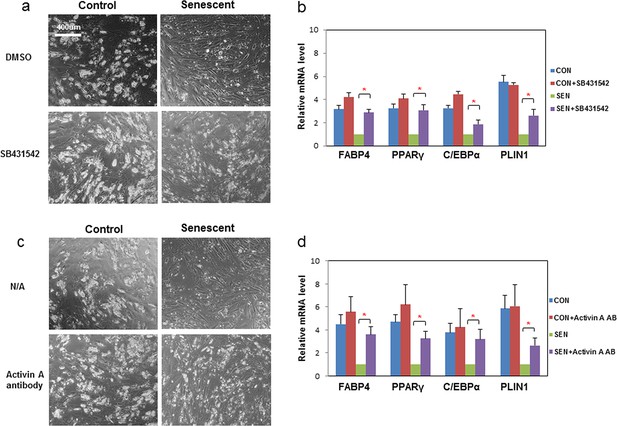
Inhibition of activin A alleviates the impairment of adipogenesis induced by senescent progenitors.
CM was collected from control (CON) and senescent (SEN) fat progenitors. Pooled human progenitors were treated with a 50:50 mixture of CM:DM in the presence of DMSO or 5μM SB431542 (SB431542). (a) Representative images are shown of differentiated cells at day 15. (b) RNA was collected 7 days after differentiation and real-time PCR was performed. Pooled human progenitors were treated with a 50:50 mixture of CM:DM in the presence or absence of 1μg/ml activin A neutralizing antibody (Activin A AB). (c) Representative images are shown of differentiated cells at day 15. (d) RNA was collected 7 days after differentiation and real-time PCR was performed. Results are shown as fold change relative to the SEN group. Results were obtained using CM from 5 strains of human primary cells from different subjects and expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 3—source data 1
Inhibition of activin A alleviates the impairment of adipogenesis induced by senescent progenitors.
- https://doi.org/10.7554/eLife.12997.009
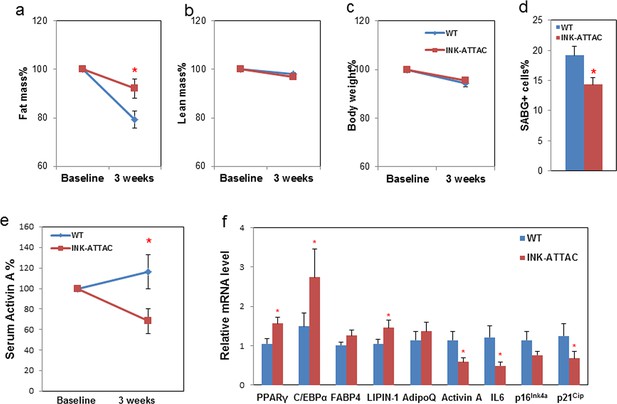
Genetic clearance of senescent cells blunts fat loss and increases adipogenic markers in fat of 18-month-old mice.
Eighteen-month-old wild-type and INK-ATTAC+/- mice were treated with AP20187 for 3 weeks (10mg/kg, three consecutive days with 14 days rest between treatments; total 6 treatments). Fat mass (a) and lean mass (b) were measured by MRI along with body weight (c) before and after treatment. The percent changes relative to baseline are shown. Results (N=8) are expressed as mean ± s.e.m. *p<0.05 for comparison between WT and INK-ATTAC+/- at 3 weeks. (d) SABG+ cells were counted in WAT and their percentages as a function of total cells (N=7) are expressed as mean ± s.e.m. *p<0.05. (e) Activin A protein in plasma was measured before and after treatment. The percent changes relative to baseline are shown. Results (N=8) are expressed as mean ± s.e.m. *p<0.05 for comparison between WT and INK-ATTAC+/- at 3 weeks. (f) RNA from white adipose tissue (WAT) was collected and real-time PCR was performed. Results (N=8) are expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 4—source data 1
Genetic clearance of senescent cells blunts fat loss and increases adipogenic markers in fat of 18-month-old mice.
- https://doi.org/10.7554/eLife.12997.011
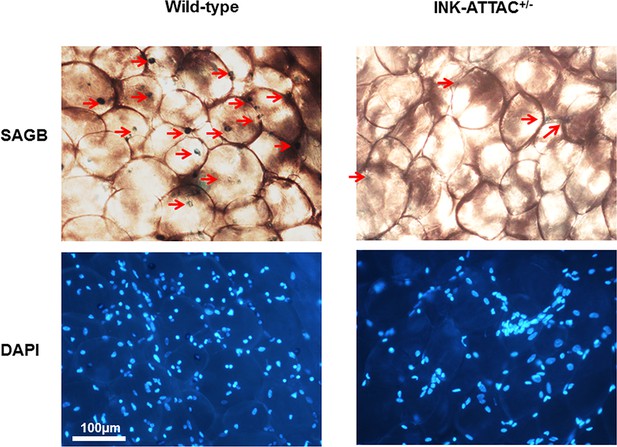
Genetic clearance of senescent cells reduced SABG+ cells in adipose tissue.
Eighteen-month-old wild-type and INK-ATTAC+/- mice were treated with AP20187 for 3 weeks (10 mg/kg, three consecutive days with 14 days rest between treatments; total 6 treatments). WAT was collected and assayed for cellular SABG activity and counterstained with DAPI. The SABG+ cells are indicated by red arrows.
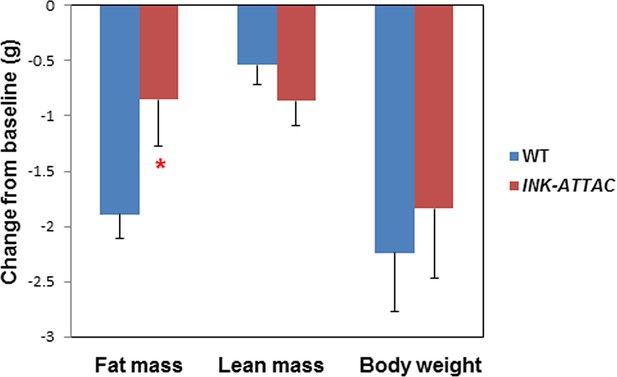
Senescent cell clearance blunts fat loss in 18-month INK-ATTAC+/- mice.
Eighteen-month-old wild-type and INK-ATTAC+/- mice were treated with AP20187 for 3 weeks (10mg/kg, three consecutive days with 14 days between treatments; total 6 treatments). Changes from baseline for fat mass, lean mass, and body weight are shown. Results (N=8) are expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
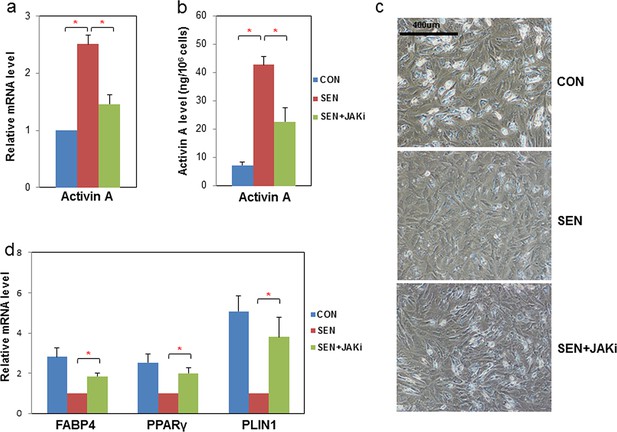
JAK inhibition suppresses activin A production by senescent fat progenitors and partially rescues adipogenesis.
Senescent human progenitors were treated with DMSO (SEN) or 0.6 μM JAK inhibitor 1 (SEN+JAKi) for 72 hours. (a) RNA was collected from control (CON), SEN, and SEN+JAKi progenitors and real-time PCR was performed. Results (N=7) are expressed as mean ± s.e.m. *p<0.05. (b) CM was collected and activin A protein was assayed by ELISA. Results (N=6) are expressed as mean ± s.e.m. *p<0.05. (c) Representative images are shown of differentiating cells at day 10. (d) RNA was collected 10 days after initiation of differentiation and real-time PCR was performed. Results are shown as fold change relative to the SEN group. Results were obtained using CM from 7 strains of human primary progenitors from different subjects and expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 5—source data 1
JAK inhibition suppresses activin A production by senescent fat progenitors and partially rescues adipogenesis.
- https://doi.org/10.7554/eLife.12997.015
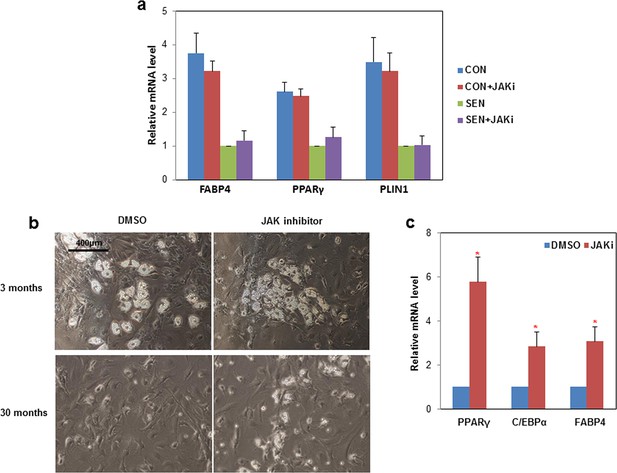
Impaired adipogenesis due to effects of senescent cells is partially rescued by JAK inhibition.
(a) CM was collected from non-senescent (CON) and senescent (SEN) fat progenitor cultures. JAK inhibitor 1(0.6 µM) was directly added into CON (CON+JAKi) and SEN (SEN+JAKi) CM. Pooled human fat progenitors were treated with 50:50% CM:DM for 10 days. Gene expression was analyzed by real-time PCR. Results were obtained using CM from 3 strains of human primary fat progenitors and expressed as mean ± s.e.m. (b) Rat fat progenitors were isolated from 3 and 30-month old rats. These cells were differentiated in presence of DMSO or 0.6µM JAK inhibitor 1. Representative pictures were shown 48 hours after initiation of differentiation. (c) Gene expression was analyzed by real-time PCR in fat progenitors from 30-month old rats. Results (N=4) are expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
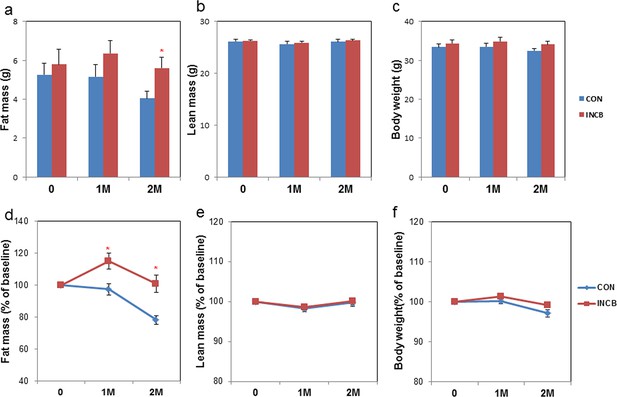
JAK inhibition reduces age-related fat loss in mice.
Twenty-two-month old male mice were treated with vehicle (CON) or ruxolitinib (INCB) for 8 weeks. Fat mass (a) and lean mass (b) were measured by MRI along with body weight (c) before treatment, as well as 1 month and 2 months after treatment. The percent changes relative to baseline are shown for fat mass (d), lean mass (e), and body weight (f). Results (N=9) are expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 6—source data 1
JAK inhibition reduces age-related fat loss in mice.
- https://doi.org/10.7554/eLife.12997.018
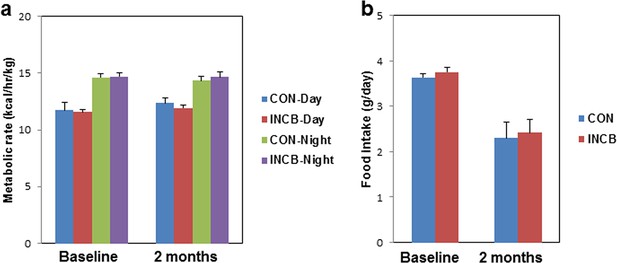
JAK inhibition did not affect metabolic rate or food intake in aged mice.
Twenty-two-month old male mice were monitored using CLAMS before and after 8 weeks of vehicle (CON) or ruxolitinib (INCB) treatment. (a) Metabolic rate and (b) food intake (N=7) are expressed as mean ± s.e.m. Two-tailed Student's t tests were used to determine statistical significance.
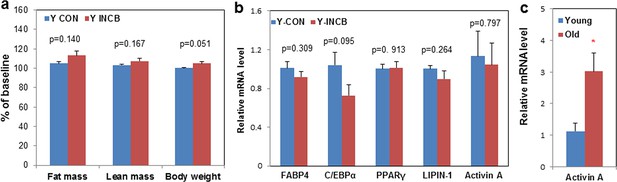
JAK inhibition had less impact on body composition and adipogenesis in 8-month old mice compared to 22-month old mice.
Eight-month old male mice were treated with vehicle (Y CON) or ruxolitinib (Y INCB) for 8 weeks. (a) Fat mass, lean mass, and body weight were measured before and one month after treatment. The percent changes relative to baseline (N=6) are expressed as mean ± s.e.m. (b) RNA from WAT was isolated and real-time PCR was performed. Results (N=6) are expressed as mean ± s.e.m. (c) WAT was collected from 8-month old (Young) and 22-month old mice (Old). RNA was isolated and real-time PCR was performed. Results (N=6 for Young, N=8 for Old) are expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
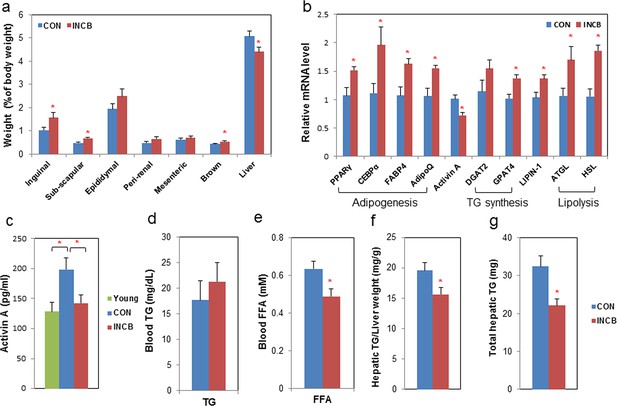
JAK inhibition increases adipogenic markers in adipose tissue and decreases circulating free fatty acids in aged mice.
Twenty-two-month old male mice were treated with vehicle (CON) or ruxolitinib (INCB) for 8 weeks. (a) Weights of different fat depots and liver are shown as percent of whole body weight. Results (N=9) are expressed as mean ± s.e.m. *p<0.05. (b) RNA from WAT was isolated and real-time PCR was performed. Results (N=8) are expressed as mean ± s.e.m. *p<0.05. (c) Plasma activin A protein levels were assayed by ELISA in parallel from 8 six-month-old male mice (Young). Results (N=15 for CON and INCB, N=8 for Young) are expressed as mean ± s.e.m. *p<0.05. Plasma TG (d) and FA (e) levels are expressed as mean ± s.e.m. (N=8). *p<0.05. (f) Hepatic TG/protein levels are expressed as mean ± s.e.m. (N=11). (g) Total hepatic TG levels are expressed as mean ± s.e.m. (N=11). Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 7—source data 1
JAK inhibition increases adipogenic markers in adipose tissue and decreases circulating free fatty acids in aged mice.
- https://doi.org/10.7554/eLife.12997.022
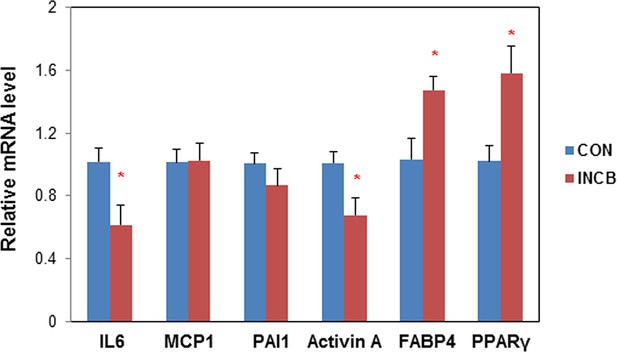
JAK inhibition in aged mice suppressed activin A expression in primary fat progenitors.
Twenty-two-month old male mice were treated with vehicle (CON) or ruxolitinib (INCB) for 8 weeks. Fat progenitors were isolated from WAT and gene expression was analyzed by real-time PCR. Some progenitors were pooled from several mice within the same treatment group due to limited yield of cells. Results (N=5 pools, each from different sets of mice) are expressed as mean ± s.e.m. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
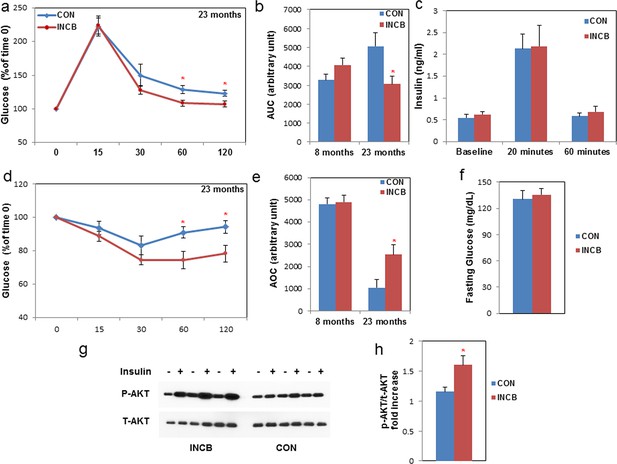
JAK inhibition increases insulin sensitivity in aged mice.
Seven-month old and twenty-two-month old male mice were treated with vehicle (CON) or ruxolitinib (INCB) daily. An oral glucose tolerance test was performed after 5 weeks of treatment. (a) Glucose level was monitored over 120 minutes for 22-month old mice (the results for 7-month old mice are shown in Figure 8—figure supplement 1) and (b) the area under the curve (AUC) was calculated. Results (N=6 for CON and INCB groups of 8-month-old mice, N=9 for CON and INCB groups of 22-month-old mice) are expressed as mean ± s.e.m. *p<0.05. (c) Plasma insulin was measured at baseline, 20 minutes, and 60 minutes after oral glucose gavage. Results (N=9) are expressed as mean ± s.e.m. *p<0.05. An insulin tolerance test was performed after 6 weeks of the treatment. (d) Glucose was monitored over 120 minutes for 22-month old mice (the results for 7-month old mice are shown in Figure 8—figure supplement 1) and (e) area over curve (AOC) was calculated. Results (N=9) are expressed as mean ± s.e.m. *p<0.05. (f) Fasting glucose levels (N=9) are expressed as mean ± s.e.m. *p<0.05. (g) WAT tissue was collected and cultured in CM with or without 5 nM insulin for 5 minutes at 37oC and tissue lysates were then prepared. p-AKT (Ser473) and total AKT protein abundance were assayed. Representative images are shown. (h) These signals were quantified by densitometry using ImageJ. The ratios of p-AKT/total AKT are expressed as mean ± s.e.m. N=6. *p<0.05. Two-tailed Student's t tests were used to determine statistical significance.
-
Figure 8—source data 1
JAK inhibition increases insulin sensitivity in aged mice.
- https://doi.org/10.7554/eLife.12997.025
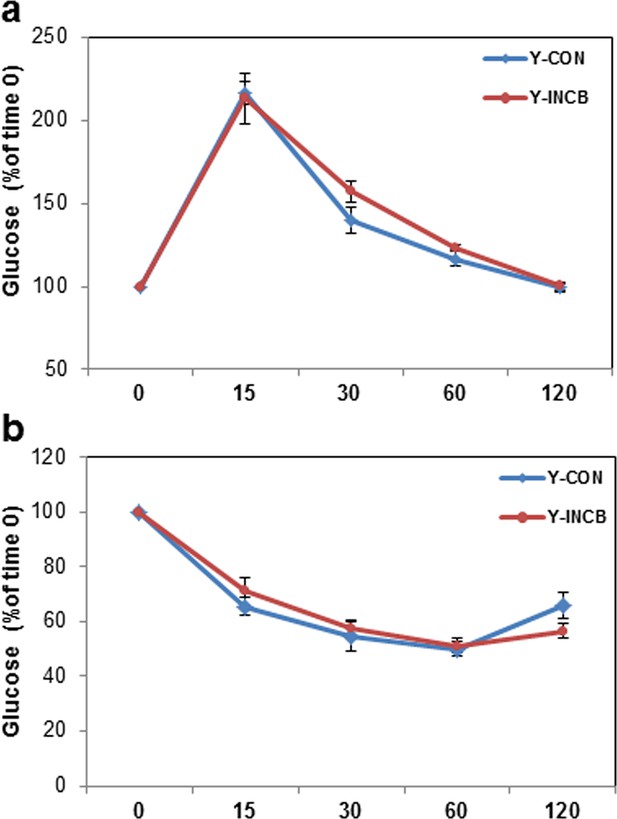
JAK inhibition had less impact on glucose tolerance and insulin sensitivity in 8-month old mice compared to 22-month old mice.
Eight-month old male mice were treated with vehicle (CON) or ruxolitinib (INCB) for 6 weeks. (a) An oral glucose tolerance test was performed after 5 weeks of treatment. Blood glucose was monitored over 120 minutes. Results (N=6) are expressed as mean ± s.e.m. (b) An insulin tolerance test was performed after 6 weeks of the treatment. Blood glucose was monitored over 120 minutes. Results (N=6) are expressed as mean ± s.e.m. Two-tailed Student's t tests were used to determine statistical significance.





